Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (2): 308-314.doi: 10.12307/2023.875
Previous Articles Next Articles
Role and mechanism of intestinal flora metabolites in obesity regulation
Yang Qihang1, Pu Rui1, 2, Chen Ziyang1, 2, Leng Siyi1, Song Yongjing1, Liu Hui3, Du Guangyou1
- 1College of Education and Sports Sciences, 2Human Science Laboratory of Exercise, 3Health Science Center, Yangtze University, Jingzhou 434023, Hubei Province, China
-
Received:2023-01-03Accepted:2023-02-02Online:2024-01-18Published:2023-06-30 -
Contact:Du Guangyou, PhD, Associate professor, Master’s supervisor, College of Education and Sports Sciences, Yangtze University, Jingzhou 434023, Hubei Province, China Pu Rui, Master, Lecturer, College of Education and Sports Sciences, Yangtze University, Jingzhou 434023, Hubei Province, China; Human Science Laboratory of Exercise, Yangtze University, Jingzhou 434023, Hubei Province, China -
About author:Yang Qihang, Master candidate, College of Education and Sports Sciences, Yangtze University, Jingzhou 434023, Hubei Province, China -
Supported by:National Natural Science Foundation of China, No. 82271514 (to LH); the Ministry of Education Humanities and Social Sciences Research Foundation Project, No. 17YJA890004 (to DGY); Key Project of Social Science Fund of Education Department of Hubei Province, No. 16D024 (to DGY)
CLC Number:
Cite this article
Yang Qihang, Pu Rui, Chen Ziyang, Leng Siyi, Song Yongjing, Liu Hui, Du Guangyou. Role and mechanism of intestinal flora metabolites in obesity regulation[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 308-314.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
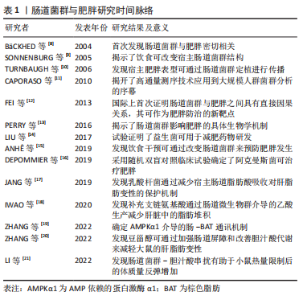
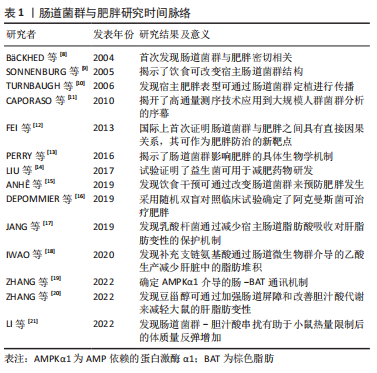
