Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (16): 2619-2624.doi: 10.12307/2024.337
Transforming growth factor beta/Smad signaling pathway and targeted therapy of keloid scars
Yao Siqi, Li Wenzheng, Wang Hong
- Department of Burn Surgery, The Second Affiliated Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China
-
Received:2023-04-09Accepted:2023-06-05Online:2024-06-08Published:2023-07-31 -
Contact:Wang Hong, Master, Chief physician, Master’s supervisor, Department of Burn Surgery, The Second Affiliated Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China -
About author:Yao Siqi, Master candidate, Department of Burn Surgery, The Second Affiliated Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China -
Supported by:Yunnan Provincial High-level Talent Training Support Program, No. YNWR-MY-2020-047 (to WH); the Postgraduate Innovation Fund of Kunming Medical University, No. 2023S326 (to YSQ)
CLC Number:
Cite this article
Yao Siqi, Li Wenzheng, Wang Hong. Transforming growth factor beta/Smad signaling pathway and targeted therapy of keloid scars[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2619-2624.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
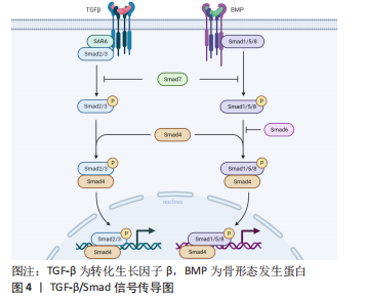
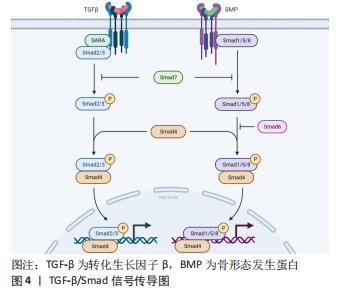
2.1 瘢痕疙瘩成纤维细胞与TGF-β/Smad信号通路 皮肤受伤后,机体便开始生理修复过程。成纤维细胞被激活并增殖,迁移到伤口中合成细胞外基质组成的修复支架。瘢痕疙瘩是皮肤创伤后深度常累及网状真皮层皮肤内结缔组织异常增生所致的病理性瘢痕,它是由成纤维细胞增殖过多、细胞外基质过度产生、生长因子过度表达所致的Ⅰ型、Ⅲ型胶原纤维异常无序增生导致的,使得瘢痕组织硬度增加,而延展性降低。并且长期的炎症浸润导致成纤维细胞活性增加,白细胞介素6是一种多效性促炎细胞因子,常在瘢痕疙瘩中强效作用[7],抑制基质金属蛋白酶的表达,靶向诱导成纤维细胞合成胶原蛋白。相较于正常细胞,瘢痕疙瘩成纤维细胞消耗更多的葡萄糖,降低基础氧化呼吸,具有更强的糖酵解能力,产生更多的乳酸堆积[8]。 成纤维细胞是瘢痕疙瘩中的主要效应细胞,承担着构建和重塑细胞外基质的功能,瘢痕疙瘩成纤维细胞中TGF-β显著增高。TGF-β有3 种亚型(TGF-β1,TGF-β2,TGF-β3),可由受损组织、成纤维细胞和M2巨噬细胞合成和释放,是一种在伤口愈合过程中普遍表达的细胞因子,更是一种具有代表性的促纤维化细胞因子,在调节炎症、伤口愈合和组织重塑中起着至关重要的作用,尤其与瘢痕疙瘩的形成密切相关。TGF-β超家族信号传导参与调节许多细胞的发生发展过程,包括细胞凋亡、炎症、纤维化等等。Ⅰ型受体(TβRⅠ)和Ⅱ型受体(TβRⅡ)由两对跨膜丝氨酸/苏氨酸蛋白激酶(serine/threonine protein kinase,RIPK)构成,形成TGF-β超家族的受体。此外,已有越来越多的Ⅲ型受体(TβRⅢ)被发现,可作为共受体与TGF-β结合并调节TGF-β信号[9]。 TGF-β的下游通路包括Smad信号通路和非Smad信号通路,非Smad信号通路包括丝裂原活化蛋白激酶(mitogen-activated protein kinases,MAPK)通路中的ERK、JNK和p38信号通路等。这些通路的激活引发后续一系列反应并形成自分泌正反馈回路进一步刺激TGF-β1的释放,导致细胞外基质过度产生最终发生纤维化。TGF-β诱导的Smad信号通路是一种有效形成瘢痕的修复系统,对人体伤口修复具有重要意义,其在伤口愈合过程中介导成纤维细胞增殖、血管生成、细胞外基质合成和再上皮化,然而瘢痕过度形成时就会导致不同程度的功能损害。TGF-β/Smad信号通路作为调节成纤维细胞中胶原蛋白形成的最典型信号通路,在瘢痕疙瘩成纤维细胞的活化中起着关键作用。Smad家族蛋白分为3 个功能类别,分别是受体调节Smad(R-Smads)、共同伴侣Smad(Co-Smads)和抑制Smad(I-Smads)。R-Smads包括Smad1、Smad2、Smad3、Smad5、Smad8,Co-Smads仅有Smad4,I-Smads包括Smad6和Smad7。 2.2 TGF-β/Smad信号通路影响瘢痕疙瘩的机制 瘢痕疙瘩成纤维细胞和损伤组织可大量分泌各TGF-β,因此TGF-β在病理性瘢痕纤维化中起着关键作用,其中,TGF-β1和TGF-β2呈现过表达状态,而TGF-β3具有抗纤维化作用。TGF-β1和TGF-β2增加了成纤维细胞的增殖、分化和基质产生,使得Ⅰ型、Ⅲ型胶原蛋白生成增多[10],并且防止其降解,在异常皮肤伤口愈合过程中导致皮肤纤维化[11],特别是TGF-β1通过转录调节各种纤维化相关蛋白,刺激增生性瘢痕中的血管生成和扩张。相较于增生性瘢痕,萎缩性瘢痕则表现出了相反的表型,这是TGF-β1的功能丧失或活性减弱导致[12]。而TGF-β3则减少结缔组织的沉积,在瘢痕疙瘩成纤维细胞中mRNA表达降低。 瘢痕疙瘩患者循环血中血管内皮生长因子升高,很可能导致血管生成不平衡以及瘢痕疙瘩的形成。血管内皮生长因子作为调节伤口愈合中血管生长的关键因子,TGF-β/Smad通路的激活可增加其表达,进而导致伤口愈合异常[13],血管内皮生长因子又被认为协助胶原蛋白的过度产生,因此抑制TGF-β信号传导可作为同时抑制瘢痕疙瘩中肌纤维和血管生成的靶标[14]。 