Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5015-5023.doi: 10.12307/2026.219
Previous Articles Next Articles
Advantages and potential of cell-derived exosomes in oral tissue regeneration
Li Jiapeng1, Zuleina·Abula1, Jia Qianqian1, Nigare·Yunusijiang1, Sun Jiaqi1, Zhao Jin2, Wu Zeyu2
- 1School of Stomatology, Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China; 2Department of Cariology and Endodontics, The First Affiliated Hospital (The Affiliated Stomatology Hospital) of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China
-
Received:2025-08-25Accepted:2025-10-17Online:2026-07-08Published:2026-02-24 -
Contact:Wu Zeyu, MD, Physician, Department of Cariology and Endodontics, The First Affiliated Hospital (The Affiliated Stomatology Hospital) of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
About author:Li Jiapeng, School of Stomatology, Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
Supported by:Xinjiang Uygur Autonomous Region "Tianchi Talents" Introduction Program - Young Doctoral Talent Fund (to WZY); Xinjiang Uygur Autonomous Region "Tianshan Talents" Science and Technology Innovation Leading Talent Project, No. 2023TSYCL0032 (to WZY)
CLC Number:
Cite this article
Li Jiapeng, Zuleina·Abula, Jia Qianqian, Nigare·Yunusijiang, Sun Jiaqi, Zhao Jin, Wu Zeyu. Advantages and potential of cell-derived exosomes in oral tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5015-5023.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 外泌体与干细胞疗法的对比分析 2.1.1 干细胞疗法的现状与挑战 干细胞包括胚胎干细胞、诱导多能干细胞和成体干细胞,凭借卓越的自我更新和多系分化能力在组织稳态维持与再生中起关键作用[9]。牙髓干细胞能够促进牙本质-牙髓复合物的再生,牙周韧带干细胞、脱落乳牙干细胞和根尖牙乳头干细胞等也表现出促牙齿修复和再生的能力[10]。 干细胞疗法存在免疫排斥和肿瘤形成等风险。免疫系统会将同种异体干细胞视为外来抗原,引起移植物排斥,进而引发破坏性的免疫反应。在临床上,防止干细胞去分化是一个重大挑战,如果细胞在移植前没有完全分化,就有形成肿瘤的风险。 2.1.2 外泌体的生物学特性 外泌体是由各种类型细胞分泌的纳米级细胞外囊泡,直径在30-150 nm之间,参与许多生理和病理过程[11],这些囊泡携带蛋白质、脂质、mRNA和miRNA等多种生物分子,在细胞间通讯中扮演着关键角色,影响细胞增殖、分化和凋亡等关键生物过程,并因外泌体在癌症进展、调节免疫反应、促进血管生成和组织再生中的作用而受到极大关注。外泌体能够继承亲本细胞的功能,规避基于细胞疗法相关的挑战,成为再生医学中有前景的治疗方法,而且外泌体具有作为靶向药物递送系统和生物标志物的潜力,这凸显了它在多种疾病诊疗中的重要性[12]。 2.1.3 外泌体的独特优势 外泌体与干细胞疗法相比具有明显优势,凭借低致瘤风险、低免疫原性及精确的靶向递送能力,有望成为干细胞疗法的替代疗法。外泌体在冻干后可以稳定储存而不失去功能性,这与干细胞严格的储存条件形成鲜明对比[12]。外泌体的无细胞特性消除了与多能干细胞相关的致瘤风险,能够从多种组织中提取,与干细胞疗法相比显著降低了免疫排斥的风险[8],且能够将外泌体内容物精确传递到体内的特定部位,从而增强治疗效果,见表1。 "

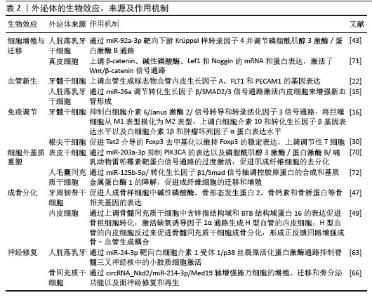
2.2 外泌体在组织再生中的作用机制 2.2.1 调控细胞增殖和迁移 调控细胞增殖和迁移是组织再生过程中的核心环节。Wnt/β-catenin信号通路是细胞增殖的关键通路,当Wnt信号激活时,β-catenin转移到细胞核,启动与分化相关基因的转录。Wnt/β-catenin与磷脂酰肌醇3激酶/蛋白激酶B通路协同发挥作用,通过促进细胞增殖、抑制细胞凋亡和阻断分化来驱动细胞扩增[13];丝裂原活化蛋白激酶/细胞外调节蛋白激酶通路激活与细胞黏附、运动相关的基因表达来促进细胞迁移[14],通过激活这些通路,外泌体能够调控细胞增殖和迁移。 2.2.2 血管新生、免疫调节及细胞外基质重塑 充足的血液供应是组织再生的必要条件,能够为损伤组织提供充足的氧气和营养并清除代谢废物,在血管生成方面,外泌体通过传递miR-26a调节转化生长因子β/SMAD2/3信号通路等方式激活内皮细胞来增强新血管形成[15]。免疫调节在组织再生中扮演着不可或缺的“指挥者”与“清道夫”双重角色,一方面免疫细胞识别损伤相关分子模式启动炎症反应,并分泌大量趋化因子和生长因子,将干细胞、成纤维细胞、内皮细胞等招募至损伤部位启动再生程序;另一方面中性粒细胞和巨噬细胞(早期M1型)等作为“清道夫”,吞噬死亡细胞、受损的细胞外基质、微生物和异物,并控制病原体入侵,避免感染破坏再生微环境。在组织损伤早期需要适度的炎症反应,但过度活跃和持续的炎症反应会阻碍组织再生并导致炎症损伤,外泌体通过将巨噬细胞极化转向抗炎的M2表型来调节免疫反应,从而减少炎症并增强组织修复[16]。细胞外基质重塑是指在生理或病理过程中,细胞主动调控细胞外基质的动态降解、合成、组装和结构重组,以改变细胞外基质成分、三维结构和生物活性的过程,旨在适应组织功能需求或修复损伤。通过影响成纤维细胞活性和基质金属蛋白酶活性等,外泌体能够调节细胞外基质重塑[17]。这些机制相互联系,外泌体释放的血管生成因子可以影响免疫细胞行为,而细胞外基质重塑则影响内皮细胞迁移和功能。 2.2.3 对干细胞与骨再生的调控 调控干细胞的核心目标是最大化干细胞的治疗潜力,而外泌体可以通过旁分泌或靶向递送,干预细胞的增殖、分化及功能表达。源自间充质干细胞的外泌体,如人牙周韧带干细胞来源外泌体可通过激活磷脂酰肌醇3激酶/蛋白激酶B信号通路提高牙骨质母细胞的活性,增加骨质生成相关基因和蛋白的表达[18]。调控口腔骨再生能够重建咀嚼功能、协同激活牙周组织再生,直接影响种植体成功率、牙周功能恢复及面部美学重建。研究表明,脱落乳牙干细胞来源外泌体能促进线粒体转录因子A的表达,从而增强谷氨酸代谢和氧化磷酸化活性以促进牙髓干细胞的骨再生[19]。骨再生是一个复杂的程序,涉及成骨与炎症之间的相互作用,人脐静脉内皮细胞衍生外泌体通过抑制X连锁死亡盒螺旋酶3/NOD样受体热蛋白结构域相关蛋白3轴以诱导巨噬细胞M2型极化,从而缓解炎症,并在海藻酸盐/明胶甲基丙烯酰互穿网络水凝胶的帮助下促进体内成骨[20]。外泌体还可以改变靶细胞的miRNAs谱,增强骨再生能力[21]。 2.3 外泌体在特定口腔组织再生中的应用 在口腔和颅面区域中软组织和硬组织共存,包括牙髓、牙周、颅面骨、唾液腺、神经等。不同来源外泌体在机体损伤修复和再生过程中通过不同的生物分子或信号通路作出反应。近年来,研究人员发现外泌体可促进口腔颅面软硬组织的再生修复,具有广泛的应用前景。 2.3.1 牙髓再生 牙髓是牙齿内唯一的软组织,位于牙髓腔内,被牙本质包围,它由结缔组织、血管和神经组成。因此,牙髓再生是一个多方面的过程,不仅包括牙髓再生和牙本质-牙髓复合物的形成,还包括牙髓血运重建。外泌体为牙髓-牙本质复合体的再生和牙髓血运重建提供了新的策略。 体外研究:GANESH等[22]研究表明5×1011 L-1的牙髓干细胞来源外泌体显著促进细胞增殖和迁移,上调血管生成标志物血管内皮生长因子A、FLT1和PECAM1的表达。WANG等[23]发现M2型巨噬细胞来源外泌体显著增强了牙髓干细胞和人脐静脉内皮细胞的增殖、迁移、成牙/成骨分化、神经生成、血管生成能力,有利于牙髓-牙本质复合体再生。