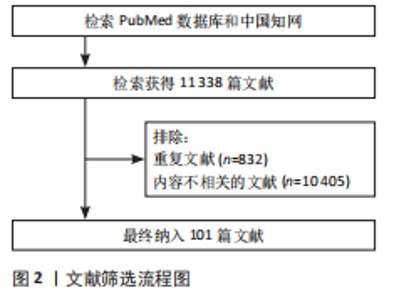
Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7370-7377.doi: 10.12307/2026.765
Previous Articles Next Articles
Signaling pathways associated with dopaminergic neuronal axonal degeneration in Parkinson’s disease
Wang Jiao, Song Wenjun, Xin Rui, Liu Wei, Ying Zhenhao
- Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China
-
Received:2025-07-22Revised:2025-10-17Online:2026-10-08Published:2026-02-24 -
Contact:Ying Zhenhao, Associate professor, Master’s supervisor, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
About author:Wang Jiao, MS candidate, Rehabilitation therapist, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
Supported by:the National Natural Science Foundation of China, No. 82174367 (to LW); Scientific Research Fund Project of Shandong University of Traditional Chinese Medicine, No. KYZK2024M08 (to YZH)
CLC Number:
Cite this article
Wang Jiao, Song Wenjun, Xin Rui, Liu Wei, Ying Zhenhao. Signaling pathways associated with dopaminergic neuronal axonal degeneration in Parkinson’s disease[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7370-7377.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

的研究并未涉及帕金森病病理机制的探索。1912年,LEWY[9]发现了路易小体,标志着帕金森病病理学研究的重大突破,为后续病理研究奠定了基础。接着,TRETIAKOFF[10]通过研究证实了黑质多巴胺能神经元的丧失,进一步确立了黑质作为帕金森病关键病变部位的地位。在后续研究中,MAROTEAUX及其团队[13]首先命名了α-突触核蛋白基因,并发现该蛋白定位于突触前末端,这一发现为理解帕金森病的分子机制提供了新的视角。BRAAK等[15]提出了帕金森病的分期理论,提出病理改变从嗅球/延髓向中脑发展,并首次提示轴突的退变可能早于神经元胞体的退变,这一理论的提出,引发了对帕金森病病理进展方式的新思考。随着研究的深入,多个独立研究团队通过尸检发现轴突的球状体病变早于神经元胞体病变,进一步提供了直接证据,显示轴突损伤可能先于神经元死亡。随之而来的是对轴突损伤病理机制的进一步探索,成为当前帕金森病研究的重要方向。基于以上研究历程,文章进一步对帕金森病中轴突相关的信号通路进行了全面综述,揭示了帕金森病病理学研究中的重要进展,并为未来的研究提供了理论支持。 2.2 帕金森病的病理机制 帕金森病是一种复杂的疾病,目前病因尚不明确,主要受遗传、环境以及老龄化因素影响。以往研究主要围绕神经元胞体内α-突触核蛋白聚集、氧化应激增加和细胞凋亡等因素展开。然而近年来的研究发现,帕金森病患者的神经元轴突常常先于胞体出现病理改变,并且这种变化在疾病的早期阶段便有所显现,这一发现使得针对轴突退行性变的帕金森病研究变得尤为关键。通过影像证据和尸检报告分析发现,帕金森病轴突退行事件大部分经历了蛋白质聚集、线粒体功能障碍、氧化应激、神经炎症等过程。 2.2.1 帕金森病早期中脑多巴胺能神经元轴突变性的影像学证据 在帕金森病初期症状的研究中,使用弥散张量成像和扩散加权磁共振成像技术发现了白质中广泛的轴突损伤[22]。影像学显示,帕金森病患者在T2加权或液体抑制反转恢复序列上出现显著的白质高信号,并且白质损伤的程度与帕金森病症状的严重性密切相关,白质损伤越严重,患者在运动、认知、执行和语言功能方面的损害也越显著[23]。这一变化与低温X射线纳米断层扫描结果一致,低温X射线纳米断层扫描结果揭示了帕金森病患者黑质轴突的肿胀和髓鞘缺陷[24],这种髓鞘脱失可能与α-突触核蛋白积聚引起"

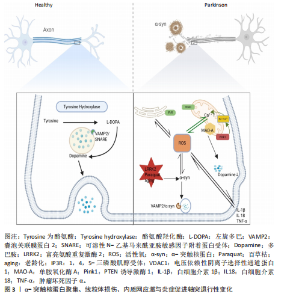
的鞘磷脂聚集有关,进而影响神经冲动传递、突触前可塑性和神经递质受体定位[24-26]。近年来,基于新型多室弥散加权成像数据分析技术,揭示了帕金森病黑质-纹状体区域的轴突分支减少和突触缩短现象;此外,神经元密度指数和取向分散指数值均表现出明显增加。尤其是白质纤维束中,神经元密度指数和取向分散指数的升高更为显著,这些结果提示,轴突运输障碍可能是导致帕金森白质微观结构变化的关键因素之一[27-28]。此外,借助单光子发射计算机断层扫描和多巴胺转运蛋白配体成像技术,在帕金森病的前驱期便能观察到纹状体轴突终末的多巴胺能纤维显著丧失[4]。这些发现进一步支持了黑质-纹状体多巴胺神经元轴突终末的丧失早于胞体丧失的假设。 2.2.2 帕金森病早期中脑多巴胺能神经元轴突变性的尸检结果 由上述影像证据可知,帕金森病中的轴突退变先于胞体退变,提示了有关帕金森病神经退行性变化的关键靶位,并强调了轴突在帕金森病早期神经生物学变化中的潜在预警作用。尸检结果进一步显示,轴突末梢的α-突触核蛋白含量显著高于神经元胞体[4,29-30],通过石蜡包埋组织印迹技术证实α-突触核蛋白主要聚集于嗅球、肠道的肠神经系统和位于延髓的迷走神经背侧运动核的突触前终端。这些发现提示,帕金森病早期病理变化可能始于α-突触核蛋白在轴突远端的聚集。DU等[28]的尸检结果及其他相关研究表明,突触前末梢的神经传导功能受帕金森病理影响最为严重,随后诱发向心性退变,继而影响轴突的连续性,最后波及远端和近端的轴突数量。遗传因素(如富亮氨酸重复激酶2突 变[31])、环境因素(如服用鱼藤酮和百草枯等[32])和衰老均是导致α-突触核蛋白增多的主要因素[33]。α-突触核蛋白积聚于突触前终末,其“朊蛋白样”传播依赖神经元之间的连接,尤其是轴突运输系统。研究表明,α-突触核蛋白与囊泡关联膜蛋白2结合抑制可溶性N-乙基马来酰亚胺敏感因子附着蛋白受体复合物的形成,导致囊泡关联膜蛋白2和可溶性N-乙基马来酰亚胺敏感因子附着蛋白受体复合物的功能异常,从而阻碍了神经递质释放,增加了轴突内的氧化应激,上调了轴突内活性氧水平,减少了ATP合成 [22,29,31-36],见图3。这些机制表明,α-突触核蛋白在轴突中的早期沉积可能是神经元变性的启动因子;此外,α-突触核蛋白的传播与轴突损伤同时发生。小鼠模型实验表明,注射预成纤维的α-突触核蛋白可诱导其沿轴突传播,导致轴突损伤和神经行为障碍,进一步证实轴突是朊样传播的媒介,同时也是这一过程的受害者[37]。 尸检结果显示,帕金森病患者的线粒体与多巴胺能神经元突触前终末之间的距离增加,同时线粒体体积明显减少[38];此外,患者多巴胺能神经元轴突内的线粒体往往集中在α-突触核蛋白聚集体周围;免疫组织化学染色进一步揭示线粒体外层存在单胺氧化酶A。单胺氧化酶A通过促进突触前神经元轴突中多巴胺的氧化分解导致活性氧的生成增加,进而引发氧化应激反应并激活炎症反应小胶质细胞,见图3。因此,单胺氧化酶A在这一过程中可能发挥关键作用,它在帕金森病患者中的异常表达和功能改变使其成为帕金森病诊断的潜在生物标志物[39]。