2.1 肠道菌群及其代谢物概述 肠道菌群是由细菌、真菌、古细菌、真核生物和病毒组成的复杂群落,在健康人体肠道内约有100万亿个微生物和1 000多种细菌[3-4]。近年来随着鸟枪法宏基因组分析的发展,研究者可从复杂的微生物群落推断其分类和功能等信息指导表型研究了解肠道菌群在促进人类健康和改善疾病中的作用。人体肠道菌群主要以厚壁菌门、拟杆菌门、放线菌门、变形菌门和疣微菌门几大类为主[5]。在肠道和机体组织器官内发现了数千种微生物代谢物[6]。研究表明,肠道菌群在促进人类健康和改善疾病方面是通过其代谢物作为信号调节因子参与机体内的生理进程而实现的。新近的研究发现,肠道菌群代谢物短链脂肪酸、胆汁酸、脂多糖、氧化三甲胺、中链脂肪酸以及色氨酸衍生物等不仅能促进人体营养物质的消化吸收,还可在维持机体代谢稳态和免疫调节等多个方面扮演重要角色。此外,肠道菌群代谢物的产生受遗传、饮食、生活方式、环境和抗生素等因素影响[7-8]。文章通过对肠道菌群代谢物调控不同生理机制介导肥胖的发生发展进行归纳总结[8-21],见表1。"
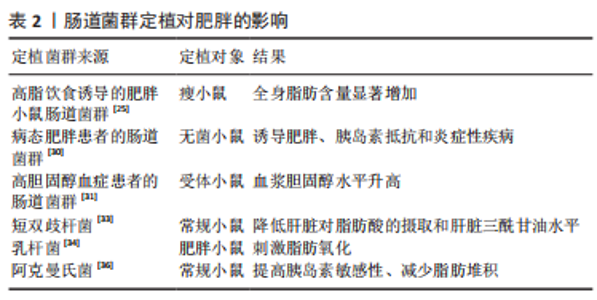
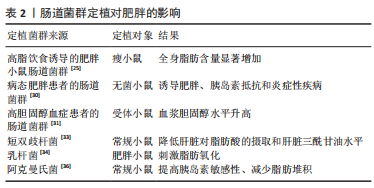
肠道菌群及其代谢物对宿主影响至关重要,肠道菌群以肠道菌群代谢物为调节因子介导参与宿主的新陈代谢和免疫反应等不同生理进程,进而与宿主健康和疾病保持密切联系,但同时又受到内外部环境及其他多种因素的影响。在未来的研究中可利用鸟枪法宏基因组、无菌对照实验和特定菌群定植等技术以肠道菌群为研究中心进行扩展和延伸,更加深入地探索肠道菌群及其代谢物对于宿主健康和防治疾病的健康效益。 2.2 肠道菌群与肥胖 研究证明,肠道菌群在肥胖的发生发展中扮演着重要角色。因此,阐明肥胖与肠道菌群的关系,揭示肠道菌群代谢物调控肥胖的生物学机制对肥胖的防治和提高机体健康水平具有重要意义。 2.2.1 肥胖患者的肠道菌群特征 肥胖会诱导机体肠道菌群发生变化,与正常人相比,肥胖人群的肠道菌群多样性和相对丰度较低[22]。YAN等[23]对41名正常体质量指数受试者进行肠道菌群测序,发现有16种细菌与内脏肥胖之间存在较强的相关性,其中大肠杆菌最强。厚壁菌门和拟杆菌门是人体肠道菌群内最丰富的两个菌门,厚壁菌门是革兰阳性细菌,被称为“肥胖微生物”,主要包括梭菌、乳酸杆菌和和长链多尔氏菌;拟杆菌门被称为“瘦微生物”,主要包括拟杆菌、普雷沃氏菌和脱硫杆菌[24]。 对遗传肥胖大鼠和瘦小鼠的肠道菌群进行分析,发现肥胖大鼠肠道菌群较瘦小鼠相比拟杆菌比值较低,厚壁菌比值较高,导致厚壁菌与拟杆菌比值增加,同时对肥胖人群的肠道菌群分析也发现同样的比值关系[25]。有学者认为,体脂增加或肥胖倾向可能由厚壁菌与拟杆菌比值决定,其比值变化可作为代谢综合征和肥胖诊断的生物标记物[26]。但另有研究发现,在超重或肥胖儿童肠道内拟杆菌丰度增加,而厚壁菌仅在一些队列中减少[27]。还有一项系统评价表明,厚壁菌与拟杆菌比值与儿童超重和肥胖无关,这可能与饮食、环境、人群和研究方法的不同有关[28]。进一步研究发现,在肥胖儿童体内厚壁菌的丰度与血清肿瘤坏死因子α水平呈正相关,表明厚壁菌丰度与肥胖炎症反应密切相关[29]。由此可见,肥胖的发生会导致机体肠道菌群发生改变,同时也证明肠道菌群的改变可作为肥胖诊断的生物标记物。 2.2.2 肠道菌群失调可诱发肥胖形成 肠道菌群的多样性下降和丰度降低均与肥胖风险的增加相关。与肠道菌群高丰度群体相比,拥有较低肠道菌群丰度的群体更易患有肥胖、血脂和代谢异常等特征的代谢性疾病,且较低肠道菌群丰度的肥胖个体会随着时间的推移伴随着体指数的增加。此外,各种不同菌群经常从不同环境进入到人体,并能在一定部位定居、不断生长、繁殖后代,这种现象通常称为“菌群定植”,而肥胖会通过肠道菌群定植进行传播。研究者将高脂饮食诱导的肥胖小鼠肠道菌群定植到瘦小鼠体内,发现定植后瘦小鼠可从食物中摄取更多的能量,从而导致全身脂肪含量显著增加,提示肠道菌群移植可诱发肥胖形成[25]。YAN等[30]将病态肥胖患者的肠道菌群移植到无菌小鼠肠道内后进行高脂饮食干预,发现与炎症、脂质、脂蛋白和甾醇代谢相关的过氧化物酶体增殖物激活受体的表达显著上调,进而诱导无菌小鼠出现肥胖、胰岛素抵抗和炎症性疾病。