在免疫调节方面,外泌体能够减轻脂多糖诱导的细胞死亡,并中和脂多糖对牙髓干细胞发育的抑制作用。AZARYAN等[24]将人乳外泌体以200 μg/mL剂量应用于脂多糖暴露的牙髓干细胞,结果表明人乳外泌体增强了细胞活力和迁移能力,并显著下调了炎症细胞因子肿瘤坏死因子α、白细胞介素1β和白细胞介素6的表达。MO等[25]探讨富血小板血浆来源外泌体对牙髓干细胞成骨分化的影响,发现富血小板血浆来源外泌体增强了成骨标志物的表达、碱性磷酸酶活性和钙化结节的形成,并改变了miRNAs的表达谱,其中Wnt信号通路相关miRNAs富集,表明富血小板血浆来源外泌体可能通过促进牙髓干细胞的成骨作用而有利于牙髓再生。有趣的是,成牙本质细胞中雷帕霉素复合体1的激活可以增加牙本质的体积和外泌体的释放,且外泌体以剂量依赖的方式负反馈抑制牙髓干细胞的成牙本质分化,这提示雷帕霉素复合体1可以作为牙髓再生的潜在调控点[26]。最新研究发现,人牙髓干细胞来源外泌体显著促进了牙髓干细胞的增殖、迁移和多系分化以及人脐静脉内皮细胞的管状结构形成,有助于牙髓-牙本质复合体再生[27]。另外,脂肪组织基质血管组分来源外泌体能够增强牙髓干细胞的迁移活性,且在0.1%的浓度下效果最佳,表明它在促进牙髓组织再生中具有潜力[28]。 体内研究:在牙髓缺损模型中使用牙髓干细胞来源外泌体显示出增强牙本质基质蛋白表达和牙髓-牙本质组织再生的能力[29]。值得注意的是,根尖牙乳头干细胞分泌的外泌体可以促进调节性T细胞转化并减少大鼠牙髓炎症,调节局部免疫环境并支持组织再生,这表明它们可能是治疗未成熟恒牙早期牙髓炎症的无细胞疗法[30]。组织学分析表明,牙髓干细胞来源外泌体处理的根管内有新的类似牙髓的组织生长,特别是与基质细胞衍生因子1结合时,显著增加血管密度并刺激根管内新组织的发生[31]。最新病例报告探讨了人脐带间充质干细胞来源外泌体对再生根管治疗牙的治疗潜力,他们将外泌体应用于患有不可逆性牙髓炎患者的根管内,治疗结果显示没有感染、肿胀或触痛,放射影像评估显示根尖周围透射性增强和牙周韧带增宽,这提示存在组织再生[32]。 2.3.2 牙周再生 牙周组织包括牙龈、牙周韧带、牙骨质和牙槽骨,是牙齿的结构支撑。牙周炎是成人牙齿丧失的主要原因。牙周再生的最终目标是促进新的牙骨质发育,并在新生的牙骨质和牙槽骨之间形成富含纤维的牙周韧带,实现真正的功能性附着。近年来,外泌体作为新兴的生物治疗手段,与机械性去除牙菌斑、再生手术、激光辅助治疗和药物治疗等疗法相比,在促进牙周再生方面表现更优。 体外研究:DKK1与LRP5/ LRP6受体特异性结合,从而抑制Wnt/β-Catenin信号通路,而人骨髓间充质干细胞来源外泌体通过过表达miR-335-5p靶向下调DKK1,促进人牙周膜干细胞成骨分化,抑制牙周炎的进展[33]。牙髓干细胞来源外泌体在急性炎症应激下通过抑制白细胞介素6/Janus激酶2/信号转导和转录活化因子3信号通路和将巨噬细胞从M1表型极化为M2表型来控制炎症,并增强牙周韧带干细胞的增殖、迁移和成骨作用从而促进牙周组织再生[16]。而M2型巨噬细胞来源外泌体能够诱导巨噬细胞从M1型重新编程为M2型,以改善慢性炎症和加速牙周修复[34]。研究表明,牙龈间充质干细胞来源外泌体可促进牙周韧带干细胞的成骨分化,抑制牙龈卟啉单胞菌脂多糖诱导的核因子κB激活,上调Wnt/β-catenin通路,并显示出对两条通路之间的交叉调控能力[35]。GAO等[36]研究结果表明M2型巨噬细胞来源外泌体通过促进趋化因子配体12表达,增强了脂多糖诱导的牙周韧带干细胞的成骨分化能力,并抑制了炎症反应。HUANG等[37]通过沉默酪蛋白激酶2相互作用蛋白1基因构建具有矿化促进作用的稳定M2型巨噬细胞来源外泌体,并发现这些外泌体能够通过传递Let-7f-5p靶向沉默酪蛋白激酶2相互作用蛋白1,激活成牙骨质细胞中过氧化物酶体增殖物激活受体γ共激活因子1α依赖的线粒体生物合成,从而挽救牙龈卟啉单胞菌抑制的成牙骨质细胞矿化。ELSAYED等[38]发现人单核细胞衍生的调节性树突状细胞外泌体可诱导CD25、Foxp3和调节性T细胞表达,并在受体成熟树突状细胞中上调程序性死亡配体1,下调共刺激分子,从而调节靶向树突状细胞和T细胞的免疫反应。 体内研究:褪黑激素+M2型巨噬细胞来源外泌体在炎症性牙周组织再生方面展现出潜力,与PBS组相比,外泌体组的破骨细胞数量减少了5倍以上,骨体积分数翻倍[34]。LU等[39]研究探讨了正常糖培养和高糖预处理的牙周韧带干细胞来源外泌体在糖尿病牙周炎中调节牙周再生的功能和机制,发现牙周韧带干细胞来源外泌体miR-31-5p通过靶向抑制破骨细胞中内皮型一氧化氮合酶蛋白表达减少牙槽骨破坏和破骨细胞数量。LIU等[40]研究探讨了骨髓间充质干细胞来源外泌体中长链非编码组织相容性白细胞抗原复合物 P5促进人牙周韧带干细胞成骨分化和缓解慢性牙周炎的作用,发现白细胞抗原复合物 P5作为miR-24-3p的海绵来上调血红素加氧酶,从而激活P38/ELK1通路,增强成骨作用并减轻炎症。最新研究发现,受到力刺激的牙囊干细胞释放的外泌体含有miR-140-3p,通过靶向DNA甲基转移酶1阻断DNA甲基转移酶1/细胞因子信号转导抑制因子1/核因子κB轴,从而抑制NOD样受体热蛋白结构域相关蛋白3介导的牙周膜细胞焦亡,进而减少M1型巨噬细胞极化和破骨细胞形成,以缓解根吸收[41]。在牙周骨缺损大鼠模型中,施万细胞来源外泌体能够促进内源性细胞募集,调节神经和血管新生,并通过调节骨愈合微环境促进牙周骨再生[42]。富含miR-92a-3p的脱落乳牙干细胞来源外泌体能够增强牙周韧带干细胞的增殖和成骨分化,减少凋亡和炎症,并通过抑制Kruppel样因子4/磷脂酰肌醇3激酶/蛋白激酶B通路缓解牙周炎[43]。临床研究发现,细胞外囊泡疗法显著改善了牙周炎患者的牙菌斑指数和探诊深度等临床参数,并将促炎细胞因子水平降低至健康水平,表明细胞外囊泡可能是恢复牙周健康的一种有前景的方法[44]。 2.3.3 颅面骨再生 骨密度因部位略有不同,而颅面骨组织含有更多孔隙,骨折的可能性较高;同时,颅面区域的骨组织大多不规则且血供丰富,这不仅意味着其抗感染能力强,也表明因损伤而引发严重炎症的概率更高。此外,创伤、感染、肿瘤术后等造成的骨缺损会严重影响颅面区域的功能和美观,因此颅面骨组织的修复和再生一直是一个复杂的临床问题。外泌体在促颅面骨修复的应用中越来越被关注,体内外研究均表明,它们能够显著促进骨组织的再生和修复。在促血管生成的研究中,中性粒细胞衍生外泌体通过miR-455-3p靶向Smad4通路介导内皮祖细胞增殖以促进内源性血管形成,从而加速内源性骨再生[45]。另有研究发现,脂肪间充质基质细胞来源外泌体能够抑制巨噬细胞M1型极化和焦亡,从而预防药物相关性颌骨坏死,作用机制涉及阻断核因子κB/ NOD样受体热蛋白结构域相关蛋白3/白细胞介素1β轴[46]。骨再生是通过骨形成(由成骨细胞完成)与骨吸收和重塑(由破骨细胞完成)之间受到严密调控的相互作用来实现的,理想情况下,再生疗法应促进成骨细胞的分化(骨形成)和/或抑制破骨细胞的分化(骨吸收)。ALBOUGHA等[47]研究探讨了人牙周韧带干细胞来源外泌体对成骨细胞样细胞和大鼠颅骨缺损的影响,发现人牙周韧带干细胞来源外泌体通过增强细胞迁移、矿化和骨相关基因表达,在体外促进成骨,而在体内则通过增加缺损部位的新骨形成来促进成骨,表明人牙周韧带干细胞来源外泌体在牙周组织修复治疗中具有潜在应用价值。HE等[48]介绍了一种使用Piezo1通道激动剂Yoda1预处理的骨髓间充质干细胞外泌体整合到甲基丙烯酰化明胶/海藻酸钠/β-磷酸三钙水凝胶中的新骨再生方法,结果表明该方法在皮下异位成骨裸鼠模型和大鼠颅骨缺损模型中增强了成骨作用,为临床骨缺损重建提供了希望。有趣的是,内皮细胞来源外泌体通过促进骨髓间充质干细胞的成骨分化和内皮细胞的血管生成,启动并增强成骨-血管生成偶联的正反馈回路,其中含锌指结构域和BTB结构域的蛋白16和缺氧诱导因子1α通路发挥关键作用[49]。外泌体与生物材料结合,例如使用渗入羟基磷灰石的小肠黏膜下层复合水凝胶同轴支架加载外泌体,能够自适应地填充拔牙窝并保持成骨,实现全方位高效的拔牙后骨修复[50]。YUAN等[51]开发了一种锶预处理系统优化滑膜间充质干细胞外泌体,通过miR-143-3p等miRNAs减少软骨细胞铁死亡和破骨细胞介导的关节疼痛,在颞下颌关节骨关节炎中显示卓越的治疗效果。 2.3.4 唾液腺活化 唾液腺在产生和分泌唾液方面发挥着至关重要的作用。唾液功能丧失会导致唾液质量和数量下降,从而增加口腔和全身感染的机会;导致吞咽困难和味觉丧失,从而导致营养不良。在干燥综合征中,牙髓干细胞来源外泌体激活跨膜G蛋白偶联雌激素受体介导的cAMP/蛋白激酶A/cAMP应答元件结合蛋白质途径以增强唾液腺上皮细胞功能,使得模型小鼠唾液流速增加、腺体炎症减轻[52]。缺氧预处理的人尿源性干细胞外泌体能够通过激活Wnt3a/糖原合成酶激酶3β途径和抑制α-平滑肌肌动蛋白和c-Kit表达来修复辐射诱导的唾液腺损伤[53]。DU等[54]发现脱落乳牙干细胞来源外泌体能够增加干燥综合征小鼠模型的唾液流量,减少淋巴细胞浸润,并降低炎性细胞因子水平,作用机制可能是通过miR-29a-3p/T-bet轴抑制Th1细胞分化有关。另有研究发现, 脱落乳牙干细胞来源外泌体通过促进闭锁小带蛋白1表达,增强腺泡上皮细胞的通透性,从而改善干燥综合征小鼠模型的唾液分泌,作用机制涉及抑制蛋白激酶B/糖原合成酶激酶3β/Slug信号通路,该通路可负调控闭锁小带蛋白1表达[55]。