Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (6): 827-832.doi: 10.3969/j.issn.2095-4344.0054
Previous Articles Next Articles
Preparation and characterization of printed magnesium scaffolds for bone tissue engineering
- School of Mechanical and Electrical Engineering, Guizhou Normal University, Guiyang 550025, Guizhou Province, China
-
Received:2017-09-30Online:2018-02-28Published:2018-02-28 -
Contact:Wu Quan, Ph.D., Associate professor, Master’s supervisor, School of Mechanical and Electrical Engineering, Guizhou Normal University, Guiyang 550025, Guizhou Province, China -
About author:Li Ying, Studying for master’s degree, School of Mechanical and Electrical Engineering, Guizhou Normal University, Guiyang 550025, Guizhou Province, China -
Supported by:the Science and Technology Foundation of Guizhou Province, No. [2014]2132; Special Science Foundation for Excellent Young Talents of Guizhou Province, No. [2015]05; the Science and Technology Projects of Guizhou Province, No. [2016]7221; Doctoral Foundation of Guizhou Normal University in 2014
CLC Number:
Cite this article
Li Ying, Wu Quan, Tang Geng, Li Hong, Shang Li-yan. Preparation and characterization of printed magnesium scaffolds for bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(6): 827-832.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

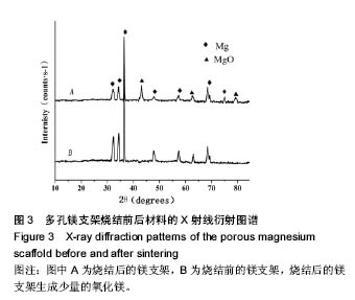
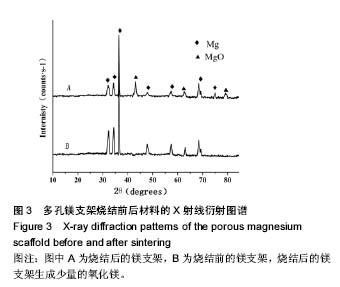
2.1 多孔镁支架的外观结构、微观形貌与孔隙率 支架的多孔结构可为细胞提供附着的场所和营养物质交换的通 道[27]。如图2所示为多孔镁支架的三维结构轮廓,观察到支架由规则圆柱状丝条堆积而成,测得丝径与孔隙均为 (450±50)μm,侧面孔为矩形,长×宽≈500 μm×300 μm,层与层之间存在(50±5)μm的重叠。宏观结构中存在规则均匀、连通性好的孔洞,形成一级孔隙;微观结构中均匀分布微米级的孔洞,形成二级孔隙,该级孔隙主要是由于支架烧结过程中,有机物挥发而镁晶粒未能及时填充造成。通过阿基米德排水法测得支架的孔隙率为(65.0±2.5)%。研究表明当支架孔隙为200-500 μm时,有利于微血管和骨细胞的长入与附着,促进新骨生成与重建,以及加快营养物质代谢[28],故实验制备的双层次孔隙结构的支架满足仿形、物质交换、细胞增殖的要求。"


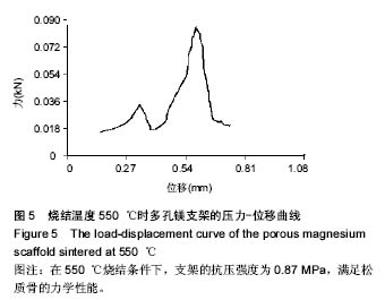
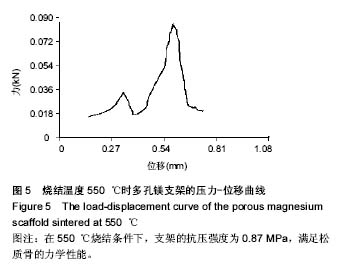
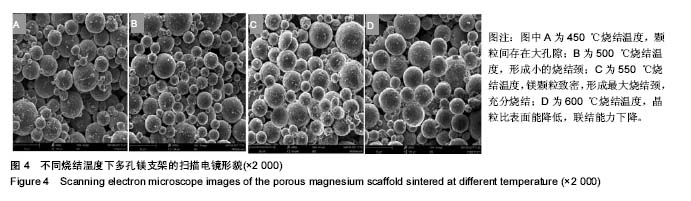
2.3 多孔镁支架的力学性能 在高温烧结过程中,支架的有机物和水分挥发,镁颗粒致密和结晶,使得支架具有一定的力学强度。图4为孔隙率为(65.0±2.5)%的支架在不同烧结温度下的扫描电镜微观形貌,在450,500 ℃较低温度下,镁颗粒粒径小,形成的烧结颈小,周围存在相当大的空隙,烧结进行得不充分(图4A,B);随着烧结温度的升高,在550 ℃条件下,镁颗粒之间结合力呈现出极大值,提高了颗粒间的联结强度,到一定保温时间,形成最大烧结颈,实现充分烧结(图4C);温度继续升高到600 ℃,超过极大值温度后,出现气孔微增的倾向,同时晶粒增大,比表面能降低,不利于颗粒之间的联结,表现为材料的致密程度下降,造成过烧的现象(图4D)。"
