Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (12): 1944-1949.doi: 10.12307/2023.927
Previous Articles Next Articles
Application of pulsed electromagnetic fields in the treatment of orthopedic diseases
Liang Hairui, Cai Zhencun, Zhang He, Duan Siyu, Chen Beibei
- Central Hospital Affiliated to Shenyang Medical College, Shenyang 110000, Liaoning Province, China
-
Received:2022-11-26Accepted:2023-02-18Online:2024-04-28Published:2023-08-23 -
Contact:Chen Beibei, Professor, Master’s supervisor, Central Hospital Affiliated to Shenyang Medical College, Shenyang 110000, Liaoning Province, China -
About author:Liang Hairui, Master candidate, Central Hospital Affiliated to Shenyang Medical College, Shenyang 110000, Liaoning Province, China -
Supported by:Science and Technology Project Fund of Shenyang, No. 22-321-32-13 (to CZC); Innovation Fund of National Clinical Medical Research Center for Orthopedics and Sports Rehabilitation, No. 2021-NCRC-CXJJ-PY-02 (to CZC)
CLC Number:
Cite this article
Liang Hairui, Cai Zhencun, Zhang He, Duan Siyu, Chen Beibei. Application of pulsed electromagnetic fields in the treatment of orthopedic diseases[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(12): 1944-1949.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
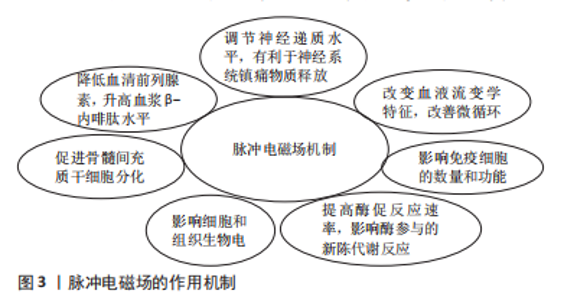
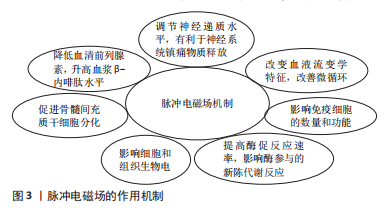
2.1 磁场的生物学效应与应用机制 磁场刺激可以调节神经递质水平,有利于神经系统镇痛物质的释放[11];特定的磁场可以改变血液流变学特性,促进血管生成,改善微循环[12];磁场刺激可以在一定程度上影响免疫细胞的数量和功能[13],可以使细胞形态、DNA、RNA及蛋白质合成发生改变[14];磁场可以促进活性中心的电荷转移,提高酶促反应速率,进而影响酶参与的新陈代谢反应[15]。外部磁场可影响细胞和组织生物电活动的相关过程,进而改变人体生物电、改善生物场,从而达到治疗的目的[6]。 20世纪50年代,研究人员Yasuda发现了骨的压电特性,在应力作用下骨发生电荷分离,张力侧带正电荷,同时破骨细胞活动加强而引起骨吸收;压缩侧带负电荷,伴随成骨细胞活动增强而引起骨沉积[16]。D’ALESSANDRO等[17]通过研究再次阐明骨的压电性作为骨和血管组织再生刺激信号的特殊贡献。自从发现骨的压电特性以来,磁场在骨科领域的研究与应用逐渐增多。脉冲电磁场是具有特定波形和幅度的低频场,特点是高变化率且磁场幅值随时间恒定变化,可以诱导组织产生特定生物电流,引起特殊的生物效应[5],改善骨代谢、增加骨密度,通过多种机制促进成骨细胞生长、分化和加快骨愈合,还可促进骨髓间充质干细胞分化和发挥促成骨作用,还可降低血清前列腺素、升高血浆β-内啡肽水平,进而产生镇痛作用,可作为炎症细胞A2a受体的腺苷激动剂,缓解关节肿胀和减少炎症[3,18-22]。随着磁场研究的深入,脉冲电磁场广泛应用于骨科临床实践,以促进骨愈合。脉冲电磁场仍是一种基于经验的治疗方法,虽然已有大量相关研究证明脉冲电磁场有可靠的疗效,但其应用机制仍然不确切[8,14,17,23-26],因此需要更深入的研究去证实脉冲电磁场应用的确切机制,推进脉冲电磁场治疗的进一步发展,见图3。"

2.2 电磁场在骨科疾病中的应用 2.2.1 脉冲电磁场在骨折愈合中的应用 骨折延迟愈合/不愈合不仅会影响患者身体健康,对患者心理也会产生极大影响[27]。脉冲电磁场在促进骨折愈合方面的有效性已在体外、体内和Ⅰ级临床研究中得到了深入研究[1,28]。在体外实验中最常用的细胞系是骨髓间充质干细胞,而在体内动物实验中最常用的模型是胫骨或股骨截骨的兔或大鼠。 文献证据表明,在体外和体内实验中脉冲电磁场使用的最常见参数如下[18,28]:强度为0.1-2 mT,频率为15-75 Hz;大多数体外实验表明,骨标志物碱性磷酸酶、骨钙素和骨桥蛋白的基因表达增加。体内研究表明,脉冲电磁场对骨折愈合有积极的影响,在不同动物模型中均观察到截骨术后愈合时间减少[18]。脉冲电磁场刺激机体产生脉冲电流,改善骨代谢,通过多种机制达到促进成骨细胞生长、分化和加快骨愈合的作用[3,18-20]。脉冲电磁场可以控制炎症微环境,促进骨髓间充质干细胞分化,发挥促成骨作用[18,29]。CALIOGNA等[30]将骨细胞暴露于脉冲电磁场,刺激细胞外基质合成以进行骨和软骨修复的研究,结果表明脉冲电磁场对骨形成起作用。BAHNEY等[29]表明脉冲电磁场通过影响血肿炎症机化、原始骨痂形成、骨痂改造塑形3个阶段质子交换膜参与的不同分子途径来促进骨折愈合,然而这些阶段有相当大的重叠,已经证实脉冲电磁场激活的通路在骨愈合全阶段起作用,同时抑制炎症阶段。VICENTI等[23]对脉冲电磁场在有骨不连风险的急性骨折、骨不连、截骨、应力性骨折和骨坏死中的应用进行了详细解读,疗效方面均得到确切的反馈。脉冲电磁场已广泛用于促进骨修复,通过促进骨痂形成来加速新近骨折的愈合过程[5]。YANG等[31]进行了一项评估不同辅助治疗骨折不愈合/延迟愈合相对疗效的随机对照试验,脉冲电磁场在治愈率和治愈时间上较自体松质骨或富血小板血浆等治疗方法显示出相对优势,见表1。虽然脉冲电磁场在骨折延迟愈合/不愈合中的应用已经颇具规模,有关于其确切机制(压电特性、生物反应、信号通路等)的报道却众说纷纭[2,26,32-33],仍需进一步研究脉冲电磁场的作用机制,为骨折延迟愈合/不愈合提供更加确实可靠的治疗依据。"


