Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (14): 2261-2266.doi: 10.12307/2024.289
Previous Articles Next Articles
Bone immunity and bone metabolism
Guo Caopei, Cheng Piaotao, Yang Chengbing, Gong Shouhang, Peng Jiaze, Zhang Lin, Peng Jiachen
- Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2023-01-17Accepted:2023-04-20Online:2024-05-18Published:2023-07-28 -
Contact:Peng Jiachen, MD, Chief physician, Doctoral supervisor, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Guo Caopei, Master candidate, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81760400 (to PJC)
CLC Number:
Cite this article
Guo Caopei, Cheng Piaotao, Yang Chengbing, Gong Shouhang, Peng Jiaze, Zhang Lin, Peng Jiachen. Bone immunity and bone metabolism[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2261-2266.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
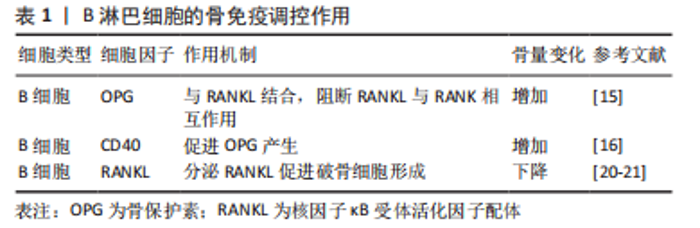
2.1 骨代谢中的免疫机制 2.1.1 B淋巴细胞参与的骨代谢调控 B细胞是来源于骨髓的多能干细胞,可对骨代谢产生调控作用(表1),其生理状态下分泌骨保护素对抗破骨细胞生成因子从而促进成骨[15]。骨保护素是一种调节骨细胞外基质稳态的相关蛋白,对骨细胞具有保护作用[16-17],其通过高亲和力与RANKL结合,阻断RANKL与RANK的相互作用,从而主动抑制骨质受到侵蚀[18]。B细胞可通过上调CD40共刺激信号促进骨保护素的产生[19]。此外,LI等[11,20]发现,在小鼠骨髓中,B细胞是骨保护素重要的来源,B细胞谱系产生的骨保护素达到总量的64%,其中45%来自成熟B细胞。而敲除B细胞的小鼠骨保护素生成障碍、骨密度下降、骨量显著减少。此外ZENG等[21]发现,在B细胞缺乏的牙周炎小鼠中,RANKL mRNA表达量显著升高,而骨保护素显著降低,使得RANKL/骨保护素比例显著升高,且RANKL阳性细胞广泛分布在骨附近炎性细胞浸润的结缔组织中,B细胞缺乏会加剧牙周炎小鼠牙槽骨丢失。"
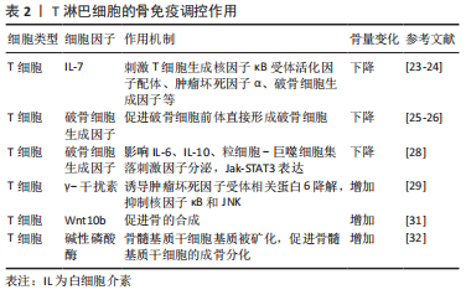
但当机体处于炎症状态下,活化的B细胞分泌RANKL促进破骨细胞形成和骨吸收[22-23]。RANKL是破骨细胞分化和骨质流失的关键调节剂。B细胞急性淋巴细胞白血病患者往往出现骨质减少和坏死等长期并发症,最新的研究发现,人原代B细胞急性淋巴细胞白血病细胞过度表达RANKL,导致破骨细胞增加、小梁骨丢失,干骺端生长板破坏以及脂肪细胞量减少等明显的骨破坏作用[24]。见表1。 2.1.2 T淋巴细胞参与的骨代谢调控 骨吸收受免疫系统的调节(表2),T细胞可以表达RANKL调节骨的吸收。此外,T细胞与多种疾病相关的病理性骨丢失有关,如类风湿性关节炎、雌激素减退、甲状旁腺功能亢进和牙周骨丢失等会产生IL-7刺激T细胞产生RANKL、TNF-α以及活化T细胞分泌破骨细胞生成因子等促进骨的吸收[25-26]。与T细胞分泌的其他因子不同,破骨细胞生成因子可促进纯化的破骨细胞前体不依赖于RANKL直接形成破骨细胞[27-28]。也有研究表明活化T细胞在条件培养基中存在独立于RANKL的破骨细胞活性[29]。此外,NAPIMOGA等[30]研究表明,破骨细胞因子在不刺激RANKL分泌的情况下,影响细胞中IL-6、IL-10、粒细胞-巨噬细胞集落刺激因子的分泌和Jak-STAT3信号分子的表达,加剧炎症反应并促进破骨细胞形成和骨质破坏。"

研究发现T细胞也可能具有促进骨骼形成的能力。T细胞产生的γ-干扰素诱导RANK衔接蛋白TNF受体相关因子6快速降解,从而抑制RANKL诱导的核因子κB和Jun氨基末端激酶的激活[31]。且在对γ-干扰素反应受损的骨组织中,破骨细胞分化增加、过度增殖[32]。表明γ-干扰素的通过干扰RANKL-RANK信号通路强烈抑制破骨细胞生成。HSU等[33]也发现T细胞产生的Wnt10b也可促进骨的合成代谢。CROES等[34]将间充质干细胞暴露于活化的T细胞或活化T细胞来源的条件培养基后,检测到碱性磷酸酶活性显著增加,随后基质被矿化,表明T细胞可以促进间充质干细胞的成骨分化。 2.1.3 其他免疫因子参与的骨代谢调控 骨代谢对IL-1高度敏感。IL-1被认为是破骨细胞活化因子,参与体外破骨细胞发育的各个时期。研究表明IL-1增强成骨细胞/基质细胞中RANKL的表达[35];它也可以通过刺激成骨细胞/基质细胞中前列腺素E2合成间接刺激破骨细胞的形成[36];此外,IL-1还诱导单核破骨细胞融合形成成熟的多核破骨细胞[37],并且IL-1也直接参与破骨细胞的存活[38]。 在骨髓中,IL-6可以增强Runx2、碱性磷酸酶、骨桥蛋白等的表达从而促进成骨细胞的生成[39]。也有研究表明IL-6还可以通过JAK/STAT3途径参与Wnt信号通路加速脂肪来源的间充质干细胞的钙化[40]。另外,IL-6通过核因子κB、ERK和JNK信号途径调节RANKL诱导的破骨细胞水平[41]。此外,IL-6还可以通过诱导IL-1的表达促进破骨细胞的形成[42]。 TNF-α是由活化T细胞产生的刺激破骨细胞增殖分化的关键因子。研究表明它可以刺激IL-6的产生,从而导致破骨细胞生成和骨吸收[43]。TNF-α也可直接或通过增加成骨细胞和基质细胞的RANKL和集落刺激因子表达来调节炎症条件下的骨吸收[44]。此外,CONSTANZE等[45]发现TNF-β可以通过调节核因子κB和沉默信息调节因子2相关酶Ⅰ途径从而抑制间充质干细胞的成骨分化。 γ-干扰素可以通过增强抗原加工和呈递、增加白细胞运输、诱导抗病毒状态、增强抗微生物功能等发挥其免疫调节作用[46]。SHASHKOVA等[47]证明了由调节性T细胞产生的γ-干扰素可以抑制破骨细胞前体形成破骨细胞,并且降解TNF受体相关因子6。