此外,将高胆固醇血症患者的肠道菌群移植到受体小鼠中,发现小鼠血浆胆固醇水平显著升高,并抑制肝胆固醇的合成[31]。上述研究表明,肥胖可通过肠道菌群定植进行传播,导致肠道菌群失调,从而诱发肥胖形成。 2.2.3 益生菌定植可延缓肥胖发生 益生菌定植作为调节机体肠道菌群稳态的一种形式可持续降低超重或肥胖患者的体质量和体质量指数,进而延缓肥胖的发生发展[32]。补充益生菌短双歧杆菌可下调脂肪酸转位酶和脂肪酸结合蛋白1的表达,并抑制脂肪酸向肝脏的运输和摄取,从而降低肝脏三酰甘油水平[33]。KIM等[34]研究发现,肠道益生菌乳杆菌丰度与大鼠的脂肪量和体质量呈负相关,定植乳杆菌可直接降低管腔内胆固醇含量,上调附睾脂肪组织中的转录因子过氧化物酶体增殖物激活受体α(peroxisome proliferators-activated receptor α,PPAR-α)、脂肪酸结合蛋白4 和肉碱棕榈酰转移酶Ⅰ的表达,并刺激脂肪氧化进而延缓肥胖形成。另有研究发现,补充大肠杆菌也可促进肠细胞中的脂肪氧化[35]。 最近的研究发现,阿克曼氏菌可上调机体胰岛素敏感性并减少肝脏损伤标志物的表达,从而抑制脂肪堆积,最终延缓1型糖尿病和肥胖等疾病的发展[36-37]。另有研究也证明了梭状芽胞杆菌及其代谢物对脂质吸收具有重要作用[38]。此外,益生菌定植有助于恢复上皮细胞之间的紧密联系,从而降低肠道通透性,还可增加肠道中瘦素、胰高糖素样肽-1和胰腺多肽的表达,从而提高饱腹感[39]。肝细胞核转录因子是肠道吸收膳食脂质的关键因子,可通过抑制转录因子肝细胞核转录因子与顺式调节DNA的序列特异性结合进而调节脂质代谢[40]。上述研究提示,短双歧杆菌、乳杆菌和阿克曼氏菌等益生菌定植可调节肠道菌群稳态,从而减少肝脏对脂肪酸的摄取、刺激脂肪氧化、上调胰岛素敏感性并提高饱腹感,进而抑制肥胖的形成,见表2。"

综上所述,肠道菌群与肥胖相互影响且关系密切。现有的研究已证明,肥胖的发生可伴随着肠道菌群的改变,如菌群多样性和丰度降低等为特征的稳态失调以及特定菌群比值的变化,故肠道菌群可作为肥胖诊断的生物标记物;此外,肠道菌群失调可诱发肥胖,而通过短双歧杆菌、乳杆菌和阿克曼氏菌等益生菌定植可防治肥胖。在后续研究中可探索益生菌联合饮食、运动或药物干预等方式的可行性方案,从而为提高肥胖的防治效果提供新的策略。 2.3 肠道菌群代谢物在肥胖中的调控作用 肠道菌群代谢物可通过调节血浆脂质水平影响宿主的代谢状态。研究表明,补充某些特定菌群代谢物可靶向调节细胞内的脂质代谢,进而治疗肥胖以及营养不良等代谢性疾病[41],如短链脂肪酸、胆汁酸、脂多糖、氧化三甲胺、中链脂肪酸和色氨酸衍生物等肠道菌群代谢物。 2.3.1 短链脂肪酸 也称为挥发性脂肪酸,是碳少于六种的有机线性羧酸,主要包括乙酸、丙酸和丁酸。短链脂肪酸主要是未被消化的膳食纤维在盲肠和大肠中经厌氧菌群发酵产生,其中拟杆菌(革兰阴性菌)主要产生乙酸盐和丙酸盐,而厚壁菌(革兰阳性菌)主要产生丁酸盐,根据饮食、微生物组的类型和数量以及在肠道中的停留时间等因素的评估,肠道每天产生500-600 mmol的短链脂肪酸[42-43]。 有研究发现,短链脂肪酸不仅能作为能量底物为宿主提供能量,还可作为调控脂质代谢的重要调节因子[44]。乙酸盐和丁酸盐可在肠上皮细胞内转化为乙酰辅酶A进入三羧酸循环产生能量。此外,乙酸盐还可作为合成棕榈酸酯和硬脂酸酯的前体来促进肝脏脂肪酸代谢,并在胞质酶系统的作用下生成棕榈酸转移到线粒体以延长碳链并与脂肪组织中储存的其他物质形成三酰甘油[45-46]。 短链脂肪酸可通过G蛋白偶联受体(G protein-coupled receptors,GPCRs)和过氧化物酶体增殖物激活受体γ(peroxisome proliferators-activated receptor γ,PPARγ)调控周围代谢组织的能量消耗和胰岛素敏感性,进而影响肥胖的发展。有研究发现,补充膳食短链脂肪酸可激活GPR41的表达改善肝脏代谢功能,从而抑制肝脏质量增加和脂质合成,延缓由小鼠高脂肪饮食诱导的肥胖发生[47]。