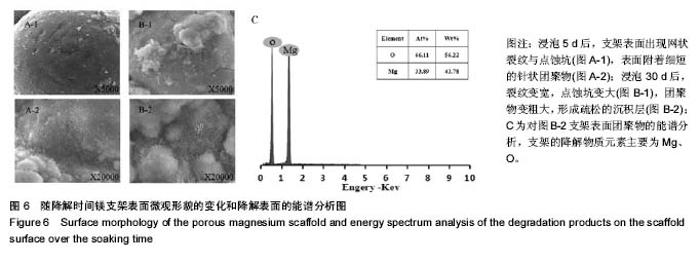
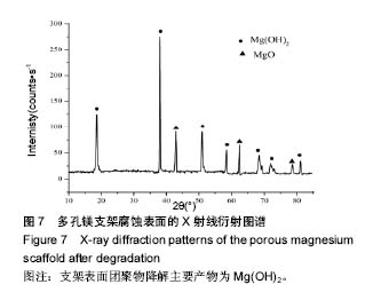
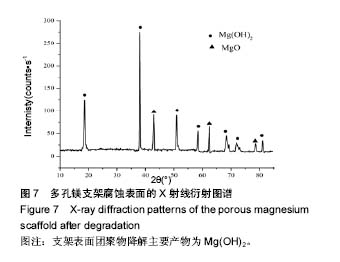
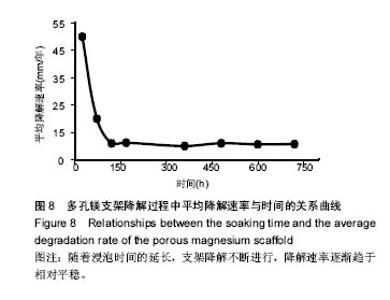
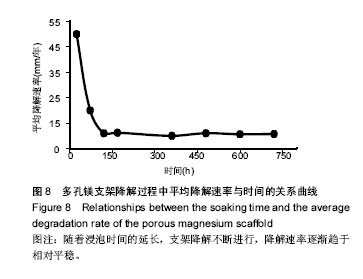
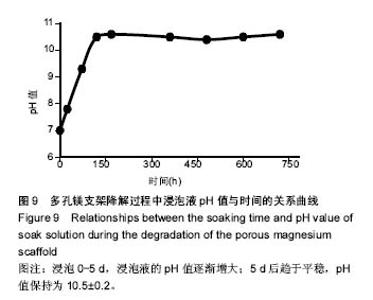
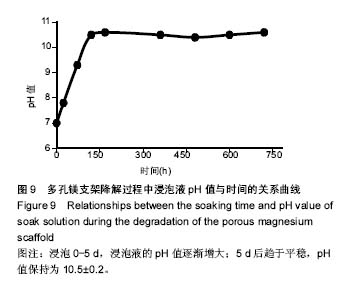
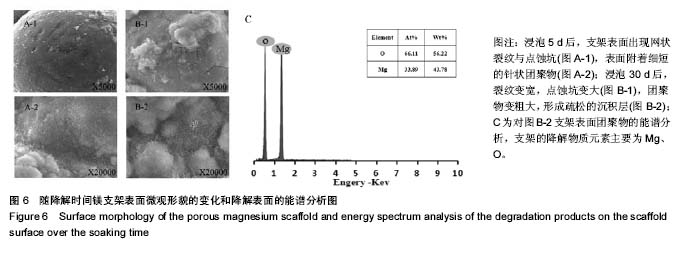
2.4 多孔镁支架的降解性能 图6显示在降解过程中,支架表面微观形貌随时间的变化情况,从第5天开始,支架表面出现网状裂纹与点蚀坑,表面同时附着细短的针状团聚物;到30 d时,裂纹变宽、点蚀坑变大,团聚物变粗大,形成疏松的沉积层,同时采用能谱测试了材料腐蚀表面的元素组成。采用X 射线衍射仪测定腐蚀表面的物相组成,表明该团聚物主要为Mg(OH)2,该疏松沉积层的面积一直随时间延长而增加,见图7。图8为依据式(2)计算得到的支架平均降解速率变化曲线,随着浸泡时间的延长,支架降解不断的进行,降解速率逐渐趋于平稳,平均降解速率为(10.0±0.2)mm/年。图9为支架降解过程中浸泡液pH值随时间的变化曲线,0-5 d,随着浸泡时间的延长,浸泡液的pH值逐渐增大,说明支架降解产生的碱性降解产物主要在这个阶段从支架扩散到溶液中;5 d后趋于平稳,pH值保持在10.5±0.2。"
| [1]Sen MK,Miclau T.Autologous iliac crest bone graft: should it still be the gold standard for treating nonunions?Injury.2007;38 Suppl 1:S75-S80.[2]Sa MW,Kim JY.Design of multi-scaffold fabrication system for various 3D scaffolds.J Mech SciTechnol.2013;27(10):2961-2966. [3]Gaikwad VV,Patil AB,Gaikwad MV.Scaffolds for Drug Delivery in Tissue Engineering.Int J Pharm Sci Nanotechnol.2008;2(1):113-122.[4]Lin Z,Zhao Y,Wong, HM,et al.Enhanced corrosion resistance and cyto-biocompatility of plasma immersion ion implanted (PIII) WE43 magnesium scaffolds for bone tissue engineering.The 4th World Congress of the Tissue Engineering and Regenerative Medicine International Society (TERMIS), Boston, MA.,2015.[5]Lewis G. Reduction in the Corrosion Rate of Magnesium and Magnesium Alloy Specimens and Implications for Plain Fully Bioresorbable Coronary Artery Stents: A Review.World J Eng Technol. 2016;4(4):597-597.[6]张兴凯,韩伟,范德增,等.聚(乳酸一羟基乙酸)共聚物涂层的释药性能[J].功能高分子学报,2014,27(2):219-223.[7]Cox SC,Thornby JA,Gibbons GJ,et al.3D printing of porous hydroxyapatite scaffolds intended for use in bone tissue engineering applications.Mater Sci Eng C.2015;47:237-247.[8]Serre CM,Papillard M,Chavassieux P,et al.Influence of magnesium substitution on a collagen-apatite biomaterial on the production of a calcifying matrix by human osteoblasts.J Biomed Mater Res.1998;42: 626-633. [9]Wiesmnn HP,Tkotz T,Joos U,et al.Magnesium in newly formed dentin mineral of rat incisor.J Bone Miner Res.1997;12:380-383.