γ-干扰素激活经典的JAK-STAT1途径启动泛素-蛋白酶体,从而增加泛素连接酶TNF受体相关因子6的降解,进而抑制TNF受体相关因子6的下游转录因子(包括核因子κB和JNK等),导致破骨细胞的形成减少[31]。研究表明γ-干扰素对成骨代谢也有一定作用。MARUHASHI等[48]发现γ-干扰素通过上调成骨细胞特异性转录因子、碱性磷酸酶和骨钙素等成骨作用因子来促进颅骨成骨细胞的形成。CROES等[34]用抗体阻断活化的CD4+ T细胞中的γ-干扰素,发现人间充质干细胞的成骨分化作用被下调。 2.2 免疫相关骨代谢疾病与骨免疫 免疫系统和骨骼代谢之间相互影响,免疫系统失衡引起炎症刺激,通过诱导破骨细胞分化和抑制成骨细胞分化导致骨骼更新失衡,从而造成骨质丢失、骨小梁变细变少等各种病理状况。免疫-骨骼界面强调骨骼与免疫系统之间的双向作用,是连接免疫系统和骨骼系统之间的关键纽带,其关键免疫成分作用于骨代谢的各种过程。并且免疫-骨骼界面与许多致使骨质流失的临床疾病息息相关,如类风湿骨关节炎、人类1型免疫缺陷病毒感染导致的骨缺失、雌激素缺乏症、甲状腺功能亢进等。 2.2.1 类风湿性关节炎引起的免疫应激和骨质流失 类风湿关节炎是一种慢性炎症性疾病,自身免疫会引起关节畸形和骨质破坏。类风湿关节炎的主要病理改变是软骨和骨组织的降解,该过程导致关节破坏,降低患者的生活质量。与类风湿关节炎相关的骨丢失有3种不同的表现:①骨骼和软骨与炎症中的滑膜直接接触诱使发炎的关节局部侵蚀;②邻近发炎部位的骨小梁和皮质周围骨丢失;③系统性骨质减少和骨质疏松症。这3种形式的骨质流失都是由于骨稳态的改变,其中破骨细胞的产生和活性增加,加速骨吸收,而成骨细胞导的作用则受到抑制[49-50]。 研究发现,破骨细胞活性增强除了细胞间的直接相互作用和炎症细胞因子的全身作用外,也与类风湿关节炎自身抗体通过直接影响破骨细胞和诱导巨噬细胞释放炎症细胞因子而发挥作用有关[51-52]。与类风湿关节炎相关的主要自身抗体有类风湿因子和抗瓜氨酸化蛋白抗体等。虽然类风湿因子在抗瓜氨酸化蛋白抗体阳性患者中与增加的骨侵蚀相关[53],但目前还没有直接数据表明其有对细胞因子产生或破骨细胞形成的直接影响。抗瓜氨酸化蛋白抗体可以在类风湿关节炎临床发作之前的10年内检测到,故抗瓜氨酸化蛋白抗体在预测类风湿关节炎患者的疾病严重程度和发生骨侵蚀可能性方面具有诊断价值[54]。此外,KELLER等[55]发现没有类风湿关节炎临床体征的抗瓜氨酸化蛋白抗体阳性个体的掌骨关节中会出现骨质流失;TEN BRINCK等[56]发现抗瓜氨酸化蛋白抗体阳性的类风湿关节炎患者仅在局部炎症的情况下才表现出骨吸收;CECCARELLI等[57]发现与其他炎症性疾病相比,抗瓜氨酸化蛋白抗体阳性类风湿关节炎患者骨质丢失最为严重。这些研究表明,骨丢失与类风湿关节炎患者的抗瓜氨酸化蛋白抗体阳性密切相关,且抗瓜氨酸化蛋白抗体可能直接或通过全身性炎症增加从而导致骨质流失。 近年来,除了抗瓜氨酸化蛋白抗体以外,还发现针对翻译后修饰的自身抗体,如抗氨甲酰化蛋白抗体(抗CarP)也可能会影响单核细胞分化成破骨细胞的能力,增强破骨细胞介导的细胞外基质吸收[58]。此外,还存在其他与抗瓜氨酸化蛋白抗体作用机制不同的自身抗体,如在类风湿关节炎和腹腔疾病中存在的针对骨保护素的自身抗体,它可以增强破骨细胞的生成[59]。 2.2.2 雌激素缺乏性骨质疏松 体外实验结果表明,雌激素可抑制T细胞和B细胞中RANKL的产生,并显著减少破骨细胞的招募以及增加破骨细胞的凋亡[60-61]。雌激素替代疗法可逆转自然或手术绝经后女性外周血单核细胞中TNF-α和IL-1的释放,当停用雌激素后,使用TNF-α阻断剂和IL-1β阻断剂可以显著阻断骨的吸收[62]。硬化蛋白是由骨细胞分泌的Wnt/β-连环蛋白信号传导的可溶性拮抗剂,研究发现雌激素可抑制硬化蛋白的产生从而增强成骨作用[63]。也有研究表明,雌激素2种受体ERRα和ERRγ会抑制骨的形成,并在衰老和雌激素缺乏造成的骨质流失中发挥作用[64-65]。 妇女在绝经后,体内雌激素缺乏造成骨稳态失调,使得骨的吸收速率大于形成速度,导致骨质流失[66]。绝经后早期妇女的雌激素缺乏症中,促炎细胞因子(TNF-α、IL-1、IL-6和IL-17等)和C-反应蛋白的血清浓度升高;RANKL、巨噬细胞集落刺激因子及其受体上调,骨保护素表达降低[67-68], 这些导致破骨细胞活性增加,骨流失加重。与绝经前妇女相比,绝经后骨质疏松妇女体内的IL-17水平显著升高,IL-17通过诱导RANKL的分泌促进破骨细胞分化[69],且升高的IL-17与sRANK配体或sRANK配体和骨保护素的比例呈正相关[70],见图3。EGHBALI-FATOURECHI等[71]发现雌激素替代治疗可以逆转雌激素缺乏症妇女体内骨髓细胞表面RANKL浓度的增加。绝经后雌激素分泌降低导致的骨质疏松症是绝经后妇女面临的一个主要的健康问题。雌激素与骨免疫系统相互串扰,在维持骨稳态中十分重要,而详细了解其作用机制,有助于开发出新的治疗靶点从而促进骨的形成。"
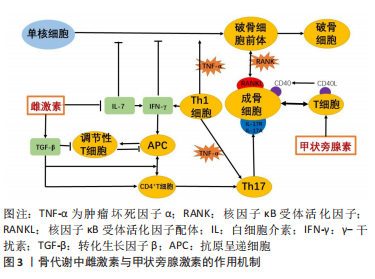

2.2.3 获得性免疫缺陷综合征引起的骨质流失 人类1型免疫缺陷病毒感染所致艾滋病的大流行是现代历史上十分重大的全球健康问题。这种感染导致机体出现慢性免疫失调相关的炎症状态,改变免疫-骨骼界面,并且其使细胞类型和细胞因子之间的深度整合,抑制骨骼更新并加剧骨丢失。因此,感染人类1型免疫缺陷病毒个体会出现骨密度下降,进行发展为骨质疏松症,从而增加骨折的患病率[72]。 人类1型免疫缺陷病毒基因编码调节蛋白(包括Tat、Rev等)、辅助蛋白(包括Nef、Vpr等)和结构蛋白。研究发现Tat蛋白通过RANKL和巨噬细胞集落刺激因子增强外周血单核细胞衍生的破骨细胞分化和活性的能力,从而增加特定破骨细胞分化标志物(如组织蛋白酶K和降钙素受体)的mRNA转录以及抗酒石酸酸性磷酸酶的表达和活性[73],这表明Tat蛋白可以刺激破骨细胞形成并增加骨吸收活性。而Vpr蛋白则上调健康个体外周血单个核细胞RANKL的表达,增强破骨细胞活性。BEAUPERE等[74]发现Tat和Nef蛋白通过影响细胞氧化应激和线粒体功能,以累积的方式减少了分化为成骨细胞的间充质干细胞数量。此外,Tat和Rev蛋白增加单核细胞向破骨细胞的分化,并通过增加破骨细胞前体中的活性氧和TNF-α的产生来增强破骨细胞的吸收功能[75]。 高效抗反转录病毒疗法可以有效控制病毒的复制,但是高效抗反转录病毒治疗并不能改善人类1型免疫缺陷病毒导致的骨骼破坏。相反,在开始治疗的一两年内,骨骼实际上会进一步恶化,骨密度的平均损失高达6%[76-77],而包括替诺福韦在内的治疗方案则导致更为严重的骨密度缺失[78]。 2.2.4 甲状旁腺功能亢进引起的骨质流失 甲状旁腺激素是体内钙磷代谢的重要调节剂,它通过促进骨的吸收来刺激骨骼中钙的释放,从而防御低钙血症。但是,原发性或继发性甲状旁腺功能亢进症会导致甲状旁腺激素持续过量产生,从而导致严重的破骨细胞骨吸收和骨骼退化[79]。 