此外,Pink1基因突变使Na+/Ca2+交换体损伤,导致线粒体内钙离子过载[40],这会消耗大量ATP以将Ca2+泵出线粒体,直到ATP耗竭,最终损害囊泡关联膜蛋白2功能,使得本应储存于囊泡中的神经递质暴露于胞浆,从而导致胞浆中多巴胺浓度升高,见图3。Ca2+过载也会通过钙蛋白酶信号传导途径增强p35的裂解产物p25与细胞周期蛋白依赖性激酶5的结合,形成p25细胞周期蛋白依赖性激酶5复合物[41],这进一步导致突触前末梢的神经传导功能受损[42]。随着突触前末梢神经传导功能的损伤,轴突的连续性也遭到破坏。Pink1基因突变引起线粒体去极化,导致突触前末梢神经传导障碍,并导致烟酰胺单核苷酸腺苷酸转移酶2的耗竭,进而破坏轴突的糖酵解功能和健康[43-44]。 这一过程导致烟酰胺单核苷酸积累和烟酰胺腺苷酸耗竭,最终引发沃勒变性,并诱发死Toll白介素受体基序蛋白1依赖性的轴突断裂[34]。随着轴突断裂的进行,轴突内的信号传导逐渐中断,影响神经功能和相关的生理活动,最终导致轴突数量减少[45]。 在Braak1期和2期尸检组织中,黑质致密部中的神经酮水平升高,这表明特定脂质的氧化损伤可能在帕金森病的早期阶段就已发生[40,46]。 大量活性氧所引起的异常蛋白聚集可触发内质网应激,进而促使Ca2+直接释放到线粒体膜[47],这有赖于1,4,5-三磷酸肌醇受体与电压依赖性阴离子选择性通道蛋白1的相互作用。而PTEN 诱导激酶1和Parkin的缺失会导致1,4,5-三磷酸肌醇受体活性失调,内质网的钙释放显著增加,进一步加剧活性氧的生成,加重线粒体损伤[48-49],见图3。此外,通过尸体核磁共振病理技术检测发现,轴突损伤后髓鞘轴突的轴质中神经丝释放量增加,并且随着神经丝含量增多,轴突损伤程度也会更加严重。因此,神经丝可能成为帕金森病的潜在诊断和预后生物标志物[50]。 轴突损伤后,小胶质细胞会迅速反应并参与局部炎症反应[51]。炎症反应在帕金森病的早期阶段发挥着关键作用。研究表明,C-反应蛋白(CRP)、脂肪酸结合蛋白、白细胞介素6、白细胞介素10、白细胞介素17A、肿瘤坏死因子α以及肿瘤坏死因子受体含量越高,患者的运动和认知功能损伤越严重,尤其是白细胞介素6和C-反应蛋白的水平升高常常预示着患者的运动功能受损更为严重[52]。这可能是由于α-突触核蛋白的异常聚集和氧化应激通过与Toll样受体的相互作用,激活小胶质细胞炎性小体中的NOD样受体蛋白3(NLRP3),并引发小胶质细胞和星形胶质细胞的激活,促进其分泌包括肿瘤坏死因子α和白细胞介素6在内的炎症因子,见图3。这些细胞因子进一步诱导抑制因子κB蛋白的磷酸化与降解,进而释放核因子κB[53],核因子κB进入细胞核后启动相关基因的转录,增加促炎症因子的表达,形成炎症级联反应,这种增强的神经炎症反应在加剧α-突触核蛋白蓄积的同时也进一步激化了脂质和线粒体的氧化应激,进而加剧了轴突损伤[54-55]。 2.2.3 帕金森病早期中脑多巴胺能神经元轴突变性的细胞分子水平证据 除了α-突触核蛋白聚集、线粒体损伤、氧化应激和炎症反应等直接导致轴突损伤的因素外,从细胞和分子生物学的角度而言,轴突损伤后修复能力的受损也会加剧轴突的进一步损伤。 (1)基础防御受损:神经系统的修复首先依赖于基础防御机制,这些机制能够在损伤发生时迅速响应并为后续的修复过程提供支持。 对抗氧化应激的能力受损:在帕金森病"

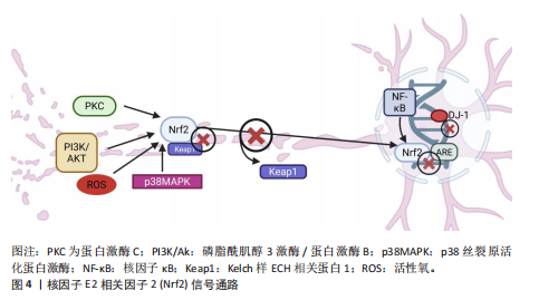
患者中,抗氧化应激的能力显著下降,导致氧化应激加剧并引发轴突变性,这一现象可能与核因子E2相关因子2 的核质穿梭失调密切相关。正常情况下,当氧化应激发生时,核因子E2相关因子2通路通过蛋白激酶C、磷脂酰肌醇3激酶/蛋白激酶B、p38丝裂原活化蛋白激酶和核因子κB等信号通路激活,从轴突中与细胞Kelch样ECH相关蛋白1解离,转入细胞核内并结合抗氧化反应元件,启动抗氧化酶的转录[56-57]。然而,在帕金森病患者的多巴胺能神经元轴突中,核因子E2相关因子2活性通常减弱或无法充分激活,导致抗氧化应激防御能力降低,见图4。DJ-1是核因子E2相关因子2下游通路中关键元件,能够通过清除轴突内的活性氧来保护线粒体免受氧化损伤,同时稳定核因子E2相关因子2,增强抗氧化防御;此外,DJ-1还能够抑制核因子κB途径,从而减轻炎症反应[58-59]。 DJ-1的缺乏对轴突的影响呈双向作用:轻度缺乏时,可能通过促进热休克蛋白水平的上升,抑制α-突触核蛋白的聚集;严重缺乏则会引发氧化应激加剧,线粒体形态改变,进而导致活性氧增多,并引发轴突损伤[60-61]。 研究证明,适量运动能够显著改善帕金森病患"

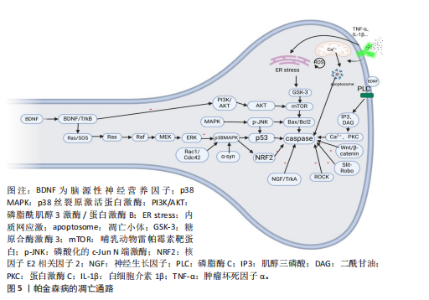
者的氧化应激状态,促进核因子E2相关因子2进入细胞核、增强DJ-1清除活性氧的能力、降低轴突内活性氧的含量,从而减轻轴突损伤[62]。 神经营养因子信号通路受损:神经营养因子如脑源性神经营养因子、神经生长因子[63]、神经营养素3和胶质细胞衍生神经营养因子[64],通过激活有丝分裂原活化蛋白激酶和磷脂酰肌醇3激酶/蛋白激酶B通路促进轴突生长、成熟和突触发育。研究表明,α-突触核蛋白过表达会下调脑源性神经营养因子的转录并损害它在轴突中的运输,导致脑源性神经营养因子表达减少并伴随活性氧增加,进而通过促凋亡蛋白Bax诱导轴突变性[65]。运动训练(如跑台运动)能够增加脑源性神经营养因子的表达,增强海马锥体神经元突触可塑性,有效减缓轴突损伤[66]。α-突触核蛋白还能够通过抑制RET转录因子核受体相关蛋白1的转录继而降低RET表达,最终加速轴突的丢失[67]。 (2)结构/功能修复:轴突损伤后的神经修复过程可按照一定的顺序进行:首先,细胞骨架的重建为神经元提供形态支持;接着,轴突再生通过信号通路促进神经纤维的恢复;最后,突触引导分子确保神经功能的精确恢复,这样的顺序保证了神经系统从结构基础到功能连接的逐步恢复。 细胞骨架重建功能下降:细胞骨架(如微管和微丝)的动态调控对轴突的生长和形态维持至关重要。许多研究表明,小GTP酶Rac1和细胞分裂控制蛋白42促进Rho GTP酶细胞骨架重组功能,而Ras同源家族成员A则起抑制作用,导致轴突和树突回缩以及棘和突触的丢失,因此,Ras同源家族成员A与小GTP酶Rac1/细胞分裂控制蛋白42之间表现出的拮抗关系能有效调节轴突的形态[68-69],而一旦这种关系失衡则有可能引发轴突损伤。小GTP酶Rac1的活化能促进α-突触核蛋白的自噬清除,从而缓解了帕金森病患者的神经病变,并提高了多巴胺能神经元的存活率。而Ras同源家族成员A对细胞骨架动力学的影响主要是通过其下游效应器Rho相关卷曲螺旋蛋白激酶的激活介导的,Rho相关卷曲螺旋蛋白激酶通过磷酸化作用抑制神经轴突的生长,使神经突收缩、棘和突触丢失并诱导神经元凋亡[70],因此,抑制Ras同源家族成员A信号传导可能是早期疾病干预的一种有前途的策略[71-72]。 轴突再生障碍:Wnt/β-连环蛋白、音猬因子和Notch等信号通路在轴突损伤后的再生过程中发挥重要作用。Wnt/β-连环蛋白信号通路激活会增加靶基因的转录,并与脑源性神经营养因子相互作用共同促进细胞的增殖、分化和轴突的生长。此外,音猬因子通路与Wnt/β-连环蛋白信号通路协同作用,激活下游靶基因,保护轴突免受氧化应激的影响,进一步促进轴突再生[73];相反,当Wnt/β-连环蛋白表达下降时,轴突的发育和再生受到影响,最终引发神经变性[74-75]。在帕金森病患者中,α-突触核蛋白的积累常伴随Notch1表达的下降,导致Notch信号在细胞迁移、形态变化、突触可塑性、神经元成熟和长时程增强等过程中的调节功能丧失,最终可能引发轴突损伤[76-77]。一项有氧运动动物实验证明,运动能显著提高Wnt/β-连环蛋白和Notch信号通路的表达、明显改善认知功能,提示运动可能作为一种潜在的治疗策略[78]。 轴突引导分子信号受损:Slit-Robo、肝配蛋白-促红细胞生成素肝细胞激酶、轴突导向因子-结直肠癌缺失蛋白/UNC5、信号素-Plexin/神经纤毛蛋白、整合素等信号通路能够促进生长锥的有效迁移[79-84]。Slit或Robo缺乏使Slit-Robo的引导作用减弱,轴突失去正常的生长导向并发生断裂。轴突损伤后,Robo4与肝配蛋白A1结合抑制了血管内皮生长因子诱导的血管生成,进一步加剧轴突的损伤[85]。随着轴突内α-突触核蛋白的积累,轴突导向因子1的表达逐渐减少,这一变化激活了细胞骨架重建通路,有助于修复受损的轴突;然而,若轴突导向因子1持续低表达且UNC5B缺乏其配体,将破坏生长锥的稳定性[86]。