邓邦利[48]研究发现,短链脂肪酸可促进小鼠肝细胞和内皮细胞AMP依赖的蛋白激酶[Adenosine 5‘- monophosphate (AMP)-activated protein kinase,AMPK]和乙酰辅酶A羧化酶(Acetyl CoA carboxylase,ACC)的磷酸化,增加脂肪组织和骨骼肌中过氧化物酶体增殖物激活受体γ的辅激活因子α(peroxisome proliferator-activated receptor γ coactivator 1α,PGC-1α)的表达,并调节转录因子PPAR-α和PPARγ的活性,进而改善脂质代谢紊乱。此外,短链脂肪酸可抑制PPARγ的表达,并激活线粒体解偶联蛋白2-AMPK-ACC信号通路,从而刺激肝脏和脂肪组织中的氧化代谢[49]。上述研究提示,短链脂肪酸可通过调节GPR41和PPARγ等信号通路改善肝脏代谢功能和调控脂肪生成,进而延缓肥胖的发生。但在上述研究中所采用的短链脂肪酸为乙酸、丙酸和丁酸的组合,现阶段受研究方法的制约,无法确定单一短链脂肪酸调控肥胖的具体生物学机制。进一步研究发现不同短链脂肪酸调控肥胖的途径不同,甚至同一短链脂肪酸经不同信号通路调控肥胖的作用呈现出矛盾性。 乙酸:PERRY等[13]通过建立肥胖小鼠模型,发现高脂饮食喂养会增加小鼠肠道内乙酸盐的含量,进而激活副交感神经系统,促进生长素释放肽(growth hormone secretagogue receptor,GHSR-1a)和葡萄糖刺激的胰岛素分泌的增加,从而产生正反馈回路,诱导食欲亢进、肝脏和骨骼肌中的脂质沉积并刺激胰岛素抵抗的发生,进而诱发肥胖。这一研究提示在高脂饮食影响下,乙酸可诱导副交感神经系统激活,导致食欲亢进和肝脏胰岛素抵抗,进而加速肥胖的生理进程。但另有研究发现,由高脂饮食喂养导致的肥胖小鼠补充乙酸盐可通过恢复肥胖抑素以及葡萄糖-6-磷酸脱氢酶-谷胱甘肽依赖性抗氧化系统功能来改善高脂饮食诱发的脂肪和肝脏代谢紊乱[50]。这两项研究表明,在高脂饮食所诱导产生的肥胖影响下,乙酸对肥胖的调控结果不同,这可能与乙酸的来源、实验对象、周期和环境不同有关。 此外,乙酸可通过其他信号通路影响肥胖的发生发展。 研究发现,乙酸可激活GPR43介导G(i/o)βγ-磷脂酶C-蛋白激酶 C信号通路,上调抑癌基因的表达并抑制蛋白激酶B磷酸化,提高全身胰岛素敏感性和改善白色脂肪组织内的脂肪堆积以及调控骨骼肌和肝脏等器官的脂质和葡萄糖代谢,提高其能量利用率,进而延缓肥胖的发生[51]。 乙酸还可通过AMPK/PGC-1α/PPARα途径促进脂质氧化[35]。FROST等[52]研究发现,乙酸还可介导脑肠轴影响肥胖,结肠内的乙酸盐通过谷氨酸-谷氨酰胺跨细胞循环和增强下丘脑控制的厌食信号传导,并增加乳酸和下丘脑中γ-氨基丁酸的分泌,提示乙酸能直接激活中枢食欲调节,抑制食欲减轻体质量。由此可见,乙酸可抑制蛋白激酶B磷酸化和介导相关信号通路,调节中枢食欲,促进脂肪酸氧化,进而延缓肥胖的进程。 丙酸:丙酸可通过激活GPR43,GPR41和GPR109A活化等不同途径增加葡萄糖耐受量、抑制食欲和增加机体能量消耗,从而影响肥胖的发生发展。FORBES等[53]发现丙酸盐可激活GPR43活化,显著刺激肠内分泌L细胞释放厌食肠道激素肽YY和胰高血糖素样肽1,抑制肠道运输,增加葡萄糖耐量,从而降低食欲。丙酸盐还可激活GPR41信号,促进Ca2+介导胰岛素分泌I/O敏感通路的表达,进而增加机体能量消耗[54]。另有研究发现,在脂肪组织中,丙酸可激活GPR109A活化刺激脂肪酶活性,降低血浆三酰甘油水平和游离脂肪酸水平[55]。上述研究提示,丙酸可通过多种途径促进能量消耗和抑制食欲,从而延缓肥胖的发生。 丁酸:现阶段的研究表明,丁酸可通过调节不同信号通路加速或延缓肥胖的病理进程。丁酸盐通过激活P38蛋白、细胞外调节蛋白激酶、c-Jun氨基末端激酶和IκB 激酶/核转录因子κB等信号通路抑制脂肪细胞的脂解,进而抑制巨噬细胞诱发的炎症反应[56]。由丁酸盐激活活化的GPR43可在白色脂肪细胞内联合抑制性偶联-磷脂酶C-蛋白激酶 C信号而抑制蛋白激酶B磷酸化并降低胰岛素敏感性,从而导致脂肪堆积。此外,GPR43在白色脂肪细胞内可联合抑制性偶联-环磷酸腺苷-蛋白激酶A信号,抑制三酰甘油分解相关酶(如激素敏感脂肪酶和脂肪甘油三酯脂肪酶)的活性,从而减少白色脂肪细胞中的脂肪分解[56-57]。