研究发现,甲状旁腺激素的合成代谢作用也涉及免疫-骨骼界面。传统观点认为甲状旁腺激素直接诱导成骨细胞增殖和分化,激活衬里成骨细胞(不活跃的成骨细胞),通过抑制细胞凋亡来延长成骨细胞寿命,以及在骨细胞中下调Wnt受体拮抗剂硬化素的表达[68,80]。尽管Wnt途径是骨合成代谢作用中甲状旁腺激素的关键靶标,但由于驱动骨形成的Wnt配体来源表征不明显,故认为其是由成骨细胞谱系细胞自主分泌的。但最近的数据表明,Wnt配体的关键来源实际上是T细胞[3],甲状旁腺激素诱导T细胞中Wnt10b的大量产生,从而促进成骨。 甲状旁腺激素和免疫-骨骼界面产生分解代谢作用,甲状旁腺激素促进T细胞分泌TNF,除了增强RANKL信号传导和抑制骨保护素的作用外。甲状旁腺激素还反馈T细胞使其分泌IL-17A的Th17亚型,从而通过成骨细胞谱系产生额外的RANKL[81]。 雌激素缺乏后使IL-7含量上升,驱动CD4+ T细胞增殖和活化。并且诱导γ-干扰素上调驱动抗原提呈细胞进一步刺激T细胞活化。Th1效应T细胞分泌TNF-α,作用于破骨细胞前体促进破骨细胞分化。TNF-α诱导Th17 T细胞分泌IL-17A与成骨细胞上IL-17R结合,进一步调节RANKL的产生。另外,雌激素缺乏导致转化生长因子β下降,使得调节性T细胞形成减少。 甲状旁腺素促进T细胞产生TNF-α,并且T细胞则通过细胞自主作用反馈以促进Th17效应细胞的形成。此外,TNF-α上调成骨细胞上的CD40受体,CD40及其配体CD40L在T细胞上的结合促进了成骨细胞的增殖和存活。"

| [1] WEITZMANN MN. Bone and the Immune System. Toxicol Pathol. 2017; 45(7):911-924. [2] ARRON JR, CHOI Y. Bone versus immune system. Nature. 2000;408(6812): 535-536. [3] TSUKASAKI M, TAKAYANAGI H. Osteoimmunology: evolving concepts in bone-immune interactions in health and disease. Nat Rev Immunol. 2019; 19(10):626-642. [4] ZHANG W, DANG K, HUAI Y, et al. Osteoimmunology: The Regulatory Roles of T Lymphocytes in Osteoporosis. Front Endocrinol (Lausanne). 2020;11:465. [5] YASUDA H. Discovery of the RANKL/RANK/OPG system. J Bone Miner Metab. 2021;39(1):2-11. [6] WANG YN, LIU S, JIA T, et al. T Cell Protein Tyrosine Phosphatase in Osteoimmunology. Front Immunol. 2021;12:620333. [7] 钱治, 仲泽远, 崔元斌, 等. T细胞与骨代谢[J]. 中国免疫学杂志,2021, 37(11):1404-1410. [8] VILACA T, EASTELL R, SCHINI M. Osteoporosis in men. Lancet Diabetes Endocrinol. 2022;10(4):273-283. [9] TITANJI K, OFOTOKUN I, WEITZMANN MN. Immature/transitional B-cell expansion is associated with bone loss in HIV-infected individuals with severe CD4+ T-cell lymphopenia. Aids. 2020;34(10):1475-1483. [10] 谭敏, 杨颜瑜, 熊琴, 等. OPG/RANKLlRANK信号通路在骨质疏松症患者骨重建中作用机制的研究进展[J]. 山东医药,2018,58(43):111-114. [11] LI Y, TORALDO G, LI A, et al. B cells and T cells are critical for the preservation of bone homeostasis and attainment of peak bone mass in vivo. Blood. 2007;109(9):3839-3848. [12] WU D, CLINE-SMITH A, SHASHKOVA E, et al. T-Cell Mediated Inflammation in Postmenopausal Osteoporosis. Front Immunol. 2021;12:687551. [13] JIANG TJ, CAO XL, LUAN S, et al. Percentage and function of CD4+CD25+ regulatory T cells in patients with hyperthyroidism. Mol Med Rep. 2018; 17(2):2137-2144. [14] MALLON PW. Aging with HIV: osteoporosis and fractures. Curr Opin HIV AIDS. 2014;9(4):428-435. [15] LI S, LIU Q, WU D, et al. PKC-δ deficiency in B cells displays osteopenia accompanied with upregulation of RANKL expression and osteoclast-osteoblast uncoupling. Cell Death Dis. 2020;11(9):762. [16] OLIVEIRA MC, PIETERS BCH, GUIMARÃES PB, et al. Bovine Milk Extracellular Vesicles Are Osteoprotective by Increasing Osteocyte Numbers and Targeting RANKL/OPG System in Experimental Models of Bone Loss. Front Bioeng Biotechnol. 