研究表明,Slit-Robo1排斥作用与结直肠癌缺失蛋白受体相互作用能够抑制轴突导向因子1的功能,这可能进一步加剧轴突修复的困难;使用静态牵伸技术能够显著降低Robo1和Robo2的表达,从而促进轴突的导向生长,这为轴突再生提供了新的潜在策略[87]。这一发现提示,合理设计和实施运动训练干预能够有效加速轴突的修复过程,促进神经功能的恢复。 (3)蛋白质的清除受损:轴突病变可通过多重机制加剧α-突触核蛋白的异常积聚及其病理传播。研究表明,轴突损伤会破坏α-突触核蛋白的正常降解途径,如泛素-蛋白酶体系统和自噬-溶酶体途径[88-90],其通过激活丝裂原活化蛋白激酶信号通路促进酪氨酸羟化酶的异常磷酸化和泛素化,进而加速α-突触核蛋白的异常聚集[88]。当蛋白酶体功能受损时,α-突触核蛋白的清除主要依赖于大自噬和伴侣介导自噬两种途径[90]。值得注意的是,自噬过程的调控涉及复杂的信号网络:一方面,神经营养因子(如脑源性神经营养因子)可通过激活磷脂酰肌醇3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号轴促进自噬发生[68];另一方面,内质网应激(由Ca2+超载、活性氧累积等因素诱发)可通过干扰素调节因子1自磷酸化激活c-Jun氨基末端激酶/p38丝裂原活化蛋白激酶通路,进而启动自噬程序。然而在病理条件下,持续的氧化应激和内质网应激会导致轴突自噬功能进行性衰退,最终引发不可逆的轴突损伤。当前的证据表明,免疫治疗可通过减缓轴突中的α-突触核蛋白传播来缓解病变:抗α-突触核蛋白单抗能够显著减少其在轴突中的扩散,减缓轴突退化和行为异常,这表明轴突传播是朊样机制中的关键环节,并且具有可干预性[91]。 2.3 帕金森病的病理结果 轴突在直接损伤后并不总是立即发生凋亡,而是通过多种修复通路延缓这一过程。例如,脑源性神经营养因子通过抑制c-Jun氨基末端激酶/p38丝裂原活化蛋白激酶通路减缓凋亡,神经生长因子通过抑制caspase活性增强抗凋亡效应,Ras同源家族成员A通过调控Rho激酶通路抑制轴突凋亡,Slit-Robo信号通路通过调节自噬减缓凋亡,从而促进轴突的生长与存活。然而,轴突修复机制的效力有限,随着氧化应激持续和脑源性神经营养因子的减少,p53被激活,启动caspase途径[92],因此,p53有望成为治疗帕金森病的潜在靶点。在此过程中,多种细胞因子或生长因子通过与质膜受体酪氨酸激酶结合触发受体二聚化,导致帕金森病患者的黑质致密部中蛋白激酶B及其磷酸化形式减少,同时糖原合成酶激酶3广泛表达;糖原合成酶激酶3激活后,通过上调轴突中的caspase-3含量进一步促进轴突凋亡[93]。此外,哺乳动物雷帕霉素靶蛋白是磷脂酰肌醇3激酶/蛋白激酶B信号转导的关键下游靶点,也是轴突生长和代谢的重要调控因子,哺乳动物雷帕霉素靶蛋白活性降低导致蛋白激酶B及其磷酸化形式减少,从而加剧神经退行性变性[94],见图5。已有研究证明,抗阻运动能够通过增强哺乳动物雷帕霉素靶蛋"
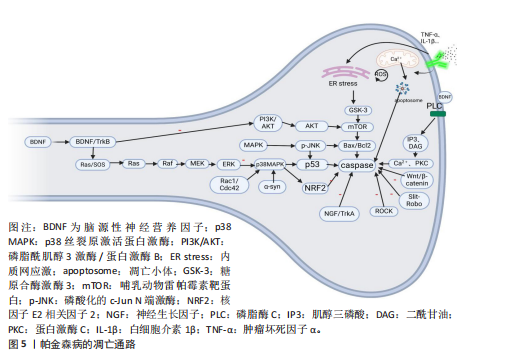
| [1] HEGARTY SV, GREEN H F, NICLIS J, et al. Editorial: The Role of Stem Cells, Epigenetics and MicroRNAs in Parkinson’s Disease. Front Neurosci. 2020; 14:515. [2] GOULDING S, SULLIVAN A, O’KEEFFE G, et al. The potential of bone morphogenetic protein 2 as a neurotrophic factor for Parkinson’s disease. Neural Regen Res. 2020;15(8):1432. [3] COSTA HN, ESTEVES AR, EMPADINHAS N, et al. Parkinson’s Disease: A Multisystem Disorder. Neurosci Bull. 2023;39(1):113-124. [4] GCWENSA NZ, RUSSELL DL, COWELL RM, et al. Molecular Mechanisms Underlying Synaptic and Axon Degeneration in Parkinson’s Disease. Front Cell Neurosci. 2021;15:626128. [5] YE H, ROBAK LA, YU M, et al. Genetics and Pathogenesis of Parkinson’s Syndrome. Annu Rev Pathol Mech Dis. 2023;18(1):95-121. [6] PARKINSON J. An Essay on the Shaking Palsy. London: Whittingham and Rowland, 1817. [7] CHARCOT JM. Leçons sur les maladies du système nerveux: tremblement parkinsonien et autres formes de la maladie de Parkinson. Paris: Bureaux du Progrès Médical, 1887. [8] BLOCQ C, MARINESCU G. Sur un cas de tremblement parkinsonien hémiplégique symptomatique d’une tumeur du pédoncle cérébral. C R Soc Biol. 1893;5:105-111. [9] LEWY F. Paralysis agitans//LEWANDOWSKY M. Handbuch der Neurologie. Berlin: Julius Springer, 1912:920-933. [10] TRETIAKOFF C. Contribution à l’étude de l’anatomie pathologique du locus niger de Soemmering avec quelques déductions relatives à la pathogénie des troubles du tonus musculaire et de la maladie de Parkinson. Paris: Université de Paris, 1919. [11] DUFFY PE, TENNYSON VM. Phase and electron microscopic observations of Lewy bodies and melanin granules in the substantia nigra and locus caeruleus in Parkinson’s disease. J Neuropathol Exp Neurol. 1966;25(3):398-414. [12] LANGSTON JW, BALLARD P, TETRUD JW, et al. Chronic parkinsonism in humans due to a product of meperidine-analog synthesis. Science. 1983;219(4587):979-980. [13] MAROTEAUX L, CAMPANELLI JT, SCHELLER RH. Synuclein: A neuron-specific protein localized to the nucleus and presynaptic nerve terminal. J Neurosci. 1988;8(8):2804-2815. [14] POLYMEROPOULOS MH, LAVEDAN C, LEROY E, et al. Mutation in the α-synuclein gene identified in families with parkinson’s disease. Science. 