骨髓炎是骨折不愈合的原因之一,也一直是骨科临床治疗的棘手问题,以往研究表明,脉冲电磁场或可用于促进骨髓炎骨愈合的治疗[34]。电磁场具有改善机体免疫功能及抗氧化和抗炎防御能力[35]。BAYIR等[36]将体外培养的细菌暴露于极低频电磁场下,结果细菌数明显减少。有研究表明脉冲电磁场在体外可以显著抑制对数生长期金黄色葡萄球菌,并且与抗生素之间具有协同抑菌作用[37]。鉴于脉冲电磁场在抑菌、杀菌、抗炎及促进骨愈合方面的作用,脉冲电磁场在骨髓炎治疗中的应用值得以继续开展与研究。 2.2.2 脉冲电磁场在骨质疏松中的应用 骨质疏松症是老年人,尤其是绝经后老年妇女的一种常见病、多发病,严重威胁老年人的身体健康,尤其是随着老龄化社会的到来,骨质疏松的问题日益突出。在确诊骨质疏松症的患者中,已被证明低骨量和低骨强度会显著增加脆性骨折的风险[38]。迄今为止,为了维持健康的骨量和强度,已经应用了各种合成代谢和抗再吸收疗法。脉冲电磁场可以控制炎症微环境,促进骨髓间充质干细胞分化;能影响成骨细胞活性,使细胞增殖或分化[18-20,39];可以激活ERK和p38MAPK信号,调节骨稳态和生理学所需的成骨和破骨活动[18]。赵琳[21]研究表明脉冲电磁场通过影响成骨及破骨细胞,可以诱导骨生长因子分泌,抑制骨吸收相关细胞因子分泌,增加细胞内Ca2+含量,降低降钙素水平,抑制骨吸收,促进钙沉积;可调节电压依赖性Ca2+通道使骨显微结构恢复,降低血清前列腺素、升高血浆β-内啡肽水平,进而产生镇痛作用。由此可见脉冲电磁场不仅可以增加骨密度,亦可缓解骨量流失导致的疼痛症状。多囊蛋白是位于细胞膜表面的新型膜蛋白,可在成骨细胞谱系中表达,参与骨发育过程,多囊蛋白2可调节钙依赖的信号传导途径[40]。HE等[41]研究发现,成骨细胞成骨性分化依赖于多囊蛋白2的存在,脉冲电磁场处理能显著提高成骨细胞内多囊蛋白2蛋白的表达量,这可能是脉冲电磁场促进成骨细胞分化以及增强骨量和强度的机制之一。WANG等[38]证实脉冲电磁场可刺激成骨细胞生成,抑制破骨细胞生成,并影响骨髓间充质干细胞和骨细胞的活性,最终导致骨量和强度的保留[42]。孙智路等[43]通过大鼠对照实验证明脉冲电磁场可促进骨形成,提高骨质疏松症模型大鼠骨密度,改善其骨质丢失症状,发挥抗骨质疏松作用,可能与 Smurf1蛋白表达下调有关,为临床骨质疏松症的治疗又提供了新的思路。 脉冲电磁场会影响骨代谢过程,导致人体正常骨组织不断进行改建活动,成骨细胞、破骨细胞及骨基质都在骨代谢过程中发挥了至关重要的作用,在此过程中骨组织吸收与合成保持着动态的平衡。脉冲电磁场可作用于骨代谢的众多环节,影响异常的生物过程,从而改善肌肉、骨骼及其他系统的病理状态,抑制骨吸收,促进骨形成[44]。ZHU等[45]进行了一项脉冲电磁场治疗老年人原发性骨质疏松有效性的对照试验,包括411名受试者,结果表明脉冲电磁场在防止腰椎和股骨颈骨矿物质密度下降方面具有不劣于一线治疗的积极作用,在平衡功能方面优于安慰剂。脉冲电磁场产生脉冲电流,亦可通过应力耦合机制改善骨代谢,近年来越来越多的研究表明脉冲电磁场对绝经后骨质疏松有抑制作用[42]。绝经后骨质疏松发病隐匿,绝大多数女性从50岁开始出现骨皮质变薄、骨小梁数量减少、骨质流失等改变,最终会引起骨的承载性能及抗压能力减弱或丧失,导致关节疼痛及脆性骨折;治疗的关键是增加骨密度,抑制骨质流失,延缓骨质疏松的进展,因此脉冲电磁场治疗开始的时机是影响疗效的重要方面[42]。 脉冲电磁场作为骨质疏松的一种治疗手段,与其他治疗方法联合应用能获得更好的效果。钱光等[46]采用卵巢切除法进行大白兔绝经后骨质疏松造模,随机将实验动物分为卵巢切除组、脉冲电磁场组、骨硬化蛋白单克隆抗体组和脉冲电磁场+骨硬化蛋白单克隆抗体组,每组10只,术后给予脉冲电磁场磁疗、骨硬化蛋白单克隆抗体皮下注射等处理,干预8周后行骨代谢指标检查、骨密度测定,结果发现脉冲电磁场+骨硬化蛋白单克隆抗体组骨微结构参数均优于其他组,联合治疗可以增强大白兔骨密度,改善骨代谢和骨微结构。吴智敏等[47]通过120例骨质疏松性桡骨远端骨折患者临床对比试验证明,淫羊藿总黄酮联合脉冲电磁场可提高骨质疏松性桡骨远端骨折患者临床疗效,改善患者骨密度及骨代谢水平,安全性高。目前临床上脉冲电磁场多与唑来膦酸、阿仑膦酸钠等膦酸盐类药物联用,与降钙素配合应用的报道也不少,都获得了很好的疗效[21]。 脉冲电磁场干预骨质疏松动物模型和临床研究汇总见表2和表3。"

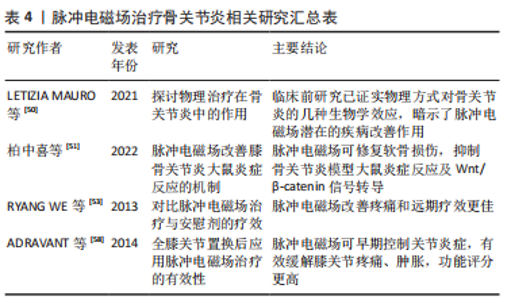
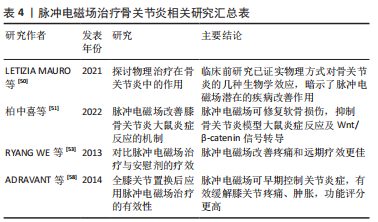
2.2.3 脉冲电磁场在骨关节炎中的应用 近年来骨关节炎发病率逐年增加,通常影响下肢关节,尤其是髋和膝关节,该病症缓慢渐进,软骨细胞、细胞基质和软骨下骨的合成与分解不平衡,关节软骨退化,最终导致关节功能障碍和慢性残疾,严重影响患者的生活质量,加重家庭及社会的经济负担,脉冲电磁场常用于骨质疏松的治疗,现也用于骨关节炎的治疗[48-49]。骨关节炎电磁场疗法主要由物理治疗师使用,可以促进骨骼、软骨的生长和修复,LETIZIA MAURO等[50]研究证明与骨关节炎造模后早期干预或延迟干预相比,造模前用脉冲电磁场对动物模型软骨下骨的保护作用更为明显。在关节形成及调控软骨细胞功能方面,Wnt/β-catenin信号转导通路发挥着至关重要的作用。柏中喜等[51]选取90只大鼠,均分为正常组、模型组和脉冲电磁场组,除正常组外均构建膝骨关节炎模型,检测各组膝关节周径、Lequesne MG评分、Mankin’s评分、Wnt/β-catenin表达,结果证明脉冲电磁场能抑制膝骨关节炎模型大鼠炎症反应,有效修复软骨损伤,降低炎症因子表达,抑制Wnt/β-catenin信号转导,延缓骨关节的进一步退化。LI等[52]检索了Cochrane数据库并进行了电磁场治疗骨关节炎的随机对照试验评价,证明电磁场治疗在缓解疼痛方面为骨关节炎患者提供中等程度的益处。