2.2.1 受体调节性Smad 由于创伤、手术等原因导致皮肤受到损伤,蛋白质的错误折叠逐渐累积,参与纤维化的主要机制TGF-β和p-Smad2/3通路被放大,使得细胞外基质沉积导致纤维化。其机制是TGF-β与TβRⅡ结合使其磷酸化,再激活TβRⅠ,TβRⅠ激活Smad通路使得Smad2/3磷酸化,蛋白水平p-Smad2/3表达增多,Smad4与磷酸化的Smad2/3结合形成Smad异源寡聚体复合物即Smad2/3/4复合物并异位到细胞核中借助多种转录因子共同转运下游促纤维蛋白的表达,在调控基因转录中起重要作用,从而引发多种生理效应。非Smad途径中的MAPK通路也参与TGF-β转录,其中p38参与Smad2/3的磷酸化,ERK和JNK则促进Smad2/3/4复合物易位[15]。骨形态发生蛋白(BMPs)属于TGF-β超家族,通过与TβRⅡ结合并活化TβRⅠ,TβRⅠ磷酸化R-Smad(Smad1/5/8),该条通路同样与Smad4结合形成复合物进入细胞核发挥作用。 2.2.2 抑制性Smad 瘢痕疙瘩成纤维细胞被认为是瘢痕疙瘩形成的关键细胞,所以寻找抑制瘢痕疙瘩成纤维细胞增殖的分子靶点或许可以抑制瘢痕疙瘩的形成。正常瘢痕组织中抑制性Smad6和Smad7的表达增加可以抑制TGF-β信号,从而阻止效应Smads的募集与磷酸化,导致正常瘢痕瘢痕疙瘩成纤维细胞增殖和胶原沉积减少[16-17]。Smad6通过与Smad1竞争和Smad4的结合起到负反馈调节作用。相较于Smad6,Smad7对TGF-β信号通路更具有特异性,其作为一种负反馈调节因子,可以与TβRⅠ结合竞争性抑制Smad2/3以及Smad1/5/8的磷酸化[18-19],阻断TGF-β/Smad信号的激活,抑制瘢痕疙瘩成纤维细胞增殖,减少细胞外胶原沉积。 TGF-β/Smad信号传导图见图4。"

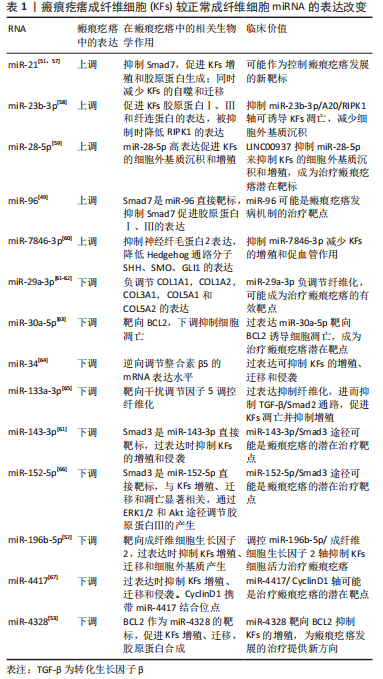
2.3 针对TGF-β/Smad信号通路的治疗措施 瘢痕疙瘩的产生通常是一个漫长的过程,往往到出现了临床症状才会引起患者的关注以及寻求治疗。虽然很难控制瘢痕疙瘩的全身性和遗传性危险因素,但还有较多的局部治疗措施,包括手术切除、病灶内注射类固醇、放射治疗、激光治疗等等,这些疗法虽都有较高的成功率,然而也会带来不可避免的不良反应和一定的复发率,并且由于随访护理的不足,治疗终点的不明确,对于治愈瘢痕疙瘩的金标准治疗方式仍缺乏共识。因此,迫切寻求早期预防瘢痕疙瘩产生以及治愈率更高的治疗方式,TGF-β/Smad信号通路的持续激活导致成纤维细胞的过度激活,这是瘢痕疙瘩中胶原过度形成的必要条件,当前许多治疗策略都集中在针对TGF-β/Smad信号通路来减轻瘢痕疙瘩的发生和发展,已有研究认为可根据血浆中的TGF-β水平作为儿童烧伤后诱发增生性瘢痕的预测因素[12]。 2.3.1 抑制TGF-β1 TGF-β1被认为是最活跃的纤维化细胞因子之一,已被证明可以通过诱导下游Smad信号通路来刺激成纤维细胞活化、增殖、抵抗凋亡,并增加金属蛋白酶组织抑制剂并减少基质金属蛋白酶的产生,从而减少胶原蛋白的降解[20],并且随着受体的上调,瘢痕疙瘩成纤维细胞对TGF-β1的作用也愈发敏感,因此作者推测衰减TGF-β1的活性能在抑制瘢痕疙瘩形成方面具有潜在优势。 当骨髓来源间充质干细胞与成纤维细胞共培养时,证明骨髓间充质干细胞分泌的因子可以阻碍TGF-β1诱导的成纤维细胞增殖和TGF-β/Smad途径中涉及的关键分子表达[21],自体脂肪移植也可通过抑制TGF-β1的表达,上调TGF-β3水平来抑制成纤维细胞的增殖[22]。KIM等[23]已经证实牛磺熊去氧胆酸通过TGF-β1/Smad信号传导以减少瘢痕疙瘩的治疗效果。乔松素[24]、氟哌酮[10]、TGF-β受体抑制剂LY2109761等也可以以剂量依赖的方式抑制TGF-β信号传导[25],降低Smad2、Smad3的磷酸化来抑制成纤维细胞活化。已被医疗美容广泛应用的A型肉毒毒素,也可使成纤维细胞在细胞周期的非增殖状态下静止,减少愈合伤口的边缘张力,从而降低TGF-β1及血管内皮生长因子基因的表达[26],上调Smad7基因和蛋白的表达[27],达到治疗瘢痕疙瘩的目的[28]。CR6相互作用因子1(CRIF1)对细胞生长和凋亡都具有调节作用,有研究发现瘢痕疙瘩中CR6相互作用因子1缺乏抑制了细胞增殖、迁移和胶原蛋白产生的能力,通过TGF-β/Smad途径抑制瘢痕疙瘩的过度生长[29],因此下调CR6相互作用因子1可能是治疗瘢痕疙瘩的潜在靶点。C1q/TNF相关蛋白3(CTRP3)作为分泌蛋白家族的一员,其过表达被证实显著控制TGF-β1诱导的瘢痕疙瘩成纤维细胞增殖和迁移,通过阻止TGF-β1刺激的Smad2和Smad核易位[30]。紫杉醇已被临床用于治疗癌症、诱导癌细胞凋亡,因此有研究将紫杉醇和胆固醇形成负载脂质体应用于瘢痕疙瘩的治疗,发现其也可以通过阻断TGF-β/Smad信号而发挥抗纤维化功能[31]。这些治疗方式的发现都表明了抑制TGF-β1对于抑制瘢痕疙瘩的发生和发展有着至关重要的作用。 2.3.2 抑制Smad4 上文提到Smad4与磷酸化的Smad2/3和Smad1/5/8结合形成复合物并转移到细胞核中共同转运下游促纤维蛋白的表达,那么如果共同伴侣Smad4被沉默时,该条通路是否能被有效抑制? 缺氧诱导因子(hypoxia inducible factor,HIF)1α是介导缺氧应激反应并调节缺氧细胞和生物学行为的核心因子。在缺氧环境下,HIF-1α不会通过pvhl介导的泛素-蛋白酶体途径立即降解,而是与HIF-1β一起形成HIF-1来激活TGF-β1/Smad信号传导通路,促进成纤维细胞中的胶原蛋白沉积[3]。 研究发现,当沉默Smad4时,HIF-1α的mRNA和蛋白质水平降低[32],这是否可以推测出HIF-1α启动子上可能存在Smad2/3/4复合物的结合位点,使得HIF-1α和Smad4存在正反馈环。若能找出该结合位点,就能靶向使用拮抗剂抑制TGF-β/Smad信号传导通路的过度激活并最终控制瘢痕疙瘩的发展。Sumoylation机制通过放大HIF-1和TGF-β/Smad信号传导通路正向调节瘢痕疙瘩的形成,SUMO1是一种类泛素修饰物,是影响HIF-1和TGF-β/Smad信号通路的关键分子,HIF-1α在瘢痕疙瘩成纤维细胞中被SUMO1修饰[33]。当SUMO特异性蛋白酶(SENP)通过去苏酰基化去除底物中的SUMO1后,游离的SUMO1增加,特别是SUMO1-RanGAP1复合物,激活HIF-1和TGF-β/Smad信号通路,胶原蛋白Ⅲ表达明显增加。 2.3.3 上调Smad7 Smad7为抑制性Smad,因此Smad7的过表达可能会减弱TGF-β1与R-Smads的磷酸化。许多RNA参与调节瘢痕疙瘩成纤维细胞增殖和迁移来介导瘢痕疙瘩的形成,miRNA-21参与多种组织纤维化过程,而Smad7是其直接靶基因。TGF-β/Smad通路激活后使miRNA-21上调,抑制Smad7,保护Smad2/3,而Smad7水平下降又正反馈作用于TGF-β/Smad通路,促进瘢痕疙瘩瘢痕疙瘩成纤维细胞胶原产生[34]。瘢痕疙瘩中miR-96表达上调,Smad7也是其直接靶标。