1997;276(5321):2045-2047. [15] BRAAK H, TREDICI KD, RÜB U, et al. Staging of brain pathology related to sporadic parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211. [16] PAISÁN-RUÍZC, JAIN S, EVANS EW, et al. Cloning of the gene containing mutations that cause PARK8-linked parkinson’s disease. Neuron. 2004; 44(4):595-600. [17] ZIMPRICH A, BISKUP S, LEITNER P, et al. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron. 2004;44(4): 601-607. [18] LI JY, ENGLUND E, HOLTON JL, et al. Lewy bodies in grafted neurons in subjects with Parkinson’s disease suggest host-to-graft disease propagation. Nat Med. 2008;14(5):501-503. [19] LUK KC, KEHM V, CARROLL J, et al. Pathological α-Synuclein transmission initiates Parkinson-like neurodegeneration in non-transgenic mice. Science. 2012; 338(6109):949-953. [20] HSIEH CH, SHALTOUKI A, GONZALEZ AE, et al. Functional impairment in Miro degradation and mitophagy is a shared feature in familial and sporadic Parkinson’s disease. Cell Stem Cell. 2016; 19(6):709-724. [21] MOLLENHAUER B, DAKNA M, KRUSE N, et al. Validation of serum neurofilament light chain as a biomarker of Parkinson’s disease progression. Mov Disord. 2020;35(11):1999-2008. [22] SHIH YC, TSENG WYI, MONTASER-KOUHSARI L. Recent advances in using diffusion tensor imaging to study white matter alterations in Parkinson’s disease: A mini review. Front Aging Neurosci. 2023;14:1018017. [23] JIANG YQ, CHEN QZ, YANG Y, et al. White matter lesions contribute to motor and non-motor disorders in parkinson’s disease: a critical review. Geroscience. 2024;47(1):591-609. [24] FATHY YY, JONKMAN LE, BOL JJ, et al. Axonal degeneration in the anterior insular cortex is associated with Alzheimer’s co-pathology in Parkinson’s disease and dementia with Lewy bodies. Transl Neurodegener. 2022;11(1):52. [25] SIGNORELLI P, CONTE C, ALBI E. The Multiple Roles of Sphingomyelin in Parkinson’s Disease. Biomolecules. 2021;11(9):1311. [26] FU Y, PICKFORD R, GALPER J, et al. A protective role of ABCA5 in response to elevated sphingomyelin levels in Parkinson’s disease. Npj Park Dis. 2024;10(1):20. [27] WEI X, WANG S, ZHANG M, et al. Gait impairment-related axonal degeneration in parkinson’s disease by neurite orientation dispersion and density imaging. Npj Park Dis. 2024;10(1):45. [28] DU R, WANG X, HE S. BDNF improves axon transportation and rescues visual function in a rodent model of acute elevation of intraocular pressure. Sci China Life Sci. 2020;63(9):1337-1346. [29] FRIGERIO I, BOUWMAN MMA, NOORDERMEER RTGMM, et al. Regional differences in synaptic degeneration are linked to alpha-synuclein burden and axonal damage in Parkinson’s disease and dementia with Lewy bodies. Acta Neuropathol Commun. 2024;12(1):4. [30] BELLUCCI A, BUBACCO L, LONGHENA F, et al. Nuclear Factor-κB Dysregulation and α-Synuclein Pathology: Critical Interplay in the Pathogenesis of Parkinson’s Disease. Front Aging Neurosci. 2020;12:68. [31] BHATIA P, BICKLE M, AGRAWAL AA, et al. Axonal lysosomal assays for characterizing the effects of LRRK2 G2019S. Biology. 2024;13(1):58. [32] YAMAMOTO Y, TANIGUCHI T, SHIMAZAKI A. Rotenone-induced optic nerve damage and retinal ganglion cell loss in rats. Biomolecules. 2024;14(9):1047. [33] KALYANARAMAN B, CHENG G, HARDY M. Gut microbiome, short-chain fatty acids, alpha-synuclein, neuroinflammation, and ROS/RNS: Relevance to Parkinson’s disease and therapeutic implications. Redox Biol. 2024;71:103092. [34] WHITE MA, LIN Z, KIM E, et al. Sarm1 deletion suppresses TDP-43-linked motor neuron degeneration and cortical spine loss. Acta Neuropathol Commun. 