Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (6): 945-951.doi: 10.3969/j.issn.2095-4344.0073
Previous Articles Next Articles
Macromolecular crowding agents for the extracellular matrix: mechanisms and unfavorable factors to be comfirmed
- Medical School of Shanghai Jiao Tong University, Shanghai 200001, China
-
Received:2017-09-21Online:2018-02-28Published:2018-02-28 -
Contact:Zhang Zhi-yong, Medical School of Shanghai Jiao Tong University, Shanghai 200001, China -
About author:Geng Ying-nan, Studying for master’s degree, Medical School of Shanghai Jiao Tong University, Shanghai 200001, China -
Supported by:the National Natural Science Foundation of China, No. 81572137
CLC Number:
Cite this article
Geng Ying-nan, Wu Ding-yu, Zhang Zhi-yong, Wei Min. Macromolecular crowding agents for the extracellular matrix: mechanisms and unfavorable factors to be comfirmed[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(6): 945-951.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
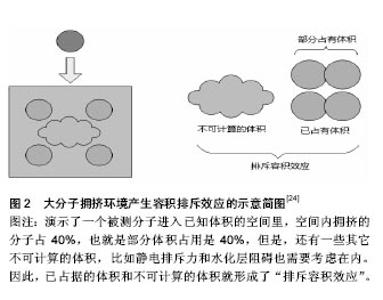
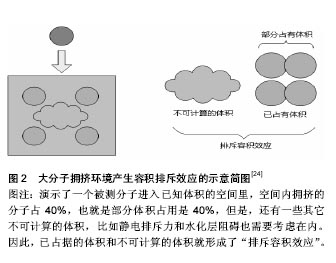
2.1 什么是大分子拥挤?它是如何作用的? 事实上,大分子拥挤就是细胞内外一种理想化的生理环境特征,是由各种各样具有多分散性的惰性大分子,在培养媒介中占据一定的空间形成,由此形成了部分体积占用[23],也就是大分子占据总细胞体积的分数,而这种现象是通过一种“排斥容积效应”(图2)发挥作用的。“排斥容积效应”最早也是由 Minton提出的,它强调源于空间排斥的纯物理非特异性效应[5,24-26],细胞总体积有很大一部分被大分子占据,因而这些被占据的体积不能被其他分子利用,从而使每一个大分子的实际有效空间显著减少,并使分子内和分子间作用受到影响,促进或减弱相应的化学反应。后来,Minton对空间排斥力和化学作用对大分子拥挤的相关作用进行了定量研究,结果发现二者是共同影响大分子拥挤的[27-28]。"
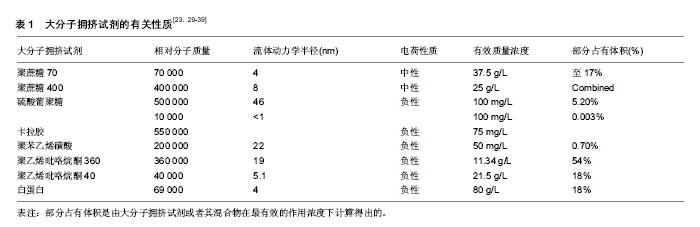
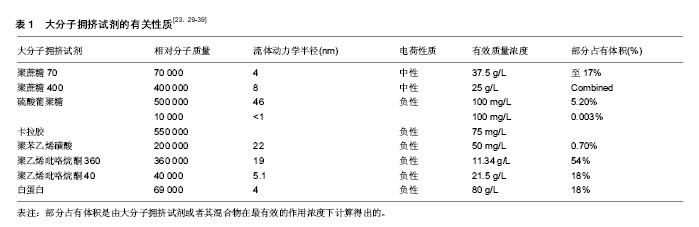
2.2 大分子拥挤试剂的应用 2.2.1 以前试验中用到过的大分子拥挤试剂 有研究证明,一些大分子比如角叉聚糖(相对分子质量550 000)、硫酸葡聚糖(相对分子质量500 000)、聚蔗糖(相对分子质量70 000/400 000)等不仅使细胞外基质成分增加了80倍[29],而且保持了永久的细胞分化表型,即使是在培养液和血清浓度很低的培养条件下也能达到这种状态。此外,4-苯乙烯磺酸钠聚酯(相对分子质量200 000)和聚乙烯吡咯烷酮(相对分子质量40 000/360 000)也都在实验研究中应用过[23,30]。其中,聚乙烯吡咯烷酮是一种比较新颖的很有趣的复合物,已被广泛应用到医药和食物加工生产中,并且在细胞培养和组织工程领域具有创新性很大的广阔前景。 为了更好地了解大分子拥挤,作者对一些大分子拥挤试剂的特性做了总结,包括相对分子质量、流体动力学半径(nm)、电荷性质、有效作用浓度及它们所产生的占有体积等,见表1。 "

2.2.2 大分子拥挤试剂被应用到的细胞或组织类型 见表2。已有很多研究用人体骨分离出来的骨髓间充质干细胞实验,证实了大分子拥挤试剂对细胞外基质的影响[7,31-32]。但是,成纤维细胞因其良好的生活习性及低要求的生长条件而得到了更广泛的应用[33-37],包括人类皮肤成纤维细胞、人肺成纤维细胞、人角膜成纤维细胞和声襞成纤维细胞[38]。此外,人成骨细胞和肌腱细胞及角质形成细胞都曾被应用到实验研究中[6,39]。而且在此之前,还有研究人员用猪的软骨细胞来研究大分子拥挤试剂对细胞外基质的作用,以及软骨细胞的生长和细胞外基质的关系[40-41]。有意思的是,有一篇文章对大分子拥挤试剂对间充质干细胞的作用做了新的阐述,证明它可通过调节细胞微环境来增强人间充质干细胞向脂肪组织的分化[42];另外一篇文章表示,大分子拥挤试剂可通过调节解偶联蛋白的表达和非偶联呼吸作用,增加人类骨髓间充质干细胞向棕色脂肪细胞的分化,并且能使人类骨髓间充质干细胞来源的白色脂肪细胞“棕色化”[43],这将为婴幼儿的产热功能缺失提供一种新的解决思路。 