上述研究提示,丁酸可抑制脂肪细胞分解,进而加速肥胖的进程。但另有研究得出不同结果,丁酸可激活GPR41和GPR43的表达从而产生有益效应,从而延缓肥胖的病理进程。丁酸可介导 GPR41和GPR43上调肝细胞中的钙/钙调蛋白依赖性蛋白激酶Ⅱ表达和抑制Hep1-6细胞中的组蛋白去乙酰化酶1表达,激活转录增强因子环磷腺苷效应元件结合蛋白磷酸化,进而改善肝脏脂肪变性、脂质代谢异常和延缓肥胖的发生[58]。此外,LI等[59]进行高脂饮食与丁酸盐+高脂饮食组合的为期9周对比实验干预,发现补充丁酸盐可降低神经肽Y的食欲性神经元、脑干孤束核和迷走神经背侧复合体的神经元活动,进而抑制食欲。丁酸盐还可促进脂肪酸氧化和激活棕色脂肪组织,减少能量摄入进而延缓肥胖发生。 综上所述,丁酸经不同信号通路,对于肥胖的调控具有差异性。丁酸可通过降低炎症因子表达,抑制脂肪细胞脂解,进而加速肥胖的生理病程;而另有研究发现丁酸介导GPR41、GPR43和降低神经肽Y的食欲性神经元活动,进而抑制肥胖发展。不同短链脂肪酸调控肥胖的路径机制见图3。"


2.3.2 胆汁酸(Bile Acids,BAs) 胆汁酸可作为脂质代谢的重要调节因子,可通过激活胆汁酸G蛋白偶联受体5(takeda G protein-couplereceptor-5,TGR5)和法尼醇X受体(farnesoid X receptor,FXR)的活化,进而延缓肥胖发生并改善高胆固醇血症[60]。胆汁酸可激活FXR和蛋白酪氨酸磷酸酶的活化,抑制肝X受体/肝受体同源物1的表达,进而介导调控甾醇调节胆固醇调节元件结合蛋白1c的表达,胆汁酸也可通过调控细胞内胆固醇含量间接调节胆固醇调节元件结合蛋白1c的表达,从而抑制脂肪合成。此外,FXR可通过上调载脂蛋白C2和载脂蛋白C3的表达,调节脂蛋白脂肪酶的活性,进而调控白色脂肪组织和骨骼肌等各种组织中脂质水解[61-62]。上述研究提示,胆汁酸可通过促进FXR活化,抑制脂肪合成和促进脂质水解,进而缓解肥胖的形成。另有研究表明,胆汁酸介导TGR5激活丝裂原活化蛋白激酶信号通路的传导,促进细胞内甲状腺激素的表达和刺激小肠中胰高血糖素样肽1的分泌,进而改善胰岛素敏感性和骨骼肌中棕色脂肪组织的能量消耗[61]。这一研究提示,胆汁酸可改善胰岛素敏感性和机体组织的能量消耗,进而延缓肥胖的发生。 2.3.3 脂多糖 脂多糖是革兰阴性细菌的细胞壁成分,在细菌死亡后分离并被肠道毛细血管吸收。研究发现,肥胖和高脂饮食可增加肠道内革兰阴性细菌丰度,导致血浆中脂多糖水平升高,进而诱发炎症反应并引发代谢性内毒素血症[63-64]。 CANI等[65]发现对小鼠皮下注射脂多糖会引发类似高脂饮食喂养的代谢反应,从而导致小鼠脂肪组织和肝脏中脂质堆积,产生胰岛素抵抗并诱发肥胖。进一步研究发现其机制可能与脂多糖通过与受体复合物(脂多糖受体CD14/Toll样受体4/可溶性髓样因子)结合来激活免疫系统,显著上调白细胞介素和肿瘤坏死因子表达,并抑制酪氨酸激酶活性和减弱胰岛素受体信号传导有关[66]。研究发现,脂多糖与Toll样受体4结合可通过上调脂肪细胞和巨噬细胞中c-Jun N末端激酶表达和激活核转录因子κB等不同途径诱发炎症反应,诱导肥胖发生[67]。另有研究表明,代谢性内毒素血症可通过脂多糖受体CD14依赖性方式直接刺激脂肪细胞的增生和促进激活素A的分泌,从而加速脂肪细胞的前体细胞增殖进程[68]。上述研究提示,脂多糖的上调可诱发炎症反应和胰岛素抵抗、并刺激脂肪细胞增生和促进激活素A分泌,进而促进肥胖的形成。由此可见,抑制脂多糖的表达可抑制胰岛素抵抗和脂质堆积,可能成为改善肥胖的新方向。而短链脂肪酸可通过不同途径发挥抗炎作用,乙酸盐可通过激活GPR43受体表达抑制脂多糖诱导促炎细胞因子肿瘤坏死因子α的增加,改善胰岛素敏感性[69]。丙酸盐和丁酸盐可降低由脂多糖刺激中性粒细胞肿瘤坏死因子α分泌水平、下调组蛋白脱乙酰酶表达和抑制核转录因子κB信号通路活化[70]。提示短链脂肪酸可抑制脂多糖诱发的炎症反应和改善胰岛素敏感性,从而发挥抑制肥胖的关键作用。 2.3.4 氧化三甲胺 氧化三甲胺是三甲胺经肝黄素单加氧酶代谢生 成[71]。