2019;7(1):166. [35] LAUTRUP S, SINCLAIR DA, MATTSON MP, et al. NAD+ in Brain Aging and Neurodegenerative Disorders. Cell Metab. 2019;30(4):630-655. [36] SHARMA M, BURRÉ J. α-Synuclein in synaptic function and dysfunction. Trends Neurosci. 2023; 46(2):153-166. [37] VOLPICELLI-DALEY LA, LUK KC, PATEL TP, et al. Exogenous α-synuclein fibrils induce lewy body pathology leading to synaptic dysfunction and neuron death. Neuron. 2011;72(1):57-71. [38] MALLACH A, WEINERT M, ARTHUR J, et al. Post mortem examination of Parkinson’s disease brains suggests decline in mitochondrial biomass, reversed by deep brain stimulation of subthalamic nucleus. FASEB J. 2019;33(6):6957-6961. [39] MUKHERJEE J, LADWA RM, LIANG C, et al. Elevated Monoamine Oxidase-A in Anterior Cingulate of Post-Mortem Human Parkinson’s Disease: A Potential Surrogate Biomarker for Lewy Bodies? Cells. 2022;11(24):4000. [40] BARAZZUOL L, GIAMOGANTE F, BRINI M, et al. PINK1/Parkin Mediated Mitophagy, Ca2+ Signalling, and ER–Mitochondria Contacts in Parkinson’s Disease. Int J Mol Sci. 2020;21(5):1772. [41] ALROUJI M, AL‐KURAISHY HM, AL‐GAREEB AI, et al. Cyclin‐dependent kinase 5 (CDK5) inhibitors in Parkinson disease. J Cell Mol Med. 2024;28(11):e18412. [42] TAN YY, JENNER P, CHEN SD. Monoamine Oxidase-B Inhibitors for the Treatment of Parkinson’s Disease: Past, Present, and Future. J Park Dis. 2022;12(2):477-493. [43] YANG S, NIOU ZX, ENRIQUEZ A, et al. NMNAT2 supports vesicular glycolysis via NAD homeostasis to fuel fast axonal transport. Mol Neurodegener. 2024;19(1):13. [44] VAN DER VLAG M, HAVEKES R, HECKMAN PRA. The contribution of parkin, PINK1 and DJ‐1 genes to selective neuronal degeneration in parkinson’s disease. Eur J Neurosci. 2020;52(4):3256-3268. [45] HINZ FI, VILLEGAS CLM, ROBERTS JT, et al. Context-Specific Stress Causes Compartmentalized SARM1 Activation and Local Degeneration in Cortical Neurons. J Neurosci. 2024;44(24):e2424232024. [46] KOEGLSPERGER T, RUMPF SL, SCHLIEßER P, et al. Neuropathology of incidental Lewy body & prodromal Parkinson’s disease. Mol Neurodegener. 2023;18(1):32. [47] BORSCHE M, PEREIRA SL, KLEIN C, et al. Mitochondria and Parkinson’s Disease: Clinical, Molecular, and Translational Aspects. J Park Dis. 2021;11(1):45-60. [48] HAM SJ, YOO H, WOO D, et al. PINK1 and Parkin regulate IP3R-mediated ER calcium release. Nat Commun. 2023;14(1):5202. [49] GHEMRAWI R, KHAIR M. Endoplasmic Reticulum Stress and Unfolded Protein Response in Neurodegenerative Diseases. Int J Mol Sci. 2020; 21(17):6127. [50] FRIGERIO I, LAANSMA MA, LIN CP, et al. Neurofilament light chain is increased in the parahippocampal cortex and associates with pathological hallmarks in parkinson’s disease dementia. Transl Neurodegener. 2023;12(1):3. [51] MCNAMARA NB, MUNRO DAD, BESTARD-CUCHE N, et al. Microglia regulate central nervous system myelin growth and integrity. Nature. 2023; 613(7942):120-129. [52] ZIMMERMANN M, BROCKMANN K. Blood and cerebrospinal fluid biomarkers of inflammation in parkinson’s disease. J Park Dis. 2022;12(s1):S183-S200. [53] SONG Z, LI W, HAN Y, et al. Association of immune cell traits with Parkinson’s disease: a Mendelian randomization study. Front Aging Neurosci. 2024;16:1340110. [54] MOHAMMED NN, TADROS MG, GEORGE MY. Empagliflozin repurposing in Parkinson’s disease; modulation of oxidative stress, neuroinflammation, AMPK/SIRT-1/PGC-1α, and wnt/β-catenin pathways. Inflammopharmacology. 