2.3 大分子拥挤试剂增加细胞外基质成分 2.3.1 大分子拥挤试剂增加细胞外基质胶原含量 迄今为止,大分子拥挤试剂已被应用于各种各样的细胞,来促进细胞外基质成分及含量的增加,主要是通过增加细胞外基质中成分的沉积、促进细胞外基质的稳定、重塑及成熟,具体来说,可能是通过以下途径作用的[23]。①增加前胶原C内肽酶增强子1酶促转化,增强前胶原C蛋白酶变构调节剂的活化形式,从而增强前胶原C蛋白酶的酶活性,进而促进胶原前体向胶原的转化,最终有利于超分子(纤连蛋白、胶原蛋白及原纤维蛋白等)的组装;②增强赖氨酰氧化酶和转谷氨酰胺酶2的活性,从而增加胶原的交联,稳定细胞外基质;③通过增加前胶原C内肽酶增强子1酶促转化来增强基质金属蛋白酶抑制剂的活性,然后增强基质金属蛋白酶/TIMP的行为及相互作用,以便减少胶原的降解或增加细胞外基质特定的重塑。总的来讲,大致经过这3个过程来促进细胞外基质的成熟。 在这里概括了这些大分子拥挤试剂及相应的细胞。首先总结了大分子拥挤试剂对细胞外基质中胶原的影响,包括Ⅰ-Ⅵ胶原蛋白。其中,聚蔗糖70与聚蔗糖400混合物应用得最多,它改变最多的是Ⅰ、Ⅳ型胶原蛋白的含量;卡拉胶应用也很广泛,而它对与Ⅰ-Ⅵ胶原蛋白都有所影响,另外,硫酸葡聚糖500增加的也主要是Ⅰ、Ⅳ型胶原蛋白。 2.3.2 大分子拥挤试剂对细胞外基质其他成分的影响 大分子拥挤试剂对间充质干细胞来源细胞外基质中糖胺聚糖的影响:糖胺聚糖在细胞外基质信号通路中扮演着很重要的角色,它调节生长因子和形态发生素的表达。加入大分子拥挤试剂后,随着Ⅰ型胶原的积累,糖胺聚糖的量也大量增加,二者共同创造了一个增厚相对无弹性的细胞外基质。未硫酸化的透明质酸是相对分子质量最大的一类糖胺聚糖,本身就占据着细胞微环境内很大一部分,因此它在含有大分子拥挤试剂的培养液中的分泌量有所减少。然而,其他硫酸化的糖胺聚糖(硫酸软骨素、硫酸皮肤素、硫酸角质素)在大分子拥挤环境中的沉积量却显著增长[32]。最后他们对细胞外基质进行消化并分析了其中硫酸化糖胺聚糖的总含量,发现它们在加入大分子拥挤试剂后显著增加了。 大分子拥挤试剂对生长因子的作用:间充质干细胞来源的细胞外基质释放了各种各样的生长因子和细胞因子,为细胞的生长分化提供营养[22]。之后,又有研究证明大分子拥挤试剂增强的细胞外基质,能提升一些生长因子如成纤维细胞生长因子和血管内皮生长因子的蓄积量,同时又能增加干细胞生长因子的释放量[32],从而证实了大分子拥挤试剂对基质中生长因子的重大调节作用。目前已有研究在探寻增加细胞外基质中生长因子和蛋白酶释放的途径,以便在组织工程中得以应用[44]。 大分子拥挤试剂加速超分子的重新组装:过去几年,人们对超分子的重新组装已进行过研究,他们应用荧光标记Ⅰ型胶原末端来隔离超分子的重新组装过程,结果发现有荧光标记的胶原数量在大分子拥挤环境下的细胞外基质中显著增加,同时也证明了大分子拥挤试剂加速胶原中超分子的重新组装[35,45-46]。此外在体外培养条件下,大分子拥挤试剂还会引起细胞外基质蛋白沉积物的直线排列,这又反过来促进细胞内肌动蛋白细胞骨架的直线排列[7]。而且,外部的震动更加有利于呈直线排列的三维胶原纤维支架的构造[47]。细胞和细胞外基质之间的相互作用的变化也会影响间充质干细胞的黏附、增殖和迁移行为。还有研究报道大分子拥挤试剂加速角膜成纤维细胞基质样组装体的形成,但容易受到表型漂移的影响[35]。 "

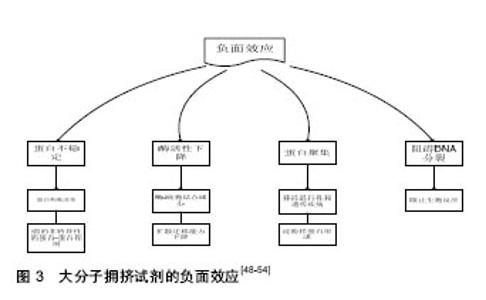
2.4 大分子拥挤试剂的一些负面效应 过去几十年,大量报道都一直在关注大分子拥挤试剂的正面作用,但事实上,它对像蛋白质和酶也有一些负面影响[48-51],例如,大分子拥挤试剂会导致细胞内环境微黏度增加、异常的扩散作用及加速蛋白的聚集[52-54],降低蛋白的弹性、限制其空间构象,妨碍生物学上蛋白质-蛋白质的相互作用,从而使机体发生异常变化,产生一系列的疾病问题。目前,有很多工业和食品生产中也会经常用到一些大分子拥挤试剂,改善食品的口感和形态,来迎合大众的生活需求。这虽然丰富了人们的物质生活,但是过量地使用这些大分子,终究会对人体的健康造成一定的危害。因此,大分子拥挤试剂并不总是有利的,需要人们理性对待。在此,通过查找一些相关文献,总结了它对其他大分子的性质及生物学过程的一些不利影响(图3)。总而言之,在拥挤的生理环境中,不同的大分子对邻近蛋白和细胞都会产生各种不一样的影响,很大可能是通过改变部分占用体积来改变拥挤程度,从而调节这些大分子的性质。因此,作者认为体内的生物大分子可很好地被调节,并且细胞内的拥挤分子也能适当地发挥生物学作用。而这个作用是可被调节的,当然方式也是多种多样的,但是,最终还是归结于我们前面提出的“部分占用体积”与“排斥容积效应”。"
| [1] Vickaryous MK,Hall BK.Human cell type diversity, evolution, development, and classi?cation with special reference to cells derived from the neural crest.Biol Rev Camb Philos Soc.2006;81:425-455. [2] Ellis RJ,Minton AP.Cell biology:join the crowd.Nature.2003;425:27-28. [3] Ebel C,Zaccai G.Crowding in extremophiles: linkage between solvation and weak protein–protein interactions, stability and dynamics, provides insight into molecular adaptation.J Mol Recognit. 