Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7364-7369.doi: 10.12307/2026.755
Previous Articles Next Articles
Mechanisms and potential therapeutic strategies for skeletal muscle extracellular matrix aging
Zhou Jing1, 2, 3, 4, 5, Su Dongming6, Yang Dan1, 2, 3, 4, 5
- Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China
-
Received:2025-07-22Revised:2025-10-17Online:2026-10-08Published:2026-02-12 -
Contact:Ying Zhenhao, Associate professor, Master’s supervisor, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
About author:Wang Jiao, MS candidate, Rehabilitation therapist, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
Supported by:the National Natural Science Foundation of China, No. 82174367 (to LW); Scientific Research Fund Project of Shandong University of Traditional Chinese Medicine, No. KYZK2024M08 (to YZH)
CLC Number:
Cite this article
Zhou Jing, Su Dongming, Yang Dan. Mechanisms and potential therapeutic strategies for skeletal muscle extracellular matrix aging[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7364-7369.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
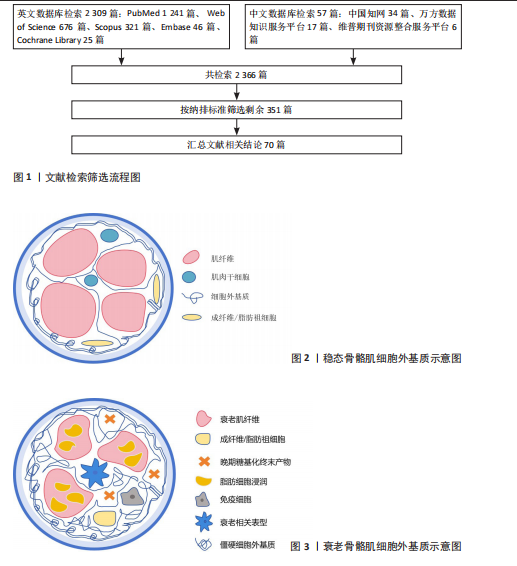
2.1 稳态骨骼肌细胞外基质的结构与动态功能 2.1.1 细胞外基质的结构组分与生理功能 细胞外基质为骨骼肌提供物理支撑、介导力学传导并构成信号通讯微环境的三维网络[8]。在组织层面,细胞外基质以结缔组织膜的形式分层包裹肌肉:最外层的肌外膜包裹整块肌肉,向内延伸形成肌束膜以分隔肌束,最内层的肌内膜则精细包裹每根肌纤维,并与肌纤维表面的基底膜无缝连接[9]。 细胞外基质的核心分子组分包括[10-12]:①结构蛋白:以Ⅰ型和Ⅲ型胶原为主,构成肌束膜与肌内膜的力学骨架,赋予组织抗张强度。而Ⅳ型胶原和层粘连蛋白则共同组建基底膜,此结构不仅是力学生物学界面,更是维持肌肉干细胞静息的关键生态位。②黏附蛋白:以纤连蛋白为代表,作为连接细胞与细胞外基质的“分子锚点”,通过整合素等受体介导细胞黏附。③蛋白聚糖:其糖胺聚糖侧链能结合大量水分子和生长因子。这不仅维持了组织的渗透压和水合状态,更使其成为一个潜在的“信号储存库”,在稳态下以低浓度螯合生长因子,防止其异常激活细胞。见图2。 细胞外基质的稳态并非静止,而是由合成与降解构成的动态平衡。骨骼肌中常驻的成纤维细胞与成纤维/脂肪祖细胞是细胞外基质分子的主要合成单元[13]。细胞外基质的降解则依赖于基质金属蛋白酶家族的精确调控,其活性又受到内源性抑制剂——基质金属蛋白酶组织抑制剂的严格控制[14]。基质金属蛋白酶与其组织抑制剂之间的平衡确保了细胞外基质在响应生理需求时能进行适应性重塑。 2.1.2 细胞外基质在骨骼肌损伤再生中的动态调控作用 当肌肉损伤时,细胞外基质从被动支架转变为主动调控再生的信号中枢,通过动态变化的生化与力学信号,精确编排细胞行为。 (1)启动并引导肌肉干细胞的再生程序[15]:损伤破坏基底膜,释放储存的生长因子,共同“唤醒”肌肉干细胞。成纤维/脂肪祖细胞快速分泌的纤连蛋白临时基质,成为活化的肌肉干细胞增殖和迁移的“轨道”。随后,新重建的层粘连蛋白基底膜则引导肌肉干细胞进行定向分化与融合。 (2)双向调节成纤维/脂肪祖细胞的行为[16]:在急性损伤初期,细胞外基质降解产物与炎症因子共同激活成纤维/脂肪祖细胞,使它们分泌胶原形成临时瘢痕以提供结构支撑。然而,若细胞外基质环境因故变得过度僵硬(即纤维化),这种持续的力学信号会反过来锁定成纤维/脂肪祖细胞的成纤维表型,阻碍功能性组织的再生。 (3)募集并调控免疫细胞的功能极化[17-19]:细胞外基质降解产生的片段,即“基质因子”,可直接募集并调控免疫细胞。例如,胶原降解产生的脯氨酸-甘氨酸-脯氨酸肽段负责募集并激活中性粒细胞;弹性蛋白源性肽则趋化单核细胞;纤连蛋白的特定片段能与Toll样受体4结合,激活先天免疫。此后,细胞外基质的动态重塑进一步引导巨噬细胞的功能极化:再生早期,临时基质有助于维持促炎(M1)巨噬细胞的吞噬功能;进入再生后期,新合成的细胞外基质组分,如透明质酸和腱生蛋白C,则促进巨噬细胞向抗炎与促修复(M2)表型转化。因此,细胞外基质通过自身的时序性重塑,确保了免疫反应从促炎清除到促修复重建的有序衔接。 2.2 衰老驱动的细胞外基质病理性重塑:从功能性基质到纤维化屏障 随着年龄增长,细胞外基质原有的精密稳态被系统性破坏,经历一系列相互关联且自我放大的病理重塑,从支持再生的动态结构转变为抑制再生的纤维化屏障。这一恶性转变的过程可以从3个层层递进的维度来理解:首先是核心分子的“成分失衡”;其次是由此引发的“物理硬化”;最终共同导致全面的“功能衰败”。见图3。 2.2.1 第一层:成分失衡——纤维化基质的形成与分子驱动力 衰老细胞外基质最显著的特"