2024;32(1):777-794. [55] ALROUJI M, AL‐KURAISHY HM, AL‐GAREEB AI, et al. NF‐κB/NLRP3 inflammasome axis and risk of Parkinson’s disease in Type 2 diabetes mellitus: A narrative review and new perspective. J Cell Mol Med. 2023;27(13):1775-1789. [56] MUSACCHIO T, YIN J, KREMER F, et al. Temporal, spatial and molecular pattern of dopaminergic neurodegeneration in the AAV-A53T α-synuclein rat model of Parkinson’s disease. Behav Brain Res. 2022;432:113968. [57] HUANG TI, HSIEH CL. Effects of Acupuncture on Oxidative Stress Amelioration via Nrf2/ARE-Related Pathways in Alzheimer and Parkinson Diseases. Evid Based Complement Alternat Med. 2021;2021:1-11. [58] LIND-HOLM MOGENSEN F, SCAFIDI A, POLI A, et al. PARK7/DJ-1 in microglia: implications in Parkinson’s disease and relevance as a therapeutic target. J Neuroinflammation. 2023;20(1):95. [59] IMBERECHTS D, KINNART I, WAUTERS F, et al. DJ-1 is an essential downstream mediator in PINK1/parkin-dependent mitophagy. Brain. 2022;145(12):4368-4384. [60] VENDREDY L, ADRIAENSSENS E, TIMMERMAN V. Small heat shock proteins in neurodegenerative diseases. Cell Stress Chaperones. 2020;25(4):679-699. [61] ALMELA P, CUENCA‐BERMEJO L, YUSTE JE, et al. Cardiac Noradrenaline Turnover and Heat Shock Protein 27 Phosphorylation in Dyskinetic Monkeys. Mov Disord. 2020; 35(4):698-703. [62] MAGAÑA JC, DEUS CM, GINÉ-GARRIGA M, et al. Exercise-Boosted Mitochondrial Remodeling in Parkinson’s Disease. Biomedicines. 2022; 10(12):3228. [63] TRIACA V, FICO E, SPOSATO V, et al. hNGF Peptides Elicit the NGF-TrkA Signalling Pathway in Cholinergic Neurons and Retain Full Neurotrophic Activity in the DRG Assay. Biomolecules. 2020; 10(2):216. [64] CONWAY J, KRAMER E. Is activation of GDNF/RET signaling the answer for successful treatment of Parkinson’s disease? A discussion of data from the culture dish to the clinic. Neural Regen Res. 2022;17(7):1462. [65] DE LEÓN A, GIBON J, BARKER PA. NGF-Dependent and BDNF-Dependent DRG Sensory Neurons Deploy Distinct Degenerative Signaling Mechanisms. Eneuro. 2021;8(1):ENEURO.0277-20.2020. [66] TANG C, LIU M, ZHOU Z, et al. Treadmill Exercise Alleviates Cognition Disorder by Activating the FNDC5: Dual Role of Integrin αV/β5 in Parkinson’s Disease. Int J Mol Sci. 2023;24(9):7830. [67] CONWAY JA, INCE S, BLACK S, et al. GDNF/RET signaling in dopamine neurons in vivo. Cell Tissue Res. 2020;382(1):135-146. [68] SCHMIDT SI, BLAABJERG M, FREUDE K, et al. RhoA Signaling in Neurodegenerative Diseases. Cells. 2022;11(9):1520. [69] IYER M, SUBRAMANIAM MD, VENKATESAN D, et al. Role of RhoA-ROCK signaling in Parkinson’s disease. Eur J Pharmacol. 2021;894:173815. [70] GUILER W, KOEHLER A, BOYKIN C, et al. Pharmacological Modulators of Small GTPases of Rho Family in Neurodegenerative Diseases. Front Cell Neurosci. 2021;15:661612. [71] ZANG CX, WANG L, YANG HY, et al. HACE1 negatively regulates neuroinflammation through ubiquitylating and degrading Rac1 in Parkinson’s disease models. Acta Pharmacol Sin. 2022;43(2): 285-294. [72] YING L, ZHAO J, YE Y, et al. Regulation of Cdc42 signaling by the dopamine D2 receptor in a mouse model of Parkinson’s disease. Aging Cell. 2022;21(5):e13588. [73] YANG C, QI Y, SUN Z. The Role of Sonic Hedgehog Pathway in the Development of the Central Nervous System and Aging-Related Neurodegenerative Diseases. Front Mol Biosci. 2021;8:711710. [74] MARCHETTI B, TIROLO C, L’EPISCOPO F, et al. Parkinson’s disease, aging and adult neurogenesis: Wnt/β‐catenin signalling as the key to unlock the mystery of endogenous brain repair. Aging Cell. 2020; 19(3):e13101. [75] RAMAKRISHNA K, NALLA LV, NARESH D, et al. WNT-β Catenin Signaling as a Potential Therapeutic Target for Neurodegenerative Diseases: Current Status and Future Perspective. Diseases. 