miRNA-21-5p促进瘢痕疙瘩角质形成细胞的迁移和侵袭,通过AKT信号诱导瘢痕疙瘩角质形成细胞的上皮-间充质转化[35]。LIU 等[36]发现沉默circPTPN12下调Smad7以通过激活靶向miRNA-21-5p的Wnt通路促进瘢痕疙瘩成纤维细胞的生长,表明circPTPN12对于瘢痕疙瘩的形成发挥了重要作用,而Smad7也可能成为miRNA-21-5p在瘢痕疙瘩中的潜在治疗靶点。 一些研究报道了某些miRNA具有抗纤维化的能力,如过表达miRNA-196b-5p可抑制瘢痕疙瘩成纤维细胞的活力、迁移和细胞外基质组分表达[35],有研究发现lnCRNA H19还可以通过调节miRNA-196b-5p/Smad5轴和靶向miRNA-769-5p/EIF3A通路促进瘢痕疙瘩成纤维细胞的增殖、迁移、侵袭、细胞外基质沉积[37-38],抑制细胞凋亡,促进瘢痕疙瘩形成,还可通过修饰下游为miR-29a和Ⅰ型胶原α1链促进成纤维细胞的增殖和转移[39],这些关于lnCRNA H19的发现对瘢痕疙瘩的调控提供了新思路。 2.3.4 减少细胞外基质的分泌与沉积 TGF-β/Smad信号转导已成为皮肤瘢痕疙瘩的关键治疗靶点,TGF-β1刺激胶原蛋白合成似乎起到了核心作用。基质金属蛋白酶和金属蛋白酶组织抑制剂分别可以增加和减少细胞外基质的降解,它们之间的不平衡是瘢痕疙瘩细胞外基质生成异常的主要原因。TGF-β1/Smad通路激活使得细胞外基质中Ⅰ型、Ⅲ型胶原生成增多,加速伤口修复,然而胶原的持续过度表达最终导致瘢痕疙瘩的发生。 在胎儿时期,Ⅲ型胶原的表达水平更高,而在成人伤口愈合过程中,则是Ⅰ型胶原占主导地位[40],而无瘢痕愈合需要高水平的基质金属蛋白酶/金属蛋白酶组织抑制剂以及Ⅲ型胶原/Ⅰ型胶原。由于Ⅰ型胶原在瘢痕中占主导地位,当外源性补充重组人源Ⅲ型胶原蛋白功能凝胶时可提高创面局部Ⅲ型胶原蛋白水平,进而抑制Ⅰ型胶原蛋白水平过度分泌,达到抑制瘢痕增生的作用[41],为临床治疗瘢痕疙瘩提供了一个重要的新方法。 用于瘢痕疙瘩治疗的激光疗法于1980年代引入,目前临床应用较多的点阵CO2激光就是通过抑制过度的胶原增生,促进胶原降解[42]、胶原纤维重新排列来治疗瘢痕疙瘩[43]。同时光敏剂Ph1[44]、氨基酮戊酸联合激光治疗也被证实通过抑制瘢痕疙瘩中的胶原来抑制瘢痕的发展,且20%氨基酮戊酸联合激光治疗抑制瘢痕效果最佳[45]。但这些方式也存在着灼伤皮肤、感染、瘙痒等不良反应,以及形成新生瘢痕的风险,因此还需要进一步研究治疗的方式、剂量、间隔时间以及疗程。 2.3.5 外泌体 外泌体(Exosomes)存在着大量遗传信息和丰富的RNA种类,具有细胞间信使的功能,在特定细胞之间传递它们的效应物或信号分子。相较于细胞,外泌体免疫原性低,与免疫系统具有更好的生物相容性,因此有着更广阔的临床治疗前景。 脂肪来源的间充质干细胞(adipoes derived mesenchymal stem cells,ADSCS)因其多能性、自我更新和促进再生细胞因子分泌的能力而成为关注的焦点[46],且相较于其他干细胞具有更大的体外增殖能力[47],已被证实可以促进伤口愈合并抑制瘢痕疙瘩成纤维细胞胶原蛋白的合成[43],脂肪间充质干细胞具有强大的抗氧化作用,可增加血管内皮细胞增殖,促进新生毛细血管生成,分泌抗纤维化细胞因子和生长因子,还能使TGF-β1表达降低。而脂肪间充质干细胞的外泌体(ADSCs-Exos)因其保留了源细胞的功能,早期应用亦可以促进伤口修复和再生,改善了Ⅰ/Ⅲ型胶原蛋白、TGF-β1表达水平、成纤维细胞的活性和迁移性升高的状态[48],协同促进细胞外基质的重建,减少瘢痕的形成。 miRNA在再生医学中具有显著作用,可以在各种过程中调节多种组织的生长,其中自然包括皮肤的创面愈合。大多数在循环中发挥作用的miRNA以外泌体为载体,外泌体可通过递送miRNA进入靶细胞来影响细胞外基质相关基因的mRNA的翻译过程,最终减少病理性瘢痕乃至瘢痕疙瘩的形成。某些关键miRNA,如miR-21,miR-181c,miR-96[49],miR-7704已被报道参与纤维化和细胞外基质代谢[50],参与介导增生性瘢痕中的胶原沉积瘢痕疙瘩成纤维细胞分泌的外泌体中miRNA21明显高于正常组织,抑制其表达后Smad7蛋白表达发生上调,并且下调了Ⅰ型胶原蛋白和Ⅲ型胶原蛋白的表达,增加了凋亡细胞的比例以及减少细胞增殖[51]。 相反的,也有一些miRNA起到了抗纤维化的作用,如miR-29,miR-10a,miR-196b-5p[52],miR-4328等[53]。miRNA-29a 修饰的ADSCs-Exos通过靶向抑制TGF-β2/Smad3信号通路抑制瘢痕疙瘩成纤维细胞的增殖和迁移并减少过度瘢痕形成[39],还能有效抑制瘢痕疙瘩中Ⅰ型胶原蛋白、Ⅲ型胶原蛋白的基因和蛋白表达,通过降低TGF-β2的表达,促进TGF-β3的表达来抑制细胞外基质的产生[46],起到抗纤维化作用。瘢痕疙瘩患者has-circ-0020792水平异常上调[54],而外周血来源的外泌体较正常人miR-193a-5p水平较低,has-circ-0020792敲低可以上调miR-193a-5p来抑制瘢痕疙瘩成纤维细胞的活力并诱导其凋亡,抑制细胞外基质合成和纤维化的产生[55]。 外泌体可以调控炎症反应、血管生成、细胞外基质沉积和细胞凋亡[56],与此同时得益于较低的免疫原性,外泌体鲜少引起排异反应,那么能否经过基因测序等方式来验证正常个体和瘢痕个体之间瘢痕疙瘩成纤维细胞的外泌体差异,人为增加或抑制具有差异性的miRNA来减轻瘢痕疙瘩的发生和发展,通过外泌体靶向给药系统进行瘢痕疙瘩的治疗将会成为控制瘢痕疙瘩形成的一个新的研究方向。见表1。"

2.3.6 改善生活方式 瘢痕疙瘩在美容形象上的改变对患者生活质量的影响远高于其自觉症状所产生的影响,有评估调查显示瘢痕疙瘩的颜色、硬度、厚度、皮损部位都与生活质量指数评分呈正相关,而与疼痛、瘙痒之间无明显相关性[68]。瘢痕疙瘩作为一种身心疾病,患者的心理健康状况受疾病因素和社会环境因素的共同影响[69],因此除了对瘢痕疙瘩的治疗之外,还应尽早进行心理干预,缓解患者因此产生的焦虑抑郁情绪,疏导其带来的心理问题,优质护理来提高患者的生活质量。阿魏酸钠具有抗纤维化作用,主要用于心脑血管疾病。有一最新研究发现,阿魏酸钠通过抑制TGF-β/Smad信号通路上的关键蛋白TGF-β1、磷酸化Smad2/3、磷酸化Smad4等,同时促进Smad7的表达,从而发挥抑制细胞增殖、促进细胞凋亡的功能[70]。而改正不良的饮食习惯,增加膳食纤维的摄入可通过肠道微生物代谢释放阿魏酸[71]。口服益生菌已被证明可有效促进健康皮肤微生物群的组成,并且在临床的一些皮肤病治疗中取得了令人满意的疗效。因此有研究已经证实了是热链球菌在纤维化皮肤模型中抵消TGF-β1诱导的促纤维化作用[72]。这表明了良好的饮食习惯和生活方式等全身性因素也能对瘢痕疙瘩的改善起到作用。"