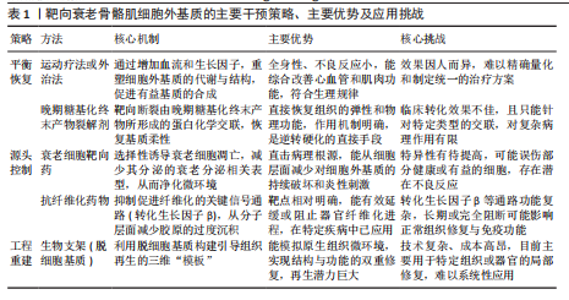
征是其组分的系统性失衡,核心表现为纤维化[20-22]。具体而言,构成力学骨架的Ⅰ型和Ⅲ型胶原被过度沉积,而对维持肌肉干细胞功能至关重要的Ⅳ型胶原和层粘连蛋白则相对减少,直接破坏了关键的干细胞生态位。此失衡是多重上游病理因素共同作用的结果: (1)衰老细胞的累积及其病理性分泌[23-25]:由于持续的DNA损伤、氧化应激以及免疫监视效率下降,衰老细胞在组织中累积。它们通过其衰老相关分泌表型,持续释放促炎细胞因子、生长因子。其中,转化生长因子β作为关键组分,是目前公认的核心促纤维化驱动因子,它直接刺激成纤维/脂肪祖细胞分化并大量合成胶原。 (2)慢性低度炎症的放大作用[26-27]:衰老分泌相关表型本身是慢性炎症的主要来源,并能招募免疫细胞,进一步放大炎症反应。这种持续的炎症环境,通过肿瘤坏死因子α等细胞因子,协同转化生长因子β增强纤维化信号。 (3)重塑酶系统的功能紊乱[28-30]:上述病理信号(如转化生长因子β)的持续刺激下,基质降解体系发生紊乱,表现为基质金属蛋白酶活性相对不足,而其组织抑制剂表达占优势。这打破了基质合成与降解的动态平衡,最终导致纤维化基质的净积累。 2.2.2 第二层:物理硬化——力学环境恶化与僵化恶性循环 由成分失衡(尤其是Ⅰ/Ⅲ型胶原过度沉积)所引发的直接后果,便是细胞外基质物理特性的剧变——基质僵硬度的显著增加[31-33]。此外,化学交联的持续累积也进一步加剧了基质的僵化,这包括由晚期糖基化终末产物介导的非酶促交联[34-35],以及由转谷氨酰胺酶2等催化的酶促交联[36]。年轻肌肉细胞外基质的生理性柔韧度(5-15 kPa)为细胞提供了最佳力学环境,而衰老肌肉的基质僵硬度可增加数倍[37]。 这种从“柔性”到“刚性”的物理转变,会通过力学转导对细胞行为产生深远的负面影响:①抑制成肌分化[38]:僵硬的基质通过激活Yes 相关蛋白/PDZ 结合基序转录共激活因子等力学敏感通路,强力抑制肌肉干细胞的成肌分化潜能;②锁定促纤维化表型,形成恶性循 环[39]:僵硬的基质亦可作为持续的物理刺激,反过来激活成纤维/脂肪祖细胞分泌更多胶原,形成“基质僵硬→细胞激活→胶原更多→基质更僵硬”的恶性正反馈循环。 2.2.3 最终结果:功能衰败——信号整合紊乱与再生潜能丧失 上述的“成分失衡”与“物理硬化”共同作用,最终导致细胞外基质作为信号整合平台的核心功能全面崩溃,即“功能衰败”。衰老的细胞外基质转变为一个充满干扰性与非生理性信号的紊乱环境,其再生支持功能基本丧失。 (1)生化信号储存与释放功能失调[40]:由于蛋白聚糖等组分的改变(成分失衡),细胞外基质储存和释放转化生长因子β等关键调控因子的能力失常,扰乱了细胞行为所需的精确时序信号。 (2)生物活性片段(基质因子)信号谱异常[41]:损伤后,纤维化的细胞外基质降解模式异常,无法产生正确的生物活性片段来引导免疫反应,导致免疫细胞调控能力下降,延滞了炎症消退与组织修复。 (3)细胞-基质通讯全面失真[42]:僵硬的物理特性(物理硬化)和异常的分子构成(成分失衡),使得基质通过整合素等受体,向细胞传递持续的、错误的力学与生化信号。这种信号串扰不仅抑制再生,还可能反过来加剧细胞衰老和凋亡,最终导致衰老肌肉在响应损伤时修复能力严重受损。 2.3 靶向衰老细胞外基质的潜在干预策略:从修复到重建 针对衰老骨骼肌细胞外基质的多维度病理性重塑,已涌现出多种旨在恢复其功能的干预策略。这些策略在逻辑上可分为3个层次:①直接纠正细胞外基质的成分与物理失衡;②靶向驱动细胞外基质恶化的上游细胞与信号源头;③通过组织工程学手段重建功能性微环境。 2.3.1 纠正失衡:恢复细胞外基质的动态平衡与物理特性 此层次策略旨在直接干预并逆转已形成的细胞外基质病理性改变,核心是恢复其动态平衡与正常的物理特性。 主动生理干预是此策略的基石。运动作为一种生理性刺激,其效果已得到广泛证实。规律的抗阻训练不仅能通过机械应力激活黏附斑激酶-丝裂原活化蛋白激酶/细胞外调节蛋白激酶通路,上调基质金属蛋白酶(如基质金属蛋白酶2、基质金属蛋白酶9)的表达与活性,从而逆转基质金属蛋白酶/质金属蛋白酶组织抑制剂失衡、降解过度沉积的胶原蛋白;还能通过激活过氧化物酶体增殖物激活受体γ共激活因子1α来抑制核心促纤维化信号转化生长因子β/Smad通路[43-44]。同时,运动被证实能促进Ⅳ型胶原、整合素α7β1等关键基底膜组分的表达,直接修复干细胞生态位,维持其功能[45]。 除主动运动外,一系列被动式的物理外治疗法也展现出潜力。这些疗法通过非侵入性方式施加能量,可作为运动干预的有力补充。例如,神经肌肉电刺激通过被动诱导肌肉收缩模拟机械应力,促进适应性基质重塑[46];光生物调节疗法利用特定波长光线减轻氧化应激,并可能通过下调转化生长因子β表达来抑制纤维化[47];体外冲击波疗法则利用机械声波松解纤维化的基质结构[48]。这些现代物理疗法为无法进行高强度主动训练的老年个体提供了替代方案。值得一提的是,传统中医外治法也为此提供了独特的实践视角。推拿通过手法直接施加机械应力,从生物力学角度调节成纤维细胞活性[49-50];针刺通过刺激穴位调节神经-内分泌-免疫网络,可能通过抑制慢性炎症反应(如衰老分泌相关表型)和改善局部微循环来减轻纤维化[51]。 与上述侧重于物理特性和基质重塑的策略不同,另一类策略则直接靶向导致细胞外基质化学僵硬化的晚期糖基化终末产物。这包括2种途径:一是通过晚期糖基化终末产物形成抑制剂(如氨基胍、吡哆胺)进行预防;二是通过晚期糖基化终末产物交联断裂剂(如阿拉格铂)进行修复[52-53]。 然而,此层次的各类干预策略均面临瓶颈。无论是主动运动还是被动物理疗法,都面临着如何将宏观效应转化为精准“细胞外基质处方”的挑战,缺乏对强度、频率和类型的标准化指导[54]。传统中医外治法的分子机制亦有待现代科学的深入阐明。而靶向晚期糖基化终末产物的药物开发,其代表性药物阿拉格铂在Ⅲ期临床试验中未能达到主要终点,揭示了从动物模型到人体的转化困境以及衰老基质化学交联的复杂性[55]。未来的突破可能依赖于更精准的靶点或结合多种机制的“鸡尾酒疗法”[56],以实现对衰老细胞外基质真正有效的重塑。 