2004;17(5):382-389 [4] Ellis RJ.Macromolecular crowding: an important but neglected aspect of the intracellular environment.Curr Opin Struct Biol. 2001;11(1):114-119. [5] Minton AP,Wilf J.Effect of macromolecular crowding upon the structure and function of an enzyme: glyceraldehyde-3-phosphate dehydrogenase.Biochemistry.1981;20(17):4821-4826. [6] Satyam A,Kumar P,Fan X,et al.Macromolecular crowding meets tissue engineering by self-assembly: a paradigm shift in regenerative medicine. Adv Mater.2014;26(19):n/a-n/a. [7] Zeiger AS,Loe FC,Li R,et al.Macromolecular crowding directs extracellular matrix organization and mesenchymal stem cell behavior. PLoS One.2012;7(5):e37904. [8] Saeidi N,Karmelek KN,Paten JA,et al.Molecular Crowding of Collagen: A Pathway to Produce Highly-Organized Collagenous Structures. Biomaterials.2012;33(30):7366. [9] Zhou HX,Rivas G,Minton AP.Macromolecular crowding and confinement: biochemical, biophysical, and potential physiological consequences.Annu Rev Biophys.2008;37:375-397. [10] Yu H,Rathore SS,Shen C,et al.Reconstituting Intracellular Vesicle Fusion Reactions: The Essential Role of Macromolecular Crowding.J Am Chem Soc.2015;137(40):12873-12883. [11] Candotti M,Orozco M.The Differential Response of Proteins to Macromolecular Crowding.PLoS Comput Biol.2016;12(7):e1005040. [12] 舒崇湘,程天民.细胞外基质的结构与功能[J].西南国防医药, 2001,11(2): 142-145. [13] 林琳,蔡光先,刘柏炎.细胞外基质对大鼠骨髓间充质干细胞生物学特性的影响[J].中国现代医学杂志, 2005,15(11):1644-1646. [14] 陈峰.骨组织工程中的细胞外基质材料及其种子细胞[J].中国组织工程研究,2010,14(38):7141-7144. [15] 李珍美玉,顾芸,易晟.细胞外基质在组织工程中的应用[J].交通医学, 2014, 28(5):425-429. [16] Silva EA,Mooney DJ.Synthetic Extracellular Matrices for Tissue Engineering and Regeneration. Curr Top Dev Biol.2004;64:181-205. [17] Lu H,Hoshiba T,Kawazoe N,et al.Cultured cell-derived extracellular matrix scaffolds for tissue engineering.Biomaterials.2011; 32(36): 9658-9666. [18] Sabetkish S,Kajbafzadeh AM,Sabetkish N,et al.Whole-organ tissue engineering: Decellularization and recellularization of three-dimensional matrix liver scaffolds.J Biomed Mater Res A. 2015;103(4):1498-1508. [19] Calle EA,Hill RC,Leiby KL,et al.Targeted Proteomics Effectively Quantifies Differences Between Native Lung and Detergent-Decellularized Lung Extracellular Matrices.Acta Biomaterialia.2016; 46:91-100. [20] Zhang Q,Johnson JA,Dunne LW,et al.Decellularized skin/adipose tissue flap matrix for engineering vascularized composite soft tissue flaps.Acta Biomaterialia.2016;35:166-184. [21] Eitan Y,Sarig U,Dahan