2020;8:891. [17] HABIBIE H, ADHYATMIKA A, SCHAAFSMA D, et al. The role of osteoprotegerin (OPG) in fibrosis: its potential as a biomarker and/or biological target for the treatment of fibrotic diseases. Pharmacol Ther. 2021;228:107941. [18] NAIK S, SAHU S, BANDYOPADHYAY D, et al. Serum levels of osteoprotegerin, RANK-L & vitamin D in different stages of osteoarthritis of the knee. Indian J Med Res. 2021;154(3):491-496. [19] OTA Y, NIIRO H, OTA S, et al. Generation mechanism of RANKL(+) effector memory B cells: relevance to the pathogenesis of rheumatoid arthritis. Arthritis Res Ther. 2016;18:67. [20] BRUNETTI G, FAIENZA MF, COLAIANNI G, et al. Impairment of Bone Remodeling in LIGHT/TNFSF14-Deficient Mice. J Bone Miner Res. 2018; 33(4):704-719. [21] ZENG W, LIU G, LUAN Q, et al. B-Cell Deficiency Exacerbates Inflammation and Bone Loss in Ligature-Induced Experimental Periodontitis in Mice. J Inflamm Res. 2021;14:5367-5380. [22] RANA AK, LI Y, DANG Q, et al. Monocytes in rheumatoid arthritis: Circulating precursors of macrophages and osteoclasts and, their heterogeneity and plasticity role in RA pathogenesis. Int Immunopharmacol. 2018;65:348-359. [23] KOMATSU N, WIN S, YAN M, et al. Plasma cells promote osteoclastogenesis and periarticular bone loss in autoimmune arthritis. J Clin Invest. 2021; 131(6):e143060. [24] RAJAKUMAR SA, PAPP E, LEE KK, et al. B cell acute lymphoblastic leukemia cells mediate RANK-RANKL-dependent bone destruction. Sci Transl Med. 2020;12(561):eaba5942. [25] WEITZMANN MN. T-cells and B-cells in osteoporosis. Curr Opin Endocrinol Diabetes Obes. 2014;21(6):461-467. [26] IHN HJ, KIM YS, LIM S, et al. PF-3845, a Fatty Acid Amide Hydrolase Inhibitor, Directly Suppresses Osteoclastogenesis through ERK and NF-κB Pathways In Vitro and Alveolar Bone Loss In Vivo. Int J Mol Sci. 2021;22(4):1915. [27] ZHANG R, PENG S, ZHU G. The role of secreted osteoclastogenic factor of activated T cells in bone remodeling. Jpn Dent Sci Rev. 2022;58:227-232. [28] RIFAS L, WEITZMANN MN. A novel T cell cytokine, secreted osteoclastogenic factor of activated T cells, induces osteoclast formation in a RANKL-independent manner. Arthritis Rheum. 2009;60(11):3324-3335. [29] WEITZMANN MN, CENCI S, RIFAS L, et al. T cell activation induces human osteoclast formation via receptor activator of nuclear factor kappaB ligand-dependent and -independent mechanisms. J Bone Miner Res. 2001;16(2): 328-337. [30] NAPIMOGA MH, DEMASI AP, JARRY CR, et al. In vitro evaluation of the biological effect of SOFAT on osteoblasts. Int Immunopharmacol. 2015; 26(2):378-383. [31] TAKAYANAGI H, OGASAWARA K, HIDA S, et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature. 2000;408(6812):600-605. [32] TSUMURA M, MIKI M, MIZOGUCHI Y, et al. Enhanced osteoclastogenesis in patients with MSMD due to impaired response to IFN-γ. J Allergy Clin Immunol. 2022;149(1):252-261.e6. [33] HSU MN, HUANG KL, YU FJ, et al. Coactivation of Endogenous Wnt10b and Foxc2 by CRISPR Activation Enhances BMSC Osteogenesis and Promotes Calvarial Bone Regeneration. Mol Ther. 2020;28(2):441-451. [34] CROES M, ÖNER FC, VAN NEERVEN D, et al. Proinflammatory T cells and IL-17 stimulate osteoblast differentiation. Bone. 2016;84:262-270. [35] LEVESCOT A, CHANG MH, SCHNELL J, et al. IL-1β-driven osteoclastogenic Tregs accelerate bone erosion in arthritis. J Clin Invest. 