2.3.2 靶向上游驱动因素:清除衰老细胞与抑制纤维化信号 此策略旨在从根源上消除驱动细胞外基质病理性重塑的细胞与分子源头。 (1)衰老细胞清除疗法[57-58]:通过清除在组织中累积的衰老细胞,根除衰老相关分泌表型这一关键“信号污染源”。目前研究最广泛的衰老细胞清除药物包括达沙替尼(一种酪氨酸激酶抑制剂)与槲皮素(一种天然黄酮类化合物)的组合、非瑟酮以及靶向Bcl-2家族抗凋亡蛋白的Navitoclax。一项关键研究证实,在老年小鼠中给予达沙替尼与槲皮素的组合能够有效清除衰老细胞,改善其运动耐力、握力等多项生理功能,其机制被认为与减轻衰老分泌相关表型相关的慢性炎症和组织功能障碍直接相关。 (2)抗纤维化疗法[59-60]:直接靶向核心的转化生长因子β/Smad信号轴是该领域的焦点。尽管尚无专门针对骨骼肌纤维化的上市药物,但已在特发性肺纤维化治疗中取得成功的吡非尼酮和尼达尼布为该方向提供了重要参考。具体而言,吡非尼酮通过下调转化生长因子β和肿瘤坏死因子α等多种促纤维化因子发挥作用;而尼达尼布则是一种多靶点酪氨酸激酶抑制剂,能同时阻断血小板衍生生长因子受体、成纤维细胞生长因子受体和血管内皮生长因子受体等与成纤维细胞活化增殖密切相关的信号通路。 这两大策略面临的共同核心挑战是靶点特异性。非特异性地清除所有衰老细胞或系统性抑制转化生长因子β信号,存在“误伤”正常生理功能(如伤口愈合)的风险[61]。因此,未来的方向是开发能够精准靶向特定组织中有害细胞群的药物递送系统,例如利用抗体-药物偶联物或特异性配体修饰的纳米颗粒[62]。 2.3.3 基于组织工程学的终极策略:重建功能性细胞外基质微环境 当前述内源性修复策略效果不佳时,终极策略是利用组织工程学原理,直接植入生物活性支架,为细胞再生提供一个“年轻化”的功能性微环境[63]。例如,源于年轻健康组织的脱细胞基质支架,因完整保留了天然细胞外基质的三维拓扑结构和多种生物活性分子(如潜在的基质因子、被螯合的生长因子),被证明可作为理想的“信号模板”,有效引导宿主干细胞的归巢、增殖与成肌分化[64-65]。 已有研究表明,将肌肉来源的脱细胞基质应用于体积性肌肉缺损模型中,能够显著促进功能性肌肉组织的再生[66]。 尽管前景广阔,但该策略在应用于肌肉衰减症这类全身性、弥散性的慢性疾病时面临巨大挑战。现有技术主要针对局部、急性的组织缺损,未来的探索可能在于开发可注射的、能原位自组装的微型生物材料。例如,基于透明质酸、胶原蛋白或聚乙二醇的可注射水凝胶[67-68],它们不仅可以模拟年轻细胞外基质的机械特性(如低硬度),还可作为衰老细胞清除药物或生长因子的缓释载体[69-70],从而实现从“宏观替换”到“微观层面局部微环境的精准时空调控”的范式跨越。各项策略的总结见表1。"

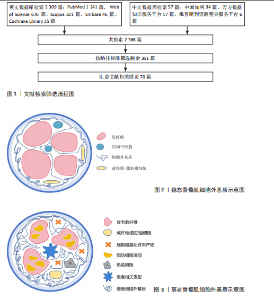
| [1] 李代萍,黄宁,葛美玲,等.中国骨骼肌衰老标志物专家共识(2024)计划书[J].中国循证医学杂志,2024,24(10):1137-1140. [2] LV J, WANG J, CHEN Q, et al. NIPSNAP1 and NIPSNAP2 facilitate healthy aging independent of mitophagy. Metabolism. 2025;170(4):156324. [3] 解瑛傲,孔健达,陈芸,等.骨骼肌中卫星细胞衰老生物学机制及潜在的应对策略[J].中国组织工程研究,2024,28(25):4094-4100. [4] MALATESTA M, CISTERNA B. Aging in the Skeletal Muscle Revealed by Molecular Immunohistochemical Imaging. Int J Mol Sci. 2025;26(13):5986. [5] KIM JT, JEON DH, LEE HJ. Molecular mechanism of skeletal muscle loss and its prevention by natural resources. Food Sci Biotechnol. 2024;33(15): 3387-3400. [6] LIBERGOLI M, ALMADA AE. Stem Cell Aging and Rejuvenation in the Skeletal Muscle System. Rejuvenation Res. 2025;3(15):1380-1390. [7] AGARWAL V, GUPTA A, CHAUDHARY R, et al. Elucidating the potential mechanism and therapeutic targets of chronic stress-induced muscle atrophy. Int Immunopharmacol. 2025; 5(10):1180-1185. [8] 甘彦明,张宇航,张学林,等.骨骼肌细胞骨架与被动刚度[J].生命科学,2023,35(9):1207-1214. [9] TSUJI R, HAYASHI T, TAKAHASHI S, et al. The multifaceted role of MyoD in adult skeletal muscle: homeostasis, regeneration, and diseases. Am J Physiol Cell Physiol. 2025;329(1):200-212. [10] 朱师宇,吕安康,赵宇星,等.基质金属蛋白酶-1、金属蛋白酶组织抑制因子-1在老年肌少症大鼠中的表达及意义[J].南方医科大学学报,2020,40(1):104-109. [11] 孔健达,穆玉晶,朱磊,等.骨骼肌再生过程中卫星细胞调控机制及其生态位信号的作用[J].