[10]Staiger MP,Pietak AM,Huadmai J,et al.Magnesiumand its alloys as orthopedic biomaterials: a review.Biomaterials.2006;27:1728-1734. [11]SchliepHake H,Welch HA,Dullin C,et a1.Mandibular bone repair by implantation of rhBMP-2 in a slow release carrier of polylactic acid——an experimental study in rats.Biomaterials.2008;29(1):103-110.[12]Lin L,Wang Z,Zhou L,et al.The influence of prefreezing temperature on pore structure in freeze-dried beta-TCP scaffolds.Proc Inst Mech Eng H.2013,227(1):50-57.[13]Guarino V,Guaccio A,Guarnieri D,et al.Binary system thermodynamics to control pore architecture of PCL scaffold via temperature-driven pHase separation process.J Biomater Appl.2012;27(3):241-254.[14]Witte F,Ulrich H,Palm C,et al.Biodegradable magnesium scaffolds:part 1I:peri-implant bone remodeling.J Biomed Mater Res Part A,2007;81A(3):757765.[15]Zhang X,Li X,Li J,et al.Processing, microstructure and mechanical properties of biomedical magnesium with a specific two-layer structure. Prog Nat Sci MaterInt.2013;23(2):183-189.[16]Tan L,Gong M,Zheng F,et a1.Study on compression behavior of porous magnesium used as bone tissue engineering scaffolds.Biomed Mater. 2009;4(1):1-7.[17]贲玥,张乐,魏帅,等.3D打印陶瓷材料研究进展[J].材料导报, 2016,30(21): 109-118.[18]Krawczak P. Additive manufacturing of plastic and polymer composite parts: Promises and challenges of 3D-printing.Exp Polym Lett.2015; 9(11):959-959.[19]利普森.3D打印:从想象到现实[M].北京:中信出版社,2013.[20]Ahlfeld T,Akkineni AR,Förster Y,et al.Design and Fabrication of Complex Scaffolds for Bone Defect Healing: Combined 3D Plotting of a Calcium Phosphate Cement and a Growth Factor-Loaded Hydrogel. Ann Biomed Eng. 2017;45(1):1-13.[21]Farzadi A,Waran V,Solati-Hashjin M,et al.Effect of layer printing delay on mechanical properties and dimensional accuracy of 3D printed porous prototypes in bone tissue engineering.Ceram Int.2015; 41(7): 8320-8330.[22]Asadi-Eydivand M,Solati-Hashjin M,Farzad A,et al.Effect of technical parameters on porous structure and strength of 3D printed calcium sulfate prototypes.Robot Com-Int Manuf. 2016;37(C):57-67.[23]Zhang J,Zhao S,Zhu Y,et al.Three-dimensional printing of strontium-containing mesoporous bioactive glass scaffolds for bone regeneration.Acta Biomaterialia.2014;10(5):2269-2281.