研究发现,大肠杆菌、鼠伤寒沙门氏菌和长双歧杆菌代谢产生的氧化三甲胺前体5-氨基戊酸甜菜碱可影响线粒体的肉碱穿梭系统,并减弱肝细胞氧化脂肪酸的能力,进而导致脂质积累[72]。SHIH等[73]研究发现,肝脏肝黄素单加氧酶在人肝癌细胞系Hep3B中的过表达可显著增加葡萄糖分泌和脂肪生成,此研究提示FMO3在调节体内葡萄糖代谢和脂质稳态中发挥重要作用。此外,氧化三甲胺可抑制合成胆汁酸的两种限速酶(CYP7A1和甾醇27 α-羟化酶)的活性,并下调肝脏中胆汁酸转运蛋白的表达,从而导致脂肪堆积促进肥胖发生[74]。另有研究发现,补充氧化三甲胺会减弱葡萄糖耐受性,阻碍肝胰岛素信号传导,并导致喂食高脂肪饮食的小鼠脂肪组织炎症发生[75]。上述研究提示,不同肠道菌群代谢物之间存在相互联系,氧化三甲胺及其相关代谢酶可影响肝脏线粒体的肉碱穿梭系统,并抑制胆汁酸合成与运输和阻碍肝胰岛素信号的传导,进而减弱肝细胞氧化脂肪酸的能力,从而促进脂质沉积,为肥胖的防治提供了新的方向。 益生菌定植可改善由氧化三甲胺引起的脂质代谢紊乱。LIANG等[76]建立高胆碱动物模型,发现动物双歧杆菌乳酸亚种F1-3-2、动物双歧杆菌乳酸杆菌亚种F1-7和植物乳杆菌F3-2等益生菌定植可显著降低血清TMA和氧化三甲胺水平,并通过调节胆固醇7α-羟化酶和3号染色体的基因表达,促进胆汁酸和胆固醇代谢,减少肝脂积累。由此可见,益生菌定植可改善由氧化三甲胺引起的脂质代谢紊乱,减少肝脂沉积,进而延缓肥胖发生。 2.3.5 中链脂肪酸 中链脂肪酸作为能量来源可直接通过胃壁吸收。最近的研究表明,己酸菌、假拉米杆菌和振荡杆菌等可产生中链脂肪酸[77]。ZHAO等[78]研究发现,中链脂肪酸可通过GPR43上调claudin-3的表达和超氧化物歧化酶的活性,下调血浆中白细胞介素1β和干扰素γ水平,促进机体能量消耗,提示中链脂肪酸可加速机体能量消耗延缓肥胖。此外,GREGOR 等[79]研究发现,中链脂肪酸可增加脂肪转录因子PPARγ的表达,从而促进脂肪合成。癸酸可促进GPR84的活化并上调过氧化物酶体增殖物激活受体的表达,诱发免疫炎症反应和刺激脂肪组织分化[80]。VAN DE HEIJNING等[81]研究发现,在幼年期增加中链脂肪酸摄入水平可以防止成年期脂肪堆积。此外,中链脂肪酸还可抑制各种肠道病原微生物的生长,如李斯特菌、产气荚膜梭菌和大肠杆菌等[82-83]。上述研究提示,中链脂肪酸在调控肥胖过程中具有重要意义,可通过不同途径发挥防治肥胖的有益效益。但目前相关研究较少,需要对不同信号通路在肥胖中的诊疗作用进行深入研究。 2.3.6 色氨酸衍生物 吲哚化合物作为色氨酸衍生物中的一种,可调节宿主肠细胞和远端组织中基因表达能力[84]。研究发现,肥胖儿童血清中吲哚-3-乳酸和吲哚-3-乙酸的浓度显著增加,而吲哚代谢物可通过作用于扩展奖励网络(特别是杏仁核-伏隔核回路和杏仁核-前岛叶回路)来影响食物的摄入和肥胖的形成[85-86]。miR-181是通过PTEN调节磷脂酰肌醇 3-激酶信号强度的重要因子,可作为机体发育和调节内环境稳态过程中细胞代谢适应性的中枢调节剂[87]。研究发现,肠道菌群产生的色氨酸衍生物可调控小鼠白色脂肪细胞中miR-181家族的表达,进而下调能量消耗和胰岛素敏感性,当肠道菌群miR-181轴失调时,会诱发胰岛素抵抗和白色脂肪组织炎症发展,并最终导致小鼠肥胖发生[88]。上述研究提示,色氨酸衍生物通过作用于扩展奖励网络和调控脂肪细胞中miR-181基因表达,从而达到调控能量的摄入与消耗的目的。因此,肠道菌群miR-181轴可在未来肥胖的防治中扮演重要角色。不同肠道菌群代谢物在肥胖中的调控作用,见表3。"

| [1] JASTREBOFF AM, KOTZ CM, KAHAN S, et al. Obesity as a disease: the obesity society 2018 position statement. Obesity (Silver Spring). 2019;27(1):7-9. [2] BLüHER M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. [3] RINNINELLA E, RAOUL P, CINTONI M, et al. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms. 2019;7(1):752-766. [4] ALMEIDA A, MITCHELL AL, BOLAND M, et al. A new genomic blueprint of the human gut microbiota. Nature. 2019;568(7753):499-504. [5] SEGATA N, HAAKE SK, MANNON P, et al. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 2012;13(6):R42. [6] UCHIMURA Y, FUHRER T, LI H, et al. Antibodies set boundaries limiting microbial metabolite penetration and the resultant mammalian host response. Immunity. 2018;49(3):545-559.e5. [7] 钱映,冯月梅,汪艳姣,等.肠道菌群与慢性病关系的研究进展[J].中国微生态学杂志,2022,34(10):1217-1221. [8] BÄCKHED F, DING H, WANG T, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A. 2004;101(44):15718-15723. [9] SONNENBURG JL, XU J, LEIP DD, et al. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science. 2005;307(5717):1955-1959. [10] TURNBAUGH PJ, LEY RE, MAHOWALD MA, et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122): 1027-1031. [11] CAPORASO JG, KUCZYNSKI J, STOMBAUGH J, et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7(5):335-336. [12] FEI N, ZHAO L. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. ISME J. 2013;7(4):880-884. [13] PERRY RJ, PENG L, BARRY NA, et al. Acetate mediates a microbiome-brain-β-cell axis to promote metabolic syndrome. Nature. 2016;534(7606):213-217. [14] LIU R, HONG J, XU X, et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23(7):859-868. [15] ANHê FF, NACHBAR RT, VARIN TV, et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut. 2019;68(3):453-464. [16] DEPOMMIER C, EVERARD A, DRUART C, et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. 