2021;131(18): e141008. [36] TOMINARI T, AKITA M, MATSUMOTO C, et al. Endosomal TLR3 signaling in stromal osteoblasts induces prostaglandin E(2)-mediated inflammatory periodontal bone resorption. J Biol Chem. 2022;298(3):101603. [37] JIMI E, NAKAMURA I, DUONG LT, et al. Interleukin 1 induces multinucleation and bone-resorbing activity of osteoclasts in the absence of osteoblasts/stromal cells. Exp Cell Res. 1999;247(1):84-93. [38] CHEN W, TANG P, FAN S, et al. A Novel Inhibitor INF 39 Promotes Osteogenesis via Blocking the NLRP3/IL-1β Axis. Biomed Res Int. 2022; 2022:7250578. [39] KUROZUMI A, NAKANO K, YAMAGATA K, et al. IL-6 and sIL-6R induces STAT3-dependent differentiation of human VSMCs into osteoblast-like cells through JMJD2B-mediated histone demethylation of RUNX2. Bone. 2019; 124:53-61. [40] FUKUYO S, YAMAOKA K, SONOMOTO K, et al. IL-6-accelerated calcification by induction of ROR2 in human adipose tissue-derived mesenchymal stem cells is STAT3 dependent. Rheumatology (Oxford). 2014;53(7):1282-1290. [41] LI N, LI X, ZHENG K, et al. Inhibition of Sirtuin 3 prevents titanium particle-induced bone resorption and osteoclastsogenesis via suppressing ERK and JNK signaling. Int J Biol Sci. 2021;17(5):1382-1394. [42] AMARASEKARA DS, YUN H, KIM S, et al. Regulation of Osteoclast Differentiation by Cytokine Networks. Immune Netw. 2018;18(1):e8. [43] SALVIO G, GIANFELICE C, FIRMANI F, et al. Bone Metabolism in SARS-CoV-2 Disease: Possible Osteoimmunology and Gender Implications. Clin Rev Bone Miner Metab. 2020;18(4):51-57. [44] MARAHLEH A, KITAURA H, OHORI F, et al. TNF-α Directly Enhances Osteocyte RANKL Expression and Promotes Osteoclast Formation. Front Immunol. 2019;10:2925. [45] CONSTANZE B, POPPER B, AGGARWAL BB, et al. Evidence that TNF-β suppresses osteoblast differentiation of mesenchymal stem cells and resveratrol reverses it through modulation of NF-κB, Sirt1 and Runx2. Cell Tissue Res. 2020;381(1):83-98. [46] CORDEIRO PAS, ASSONE T, PRATES G, et al. The role of IFN-γ production during retroviral infections: an important cytokine involved in chronic inflammation and pathogenesis. Rev Inst Med Trop Sao Paulo. 2022;64:e64. [47] SHASHKOVA EV, TRIVEDI J, CLINE-SMITH AB, et al. Osteoclast-Primed Foxp3+ CD8 T Cells Induce T-bet, Eomesodermin, and IFN-γ To Regulate Bone Resorption. J Immunol. 2016;197(3):726-735. [48] MARUHASHI T, KAIFU T, YABE R, et al. DCIR maintains bone homeostasis by regulating IFN-γ production in T cells. J Immunol. 2015;194(12):5681-5691. [49] JANG S, KWON EJ, LEE JJ. Rheumatoid Arthritis: Pathogenic Roles of Diverse Immune Cells. Int J Mol Sci. 2022;23(2):905. [50] ZERBINI CAF, CLARK P, MENDEZ-SANCHEZ L, et al. Biologic therapies and bone loss in rheumatoid arthritis. Osteoporos Int. 2017; 28(2): 429-446. [51] WANG X, SUN L, HE N, et al. Increased expression of CXCL2 in ACPA-positive rheumatoid arthritis and its role in osteoclastogenesis. Clin Exp Immunol. 