| [1] OGAWA R. Keloid and Hypertrophic Scars Are the Result of Chronic Inflammation in the Reticular Dermis. Int J Mol Sci. 2017;18(3):606. [2] KIM SW. Management of keloid scars: noninvasive and invasive treatments. Arch Plast Surg. 2021;48(2):149-157. [3] LEI R, LI J, LIU F, et al. HIF-1alpha promotes the keloid development through the activation of TGF-beta/Smad and TLR4/MyD88/NF-kappaB pathways. Cell Cycle. 2019;18(23):3239-3250. [4] STONE RC, CHEN V, BURGESS J, et al. Genomics of Human Fibrotic Diseases: Disordered Wound Healing Response. Int J Mol Sci. 2020;21(22): 8590. [5] LIU R, XIAO H, WANG R, et al. Risk factors associated with the progression from keloids to severe keloids. Chin Med J (Engl). 2022;135(7):828-836. [6] LU YY, TU HP, WU CH, et al. Risk of cancer development in patients with keloids. Sci Rep. 2021;11(1):9390. [7] HUGUIER V, GIOT JP, SIMONNEAU M, et al. Oncostatin M exerts a protective effect against excessive scarring by counteracting the inductive effect of TGFbeta1 on fibrosis markers. Sci Rep. 2019;9(1):2113. [8] LU YY, WU CH, HONG CH, et al. GLUT-1 Enhances Glycolysis, Oxidative Stress, and Fibroblast Proliferation in Keloid. Life (Basel). 2021;11(6):505. [9] ZHANG T, WANG XF, WANG ZC, et al. Current potential therapeutic strategies targeting the TGF-beta/Smad signaling pathway to attenuate keloid and hypertrophic scar formation. Biomed Pharmacother. 2020;129:110287. [10] MARTY P, CHATELAIN B, LIHOREAU T, et al. Halofuginone regulates keloid fibroblast fibrotic response to TGF-beta induction. Biomed Pharmacother. 2021; 135:111182. [11] KIM J, KIM B, KIM SM, et al. Hypoxia-Induced Epithelial-To-Mesenchymal Transition Mediates Fibroblast Abnormalities via ERK Activation in Cutaneous Wound Healing. Int J Mol Sci. 2019;20(10):2546. [12] ILIES RF, AIOANEI CS, CATANA A, et al. Involvement of COL5A2 and TGF-beta1 in pathological scarring. Exp Ther Med. 2021;22(4):1067. [13] WANG XM, LIU XM, WANG Y, et al. Activating transcription factor 3 (ATF3) regulates cell growth, apoptosis, invasion and collagen synthesis in keloid fibroblast through transforming growth factor beta (TGF-beta)/SMAD signaling pathway. Bioengineered. 2021;12(1):117-126. [14] LIU X, CHEN W, ZENG Q, et al. Single-Cell RNA-Sequencing Reveals Lineage-Specific Regulatory Changes of Fibroblasts and Vascular Endothelial Cells in Keloids. J Invest Dermatol. 2022;142(1):124-135 e11. [15] ZHOU BY, WANG WB, WU XL, et al. Nintedanib inhibits keloid fibroblast functions by blocking the phosphorylation of multiple kinases and enhancing receptor internalization. Acta Pharmacol Sin. 2020;41(9):1234-1245. [16] LI XY, WENG XJ, LI XJ, et al. TSG-6 Inhibits the Growth of Keloid Fibroblasts Via Mediating the TGF-beta1/Smad Signaling Pathway. J Invest Surg. 2021;34(9): 947-956. [17] DOLIVO D, WEATHERS P, DOMINKO T. Artemisinin and artemisinin derivatives as anti-fibrotic therapeutics. Acta Pharm Sin B. 2021;11(2):322-339. [18] CUI J, JIN S, JIN C, et al. Syndecan-1 regulates extracellular matrix expression in keloid fibroblasts via TGF-beta1/Smad and MAPK signaling pathways. Life Sci. 2020;254:117326. [19] LI SN, WU JF. TGF-beta/SMAD signaling regulation of mesenchymal stem cells in adipocyte commitment. Stem Cell Res Ther. 2020;11(1):41. [20] BETARBET U, BLALOCK TW. Keloids: A Review of Etiology, Prevention, and Treatment. J Clin Aesthet Dermatol. 