2023;11(3):89. [76] KIM S, LEE M, CHOI YK. The Role of a Neurovascular Signaling Pathway Involving Hypoxia-Inducible Factor and Notch in the Function of the Central Nervous System. Biomol Ther. 2020;28(1):45-57. [77] KATOLIKOVA NV, KHUDIAKOV AA, SHAFRANSKAYA DD, et al. Modulation of Notch Signaling at Early Stages of Differentiation of Human Induced Pluripotent Stem Cells to Dopaminergic Neurons. Int J Mol Sci. 2023;24(2):1429. [78] METHI A, ISLAM MR, KAURANI L, et al. A single-cell transcriptomic analysis of the mouse hippocampus after voluntary exercise. Mol Neurobiol. 2024;61(8):5628-5645. [79] TOMIYAMA H. A Commentary on Axon guidance pathway genes and Parkinson’s disease. J Hum Genet. 2011;56(2):102-103. [80] BASTIOLI G, ARNOLD JC, MANCINI M, et al. Voluntary Exercise Boosts Striatal Dopamine Release: Evidence for the Necessary and Sufficient Role of BDNF. J Neurosci. 2022;42(23):4725-4736. [81] GONDA Y, NAMBA T, HANASHIMA C. Beyond Axon Guidance: Roles of Slit-Robo Signaling in Neocortical Formation. Front Cell Dev Biol. 2020;8:607415. [82] AHN EH, KANG SS, QI Q, et al. Netrin1 deficiency activates MST1 via UNC5B receptor, promoting dopaminergic apoptosis in Parkinson’s disease. Proc Natl Acad Sci. 2020;117(39):24503-24513. [83] HARREGUY MB, TANVIR Z, SHAH E, et al. Semaphorin signaling restricts neuronal regeneration in C. elegans. Front Cell Dev Biol. 2022;10:814160. [84] DAVIS-LUNN M, GOULT BT, ANDREWS MR. Clutching at Guidance Cues: The Integrin–FAK Axis Steers Axon Outgrowth. Biology. 2023;12(7):954. [85] HECKING I, STEGEMANN LN, THEIS V, et al. Neuroprotective Effects of VEGF in the Enteric Nervous System. Int J Mol Sci. 2022;23(12):6756. [86] LO P, RYMAR VV, KENNEDY TE, et al. The netrin‐1 receptor DCC promotes the survival of a subpopulation of midbrain dopaminergic neurons: Relevance for ageing and Parkinson’s disease. J Neurochem, 2022;161(3):254-265. [87] 肖宇辰,安子通,阿如汗,等.静态性牵张诱导神经突起导向生长的Slit/Robo分子机制的研究[J].医用生物力学,2024,39(S01):91-92. [88] FRANIĆ D, ZUBČIĆ K, BOBAN M. Nuclear ubiquitin-proteasome pathways in proteostasis maintenance. Biomolecules. 2021;11(1):54. [89] SHIN WH, PARK JH, CHUNG KC. The central regulator p62 between ubiquitin proteasome system and autophagy and its role in the mitophagy and parkinson’s disease. BMB Rep. 2020;53(1):56-63. [90] KAWAHATA I, FUKUNAGA K. Degradation of tyrosine hydroxylase by the ubiquitin-proteasome system in the pathogenesis of parkinson’s disease and dopa-responsive dystonia. Int J Mol Sci. 2020; 21(11):3779. [91] SPENCER B, VALERA E, ROCKENSTEIN E, et al. Anti-α-synuclein immunotherapy reduces α-synuclein propagation in the axon and degeneration in a combined viral vector and transgenic model of synucleinopathy. Acta Neuropathol Commun. 2017;5(1):7. [92] KANG SS, WU Z, LIU X, et al. Treating parkinson’s disease via activation of BDNF/TrkB signaling pathways and inhibition of delta-secretase. Neurotherapeutics. 2022;19(4):1283-1297. [93] LONG HZ, CHENG Y, ZHOU ZW, et al. PI3K/AKT Signal Pathway: A Target of Natural Products in the Prevention and Treatment of Alzheimer’s Disease and Parkinson’s Disease. Front Pharmacol. 2021;12:648636. [94] LUO Q, SUN W, WANG YF, et al. Association of p53 with Neurodegeneration in Parkinson’s Disease. Park Dis. 2022;2022:1-11. [95] LEVITT D, LUK HY, VINGREN J. Alcohol, resistance exercise, and mTOR pathway signaling: an evidence-based narrative review. Biomolecules. 