[24]Yang Y,Yang S,Wang Y,et al.Anti-infective efficacy, cytocompatibility and biocompatibility of a 3D-printed osteoconductive composite scaffold functionalized with quaternized chitosan.Acta Biomaterialia. 2016;46:112.[25]Standard Test Method for Density of Powder Metallurgy (PM) Materials Containing Less Than Two Percent Porosity.Astm B311-2013.[26]Pu SY.Embedded Metal Materials and their Corrosion.Beijing:Press of Beijing University of Aeronauties and Astronautics,l990:287.[27]Tsuruga E,Takita H,Itoh H,et al.Pore size of porous hydroxyapatite as the cell-substratum controls BMP-induced osteogenesis.J Biochem. 1997;121(2):317-324. [28]Clemow AJT,Weinstein AM,Klawitter JJ,et al.Interface mechanics of porous titanium implants. J Biomed Mater Res Part A. 1981;15(1): 73-82.[29]Gibson LJ.The mechanical behaviour of cancellous bone.J Biomech. 1985;18(5):317-328.[30]Levrero-Florencio F,Margetts L,Sales E,et al.Evaluating the macroscopic yield behaviour of trabecular bone using a nonlinear homogenisation approach.J Mech Behav Biomed Mater.2016;61:384. [31]Babiker H,Ding M,Overgaard S.Demineralized bone matrix and human cancellous bone enhance fixation of porous-coated titanium implants in sheep.J Tissue Eng Regene Med.2016;10(3):245.[32]Almeida HA,Bártolo PJ.Design of tissue engineering scaffolds based on hyperbolic surfaces: structural numerical evaluation.Med Eng Phys. 2014;36(8):1033.[33]Xiao D,Yang Y,Su X,et al.An integrated approach of topology optimized design and selective laser melting process for titanium implants materials.Biomed Mater Eng.2013;23(5):433-445.[34]Makar GL,Kruger J.Corrosion of Magnesium.Int Mater Rev. 1993; 38(3):138-153.[35]Kotoka R,Yamoah NK,Mensah-Darkwa K,et al. Electrochemical corrosion behavior of silver doped tricalcium pHospHate coatings on magnesium for biomedical application.Surf Coat Technol. 2016;292: 99-109.[36]Tekumalla S,Seetharaman S,Nguyen QB,et al.Influence of Cerium on the Deformation and Corrosion of Magnesium.J Eng Mater Technol. 2016;138(3):031011 .[37]Li X,Wang L,Fan Y,et al.Nanostructured scaffolds for bone tissue engineering.J Biomed Mater Res Part A.2013;101(8):2424. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||