2021;203(2):194-208. [52] STEFFEN U, SCHETT G, BOZEC A. How Autoantibodies Regulate Osteoclast Induced Bone Loss in Rheumatoid Arthritis. Front Immunol. 2019;10:1483. [53] HECHT C, ENGLBRECHT M, RECH J, et al. Additive effect of anti-citrullinated protein antibodies and rheumatoid factor on bone erosions in patients with RA. Ann Rheum Dis. 2015;74(12):2151-2156. [54] PETROVSKÁ N, PRAJZLEROVÁ K, VENCOVSKÝ J, et al. The pre-clinical phase of rheumatoid arthritis: From risk factors to prevention of arthritis. Autoimmun Rev. 2021;20(5):102797. [55] KELLER KK, THOMSEN JS, STENGAARD-PEDERSEN K, et al. Local bone loss in patients with anti-citrullinated peptide antibody and arthralgia, evaluated with high-resolution peripheral quantitative computed tomography. Scand J Rheumatol. 2018;47(2):110-116. [56] TEN BRINCK RM, TOES REM, Van Der Helm-Van Mil AHM. Inflammation functions as a key mediator in the link between ACPA and erosion development: an association study in Clinically Suspect Arthralgia. Arthritis Res Ther. 2018;20(1):89. [57] CECCARELLI F, PERRICONE C, COLASANTI T, et al. Anti-carbamylated protein antibodies as a new biomarker of erosive joint damage in systemic lupus erythematosus. Arthritis Res Ther. 2018;20(1):126. [58] O’NEIL LJ, OLIVEIRA CB, SANDOVAL-HEGLUND D, et al. Anti-Carbamylated LL37 Antibodies Promote Pathogenic Bone Resorption in Rheumatoid Arthritis. Front Immunol. 2021;12:715997. [59] HAUSER B, HARRE U. The Role of Autoantibodies in Bone Metabolism and Bone Loss. Calcif Tissue Int. 2018;102(5):522-532. [60] SRIVASTAVA S, TORALDO G, WEITZMANN MN, et al. Estrogen decreases osteoclast formation by down-regulating receptor activator of NF-kappa B ligand (RANKL)-induced JNK activation. J Biol Chem. 2001;276(12): 8836-8840. [61] UEHARA IA, SOLDI LR, SILVA MJB. Current perspectives of osteoclastogenesis through estrogen modulated immune cell cytokines. Life Sci. 2020;256: 117921. [62] MANOLAGAS SC, O’BRIEN CA, ALMEIDA M. The role of estrogen and androgen receptors in bone health and disease. Nat Rev Endocrinol. 2013; 9(12):699-712. [63] DRAKE MT, KHOSLA S. Hormonal and systemic regulation of sclerostin. Bone. 2017;96:8-17. [64] HUANG T, FU X, WANG N, et al. Andrographolide prevents bone loss via targeting estrogen-related receptor-α-regulated metabolic adaption of osteoclastogenesis. Br J Pharmacol. 2021;178(21):4352-4367. [65] TANG J, LIU T, WEN X, et al. Estrogen-related receptors: novel potential regulators of osteoarthritis pathogenesis. Mol Med. 2021;27(1):5. [66] CHENG CH, CHEN LR, CHEN KH. Osteoporosis Due to Hormone Imbalance: An Overview of the Effects of Estrogen Deficiency and Glucocorticoid Overuse on Bone Turnover. Int J Mol Sci. 2022;23(3):1376. [67] STEPAN JJ, HRUSKOVA H, KVERKA M. Update on Menopausal Hormone Therapy for Fracture Prevention. Curr Osteoporos Rep. 2019;17(6):465-473. [68] 张晓红, 高艳丽, 袁征, 等. 