RYANG WE等[53]对482例应用脉冲电磁场和448例应用安慰剂治疗的骨关节炎患者使用高质量方法进行分析,结果表明脉冲电磁场应用4周和8周时在缓解疼痛方面明显比安慰剂更有效,在治疗开始后8周观察到疗效显著改善。向小娜等[54]研究证明:脉冲电磁场治疗能增加细胞中 Ca2+浓度,改变膜电位,使钙在骨形成点沉积,加速骨与软骨的形成;脉冲电磁场能抑制髓核细胞分泌白细胞介素1和肿瘤坏死因子α;脉冲电磁场治疗后骨关节炎患者疼痛缓解,治疗结果与选择的磁场参数有关,向小娜等[54]还对脉冲电磁场联合富血小板血浆治疗骨关节炎进行研究,可能机制为抑制白细胞介素1和肿瘤坏死因子α等炎症因子表达、调控Toll样受体表达、通过Wnt/β-catenin信号通路协同促进血管内皮生长因子分泌有关,为骨关节炎早期治疗提供了新思路。脉冲电磁场刺激软骨细胞增殖,对分解代谢环境产生保护作用,有利于维持软骨下小梁骨微结构和预防软骨下骨质流失,增加骨和软骨的合成,从而阻断骨关节炎进展,对缓解疼痛症状也有较好疗效,能起到推迟进一步手术治疗的作用,有望成为骨关节炎的一种有前途的治疗选择[49,55]。 全膝关节置换术是一种既定的标准程序,用于缓解晚期膝骨关节炎引起的问题[56]。全髋关节置换术和全膝关节置换术后局部关节肿胀、炎症和疼痛是影响患者康复和关节功能的重要因素,FRANKLIN等[57]临床研究显示,全膝关节置换术后部分患者肌力下降,运动功能受限,并可能会留有长期疼痛后遗症,因此全膝关节置换术后恢复尤为重要。脉冲电磁场可促进内源性骨骼修复,可作为炎症细胞A2a受体的腺苷激动剂(具有最强的抗炎作用),可缓解关节肿胀和减少炎症,亦可减少非类固醇抗炎药和止痛药的使用,促进功能恢复,很好地被患者接受[22]。ADRAVANTI等[58]比较了33例全膝关节置换术后脉冲电磁场治疗和对照安慰剂治疗患者的临床指标,随访结果表明在全膝关节置换术后1-6个月,脉冲电磁场治疗组疼痛、膝关节肿胀、功能评分均明显优于对照组,术后3年,脉冲电磁场组剧烈疼痛和偶尔行走受限的患者数明显减少,可见在围术期及术后使用脉冲电磁场治疗是有必要的,应该在临床中合理应用。随着研究深入,基于磁场的磁导航技术在全膝关节置换术、全髋关节置换术假体安放中也逐渐被应用。 脉冲电磁场治疗骨关节炎相关研究汇总见表4。"

2.2.4 电磁场在骨科其他疾病中的应用 肩关节撞击综合征的病理机制是肩峰下空间结构狭窄[59]。近期由于电磁场研究的发展,脉冲电磁场也应用于肩关节撞击综合征的治疗。GALACE DE FREITAS等[60]进行了一项随机双盲对照试验,将诊断肩关节撞击综合征的患者随机分配接受脉冲电磁场或安慰剂治疗,结果表明脉冲电磁场联合肩关节功能锻炼能有效改善肩关节撞击综合征患者的功能和肌力,减轻疼痛。 多种血管生成因素促进了类风湿性关节炎形成,因此抗血管生成治疗是类风湿性关节炎治疗的重点。ROSS等[61]研究表明,极低频脉冲电磁场可以增加间充质干细胞的功能活性,促进间充质干细胞的分化而具有抗炎作用,同时可以增加胶原沉积,并可使血管功能障碍恢复到稳态。脉冲电磁场可以在一定程度上影响免疫细胞的数量和功能[13]。以上都表明脉冲电磁场在治疗类风湿性关节炎中的潜在作用。 股骨头坏死通常累及50岁以下人群且部分患者可能要经历多次手术。侵入性较小的技术将有助于减少手术次数并对疾病的正常发展过程产生积极影响[62]。HINES等[63]更新了股骨头坏死的发病机制、分类标准、分期系统和治疗方面的知识,提及脉冲电磁场新型治疗措施。脉冲电磁场能加速新骨形成,可用于股骨头坏死的早期治疗[62]。洪志楠等[64]表明脉冲电磁场可改善人体微循环,加速坏死灶内血管生长,促进成骨活性,预防骨小梁骨折和软骨下骨塌陷,可以延迟关节置换术的时间,对缓解疼痛症状也有较好疗效,可作为早期股骨头坏死治疗的辅助手段。 磁场发展大事记时线图,见图4。"


2.3 电磁场在骨科器械方面的应用进展 2.3.1 电磁导航 作为新的临床辅助导航技术,主要用于降低手术的侵入性,提高操作精度,缩短手术时间及增加手术的安全性。使用电磁方法获得的坐标一般会与磁共振或计算机断层扫描获得的患部三维图像相结合,使得手术器械相对于患者身体解剖结构的实际位置变得更加可视化,通过了解产生磁场的模块位置,确定放置在仪器上的磁场传感器的位置和空间方向,使操作更加方便安全,电磁导航在医学领域的应用越来越广泛[65]。 2.3.2 电磁导航系统(电磁定位装置)在骨折锁钉及内植物安放中的应用 BLYTH等[66]评价了使用传统仪器或电磁计算机辅助手术技术进行的全膝关节置换术,在5年随访时,与传统组相比,导航组植入物存活率及存活时间明显改善,表明导航技术在全膝关节置换术中不仅是一个挑战,也是一个机遇,需要也有必要继续进行研究。电磁导航技术在全髋关节置换术假体安放中也有一定的应用,MIHALI?等[67]进行了电磁导航系统与徒手技术用于全髋关节置换术中髋臼组件放置准确度及时间的随机临床对照试验,结果显示在电磁导航技术下,在不影响手术持续时间的同时,髋臼杯放置位置更精确。 胫骨骨折非常常见,对于骨科医生来说,胫骨髓内钉远端螺钉传统通过C型臂多角度透视进行锁钉很容易穿透骨皮质,不仅辐射量大、费时费力,失败率还较高。李军等[68]对45例患者进行了TRIGEN-SURESHOT导航系统与传统C型臂透视锁钉的对照研究,结果表明TRIGEN-SURESHOT导航系统在操作、治疗时间及患者预后情况各方面结果更令人满意。同年 CHAN等[69]对使用C型臂徒手透视辅助锁钉和基于电磁的导航系统插入髓内钉锁螺钉操作的准确性、时间及辐射暴露进行了比较研究,结果表明使用电磁导航锁螺钉不仅显著缩短了总操作时间,同时完全消除了辐射暴露。这种无辐射、耗时短、治疗效果更好的导航系统值得在以后骨折患者治疗中广泛应用。"