中国组织工程研究,2024,28(7):1105-1111. [12] ELENA ZK, REIMER C, HERRMANN D, et al. Substrate stiffness modifies gene expression and transcriptional response of equine endometrial fibroblasts to TGF-β1. Anim Reprod Sci. 2025; 278(2):107873. [13] 靳庆瑞,边汝涛,徐学功.成纤维细胞与巨噬细胞交互作用在心肌纤维化中的研究进展[J].中华老年心脑血管病杂志,2025,27(3):389-391. [14] KANAZAWA Y, TAKAHASHI T, INOUE T, et al. Effects of Aging on Intramuscular Collagen-Related Factors After Injury to Mouse Tibialis Anterior Muscle. Int J Mol Sci. 2025;26(2):801. [15] KAMAL M, SHANMUGANATHAN M, KROEZEN Z, et al. Senescent myoblasts exhibit an altered exometabolome that is linked to senescence-associated secretory phenotype signaling. Am J Physiol Cell Physiol. 2025;328(2):440-451. [16] OLSON LC, JAWAD AY, CROCKER ES, et al. Glycations on Decellularized Muscle Matrix Reduce Muscle Regeneration and Increase Inflammation. Tissue Eng Part A. 2025;24(4):319-325. [17] CHANDRA P, FAIZAN M, PORWAL M, et al. An Overview and Review of Growth Factors in Wound Healing: Emerging Trends and Innovations. Curr Diabetes Rev. 2025;14(2):1219-1225. [18] YAO Y, LUO Y, LIANG X, et al. The role of oxidative stress-mediated fibro-adipogenic progenitor senescence in skeletal muscle regeneration and repair. Stem Cell Res Ther. 2025;16(1):104. [19] RODA MA, XU X, ABDALLA TH, et al. Proline-Glycine-Proline Peptides Are Critical in the Development of Smoke-induced Emphysema. Am J Respir Cell Mol Biol. 2019;61(5):560-566. [20] DE SAEYTYD L, WANG Z, BLOEMEN M, et al. Effect of transforming growth factor beta receptor I inhibitors on myotube formation in vitro. Sci Rep. 2025;15(1):23692. [21] COLLAO N, JOHANNSEN EB, JUST J, et al. Single-cell transcriptomic analysis reveals alterations to cellular dynamics and paracrine signaling in radiation-induced muscle pathology. Am J Physiol Cell Physiol. 2025;328(6):1995-2012. [22] MA X, LIU B, JIANG Z, et al. Physical Exercise: A Promising Treatment Against Organ Fibrosis. Int J Mol Sci. 2025;26(1):343. [23] KASPRZYCKA P, CIEMERYCH MA, ZIMOWSKA M. Differential regulation of MMP activity by TGFβ1 in fast- and slow- twitch muscle repair: insights from EDL and soleus muscle-derived myoblasts. Front Cell Dev Biol. 2025;328(2):440-451. [24] BAGHY K, SZAKADATI H, KOVALSZKY I. Decorin the antifibrotic proteoglycan and its progression in therapy. Am J Physiol Cell Physiol. 2025;328(6):1853-1865. [25] ZHONG W, JIA H, ZHU H, et al. Sarcopenia is attenuated by mairin in SAMP8 mice via the inhibition of FAPs fibrosis through the AMPK-TGF-β-SMAD axis. Gene. 2024;931(1):148873. [26] ATLANTE S, GOTTARDI ZAMPERLA M, CIS L, et al. Senolytic therapies for cardiovascular aging: tackling fibrosis and metabolic dysfunction. Eur J Intern Med. 2025;318(6):1153-1165. [27] ALEXAKOU E, BAKOPOULOU