N,et al.Acellular cardiac extracellular matrix as a scaffold for tissue engineering: in vitro cell support, remodeling, and biocompatibility.Tissue Eng Part C Methods.2009;16(4):671-683. [22] Rozario T,Desimone DW.The Extracellular Matrix In Development and Morphogenesis: A Dynamic View. Dev Biol.2009;341(1):126-140. [23] Chen C,Loe F,Blocki A,et al.Applying macromolecular crowding to enhance extracellular matrix deposition and its remodeling in vitro for tissue engineering and cell-based therapies.Adv Drug Deliv Rev. 2011;63(4-5):277–290. [24] 李剑,王志珍.细胞内的大分子拥挤环境[J].生物化学与生物物理进展, 2001,28(6):788-792. [25] Minton AP.The influence of macromolecular crowding and macromolecular confinement on biochemical reactions in physiological media.J Biol Chem.2001;276(14):10577. [26] Minton AP.Macromolecular Crowding.Curr Biol.2006;16(16):R269-71. [27] Minton AP.Quantitative assessment of the relative contributions of steric repulsion and chemical interactions to macromolecular crowding. Biopolymers.2013;99(4):239. [28] Mukherjee SK,Gautam S,Biswas S,et al.Do Macromolecular Crowding Agents Exert Only an Excluded Volume Effect? A Protein Solvation Study.J Phys Chem B.2015;119(44):14145-14156. [29] Satyam A,Kumar P,Fan X,et al.Macromolecular crowding meets tissue engineering by self-assembly: a paradigm shift in regenerative medicine. Adv Mater.2014;26(19):n/a-n/a. [30] Rashid R,Lim NS,Chee SM,et al.Novel use for polyvinylpyrrolidone as a macromolecular crowder for enhanced extracellular matrix deposition and cell proliferation.Tissue Eng Part C Methods. 2014; 20(12):994. [31] Cigognini D,Gaspar D,Kumar P,et al.Macromolecular crowding meets oxygen tension in human mesenchymal stem cell culture - A step closer to physiologically relevant in vitro organogenesis.Sci Rep. 2016;6:30746. [32] Prewitz MC,Stißel A,Friedrichs J,et al.Extracellular matrix deposition of bone marrow stroma enhanced by macromolecular crowding. Biomaterials.2015;73:60-69. [33] Satyam A,Kumar P,Cigognini D,et al.Low, but not too low, oxygen tension and macromolecular crowding accelerate extracellular matrix deposition in human dermal fibroblast culture.Acta Biomaterialia. 2016;44:221. [34] Kumar P,Satyam A,Cigognini D,et al.Low oxygen tension and macromolecular crowding accelerate extracellular matrix deposition in human corneal fibroblast culture.J Tissue Eng Regen Med.2016.doi: 10.1002/term.2283.