免疫系统在绝经后骨质疏松症中的作用[J]. 中国骨质疏松杂志,2020,26(10):1538-1541. [69] SCHEFFLER JM, GRAHNEMO L, ENGDAHL C, et al. Interleukin 17A: a Janus-faced regulator of osteoporosis. Sci Rep. 2020;10(1):5692. [70] MOLNÁR I, BOHATY I, SOMOGYINÉ-VÁRI É. IL-17A-mediated sRANK ligand elevation involved in postmenopausal osteoporosis. Osteoporos Int. 2014; 25(2):783-786. [71] EGHBALI-FATOURECHI G, KHOSLA S, SANYAL A, et al. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J Clin Invest. 2003;111(8):1221-1230. [72] DELPINO MV, QUARLERI J. Influence of HIV Infection and Antiretroviral Therapy on Bone Homeostasis. Front Endocrinol (Lausanne). 2020;11:502. [73] COTTER EJ, CHEW N, POWDERLY WG, et al. HIV type 1 alters mesenchymal stem cell differentiation potential and cell phenotype ex vivo. AIDS Res Hum Retroviruses. 2011;27(2):187-199. [74] BEAUPERE C, GARCIA M, LARGHERO J, et al. The HIV proteins Tat and Nef promote human bone marrow mesenchymal stem cell senescence and alter osteoblastic differentiation. Aging Cell. 2015;14(4):534-546. [75] AGIDIGBI TS, KIM C. Reactive Oxygen Species in Osteoclast Differentiation and Possible Pharmaceutical Targets of ROS-Mediated Osteoclast Diseases. Int J Mol Sci. 2019;20(14):3576. [76] MCCOMSEY GA, TEBAS P, SHANE E, et al. Bone disease in HIV infection: a practical review and recommendations for HIV care providers. Clin Infect Dis. 2010;51(8):937-946. [77] MATUSZEWSKA A, NOWAK B, NIŻAŃSKI W, et al. Long-Term Administration of Abacavir and Etravirine Impairs Semen Quality and Alters Redox System and Bone Metabolism in Growing Male Wistar Rats. Oxid Med Cell Longev. 2021;2021:5596090. [78] CHISATI EM, CONSTANTINOU D, LAMPIAO F. Reduced bone mineral density among HIV infected patients on anti-retroviral therapy in Blantyre, Malawi: Prevalence and associated factors. PLoS One. 2020;15(1):e0227893. [79] CORMIER C, KOUMAKIS E. Bone and primary hyperparathyroidism. Joint Bone Spine. 2022;89(1):105129. [80] PACIFICI R. Role of T cells in the modulation of PTH action: physiological and clinical significance. Endocrine. 2013;44(3):576-582. [81] PACIFICI R. Role of Gut Microbiota in the Skeletal Response to PTH. J Clin Endocrinol Metab. 2021;106(3):636-645. |
| [1] | Chen Shuai, Jin Jie, Han Huawei, Tian Ningsheng, Li Zhiwei . Causal relationship between circulating inflammatory cytokines and bone mineral density based on two-sample Mendelian randomization [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1556-1564. |
| [2] | Zhu Hanmin, Wang Song, Xiao Wenlin, Zhang Wenjing, Zhou Xi, He Ye, Li Wei, . Mitophagy regulates bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1676-1683. |
| [3] | Zhao Jiacheng, Ren Shiqi, Zhu Qin, Liu Jiajia, Zhu Xiang, Yang Yang. Bioinformatics analysis of potential biomarkers for primary osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1741-1750. |