A, APATZIDOU DA, et al. Biological Effects of “Inflammageing” on Human Oral Cells: Insights into a Potential Confounder of Age-Related Diseases. Int J Mol Sci. 2023;25(1):5. [28] KIM IS, YANG WS, KIM CH. Physiological Properties, Functions, and Trends in the Matrix Metalloproteinase Inhibitors in Inflammation-Mediated Human Diseases. Curr Med Chem. 2023;30(18):2075-2112. [29] CHEN WJ, LIN IH, LEE CW, et al. Aged Skeletal Muscle Retains the Ability to Remodel Extracellular Matrix for Degradation of Collagen Deposition after Muscle Injury. Int J Mol Sci. 2021;22(4):2123. [30] BONNEMA DD, WEBB CS, PENNINGTON WR, et al. Effects of age on plasma matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinases (TIMPs). J Card Fail. 2007;13(7):530-540. [31] ALAKHDAR AA, SIVAKUMAR S, KOPCHAK RM, et al. Age-Related ECM Stiffness Mediates TRAIL Activation in Muscle Stem Cell Differentiation. Adv Biol (Weinh). 2024;8(12):e2400334. [32] DIll MN, TURNER Z, KAPUSCINSKA PW, et al. Exploring the Unique Extracellular Matrix Composition of Acomys as a Potential Key to Resisting Fibrosis. ACS Biomater Sci Eng. 2025; 11(6):3616-3633. [33] GRIGORIEVA O, BASALOVA N, DYACHKOVA U, et al. Modeling the profibrotic microenvironment in vitro: Model validation. Biochem Biophys Res Commun. 2024;733(2):150574. [34] MARCUCCI L, REGGIANI C. Increase of resting muscle stiffness, a less considered component of age-related skeletal muscle impairment. Eur J Transl Myol. 2020;30(2):8982. [35] MADI CM, WANG YX, HOLBROOK CA, et al. Hydrogel biomaterials that stiffen and soften on demand reveal that skeletal muscle stem cells harbor a mechanical memory. Proc Natl Acad Sci U S A. 2024;121(35):e2406787121. [36] GISH P, STEWART M, KHUU B, et al. The impact of extracellular matrix proteins on bovine fibro-adipogenic progenitor cell adhesion, proliferation, and differentiation in vitro. Physiol Rep. 2025; 13(9):e70283. [37] KIM J, KIM IU, LEE ZF, et al. Detrimental effects of advanced glycation end-products (AGEs) on a 3D skeletal muscle model in microphysiological system. Biosens Bioelectron. 2025;12(278):117316. [38] GUO Z, LI H, JIANG S, et al. The role of AGEs in muscle ageing and sarcopenia. Bone Joint Res. 2025;14(3):185-198. [39] GUVATOVA ZG, KUDASHEVA ER, EFREMOV YM, et al. Changes in Gene Expression Patterns in Young and Senescent Fibroblasts in Glycated Three-Dimensional Collagen Matrices. Int J Mol Sci. 2025;26(10):4769. [40] KOSUGE H, NAKAKIDO M, DE VEGA S, et al. Prelp functions via multiple interactions with intrinsically weak affinity relying on ECM anchoring and remodeling. Sci Rep. 2025;15(1):24634. [41] SANKHE CS, SACCO JL, CRUNKLETON VL, et al. Matrix Stiffness Regulates TGFβ1-Induced αSMA Expression via a G9a-LATS-YAP Signaling Cascade. FASEB Bioadv. 