脱落乳牙干细胞来源外泌体还能够限制干燥综合征炎症诱导的上皮细胞凋亡,通过抑制磷酸化细胞外调节蛋白激酶1/2的激活以及阻断多条细胞死亡通路,从而促进唾液分泌[56]。对于老年小鼠,衰老通过诱导下颌下腺衰老和破坏紧密连接分子的表达来减少唾液分泌,注射牙髓干细胞外泌体可以改善老年小鼠的唾液流量、减少腺泡萎缩、降低β-半乳糖苷酶活性并延缓衰老[57]。GUO等[58]发现脂肪干细胞来源外泌体通过增加唾液分泌、促进细胞增殖和减少纤维化,恢复了小鼠辐射损伤的唾液腺功能,主要机制是通过miR-199a-3p靶向Twist1来抑制上皮-间充质转化,并阻断转化生长因子β1/Smad3信号通路。在免疫调节方面,人脐带间充质干细胞来源外泌体能够通过自噬途径调节原发性干燥综合征小鼠CD4+ T细胞的功能,抑制过度增殖、凋亡和Th17细胞分化,促进调节性T细胞分化,从而恢复Th17细胞和调节性T细胞比例[59]。ZAKARIA等[60]评估了骨髓间充质干细胞及骨髓间充质干细胞来源外泌体对顺铂诱导的大鼠腮腺损伤的影响,发现骨髓间充质干细胞及其来源外泌体均改善了组织学特征,增加了增殖细胞核抗原阳性细胞,减少了氧化应激和凋亡,且外泌体治疗比细胞治疗取得了更好的效果。 2.3.5 神经再生 颌面神经系统疾病包括一系列影响颅神经的疾病,这些疾病可能由多种原因引起,包括肿瘤、感染、创伤和手术并发症等,导致严重的功能障碍,研究治疗这些疾病的新方法对于恢复面部的感觉和运动功能至关重要。外泌体能够通过多种机制减轻炎症反应、抑制细胞凋亡并促进轴突生长和髓鞘化,在神经炎症调节和神经修复中展现出显著的治疗潜力。例如,经肿瘤坏死因子α刺激的牙龈间充质干细胞通过母系表达基因3/miR-21-5p/程序性细胞死亡因子4轴递送富含miR-21-5p的外泌体显著减少了炎性小胶质细胞并抑制了视网膜神经节细胞凋亡[61]。研究表明,叉头转录因子O3的3’非翻译区与miR-197-3p结合,可抑制其转录,而牙髓干细胞外泌体通过抑制叉头转录因子O3的表达从而增强miR-197-3p的保护作用并抑制NOD样受体热蛋白结构域相关蛋白3炎性小体的激活和相关细胞因子的释放,抑制小胶质细胞焦亡,从而减轻神经炎症[62]。脱落乳牙干细胞来源外泌体通过miR-24-3p靶向白细胞介素1受体1阻断磷酸化p38丝裂原活化蛋白激酶通路抑制脊髓三叉神经核中的小胶质细胞激活,减轻了眶下神经慢性缩窄损伤小鼠的三叉神经痛[63]。YU等[64]将人脐带间充质干细胞来源细胞外囊泡注入患有慢性眼内高压的大鼠玻璃体腔中,显著抑制半胱天冬酶3的激活,增加视网膜神经节细胞的数量,表明这些囊泡可能通过抑制凋亡来减轻视神经损伤。在促进大鼠面神经修复方面,脂肪干细胞来源外泌体通过增强轴突再生和髓鞘化,减少炎症,驱动巨噬细胞向M2型极化,降低M1型与M2型巨噬细胞的比例,并通过p38丝裂原活化蛋白激酶/核因子κB通路控制巨噬细胞的异质性来抑制炎症,提示脂肪干细胞来源外泌体作为面神经损伤潜在治疗方法的可能性[65]。缺氧预处理的骨髓间充质干细胞来源外泌体通过促进施万细胞的增殖、迁移和旁分泌功能,增强大鼠面神经的修复,其中外泌体中的circRNA_Nkd2通过吸附miR-214-3p来上调MED19,表明缺氧预处理可改善治疗效果[66]。脐带间充质干细胞来源外泌体通过传递特定的miRNAs( miR-222-3p、miR-22-3p),在视网膜神经节细胞中激活雷帕霉素复合体1信号通路以增强神经突生长和轴突再生,从而促进视神经再生和视网膜神经节细胞存活[67]。SALEM等[68]评估了骨髓间充质干细胞来源外泌体(有或没有氯化锌)在大鼠双侧舌咽神经切断术后促进轮廓乳头再生的潜力,结果显示,单独使用骨髓间充质干细胞来源外泌体与生理盐水组相比显著增强了味蕾的恢复并减少了氧化应激,而与氯化锌联合使用则产生了较差的结果,表明外泌体具有更优越的再生能力。 2.3.6 皮肤愈合 皮肤损伤是紫外线、创伤和大肿瘤切除等引起的常见疾病。然而,较长时间的愈合通常会导致过度的瘢痕形成,这给患者的心理和生理带来负担,尤其是当损伤暴露于外界的口腔颜面部时。外泌体通过递送功能性分子、调节细胞行为,为肥厚性瘢痕、毛囊再生及皮肤光老化等皮肤损伤疾病提供了创新性的无细胞治疗策略。研究发现,脂肪干细胞来源外泌体通过特异性递送miR-125b-5p来抑制Smad2的表达,从而抑制肌成纤维细胞的转分化以及纤维化相关分子的表达,进而减轻肥厚性瘢痕[69]。表皮干细胞来源外泌体通过递送miR-203a-3p来抑制PIK3CA的表达和磷脂酰肌醇3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路的过度激活,从而促进肌成纤维细胞的去分化,减轻肥厚性瘢痕[70]。在毛囊再生方面,毛乳头细胞来源外泌体通过激活Wnt/β-catenin信号通路促进成纤维细胞的增殖、迁移和毛发诱导能力,在伤口愈合过程中增强毛囊再生[71]。CUI等[72]研究表明,人毛囊间充质干细胞来源外泌体通过增强胶原蛋白形成、促进成纤维细胞增殖和迁移以及逆转紫外线诱导的衰老,有效缓解皮肤光老化,从机制上讲,人毛囊间充质干细胞来源外泌体通过传递miR-125b-5p来上调转化生长因子β1和Smad通路活性,下调基质金属蛋白酶1,凸显了miR-125b-5p/转化生长因子β1/Smad轴作为皮肤光老化治疗靶点的潜力。在皮下伤口模型中,3D生物打印的 COL@d-ECM/M2-Exo水凝胶显示出强大的伤口重塑和毛囊诱导作用[73]。 外泌体的来源及作用机制,见表2。"


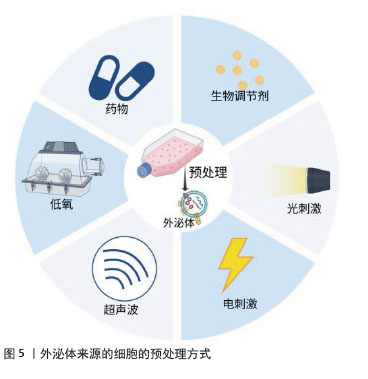
间充质干细胞在体内处于缺氧生态位,并且大多数间充质干细胞存在于体内氧体积分数为2%-8%(甚至更低)的环境中。因此,在低氧环境中培养间充质干细胞将更紧密地与生理环境保持一致,同时增强外泌体对特定生物活性分子的表达,这些分子在促进血管生成和修复、调节免疫反应和组织修复中扮演关键角色[74]。ZUO等[75]的体外实验表明,缺氧预处理的间充质干细胞外泌体在多个方面显著优于常氧间充质干细胞外泌体,包括缓解缺氧诱导的异常血管生成、维持血管内皮细胞活力以及抑制缺氧应激下的活性氧积累和凋亡信号传导。 外泌体的产生和功能可以通过生化因子(如促炎因子和生长因子)预处理亲本细胞来调节,在培养基中加入肿瘤坏死因子α、干扰素γ及脂多糖等生物调节剂能够改变外泌体内容物(蛋白质组成、细胞因子和RNA含量等)的组成,增强外泌体治疗相关疾病的潜力[76]。研究证实,肿瘤坏死因子α预处理能够激活间充质干细胞的磷脂酰肌醇3激酶/蛋白激酶B信号通路,使自噬相关蛋白16样1水平上调,从而促进外泌体分泌,并上调外泌体中低密度脂蛋白受体相关蛋白1水平增强外泌体的组织保护作用[77]。 褪黑激素是一种有效的自由基清除剂和金属螯合剂,能够缓解氧化应激和炎症反应并稳定细胞膜,作为一种新的药物预处理方法,在慢性肾病、糖尿病伤口愈合和缺血再灌注等疾病的治疗中可有效增强外泌体的抗氧化、抗炎和抗凋亡等功能[78]。例如,CHEN等[79]观察到工程化褪黑激素预处理能够促进血浆外泌体的释放,并通过递送miR-138-5p降低了SOX4的表达从而促进小胶质细胞从M1型向M2型转化以增强抗炎功能。 大量研究证实,外泌体的产生和生物学特性都可以通过暴露于各种物理刺激来调节。此外,也可以将物理特性引入外泌体以增强外泌体的功能。例如,CHEN等[80]在体内和体外实验中均证实电刺激预处理的骨髓间充质干细胞衍生外泌体能够显著促进成骨细胞增殖和骨髓间充质干细胞成骨分化,表现出良好的骨再生效果;外泌体还可以通过加载磁性纳米颗粒来赋予磁性,进而控制外泌体的靶向递送路径并通过磁性标记跟踪外泌体的生物分布。物理刺激一方面可以模拟外泌体所在微环境动力学,提高它们的分泌及生物学效应;另一方面可以作为工程外泌体的技术,最终促进临床转化[81]。 2.4.2 分离与纯化 从复杂的生物成分中分离和富集外泌体是研究外泌体及其生物医学意义的前提[82]。随着科学研究的快速进步,已经开发出许多方法来分离大量和高纯度的外泌体,包括超速离心、体积排阻色谱、免疫亲和捕获、聚合物共沉淀、超滤和微流控技术等。有效的分离方法必须在避免其他分子(如脂蛋白和蛋白质聚集体)污染的同时,浓缩外泌体信号以便于分析和后续研究。超速离心是传统的黄金标准,但获得的样品量少,常常与可重复性好、样品量大的体积排阻色谱联合使用以提高效率。免疫亲和捕获法分离的外泌体纯度高、特异性高且对结构和形态无影响,但存在分离成本高、需要额外的分离和纯化程序等缺点。超滤得到的外泌体纯度高且速度快,但过滤时的压力和剪切力可能导致外泌体变形和损坏,这显著降低了外泌体产量。微流控芯片是小型轻量级系统,能够高效、高通量地分离纯化外泌体,其应用需要按具体要求进行定制、价格昂贵,在临床推广上存在挑战[83]。每种外泌体分离技术存在的各自优势和局限性,见表3。 "