| [1] FERSON ND, UHL AM, ANDREW JS. Piezoelectric and Magnetoelectric Scaffolds for Tissue Regeneration and Biomedicine: A Review. IEEE Trans Ultrason Ferroelectr Freq Control. 2021;68(2):229-241. [2] RAJABI AH, JAFFE M, ARINZEH TL. Piezoelectric materials for tissue regeneration: A review. Acta Biomater. 2015;24:12-23. [3] SAFAVI AS, SENDERA A, HAGHIGHIPOUR N, et al. The Role of Low-Frequency Electromagnetic Fields on Mesenchymal Stem Cells Differentiation: A Systematic Review. Tissue Eng Regen Med. 2022;19(6):1147-1160. [4] PAOLUCCI T, PEZZI L, CENTRA AM, et al. Electromagnetic Field Therapy: A Rehabilitative Perspective in the Management of Musculoskeletal Pain - A Systematic Review. J Pain Res. 2020;13:1385-1400. [5] HU H, YANG W, ZENG Q, et al. Promising application of Pulsed Electromagnetic Fields (PEMFs) in musculoskeletal disorders. Biomed Pharmacother. 2020;131:110767. [6] 李天乐,卢倩,苗辉,等.低频脉冲电磁场对骨骼相关疾病的生物学效应研究进展[J].医学研究杂志,2022,51(7):7-10. [7] ROSSO F, BONASIA DE, MARMOTTI A, et al. Mechanical Stimulation (Pulsed Electromagnetic Fields “PEMF” and Extracorporeal Shock Wave Therapy “ESWT”) and Tendon Regeneration: A Possible Alternative. Front Aging Neurosci. 2015;7:211. [8] SCHUERMANN D, MEVISSEN M. Manmade Electromagnetic Fields and Oxidative Stress-Biological Effects and Consequences for Health. Int J Mol Sci. 2021;22(7):3772. [9] CIFRA M, APOLLONIO F, LIBERTI M, et al. Possible molecular and cellular mechanisms at the basis of atmospheric electromagnetic field bioeffects. Int J Biometeorol. 2021;65(1):59-67. [10] NIMPF S, KEAYS DA. Myths in magnetosensation. iScience. 2022;25(6): 104454. [11] DEL SEPPIA C, GHIONE S, LUSCHI P, et al. Pain perception and electromagnetic fields. Neurosci Biobehav Rev. 2007;31(4):619-642. [12] PENG L, FU C, WANG L, et al. The Effect of Pulsed Electromagnetic Fields on Angiogenesis. Bioelectromagnetics. 2021;42(3):250-258. [13] YAO C, ZHAO L, PENG R. The biological effects of electromagnetic exposure on immune cells and potential mechanisms. Electromagn Biol Med. 2022; 41(1):108-117. [14] PANAGOPOULOS DJ, KARABARBOUNIS A, YAKYMENKO I, et al. Human‑made electromagnetic fields: Ion forced‑oscillation and voltage‑gated ion channel dysfunction, oxidative stress and DNA damage (Review). Int J Oncol. 2021; 59(5):92. [15] WANG F, LIU Y, DU C, et al. Current Strategies for Real-Time Enzyme Activation. Biomolecules. 2022;12(5):599. [16] The classic: Fundamental aspects of fracture treatment by Iwao Yasuda, reprinted from J. Kyoto Med. Soc., 4:395-406, 1953. Clin Orthop Relat Res. 1977;(124):5-8. [17] D’ALESSANDRO D, RICCI C, MILAZZO M, et al. Piezoelectric Signals in Vascularized Bone Regeneration. Biomolecules. 2021;11(11):1731. [18] CALIOGNA L, MEDETTI M, BINA V, et al. Pulsed Electromagnetic Fields in Bone Healing: Molecular Pathways and Clinical Applications. Int J Mol Sci. 2021;22(14):7403. [19] MARTINI F, PELLATI A, MAZZONI E, et al. Bone Morphogenetic Protein-2 Signaling in the Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells Induced by Pulsed Electromagnetic Fields. Int J Mol Sci. 2020;21(6):2104. [20] VARANI K, VINCENZI F, PASQUINI S, et al. Pulsed Electromagnetic Field Stimulation in Osteogenesis and Chondrogenesis: Signaling Pathways and Therapeutic Implications. Int J Mol Sci. 2021;22(2):809. [21] 赵琳.