2025;7(7):e70035. [42] NELSON AC, MOLLEY TG, GONZALEZ G, et al. Vinculin haploinsufficiency impairs integrin-mediated costamere remodeling on stiffer microenvironments. J Mol Cell Cardiol. 2025;200: 1-10. [43] YOUSEFI F, FOSTER LA, SELIM OA, et al. Integrating Physical and Biochemical Cues for Muscle Engineering: Scaffolds and Graft Durability. Bioengineering (Basel). 2024;11(12):1245. [44] PODRAZA-FARHANIEH A, SPINELLI R, ZATTERALE F, et al. Physical training reduces cell senescence and associated insulin resistance in skeletal muscle. Mol Metab. 2025;95:102130. [45] SCHWEITZER AM, KOEHLE MS, FLISS MD, et al. Collagen remodeling increases after acute resistance exercise in healthy skeletal muscle irrespective of age. Am J Physiol Cell Physiol. 2025; 329(1):68-81. [46] CAVANAUGH CA, MOORE AE, FITZ NF, et al. Transcriptomic Response to Neuromuscular Electrical Stimulation in Muscle, Brain, and Plasma EVs in WT and Klotho-Deficient Mice. Int J Mol Sci. 2025;26(16):7849. [47] KUMAR P, UMAKANTH S, N G. Photobiomodulation therapy as an adjunct to resistance exercises on muscle metrics, functional balance, functional capacity, and physical performance among older adults: A systematic scoping review. Lasers Med Sci. 2024;39(1):232. [48] PARK SW, SHIN J, JEONG BK, et al. The Effects of Extracorporeal Shock Wave Therapy on Cutaneous Radiation Injury in a Mouse Model. Plast Reconstr Surg. 2025;155(5):813-825. [49] 赵娜,张玮,庞赓,等.基于TGF-β1/CTGF作用途径探讨推拿干预骨骼肌纤维化的作用机制[J].辽宁中医杂志,2016,43(12):2539-2541. [50] 庞赓,李沙,唐新月,等.推拿疗法对骨骼肌纤维化大鼠MMP-1和TIMP-1表达的影响[J].天津中医药大学学报,2020,39(2):209-214. [51] 陈玉佩,刘通,许玥,等.电针“委中”穴对大鼠腰多裂肌损伤后细胞外基质中相关蛋白表达的影响[J].针刺研究,2019,44(5):341-346. [52] ANCEL S, MICHAUD J, MIGLIAVACCA E, et al. Nicotinamide and pyridoxine stimulate muscle stem cell expansion and enhance regenerative capacity during aging. J Clin Invest. 2024;134(24):e163648. [53] BANG S, KIM DE, KANG HT, et al. Metformin restores autophagic flux and mitochondrial function in late passage myoblast to impede age-related muscle loss. Biomed Pharmacother. 2024;180:116981. [54] JI F, LEE HS, LEE H, et al. The impact of frailty syndrome on skeletal muscle histology: preventive effects of exercise. FEBS Open Bio. 2025;329(1): 68-81. [55] CARRICK-RANSON G, FUJIMOTO N, SHAFER KM, et al. The effect of 1 year of Alagebrium and moderate-intensity exercise training on left ventricular function during exercise in seniors: a randomized controlled trial. J Appl Physiol (1985). 