| [1] GUARNIZO-HERREÑO CC, CELESTE RK, PERES MA. The ongoing fight for population oral health. Lancet. 2024;404(10453):635-638. [2] LI C, DU L, XIAO Y, et al. Multi-active phlorotannins boost antimicrobial peptide LL-37 to promote periodontal tissue regeneration in diabetic periodontitis. Mater Today Bio. 2025;31:101535. [3] HUANG D, WANG YY, LI BH, et al. Association between periodontal disease and systemic diseases: a cross-sectional analysis of current evidence. Mil Med Res. 2024;11(1):74. [4] DAMIANI BAM, PINTO KP, FERREIRA CMA, et al. Apical periodontitis as an aggravating factor for the severity of rheumatoid arthritis: An animal study. Int Endod J. 2024;57(11):1669-1681. [5] ZHU Y, ZHANG X, CHANG G, et al. Bioactive Glass in Tissue Regeneration: Unveiling Recent Advances in Regenerative Strategies and Applications. Adv Mater. 2025;37(2):e2312964. [6] YOU J, ZHANG Q, QIAN L, et al. Antibacterial periodontal ligament stem cells enhance periodontal regeneration and regulate the oral microbiome. Stem Cell Res Ther. 2024;15(1):334. [7] PAN BT, JOHNSTONE RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983; 33(3):967-978. [8] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020; 367(6478):eaau6977. [9] DE MORREE A, RANDO TA. Regulation of adult stem cell quiescence and its functions in the maintenance of tissue integrity. Nat Rev Mol Cell Biol. 2023;24(5):334-354. [10] ZHANG W, YELICK PC. Tooth Repair and Regeneration: Potential of Dental Stem Cells. Trends Mol Med. 2021;27(5):501-511. [11] LI J, WANG J, CHEN Z. Emerging role of exosomes in cancer therapy: progress and challenges. Mol Cancer. 2025;24(1):13. [12] CHOUDHERY MS, ARIF T, MAHMOOD R, et al. Stem Cell-Based Acellular Therapy: Insight into Biogenesis, Bioengineering and Therapeutic Applications of Exosomes. Biomolecules. 2024; 14(7):792. [13] PERRY JM, HE XC, SUGIMURA R, et al. Cooperation between both Wnt/{beta}-catenin and PTEN/PI3K/Akt signaling promotes primitive hematopoietic stem cell self-renewal and expansion. Genes Dev. 2011;25(18):1928-1942. [14] QIU S, XIE L, LU C, et al. Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis. J Exp Clin Cancer Res. 2022; 41(1):296. [15] WU M, LIU X, LI Z, et al. SHED aggregate exosomes shuttled miR-26a promote angiogenesis in pulp regeneration via TGF-β/SMAD2/3 signalling. Cell Prolif. 2021;54(7):e13074. [16] QIAO X, TANG J, DOU L, et al. Dental Pulp Stem Cell-Derived Exosomes Regulate Anti-Inflammatory and Osteogenesis in Periodontal Ligament Stem Cells and Promote the Repair of Experimental Periodontitis in Rats. Int J Nanomedicine. 2023;18:4683-4703. [17] PAPADOPOULOS KS, PIPERI C, KORKOLOPOULOU P. Clinical Applications of Adipose-Derived Stem Cell (ADSC) Exosomes in Tissue Regeneration. Int J Mol Sci. 2024;25(11):5916. [18] LI S, GUAN X, YU W, et al. Effect of human periodontal ligament stem cell-derived exosomes on cementoblast activity. Oral Dis. 2024;30(4): 2511-2522. [19] GUO J, ZHOU F, LIU Z, et al. Exosome-shuttled mitochondrial transcription factor A mRNA promotes the osteogenesis of dental pulp stem cells through mitochondrial oxidative phosphorylation activation. Cell Prolif. 2022; 55(12):e13324. [20] CHEN Y, WU Y, GUO L, et al. Exosomal Lnc NEAT1 from endothelial cells promote bone regeneration by regulating macrophage polarization via DDX3X/NLRP3 axis. J Nanobiotechnology. 2023;21(1):98. [21] LI K, GU X, ZHU Y, et al. Human Umbilical Cord Mesenchymal Stem Cells-Derived Exosomes Attenuates Experimental Periodontitis in Mice Partly by Delivering miRNAs. Int J Nanomedicine. 2025;20:2879-2899. [22] GANESH V, SEOL D, GOMEZ-CONTRERAS PC, et al. Exosome-Based Cell Homing and Angiogenic Differentiation for Dental Pulp Regeneration. Int J Mol Sci. 2022;24(1):466. [23] WANG Y, MAO J, WANG Y, et al. Multifunctional Exosomes Derived from M2 Macrophages with Enhanced Odontogenesis, Neurogenesis and Angiogenesis for Regenerative Endodontic Therapy: An In Vitro and In Vivo Investigation. Biomedicines. 2024;12(2):441. [24] AZARYAN E, KARBASI S, SAHARKHIZ M, et al. Effect of HM-Exos on the migration and inflammatory response of LPS-exposed dental pulp stem cells. BMC Oral Health. 2023;23(1):95. [25] MO C, LIU Z, LIN Y, et al. Effects of exosomes derived from platelet-rich plasma on osteogenic differentiation of dental pulp stem cells. Cell Mol Biol (Noisy-le-grand). 