低频脉冲电磁场治疗骨质疏松的机制及临床应用[J].中国组织工程研究,2016,20(8):1210-1216. [22] IBRAHIM MS, KHAN MA, NIZAM I, et al. Peri-operative interventions producing better functional outcomes and enhanced recovery following total hip and knee arthroplasty: an evidence-based review. BMC Med. 2013;11:37. [23] VICENTI G, BIZZOCA D, SOLARINO G, et al. The role of biophysical stimulation with pemfs in fracture healing: from bench to bedside. J Biol Regul Homeost Agents. 2020;34(5 Suppl. 1):131-135. [24] NICKSIC PJ, DONNELLY DT, HESSE M, et al. Electronic Bone Growth Stimulators for Augmentation of Osteogenesis in In Vitro and In Vivo Models: A Narrative Review of Electrical Stimulation Mechanisms and Device Specifications. Front Bioeng Biotechnol. 2022;10:793945. [25] VARANI K, VINCENZI F, RAVANI A, et al. Adenosine Receptors as a Biological Pathway for the Anti-Inflammatory and Beneficial Effects of Low Frequency Low Energy Pulsed Electromagnetic Fields. Mediators Inflamm. 2017;2017:2740963. [26] MOHAMMADKHAH M, MARINKOVIC D, ZEHN M, et al. A review on computer modeling of bone piezoelectricity and its application to bone adaptation and regeneration. Bone. 2019;127:544-555. [27] GRIFFIN XL, COSTA ML, PARSONS N, et al. Electromagnetic field stimulation for treating delayed union or non-union of long bone fractures in adults. Cochrane Database Syst Rev. 2011;(4):CD008471. [28] DAISH C, BLANCHARD R, FOX K, et al. The Application of Pulsed Electromagnetic Fields (PEMFs) for Bone Fracture Repair: Past and Perspective Findings. Ann Biomed Eng. 2018;46(4):525-542. [29] BAHNEY CS, ZONDERVAN RL, ALLISON P, et al. Cellular biology of fracture healing. J Orthop Res. 2019;37(1):35-50. [30] CALIOGNA L, BINA V, BRANCATO AM, et al. The Role of PEMFs on Bone Healing: An In Vitro Study. Int J Mol Sci. 2022;23(22):14298. [31] YANG J, ZHANG X, LIANG W, et al. Efficacy of adjuvant treatment for fracture nonunion/delayed union: a network meta-analysis of randomized controlled trials. BMC Musculoskelet Disord. 2022;23(1):481. [32] SISKEN BF, WALKER J, ORGEL M. Prospects on clinical applications of electrical stimulation for nerve regeneration. J Cell Biochem. 1993;51(4): 404-409. [33] TANDON B, BLAKER JJ, CARTMELL SH. Piezoelectric materials as stimulatory biomedical materials and scaffolds for bone repair. Acta Biomater. 2018; 73:1-20. [34] 王震,张岩,朱彦丞,等.骨髓炎辅助治疗的研究进展[J].中华创伤骨科杂志,2016,18(12):1064-1068. [35] DÍAZ-DEL CERRO E, VIDA C, MARTÍNEZ DE TODA I, et al. The use of a bed with an insulating system of electromagnetic fields improves immune function, redox and inflammatory states, and decrease the rate of aging. Environ Health. 