2020;13(2):33-43. [21] XU JH, XU SQ, DING SL, et al. Bone marrow mesenchymal stem cells alleviate the formation of pathological scars in rats. Regen Ther. 2022;20:86-94. [22] 贾文斌,谭谦.病理性瘢痕的非手术治疗新进展[J].中国美容医学,2023, 32(1):183-189. [23] KIM S, LEE SE, YI S, et al. Tauroursodeoxycholic Acid Decreases Keloid Formation by Reducing Endoplasmic Reticulum Stress as Implicated in the Pathogenesis of Keloid. Int J Mol Sci. 2021;22(19):10765. [24] LI X, ZHAI Y, XI B, et al. Pinocembrin Ameliorates Skin Fibrosis via Inhibiting TGF-beta1 Signaling Pathway. Biomolecules. 2021;11(8):1240. [25] WANG X, GU C, SHANG F, et al. Inhibitory Effect of the LY2109761 on the Development of Human Keloid Fibroblasts. Anal Cell Pathol (Amst). 2021;2021: 8883427. [26] YANG S, LUO YJ, LUO C. Network Meta-Analysis of Different Clinical Commonly Used Drugs for the Treatment of Hypertrophic Scar and Keloid. Front Med (Lausanne). 2021; 8:691628. [27] 尚念胜,牛燕英. A型肉毒毒素对瘢痕疙瘩成纤维细胞TGF-β/Smad通路和ERK通路表达的影响[J].中国美容医学,2020,29(5):104-109. [28] 黄立军,王瑞肖,徐瑞,等. A型肉毒素治疗瘢痕疙瘩的疗效[J].安徽医学, 2023,44(2):162-165. [29] NAGAR H, KIM S, LEE I, et al. Downregulation of CR6-interacting factor 1 suppresses keloid fibroblast growth via the TGF-beta/Smad signaling pathway. Sci Rep. 2021;11(1):500. [30] HE L, ZHU C, DOU H, et al. Keloid Core Factor CTRP3 Overexpression Significantly Controlled TGF-beta1-Induced Propagation and Migration in Keloid Fibroblasts. Dis Markers. 2023;2023:9638322. [31] WANG M, CHEN L, HUANG W, et al. Improving the anti-keloid outcomes through liposomes loading paclitaxel-cholesterol complexes. Int J Nanomedicine. 2019; 14:1385-1400. [32] MINGYUAN X, QIANQIAN P, SHENGQUAN X, et al. Hypoxia-inducible factor-1alpha activates transforming growth factor-beta1/Smad signaling and increases collagen deposition in dermal fibroblasts. Oncotarget. 2018;9(3):3188-3197. [33] LIN X, WANG Y, JIANG Y, et al. Sumoylation enhances the activity of the TGF-beta/SMAD and HIF-1 signaling pathways in keloids. Life Sci. 2020;255:117859. [34] ZHOU R, WANG C, WEN C, et al. miR-21 promotes collagen production in keloid via Smad7. Burns. 2017;43(3):555-561. [35] STEVENSON AW, DENG Z, ALLAHHAM A, et al. The epigenetics of keloids. Exp Dermatol. 2021;30(8):1099-1114. [36] LIU F, LI T, ZHAN X. Silencing circular RNAPTPN12 promoted the growth of keloid fibroblasts by activating Wnt signaling pathway via targeting microRNA-21-5p. Bioengineered. 2022;13(2):3503-3515. [37] LI Z, GONG C, WEI H. Long non-coding RNA H19 aggravates keloid progression by upregulating SMAD family member 5 expression via miR-196b-5p. Bioengineered. 2022;13(1):1447-1458. [38] XU L, SUN N, LI G, et al. LncRNA H19 promotes keloid formation through targeting the miR-769-5p/EIF3A pathway. Mol Cell Biochem. 