2024;70(2):227-234. [26] LUO X, FENG W, HUANG S, et al. Odontoblasts release exosomes to regulate the odontoblastic differentiation of dental pulp stem cells. Stem Cell Res Ther. 2023;14(1):176. [27] WANG Y, MAO J, WANG Y, et al. Odontogenic exosomes simulating the developmental microenvironment promote complete regeneration of pulp-dentin complex in vivo. J Adv Res. 2025. doi: 10.1016/j.jare.2024.12.048. [28] ALINDA SD, MARGONO A, YULIANTO I, et al. The potential of exosomes from adipose-derived stromal-vascular fraction in Increasing Migration Activity of Human Dental Pulp Stromal Cells (in vitro study). Saudi Dent J. 2024;36(11):1425-1431. [29] WANG S, XING X, PENG W, et al. Fabrication of an exosome-loaded thermosensitive chitin-based hydrogel for dental pulp regeneration. J Mater Chem B. 2023;11(7):1580-1590. [30] YU S, CHEN X, LIU Y, et al. Exosomes derived from stem cells from the apical papilla alleviate inflammation in rat pulpitis by upregulating regulatory T cells. Int Endod J. 2022;55(5):517-530. [31] 蓝彬园,林熹,陈文瑨,等.脂多糖刺激人牙髓干细胞分泌的外泌体联合基质细胞衍生因子-1对牙髓再生的影响[J].中华口腔医学杂志,2022,57(1):60-67. [32] JAFARI N, SEYED HABASHI M, AFSHAR A, et al. The Effect of Exosomes Derived from Human Umbilical Cord Mesenchymal Stromal/Stem Cells on the Regeneration of Human Pulpectomized Tooth: A Case Report. Iran J Med Sci. 2025;50(1):54-58. [33] 刘屿,曾莲,王卫红,等.人骨髓间充质干细胞外泌体来源的miR-335-5p促进人牙周膜干细胞的成骨分化:基于下调DKK1表达[J].南方医科大学学报,2023,43(3):420-427. [34] CUI Y, HONG S, XIA Y, et al. Melatonin Engineering M2 Macrophage-Derived Exosomes Mediate Endoplasmic Reticulum Stress and Immune Reprogramming for Periodontitis Therapy. Adv Sci (Weinh). 2023;10(27):e2302029. [35] HU Y, WANG Z, FAN C, et al. Human gingival mesenchymal stem cell-derived exosomes cross-regulate the Wnt/β-catenin and NF-κB signalling pathways in the periodontal inflammation microenvironment. J Clin Periodontol. 2023;50(6): 796-806. [36] GAO J, WU Z. M2 macrophage-derived exosomes enable osteogenic differentiation and inhibit inflammation in human periodontal ligament stem cells through promotion of CXCL12 expression. BMC Oral Health. 2024;24(1):1070. [37] HUANG X, DENG Y, XIAO J, et al. Genetically engineered M2-like macrophage-derived exosomes for P. gingivalis-suppressed cementum regeneration: From mechanism to therapy. Bioact Mater. 2023;32:473-487. [38] ELSAYED R, ELASHIRY M, TRAN C, et al. Engineered Human Dendritic Cell Exosomes as Effective Delivery System for Immune Modulation. Int J Mol Sci. 2023;24(14):11306. [39] LU J, YU N, LIU Q, et al. Periodontal Ligament Stem Cell Exosomes Key to Regulate Periodontal Regeneration by miR-31-5p in Mice Model. Int J Nanomedicine. 2023;18:5327-5342. [40] LIU Y, ZHU J, WANG WH, et al. Exosomal lncRNA HCP5 derived from human bone marrow mesenchymal stem cells improves chronic periodontitis by miR-24-3p/HO1/P38/ELK1 pathway. Heliyon. 2024;10(14):e34203. [41] LI X, LIU X, ZHOU J, et al. Human dental follicle stem cell-derived exosomes reduce root resorption by inhibiting periodontal ligament cell pyroptosis. Stem Cell Res Ther. 2025;16(1):79. [42] CUI Y, LI X, HE X, et al. Schwann cell-derived exosomes accelerate periodontal bone regeneration with osteogenesis, angiogenesis, and neurogenesis. J Mater Chem B. 2025;13(12): 4020-4029. [43] YU T, MI N, SONG Y, et al. Exosomes miR-92a-3p from human exfoliated deciduous teeth inhibits periodontitis progression via the KLF4/PI3K/AKT pathway. J Periodontal Res. 2024;59(4):771-782. [44] PULETIC M, VELIKIC G, MARIC DM, et al. Clinical Efficacy of Extracellular Vesicle Therapy in Periodontitis: Reduced Inflammation and Enhanced Regeneration. Int J Mol Sci. 2024; 25(11):5753. [45] WANG L, YANG L, TIAN L, et al. Exosome-capturing scaffold promotes endogenous bone regeneration through neutrophil-derived exosomes by enhancing fast vascularization. Biomaterials. 2025;319:123215. [46] ZHENG Y, WANG X, HE Y, et al. Exosomes from Adipose-Derived Mesenchymal Stromal Cells Prevent Medication-Related Osteonecrosis of the Jaw by Inhibiting Macrophage M1 Polarization and Pyroptosis. Int J Nanomedicine. 