2020;19(1):118. [36] BAYIR E, BILGI E, ŞENDEMIR-ÜRKMEZ A, et al. The effects of different intensities, frequencies and exposure times of extremely low-frequency electromagnetic fields on the growth of Staphylococcus aureus and Escherichia coli O157:H7. Electromagn Biol Med. 2015;34(1):14-18. [37] 李旭纲,陈一心,邱旭升.低能量脉冲电磁场对金黄色葡萄球菌生长的影响及其与抗生素的协同抑菌作用[J].中国医学物理学杂志,2021, 38(10):1294-1298. [38] WANG T, YANG L, JIANG J, et al. Pulsed electromagnetic fields: promising treatment for osteoporosis. Osteoporos Int. 2019;30(2):267-276. [39] 张璟岚,张滨婧,陈艺菲,等.磁场对间充质干细胞成骨的生物学效应[J].中国组织工程研究,2023,27(1):145-151. [40] BRILL AL, EHRLICH BE. Polycystin 2: A calcium channel, channel partner, and regulator of calcium homeostasis in ADPKD. Cell Signal. 2020;66:109490. [41] HE Y, YANG M, CHEN Z, et al. Effect of polycystin2 on differentiation and maturation of osteoblasts promoted by low-frequency pulsed electromagnetic fields. Sheng Wu Gong Cheng Xue Bao. 2022;38(3):1159-1172. [42] 肖豪,刘静,周君.脉冲电磁场治疗绝经后骨质疏松症的研究进展[J].中国组织工程研究,2022,26(8):1266-1271. [43] 孙智路,尹剑,黄开亮,等.脉冲电磁场抗骨质疏松作用及对Smurf1表达的影响[J].中国骨质疏松杂志,2020,26(8):1093-1097. [44] 刘勇华,涂萍,吴和平.脉冲电磁场辅助治疗原发性骨质疏松症疗效分析[J].中国骨质疏松杂志,2014,20(3):275-278. [45] ZHU S, LI Y, WANG L, et al. Pulsed Electromagnetic Fields May Be Effective for the Management of Primary Osteoporosis: A Systematic Review and Meta-Analysis. IEEE Trans Neural Syst Rehabil Eng. 2022;30:321-328. [46] 钱光,余月明,董有海,等.脉冲电磁场磁疗与骨硬化蛋白单克隆抗体联合干预绝经后新西兰大白兔骨代谢和骨微结构的变化[J].中国组织工程研究,2020,24(20):3130-3134. [47] 吴智敏,吴明,胡关彪,等.淫羊藿总黄酮联合低频脉冲电磁场治疗骨质疏松性桡骨远端骨折[J].中国新药与临床杂志,2019,38(11):665-668. [48] 李兰,周君.骨关节炎的物理治疗进展[J].中国康复理论与实践,2019, 25(8):918-921. [49] WANG T, XIE W, YE W, et al. Effects of electromagnetic fields on osteoarthritis. Biomed Pharmacother. 2019;118:109282. [50] LETIZIA MAURO G, SCATURRO D, GIMIGLIANO F, et al. Physical Agent Modalities in Early Osteoarthritis: A Scoping Review. Medicina (Kaunas). 2021;57(11):1165. [51] 柏中喜,方兴刚,马龙祥,等.脉冲电磁场通过Wntβ-catenin信号通路改善膝骨关节炎大鼠炎症反应的机制[J].中国医学物理学杂志,2022, 39(5):617-622. [52] LI S, YU B, ZHOU D, et al. Electromagnetic fields for treating osteoarthritis. Cochrane Database Syst Rev. 2013;(12):CD003523. [53] RYANG WE S, KOOG YH, JEONG KI, et al. Effects of pulsed electromagnetic field on knee osteoarthritis: a systematic review. Rheumatology (Oxford). 2013;52(5):815-824. [54] 向小娜,余曦,刘岩,等.富血小板血浆与脉冲电磁场联合应用治疗骨关节炎:理论与临床研究的进展[J].中国组织工程研究,2019,23(30): 4868-4874. [55] 郑晓芬.骨关节炎发病机制和治疗的最新进展[J].中国组织工程研究, 2017,21(20):3255-3262. [56] ALRAWASHDEH W, ESCHWEILER J, MIGLIORINI F, et al. Effectiveness of total knee arthroplasty rehabilitation programmes: A systematic review and meta-analysis. J Rehabil Med. 2021;53(6):jrm00200. [57] FRANKLIN PD, LI W, AYERS DC. The Chitranjan Ranawat Award: functional outcome after total knee replacement varies with patient attributes. Clin Orthop Relat Res. 2008;466(11):2597-2604. [58] ADRAVANTI P, NICOLETTI S, SETTI S, et al. Effect of pulsed electromagnetic field therapy in patients undergoing total knee arthroplasty: a randomised controlled trial. Int Orthop. 