[Epub ahead of print] [35] Kumar P,Satyam A,Fan X,et al.Accelerated Development of Supramolecular Corneal Stromal-Like Assemblies from Corneal Fibroblasts in the Presence of Macromolecular Crowders.Tissue Eng Part C Methods.2015;21(7):660-670. [36] Peng Y,Bocker MT,Holm J,et al.Human fibroblast matrices bio-assembled under macromolecular crowding support stable propagation of human embryonic stem cells.J Tissue Eng Regen Med. 2012;6(10):e74-86. [37] Lareu RR,Subramhanya KH,Peng Y,et al.Collagen matrix deposition is dramatically enhanced in vitro, when crowded with charged macromolecules: The biological relevance of the excluded volume effect. FEBS Lett.2007;581(14):2709-2714. [38] Graupp M,Gruber H,Weiss G,et al.Establishing principles of macromolecular crowding for in vitro fibrosis research of the vocal fold lamina propria.Laryngoscope.2014;125(6):203-209. [39] Benny P,Badowski C,Lane EB,et al.Making More Matrix: Enhancing the Deposition of Dermal–Epidermal Junction Components In Vitro and Accelerating Organotypic Skin Culture Development, Using Macromolecular Crowding.Tissue Eng Part A.2015;21(1-2):183-192. [40] Bo C,Wang B,Wen JZ,et al.Making more matrix: enhancing the deposition of dermal-epidermal junction components in vitro and accelerating organotypic skin culture development, using macromolecular crowding.Tissue Eng Part C Methods. 2013;19(8): 586-595. [41] Behonick DJ,Werb Z.A bit of give and take: the relationship between the extracellular matrix and the developing chondrocyte.Mech Dev. 2003;120(11):1327-1336. [42] Ang XM,Lee MH,Blocki A,et al.Macromolecular Crowding Amplifies Adipogenesis of Human Bone Marrow-Derived Mesenchymal Stem Cells by Enhancing the Pro-Adipogenic Microenvironment.Tissue Eng Part A.2014;20(5-6):966-981. [43] Lee MH,Goralczyk AG,Kriszt R,et al.ECM microenvironment unlocks brown adipogenic potential of adult human bone marrow-derived MSCs.Sci Rep.2016;6:21173. [44] Zhang X,Battiston KG,Labow RS,et al.Generating favorable growth factor and protease release profiles to enable extracellular matrix accumulation within an in vitro tissue engineering environment.Acta Biomaterialia.2017;54:81-94. [45] Kumar P,Satyam A,Fan X,et al.Macromolecularly crowded in vitro microenvironments accelerate the production of extracellular matrix-rich supramolecular assemblies.Sci Rep.2015;5:8729. [46] Minton AP.Implications of macromolecular crowding for protein assembly.Curr Opin Struct Biol. 2000;10(10):34-39. [47] Hsu HT,Rau LR,Zeng YN,et al.External vibration enhances macromolecular crowding for construction of aligned three-dimensional collagen fibril scaffolds.Biofabrication. 