2024;19:12675-12693. [47] ALBOUGHA MS, SUGII H, ADACHI O, et al. Exosomes from Human Periodontal Ligament Stem Cells Promote Differentiation of Osteoblast-like Cells and Bone Healing in Rat Calvarial Bone. Biomolecules. 2024;14(11):1455. [48] HE X, LIU Y, DAI Z, et al. Yoda1 pretreated BMSC derived exosomes accelerate osteogenesis by activating phospho-ErK signaling via Yoda1-mediated signal transmission. J Nanobiotechnology. 2024;22(1):407. [49] LIU L, ZHOU N, FU S, et al. Endothelial cell-derived exosomes trigger a positive feedback loop in osteogenesis-angiogenesis coupling via up-regulating zinc finger and BTB domain containing 16 in bone marrow mesenchymal stem cell. J Nanobiotechnology. 2024;22(1):721. [50] MA S, LI Y, YAO S, et al. A deformable SIS/HA composite hydrogel coaxial scaffold promotes alveolar bone regeneration after tooth extraction. Bioact Mater. 2024;46:97-117. [51] YUAN W, LIU J, ZHANG Z, et al. Strontium-Alix interaction enhances exosomal miRNA selectively loading in synovial MSCs for temporomandibular joint osteoarthritis treatment. Int J Oral Sci. 2025; 17(1):6. [52] HU S, CHEN B, ZHOU J, et al. Dental pulp stem cell-derived exosomes revitalize salivary gland epithelial cell function in NOD mice via the GPER-mediated cAMP/PKA/CREB signaling pathway. J Transl Med. 2023;21(1):361. [53] XIAO XY, ZHANG NN, LONG YZ, et al. Repair mechanism of radiation-induced salivary gland injury by hypoxia-pretreated human urine-derived stem cell exosomes. Oral Dis. 2024;30(3):1234-1241. [54] DU ZH, CHU WX, PENG X, et al. SHED-Derived Exosomes Ameliorate Sjögren’s Syndrome-Induced Hyposalivation by Suppressing Th1 Cell Response via the miR-29a-3p/T-bet Axis. ACS Appl Mater Interfaces. 2025;17(4): 5752-5761. [55] DU Z, WEI P, JIANG N, et al. SHED-derived exosomes ameliorate hyposalivation caused by Sjögren’s syndrome via Akt/GSK-3β/Slug-mediated ZO-1 expression. Chin Med J (Engl). 2023;136(21):2596-2608. [56] CHU WX, DING C, DU ZH, et al. SHED-exos promote saliva secretion by suppressing p-ERK1/2-mediated apoptosis in glandular cells. Oral Dis. 2024;30(5):3066-3080. [57] CHEN Z, MAO QY, ZHANG JY, et al. Cellular Senescence Contributes to the Dysfunction of Tight Junctions in Submandibular Glands of Aging Mice. Aging Cell. 2025;24(5):e14470. [58] GUO X, HUANG Z, WU F, et al. Exosomes of human adipose stem cells mitigate irradiation injury to salivary glands by inhibiting epithelial-mesenchymal transition through miR-199a-3p targeting Twist1 and regulating TGFβ1/Smad3 pathway. Theranostics. 2025;15(5):1622-1641. [59] MA D, WU Z, ZHAO X, et al. Immunomodulatory effects of umbilical mesenchymal stem cell-derived exosomes on CD4 T cells in patients with primary Sjögren’s syndrome. Inflammopharmacology. 2023;31(4):1823-1838. [60] ZAKARIA A, SULTAN N, NABIL N, et al. Exosomes derived from bone marrow mesenchymal stem cells ameliorate chemotherapeutically induced damage in rats’ parotid salivary gland. Oral Maxillofac Surg. 2025;29(1):39. [61] YU Z, WEN Y, JIANG N, et al. TNF-α stimulation enhances the neuroprotective effects of gingival MSCs derived exosomes in retinal ischemia-reperfusion injury via the MEG3/miR-21a-5p axis. Biomaterials. 2022;284:121484. [62] LIANG X, MIAO Y, TONG X, et al. Dental pulp mesenchymal stem cell-derived exosomes inhibit neuroinflammation and microglial pyroptosis in subarachnoid hemorrhage via the miRNA-197-3p/FOXO3 axis. J Nanobiotechnology. 2024;22(1):426. [63] GUO R, FANG Y, ZHANG Y, et al. SHED-derived exosomes attenuate trigeminal neuralgia after CCI of the infraorbital nerve in mice via the miR-24-3p/IL-1R1/p-p38 MAPK pathway. J Nanobiotechnology. 