2021;476(3):1477-1487. [39] YUAN R, DAI X, LI Y, et al. Exosomes from miR-29a-modified adipose-derived mesenchymal stem cells reduce excessive scar formation by inhibiting TGF-beta2/Smad3 signaling. Mol Med Rep. 2021;24(5):758. [40] MORETTI L, STALFORT J, BARKER TH, et al. The interplay of fibroblasts, the extracellular matrix, and inflammation in scar formation. J Biol Chem. 2022; 298(2):101530. [41] 羊剑秋,高以红,朱红柳.重组人源Ⅲ型胶原蛋白功能凝胶对皮肤创口愈合的疗效及其机制[J].山东医药,2021,61(34):80-83. [42] 杨帅,蒋小姣,马海燕.超脉冲点阵CO2激光联合窄谱强脉冲光治疗创伤后早期增生性瘢痕效果分析[J].中国美容医学,2022,31(2):58-61. [43] OJEH N, BHARATHA A, GAUR U, et al. Keloids: Current and emerging therapies. Scars Burn Heal. 2020;6:2059513120940499. [44] ZHENG W, WU H, LI Y, et al. Phenalen-1-one-mediated photodynamic therapy inhibits keloid graft progression by reducing vessel formation and promoting fibroblast apoptosis. Adv Clin Exp Med. 2021;30(4):431-439. [45] 游传华,王一贺,陆雪飞.光动力对瘢痕疙瘩的抑制效果研究[J].中国医疗美容,2019,9(5):55-58. [46] LI J, LI Z, WANG S, et al. Exosomes from human adipose-derived mesenchymal stem cells inhibit production of extracellular matrix in keloid fibroblasts via downregulating transforming growth factor-beta2 and Notch-1 expression. Bioengineered. 2022;13(4):8515-8525. [47] BOJANIC C, TO K, HATOUM A, et al. Mesenchymal stem cell therapy in hypertrophic and keloid scars. Cell Tissue Res. 2021;383(3):915-930. [48] ZHOU C, ZHANG B, YANG Y, et al. Stem cell-derived exosomes: emerging therapeutic opportunities for wound healing. Stem Cell Res Ther. 2023;14(1):107. [49] CHAO L, HUA-YU Z, WEN-DONG B, et al. miR-96 promotes collagen deposition in keloids by targeting Smad7. Exp Ther Med. 2019;17(1):773-781. [50] SHEN Z, SHAO J, SUN J, et al. Exosomes released by melanocytes modulate fibroblasts to promote keloid formation: a pilot study. J Zhejiang Univ Sci B. 2022;23(8):699-704. [51] LI Q, FANG L, CHEN J, et al. Exosomal MicroRNA-21 Promotes Keloid Fibroblast Proliferation and Collagen Production by Inhibiting Smad7. J Burn Care Res. 2021;42(6):1266-1274. [52] YANG J, DENG P, QI Y, et al. NEAT1 Knockdown Inhibits Keloid Fibroblast Progression by miR-196b-5p/FGF2 Axis. J Surg Res. 2021;259:261-270. [53] TANG H, CHEN Q, YU W, et al. MiR-4328 inhibits proliferation, metastasis and induces apoptosis in keloid fibroblasts by targeting BCL2 expression. Open Life Sci. 2020;15(1):638-646. [54] SHI J, YAO S, CHEN P, et al. The integrative regulatory network of circRNA and microRNA in keloid scarring. Mol Biol Rep. 2020;47(1):201-209. [55] HU H, MAO G, ZHENG J, et al. Keloid Patient Plasma-Derived Exosomal hsa_circ_0020792 Promotes Normal Skin Fibroblasts Proliferation, Migration, and Fibrogenesis via Modulating miR-193a-5p and Activating TGF-beta1/Smad2/3 Signaling. Drug Des Devel Ther. 2022;16:4223-4234. [56] 陶瑞,谭植襄,李思成,等.外泌体在增生性瘢痕和瘢痕疙瘩治疗中的研究进展[J].临床外科杂志,2022,30(12):1196-1199. [57] YAN L, WANG LZ, XIAO R, et al. Inhibition of microRNA-21-5p reduces keloid fibroblast autophagy and migration by targeting PTEN after electron beam irradiation. Lab Invest. 