2014;38(2):397-403. [59] GARVING C, JAKOB S, BAUER I, et al. Impingement Syndrome of the Shoulder. Dtsch Arztebl Int. 2017;114(45):765-776. [60] GALACE DE FREITAS D, MARCONDES FB, MONTEIRO RL, et al. Pulsed electromagnetic field and exercises in patients with shoulder impingement syndrome: a randomized, double-blind, placebo-controlled clinical trial. Arch Phys Med Rehabil. 2014;95(2):345-352. [61] ROSS CL, ANG DC, ALMEIDA-PORADA G. Targeting Mesenchymal Stromal Cells/Pericytes (MSCs) With Pulsed Electromagnetic Field (PEMF) Has the Potential to Treat Rheumatoid Arthritis. Front Immunol. 2019;10:266. [62] AL-JABRI T, TAN JYQ, TONG GY, et al. The role of electrical stimulation in the management of avascular necrosis of the femoral head in adults: a systematic review. BMC Musculoskelet Disord. 2017;18(1):319. [63] HINES JT, JO WL, CUI Q, et al. Osteonecrosis of the Femoral Head: an Updated Review of ARCO on Pathogenesis, Staging and Treatment. J Korean Med Sci. 2021;36(24):e177. [64] 洪志楠,何伟,魏秋实,等.股骨头坏死物理治疗的研究进展[J].中国矫形外科杂志,2017,25(23):2160-2164. [65] EWURUM CH, GUO Y, PAGNHA S, et al. Surgical Navigation in Orthopedics: Workflow and System Review. Adv Exp Med Biol. 2018;1093:47-63. [66] BLYTH MJ, SMITH JR, ANTHONY IC, et al. Electromagnetic navigation in total knee arthroplasty-a single center, randomized, single-blind study comparing the results with conventional techniques. J Arthroplasty. 2015;30(2):199-205. [67] MIHALIČ R, ZDOVC J, MOHAR J, et al. Electromagnetic navigation system for acetabular component placement in total hip arthroplasty is more precise and accurate than the freehand technique: a randomized, controlled trial with 84 patients. Acta Orthop. 2020;91(6):675-681. [68] 李军,詹俊锋,许新忠,等.一种新型导航系统在胫骨髓内钉远端锁钉应用中的评价[J].中国组织工程研究,2017,21(27):4342-4347. [69] CHAN DS, BURRIS RB, ERDOGAN M, et al. The insertion of intramedullary nail locking screws without fluoroscopy: a faster and safer technique. J Orthop Trauma. 2013;27(7):363-366. |
| [1] | Guo Sutong, Feng Dehong, Guo Yu, Wang Ling, Ding Yujian, Liu Yi, Qian Zhengying, Li Mingyang. Construction and finite element analysis of normal and osteoporotic hip models [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1342-1346. |
| [2] | Li Yongjie, Fu Shenyu, Xia Yuan, Zhang Dakuan, Liu Hongju. Correlation of knee extensor muscle strength and spatiotemporal gait parameters with peak knee flexion/adduction moment in female patients with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1354-1358. |
| [3] | Qi Haodong, Lu Chao, Xu Hanbo, Wang Mengfei, Hao Yangquan. Effect of diabetes mellitus on perioperative blood loss and pain after primary total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1383-1387. |