2016;121(2):528-536. [56] BALAYAN A, DEBOUTRAY M, MOLLEY TG, et al. Dispase/collagenase cocktail allows for coisolation of satellite cells and fibroadipogenic progenitors from human skeletal muscle. Am J Physiol Cell Physiol. 2024;326(4):C1193-C1202. [57] SEARA FAC, KASAI-BRUNSWICK TH, NASCIMENTO JHM, et al. Anthracycline-induced cardiotoxicity and cell senescence: new therapeutic option? Cell Mol Life Sci. 2022;79(11):568. [58] NOVAIS EJ, TRAN VA, JOHNSTON SN, et al. Long-term treatment with senolytic drugs Dasatinib and Quercetin ameliorates age-dependent intervertebral disc degeneration in mice. Nat Commun. 2021;12(1):5213. [59] GAN C, WEI W, XUE T, et al. Focal adhesion kinase inhibitors in fibrotic diseases therapy: Development and therapeutic potential. Eur J Med Chem. 2025;296:117882. [60] BHATT J, GHIGO A, HIRSCH E. PI3K/Akt in IPF: untangling fibrosis and charting therapies. Front Immunol. 2025;16:1549277. [61] LUO YE, ABE-TEH Z, ALSAGHIR TY, et al. Fibro-Adipogenic Progenitors require autocrine IGF-I in homeostatic and regenerating skeletal muscle. Preprint. bioRxiv. 2025;2025.04.11.648330. [62] DEWI NM, MEILIANA A, DEFI IR, et al. Targeted Therapy for Skeletal Muscle Fibrosis: Regulation of Myostatin, TGF-β, MMP, and TIMP to Maintain Extracellular Matrix Homeostasis. Biologics. 2025;17(19):213-229. [63] OSTASZEWSKA A, MICHALSKA Z, DZIERZYNSKA M, et al. Beneficial but diverse influence of custom-designed hydrogels modified with IL-4 and SDF-1 peptides on selected populations of cells essential for skeletal muscle regeneration. Int J Biol Macromol. 2025;317(1):144282. [64] MORAIS LIMA V, CARRE A, POQUE E, et al. Biohybrid microstructured hydrogels obtained via in situ extracellular matrix deposition and decellularization using supercritical CO2. Biofabrication. 2025;213(2):123-134. [65] ZHANG C, LIU J. Engineered tumor microspheres via microfluidics and decellularized extracellular matrix for high-throughput organoid-based drug screening. Biofabrication. 2025;33(15):3387-3400. [66] TAN YH, ALCAZAR-DALEO CA, HOLBROOK JG, et al. Shear-Induced Patterning of Decellularized Skeletal Muscle Extracellular Matrix for Enhanced Myogenesis. Adv Healthc Mater. 2025;67(5): 103-112. [67] CHOI S, LEE MJ, KIM M, et al. Durable Muscle Extracellular Matrix Engineered with Adhesive Phenolic Moieties for Effective Skeletal Muscle Regeneration in Muscle Atrophy. Adv Healthc Mater. 2024;13(32):e2401826. [68] MENDES MC, SANTOS SC, CUSTODIO CA, et al. Advanced Injectable Human-Derived Microgels for Improved Cell Delivery and Tissue Regeneration. Adv Healthc Mater. 2025;21(2):65-76. [69] LI Y, ZHANG Y, WANG S, et al. Synergistic reversal of inflammation-mediated degeneration in intervertebral discs: Phenylboric acid-grafted hyaluronic acid hydrogel as an anti-oxidative vehicle for Timp-3 delivery and promotion of extracellular matrix synthesis. Acta Biomater. 2025;201:156-170. [70] LV Y, LIANG L, QIN M, et al. RGD peptide hydrogel downregulates mechanosignal YAP to inhibit postoperative scarring. Acta Biomater. 2025;199:132-141. |