2020;100(3):387-399. [58] KUAI Q, JIAN X. Inhibition of miR-23b-3p Ameliorates Scar-Like Phenotypes of Keloid Fibroblasts by Facilitating A20 Expression. Clin Cosmet Investig Dermatol. 2022;15:1549-1559. [59] WAN J, HE XL, JIAN QC, et al. LINC00937 suppresses keloid fibroblast proliferation and extracellular matrix deposition by targeting the miR-28-5p/MC1R axis. Histol Histopathol. 2021;36(9):995-1005. [60] WU D, LIU X, JIN Z. Adipose-derived mesenchymal stem cells-sourced exosomal microRNA-7846-3p suppresses proliferation and pro-angiogenic role of keloid fibroblasts by suppressing neuropilin 2. J Cosmet Dermatol. 2023. doi: 10.1111/jocd.15721. [61] HE Y, ZHANG Z, YIN B, et al. Identifying miRNAs Associated with the Progression of Keloid through mRNA-miRNA Network Analysis and Validating the Targets of miR-29a-3p in Keloid Fibroblasts. Biomed Res Int. 2022;2022:6487989. [62] GALLANT-BEHM CL, PIPER J, LYNCH JM, et al. A MicroRNA-29 Mimic (Remlarsen) Represses Extracellular Matrix Expression and Fibroplasia in the Skin. J Invest Dermatol. 2019;139(5):1073-1081. [63] JIAN X, QU L, WANG Y, et al. Trichostatin A‑induced miR‑30a‑5p regulates apoptosis and proliferation of keloid fibroblasts via targeting BCL2. Mol Med Rep. 2019;19(6):5251-5262. [64] JIANG L, SHI X, WANG M, et al. Study on the Mechanism of miR-34a Affecting the Proliferation, Migration, and Invasion of Human Keloid Fibroblasts by Regulating the Expression of SATB1. J Healthc Eng. 2021;2021:8741512. [65] HUANG Y, WANG Y, LIN L, et al. Overexpression of miR-133a-3p inhibits fibrosis and proliferation of keloid fibroblasts by regulating IRF5 to inhibit the TGF-beta/Smad2 pathway. Mol Cell Probes. 2020;52:101563. [66] PANG Q, WANG Y, XU M, et al. MicroRNA-152-5p inhibits proliferation and migration and promotes apoptosis by regulating expression of Smad3 in human keloid fibroblasts. BMB Rep. 2019;52(3):202-207. [67] LIU P, HU Y, XIA L, et al. miR-4417 suppresses keloid fibrosis growth by inhibiting CyclinD1. J Biosci. 2020;45:47. [68] 谭欢,张远理,唐原,等.瘢痕疙瘩患者128例生活质量评估及影响因素分析[J].中国皮肤性病学杂志,2021,35(2):200-204. [69] LU W, CHU H, ZHENG X. Effects on quality of life and psychosocial wellbeing in Chinese patients with keloids. Am J Transl Res. 2021;13(3):1636-1642. [70] 王畅,陈炜,王宝家.阿魏酸钠对人皮肤增生性瘢痕成纤维细胞增殖和凋亡的调控作用及其信号机制[J].中华烧伤与创面修复杂志,2022,38(5):471-480. [71] 徐琦,刘伟.不良饮食习惯影响瘢痕疙瘩形成的研究进展[J].中华烧伤与创面修复杂志,2022,38(4):389. [72] LOMBARDI F, AUGELLO FR, ARTONE S, et al. Efficacy of probiotic Streptococcus thermophilus in counteracting TGF-beta1-induced fibrotic response in normal human dermal fibroblasts. J Inflamm (Lond). 2022;19(1):27. |
| [1] | Shen Jiangyong, He Xi, Tang Yuting, Wang Jianjun, Liu Jinyi, Chen Yuanyuan, Wang Xinyi, Liu Tong, Sun Haoyuan. RAS-selective lethal small molecule 3 inhibits the fibrosis of pathological scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1168-1173. |
| [2] | Ran Lei, Han Haihui, Xu Bo, Wang Jianye, Shen Jun, Xiao Lianbo, Shi Qi. Molecular docking analysis of the anti-inflammatory mechanism of Cibotium barometz and Epimedium for rheumatoid arthritis: animal experiment validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 208-215. |