2023;21(1):458. [64] YU F, WANG Y, HUANG CQ, et al. Neuroprotective effect of mesenchymal stem cell-derived extracellular vesicles on optic nerve injury in chronic ocular hypertension. Neural Regen Res. 2023;18(10):2301-2306. [65] XUE R, XIE M, WU Z, et al. Mesenchymal Stem Cell-Derived Exosomes Promote Recovery of The Facial Nerve Injury through Regulating Macrophage M1 and M2 Polarization by Targeting the P38 MAPK/NF-Κb Pathway. Aging Dis. 2024;15(2):851-868. [66] WANG H, ZHAO H, CHEN Z, et al. Hypoxic Bone Mesenchymal Stem Cell-Derived Exosomes Direct Schwann Cells Proliferation, Migration, and Paracrine to Accelerate Facial Nerve Regeneration via circRNA_Nkd2/miR-214-3p/MED19 Axis. Int J Nanomedicine. 2024; 19:1409-1429. [67] SANG X, TANG L, ZHAO L, et al. Umbilical cord mesenchymal stem cell-derived exosomes promote axon regeneration during optic nerve injury through microRNA-dependent mTORC1 signalling. Clin Transl Med. 2023;13(7):e1319. [68] SALEM EM, RIZK H, ABOUELELA YS, et al. Regenerative potentials of bone marrow mesenchymal stem cells derived exosomes or its combination with zinc in recovery of degenerated circumvallate papilla following surgical bilateral transection of glossopharyngeal nerve in rats. BMC Oral Health. 2024;24(1):1320. [69] XU C, ZHANG H, YANG C, et al. miR-125b-5p delivered by adipose-derived stem cell exosomes alleviates hypertrophic scarring by suppressing Smad2. Burns Trauma. 2024;12:tkad064. [70] ZHAO S, KONG H, QI D, et al. Epidermal stem cell derived exosomes-induced dedifferentiation of myofibroblasts inhibits scarring via the miR-203a-3p/PIK3CA axis. J Nanobiotechnology. 2025; 23(1):56. [71] SHANG Y, LI M, ZHANG L, et al. Exosomes derived from mouse vibrissa dermal papilla cells promote hair follicle regeneration during wound healing by activating Wnt/β-catenin signaling pathway. J Nanobiotechnology. 2024;22(1):425. [72] CUI H, FU LQ, TENG Y, et al. Human Hair Follicle Mesenchymal Stem Cell-Derived Exosomes Attenuate UVB-Induced Photoaging via the miR-125b-5p/TGF-β1/Smad Axis. Biomater Res. 2025;29:0121. [73] DUTTA SD, AN JM, HEXIU J, et al. 3D bioprinting of engineered exosomes secreted from M2-polarized macrophages through immunomodulatory biomaterial promotes in vivo wound healing and angiogenesis. Bioact Mater. 2024;45:345-362. [74] ZHUO H, CHEN Y, ZHAO G. Advances in application of hypoxia-preconditioned mesenchymal stem cell-derived exosomes. Front Cell Dev Biol. 2024; 12:1446050. [75] ZUO JC, LIANG J, HU N, et al. Hypoxia preconditioned MSC exosomes attenuate high-altitude cerebral edema via the miR-125a-5p/RTEF-1 axis to protect vascular endothelial cells. Bioact Mater. 2025;52:541-563. [76] LI Q, FU X, KOU Y, et al. Engineering strategies and optimized delivery of exosomes for theranostic application in nerve tissue. Theranostics. 2023; 13(12):4266-4286. [77] WU J, WU J, XIANG W, et al. Engineering exosomes derived from TNF-α preconditioned IPFP-MSCs enhance both yield and therapeutic efficacy for osteoarthritis. J Nanobiotechnology. 2024;22(1):555. [78] ZHOU Z, WANG R, WANG J, et al. Melatonin pretreatment on exosomes: Heterogeneity, therapeutic effects, and usage. Front Immunol. 2022;13:933736. [79] CHEN H, SUN H, YANG Y, et al. Engineered melatonin-pretreated plasma exosomes repair traumatic spinal cord injury by regulating miR-138-5p/SOX4 axis mediated microglia polarization. J Orthop Translat. 2024;49:230-245. [80] CHEN J, CHEN J, CHEN J, et al. Pretreated exosomes by electrical stimulation accelerate bone regeneration. Bioact Mater. 2025;51:383-398. [81] WU D, ZHAO X, XIE J, et al. Physical modulation of mesenchymal stem cell exosomes: A new perspective for regenerative medicine. Cell Prolif. 2024;57(8):e13630. [82] WANG W, SUN H, DUAN H, et al. Isolation and usage of exosomes in central nervous system diseases. CNS Neurosci Ther. 2024; 30(3):e14677. [83] WANG C, QIU J, LIU M, et al. Microfluidic Biochips for Single-Cell Isolation and Single-Cell Analysis of Multiomics and Exosomes. Adv Sci (Weinh). 2024;11(28):e2401263. [84] ZHAO J, LV X, LU Q, et al. Engineered platelet-derived exosomal spheres for enhanced tumor penetration and extended circulation in melanoma immunotherapy. Acta Pharm Sin B. 2025;15(7):3756-3766. [85] LIANG Y, IQBAL Z, LU J, et al. Cell-derived nanovesicle-mediated drug delivery to the brain: Principles and strategies for vesicle engineering. Mol Ther. 2023;31(5):1207-1224. [86] LIU JJJ, LIU D, TO SKY, et al. Exosomes in cancer nanomedicine: biotechnological advancements and innovations. Mol Cancer. 