2015;7(2):025004. [48] Mittal S,Chowhan RK,Singh LR.Macromolecular crowding: Macromolecules friend or foe.Biochimica Et Biophysica Acta. 2015;1850(9):1822-1831. [49] Hatters DM,Minton AP,Howlett GJ.Macromolecular crowding accelerates amyloid formation by human apolipoprotein C-II.J Biol Chem.2002;277(10):7824-7830. [50] Wu Y,Teng N,Li S.Effects of macromolecular crowding and osmolyte on human Tau fibrillation. Int J Biol Macromol.2016;90:27-36. [51] Luo XD,Kong FL,Dang HB,et al.Macromolecular crowding favors the fibrillization of β2-microglobulin by accelerating the nucleation step and inhibiting fibril disassembly.Biochim Biophys Acta. 2016;1864(11): 1609-1619. [52] Drenckhahn D,Pollard TD.Elongation of actin filaments is a diffusion-limited reaction at the barbed end and is accelerated by inert macromolecules.J Biol Chem.1986;261(27):12754-12758. [53] Golkaram M,Hellander S,Drawert B,et al.Macromolecular Crowding Regulates the Gene Expression Profile by Limiting Diffusion.PLoS Comput Biol.2016;12(11):e1005122. [54] Rashid R,Chee SM,Raghunath M,et al.Macromolecular crowding gives rise to microviscosity, anomalous diffusion and accelerated actin polymerization.Phys Biol.2015;12(3):034001. [55] Puxkandl R,Zizak I,Paris O,et al.Viscoelastic properties of collagen: synchrotron radiation investigations and structural model.Philos Trans R Soc Lond B Biol Sci.2002;357(1418):191-197. [56] 朱梅湘,穆畅道,林炜,等.胶原作为生物医学材料的优势与应用[J].化学世界,2003,44(3):161-164. [57] Abou Neel EA,Bozec L,Knowles JC,et al.Collagen--emerging collagen based therapies hit the patient.Adv Drug Deliv Rev. 2013;65(4): 429-456. [58] Saeidi N,Karmelek KN,Paten JA,et al.Molecular Crowding of Collagen: A Pathway to Produce Highly-Organized Collagenous Structures. Biomaterials.2012;33(30):7366. [59] Dewavrin JY,Hamzavi N,Shim VP,et al.Tuning the architecture of three-dimensional collagen hydrogels by physiological macromolecular crowding.Acta Biomaterialia.2014;10(10):4351. [60] Ferreira AM,Gentile P,Chiono V,et al.Collagen for bone tissue regeneration.Acta Biomaterialia. 2012;8(9):3191-3200. [61] Nguyen BB,Moriarty RA,Kamalitdinov T,et al.Collagen hydrogel scaffold promotes mesenchymal stem cell and endothelial cell coculture for bone tissue engineering.J Biomed Mater Res A. 2017;105(4):1123-1131. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||