| [4] | Zhang Zhenyu, Liang Qiujian, Yang Jun, Wei Xiangyu, Jiang Jie, Huang Linke, Tan Zhen. Target of neohesperidin in treatment of osteoporosis and its effect on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1437-1447. |
| [5] | Qian Kun, Li Ziqing, Sun Shui . Endoplasmic reticulum stress in the occurrence and development of common degenerative bone diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1285-1295. |
| [6] | Li Yueyao, Zhang Min, Yang Jiaju. Cistanoside A mediates p38/MAPK pathway to inhibit osteoclast activity [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1144-1151. |
| [7] | Zheng Lin, Jin Wenjun, Luo Shanshan, Huang Rui, Wang Jie, Cheng Yuting, An Zheqing, Xiong Yue, Gong Zipeng, Liao Jian. Eucommia ulmoides promotes alveolar bone formation in ovariectomized rats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1159-1167. |
| [8] |
Huang Xiaobin, Ge Jirong, Li Shengqiang, Xie Lihua, Huang Jingwen, He Yanyan, Xue Lipeng.
Mechanisms of different yin nourishing and kidney tonifying methods on osteoclastysis pathway in ovariectomized rats #br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1214-1219.
|
| [9] | Lan Shuangli, Xiang Feifan, Deng Guanghui, Xiao Yukun, Yang Yunkang, Liang Jie. Naringin inhibits iron deposition and cell apoptosis in bone tissue of osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 888-898. |
| [10] | Niu Yongkang, Feng Zhiwei, Wang Yaobin, Liu Zhongcheng, Xiang Dejian, Liang Xiaoyuan, Yi Zhi, Zhan Hongwei, Geng Bin, Xia Yayi. Resveratrol activates extracellular-regulated protein kinase 5 signaling protein to promote proliferation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 908-916. |
| [11] | Wang Dongyang, Yang Qiaohui, Lin Xinchao. Relationship between vitamin D levels and reproductive characteristics and exercise dietary situation in postmenopausal women [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1021-1025. |
| [12] | Zhang Lichuang, Yang Wen, Ding Guangjiang, Li Peikun, Xiao Zhongyu, Chen Ying, Fang Xue, Zhang Teng. Dispersion effect of bone cement after vertebroplasty using individualized unilateral external pedicle approach and bilateral pedicle approach [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 800-808. |
| [13] | Liu Mengfei, Chen Gang, Shi Yihan, Zeng Lin, Jiang Kan, Yilihamujiang•Wusiman. Finite element analysis of optimization of femoral prosthesis implantation position in unicompartmental knee arthroplasty in osteoporotic patients [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 464-470. |
| [14] | Yang Cheng, Li Weimin, Ran Dongcheng, Xu Jiamu, Wu Wangxiang, Xu Jiafu, Chen Jingjing, Jiang Guangfu, Wang Chunqing. Ferroptosis and osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 554-562. |
| [15] | Zhao Xingcheng, Wang Jun, Lu Ming. Repair strategies for nonunion in old osteoporotic vertebral compression fractures: a case analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(3): 538-546. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||