2025;24(1):166. [87] ZHANG YX, WANG M, XU LL, et al. An integrated microfluidic chip for synchronous drug loading, separation and detection of plasma exosomes. Lab Chip. 2025;25(13):3185-3196. [88] SON G, SONG J, PARK JC, et al. Fusogenic lipid nanoparticles for rapid delivery of large therapeutic molecules to exosomes. Nat Commun. 2025;16(1):4799. [89] DENG D, LI X, ZHANG JJ, et al. Biotin-Avidin System-Based Delivery Enhances the Therapeutic Performance of MSC-Derived Exosomes. ACS Nano. 2023;17(9):8530-8550. [90] ZHENG J, HE J, WU J, et al. Polyphenol-Mediated Electroactive Hydrogel with Armored Exosomes Delivery for Bone Regeneration. ACS Nano. 2025; 19(18):17796-17812. [91] TAN F, LI X, WANG Z, et al. Clinical applications of stem cell-derived exosomes. Signal Transduct Target Ther. 2024;9(1):17. [92] CALLAWAY E. Chemistry Nobel goes to developers of AlphaFold AI that predicts protein structures. Nature. 2024;634(8034):525-526. [93] GUO SB, MENG Y, LIN L, et al. Artificial intelligence alphafold model for molecular biology and drug discovery: a machine-learning-driven informatics investigation. Mol Cancer. 2024;23(1):223. [94] KRUSE T, GARVANSKA DH, VARGA JK, et al. Substrate recognition principles for the PP2A-B55 protein phosphatase. Sci Adv. 2024; 10(40):eadp5491. |
| [1] | Rao Jingcheng, Li Yuwan, Zheng Hongbing, Xu Zhi, Zhu Aixiang, Shi Ce, Wang Bing, Yang Chun, Kong Xiangru, Zhu Dawei. Biomechanical differences between the new proximal femoral stable intramedullary nail and traditional intramedullary nail#br# [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2217-2225. |
| [2] | Zhao Feifan, Cao Yujing. Risk factors and coping strategies of internal fixation failure in treatment of intertrochanteric fracture with proximal femoral nail antirotation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2323-2333. |
| [3] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [4] | Min Changqin, Huang Ying. Construction of pH/near-infrared laser stimuli-responsive drug delivery system and its application in treatment of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1940-1951. |
| [5] | Xu Chang, Jiang Mingzhu, Liu Xin, Yan Weijun. Three-dimensional finite element analysis of implant anchorage combined with torque auxiliary arch to lower anterior teeth [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1971-1978. |
| [6] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [7] | Han Teng, Ma Hong, Yang Ruoyi, Luo Yi, Li Chao. Oral squamous cell carcinoma-derived exosomal delivery of angiopoietin-2 is involved in tumor angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1755-1767. |
| [8] | Huang Jiawen, Pan Zhiyi, Xue Wenjun, Lian Yuanpei, Xu Jianda. Plant-derived vesicles and malignant tumor therapy: cross-species communication and modulation of host cell responses [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1828-1838. |
| [9] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [10] | Fan Yongjing, Jin Wulong, Bai Haoyu, Ma Ping, Wang Shu. Role and mechanism of stem cells from human exfoliated deciduous teeth in tissue regeneration and disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1850-1857. |
| [11] | Wang Zhenze, Liu Fende, Zhang Rui, Li Wujun. Mesenchymal stem cells in treatment of arteriosclerosis obliterans of lower extremities: systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1869-1876. |
| [12] | Song Puzhen, Ma Hebin, Chen Hongguang, Zhang Yadong. Effect of bone marrow mesenchymal stem cell-derived exosomes combined with transforming growth factor beta 1 on macrophages [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1616-1623. |
| [13] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [14] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [15] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||