| [4] | Du Changling, Shi Hui, Zhang Shoutao, Meng Tao, Liu Dong, Li Jian, Cao Heng, Xu Chuang. Efficacy and safety of different applications of tranexamic acid in high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1409-1413. |
| [5] | Zhang Xiaoyun, Liu Hua, Chai Yuan, Chen Feng, Zeng Hao, Gao Zhengang, Huang Yourong. Effect of Yishen Gushu Formula on bone metabolic markers and clinical efficacyn in patients with osteoporosis of kidney deficiency and blood stasis type [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1155-1160. |
| [6] | Huang Xiarong, Hu Lizhi, Sun Guanghua, Peng Xinke, Liao Ying, Liao Yuan, Liu Jing, Yin Linwei, Zhong Peirui, Peng Ting, Zhou Jun, Qu Mengjian. Effect of electroacupuncture on the expression of P53 and P21 in articular cartilage and subchondral bone of aged rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1174-1179. |
| [7] | Zhao Garida, Ren Yizhong, Han Changxu, Kong Lingyue, Jia Yanbo. Mechanism of Mongolian Medicine Erden-uril on osteoarthritis in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1193-1199. |
| [8] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| [9] | Li Rui, Zhang Guihong, Wang Tao, Fan Ping. Effect of ginseng polysaccharide on the expression of prostaglandin E2/6-keto-prostaglandin 1alpha in traumatic osteoarthritis model rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1235-1240. |
| [10] | Zhang Min, Peng Jing, Zhang Qiang, Chen Dewang. Mechanical properties of L3/4 laminar decompression and intervertebral fusion in elderly osteoporosis patients analyzed by finite element method [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 847-851. |
| [11] | Xue Xiaofeng, Wei Yongkang, Qiao Xiaohong, Du Yuyong, Niu Jianjun, Ren Lixin, Yang Huifeng, Zhang Zhimin, Guo Yuan, Chen Weiyi. Finite element analysis of osteoporosis in proximal femur after cannulated screw fixation for femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 862-867. |
| [12] | Kaiyisaier•Abudukelimu, Maimaitimin•Abulimiti, Li Lei, Yang Xiaokai, Zhang Yukun, Liu Shuai. Effect of lumbar CT values in the diagnosis of osteoporosis in women patients with lumbar degenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 945-949. |
| [13] | Wang Liping, Lian Tianxing, Hu Yongrong, Yang Hongsheng, Zeng Zhimou, Liu Hao, Qu Bo. HU value of chest CT vertebral body in the opportunistic screening of type 2 diabetes mellitus osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 950-954. |
| [14] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [15] | Zhang Zeyi, Yang Yimin, Li Wenyan, Zhang Meizhen. Effect of foot progression angle on lower extremity kinetics of knee osteoarthritis patients of different ages: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 968-975. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||