| [3] | Zuo Jun, Ma Shaolin. Mechanism of beta-sitosterol on hypertrophic scar fibroblasts: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 216-223. |
| [4] | Chen Ligang, He Xiaoming, Tan Yu, Xiao Yuzhi, Ma Chuntao, Guo Liang. Bone metabolism in patients with osteonecrosis of the femoral head based on etiology and Association Research Circulation Osseous staging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2461-2466. |
| [5] | Zhao Shasha, He Qing, Li Jia, Wu Ying. Effects of recombinant human collagen supplementation on extracellular matrix remodeling in mouse skeletal muscle after eccentric exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2542-2549. |
| [6] | Zuo Jun, Ma Shaolin. Bioinformatics analysis and validation of differentially expressed genes and small molecule drug prediction in proliferative scar [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2166-2172. |
| [7] | Zhang Hongxia, Li Xingchao, Gao Xixin, Zhang Xiao, Mei Shuang, Ma Hanxi, Zhang Tian. The histological and thickness changes of attached gingiva following grafting with different soft tissue substitutes in the labial region of the cuspids in Beagles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(11): 1660-1665. |
| [8] | Hu Mengfan, Yan Qiuhui, Deng Mengran, Liang Meimei, Liang Liang, Yi Sisi, Deng Jiagang, Yun Chenxia. Mangiferin inhibits proliferation, migration and inflammatory factor expression of fibroblast-like synoviocytes in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(11): 1690-1695. |
| [9] | He Xi, Wan Yu, Tang Yuting, Yang Anning, Wu Kai, Jiao Yun, Bai Zhigang, Jiang Yideng, Shen Jiangyong. Erastin inhibits proliferation of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-. |
| [10] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [11] | Ke Weiqiang, Chen Xianghui, Chen Xiaoling, Meng Jie, Ma Yanlin. Rituximab combined with autologous peripheral blood stem cell transplantation in the treatment of diffuse large B-cell lymphoma and the expression of related factors [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 915-920. |
| [12] | Hu Xinming, Qiao Yanhua, Wang Xiaofan, Li Linyu, Zhao Bing. Mechanism of long non-coding RNA plasmacytoma variant translocation 1 involved in pelvic organ prolapse [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 669-675. |
| [13] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [14] | Liang Weidong, Zhang Shuwen, Cai Xiaoyu, Xun Chuanhui, Sheng Jun, Cao Rui, Hao Honggang, Sheng Weibin. Differentially expressed galectin-1 during intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5610-5615. |
| [15] | Tang Yuting, He Xi, Wan Yu, Wang Jianjun, Yang Anning, Wu Kai, Jiao Yun, Bai Zhigang, Jiang Yideng, Shen Jiangyong. Shikonin inhibits fibrosis of human hypertrophic scar fibroblasts via MicroRNA-382-5p [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5642-5648. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||