2022;13(1):2. [96] CHEN Y, DENG J, WANG P, et al. PINK1 and Parkin are genetic modifiers for FUS-induced neurodegeneration. Hum Mol Genet. 2016;25(23): 5059-5068. [97] GOULDING SR, LÉVESQUE M, SULLIVAN AM, et al. Quinacrine and Niclosamide Promote Neurite Growth in Midbrain Dopaminergic Neurons Through the Canonical BMP-Smad Pathway and Protect Against Neurotoxin and α-Synuclein-Induced Neurodegeneration. Mol Neurobiol. 2021;58(7): 3405-3416. [98] KAAGMAN DGM, VAN WEGEN EEH, CIGNETTI N, et al. Effects and mechanisms of exercise on brain-derived neurotrophic factor (BDNF) levels and clinical outcomes in people with parkinson’s disease: a systematic review and meta-analysis. Brain Sci. 2024;14(3):194. [99] GHOWEBA RE, KHOWAILED AA, ABOULHODA BE, et al. Synergistic role of resveratrol and exercise training in management of diabetic neuropathy and myopathy via SIRT1/NGF/GAP43 linkage. Tissue Cell. 2023;81:102014. [100] KEIKHAEI R, ABDI E, DARVISHI M, et al. Combined treatment of high‐intensity interval training with neural stem cell generation on contusive model of spinal cord injury in rats. Brain Behav. 2023;13(7):e3043. [101] KILLACKEY SA, BI Y, PHILPOTT DJ, et al. Mitochondria-ER cooperation: NLRX1 detects tochondrial protein import stress and promotes mitophagy through the ER protein RRBP1. Autophagy. 2023;19(5):1601-1603. |
| [1] | Zhang Nan, Meng Qinghua, Bao Chunyu. Characteristics and clinical application of ankle joint finite element models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2343-2349. |
| [2] | Jiang Xianglong, Li Zhongshan, Che Tongtong. Application effects and mechanisms of low-frequency pulsed electromagnetic fields in muscle repair and growth [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2350-2360. |
| [3] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [4] | Tan Jing, Li Li, Wang Liangliang, Qin Xiangyu. Bionic functional coating improves the integration of titanium implants and skin tissue interface [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2014-2022. |
| [5] | Wang Songpeng, Liu Yusan, Yu Huanying, Gao Xiaoli, Xu Yingjiang, Zhang Xiaoming, Liu Min. Bidirectional regulation of reactive oxygen species based on zeolitic imidazolate framework-8 nanomaterials: from tumor therapy and antibacterial activity to cytoprotection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2033-2013. |
| [6] | Chen Haojie, Wang Dai, Shen Shan. Immune inflammatory microenvironment mechanisms in peri-implantitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2054-2062. |
| [7] | Liu Dawei, Cui Yingying, Wang Fanghui, Wang Zixuan, Chen Yuhan, Li Yourui, Zhang Ronghe. Epigallocatechin gallate-mediated bidirectional regulation of reactive oxygen species and its application in nanomaterials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2101-2112. |
| [8] | Fu Lyupeng, Yu Peng, Liang Guoyan, Chang Yunbing. Electroactive materials applied in spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2113-2123. |
| [9] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [10] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [11] | Han Nianrong, Huang Yifei, Akram · Osman, Liu Yanlu, Hu Wei . Programmed cell death receptor-1 suppresses osteogenic differentiation of rat bone marrow mesenchymal stem cells in a high-glucose microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1649-1657. |
| [12] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [13] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [14] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [15] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||