| [1] | Zhang Nan, Meng Qinghua, Bao Chunyu. Characteristics and clinical application of ankle joint finite element models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2343-2349. |
| [2] | Jiang Xianglong, Li Zhongshan, Che Tongtong. Application effects and mechanisms of low-frequency pulsed electromagnetic fields in muscle repair and growth [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2350-2360. |
| [3] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [4] | Tan Jing, Li Li, Wang Liangliang, Qin Xiangyu. Bionic functional coating improves the integration of titanium implants and skin tissue interface [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2014-2022. |
| [5] | Wang Songpeng, Liu Yusan, Yu Huanying, Gao Xiaoli, Xu Yingjiang, Zhang Xiaoming, Liu Min. Bidirectional regulation of reactive oxygen species based on zeolitic imidazolate framework-8 nanomaterials: from tumor therapy and antibacterial activity to cytoprotection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2033-2013. |
| [6] | Chen Haojie, Wang Dai, Shen Shan. Immune inflammatory microenvironment mechanisms in peri-implantitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2054-2062. |
| [7] | Liu Dawei, Cui Yingying, Wang Fanghui, Wang Zixuan, Chen Yuhan, Li Yourui, Zhang Ronghe. Epigallocatechin gallate-mediated bidirectional regulation of reactive oxygen species and its application in nanomaterials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2101-2112. |
| [8] | Fu Lyupeng, Yu Peng, Liang Guoyan, Chang Yunbing. Electroactive materials applied in spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2113-2123. |
| [9] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [10] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [11] | Han Nianrong, Huang Yifei, Akram · Osman, Liu Yanlu, Hu Wei . Programmed cell death receptor-1 suppresses osteogenic differentiation of rat bone marrow mesenchymal stem cells in a high-glucose microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1649-1657. |
| [12] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [13] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [14] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [15] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||