2019;25(7):1096-1103. [17] JANG HR, PARK HJ, KANG D, et al. A protective mechanism of probiotic Lactobacillus against hepatic steatosis via reducing host intestinal fatty acid absorption. Exp Mol Med. 2019;51(8):1-14. [18] IWAO M, GOTOH K, ARAKAWA M, et al. Supplementation of branched-chain amino acids decreases fat accumulation in the liver through intestinal microbiota-mediated production of acetic acid. Sci Rep. 2020;10(1):18768. [19] ZHANG E, JIN L, WANG Y, et al. Intestinal AMPK modulation of microbiota mediates crosstalk with brown fat to control thermogenesis. Nat Commun. 2022; 13(1):1135. [20] ZHANG Y, GU Y, JIANG J, et al. Stigmasterol attenuates hepatic steatosis in rats by strengthening the intestinal barrier and improving bile acid metabolism. NPJ Sci Food. 2022;6(1):38. [21] LI M, WANG S, LI Y, et al. Gut microbiota-bile acid crosstalk contributes to the rebound weight gain after calorie restriction in mice. Nat Commun. 2022;13(1):2060. [22] LE CHATELIER E, NIELSEN T, QIN J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500(7464):541-546. [23] YAN H, QIN Q, CHEN J, et al. Gut microbiome alterations in patients with visceral obesity based on quantitative computed tomography. Front Cell Infect Microbiol. 2021;11(8)232-262. [24] ZHANG S, DANG Y. Roles of gut microbiota and metabolites in overweight and obesity of children. Front Endocrinol (Lausanne). 2022;13(99):49-70. [25] LEY RE, TURNBAUGH PJ, KLEIN S, et al. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022-1023. [26] VAISERMAN A, ROMANENKO M, PIVEN L, et al. Differences in the gut Firmicutes to Bacteroidetes ratio across age groups in healthy Ukrainian population. BMC Microbiol. 2020;20(1):221. [27] INDIANI C, RIZZARDI KF, CASTELO PM, et al. Childhood obesity and firmicutes/bacteroidetes ratio in the gut microbiota: a systematic review. Child Obes. 2018; 14(8):501-509. [28] VANDER WYST KB, ORTEGA-SANTOS CP, TOFFOLI SN, et al. Diet, adiposity, and the gut microbiota from infancy to adolescence: a systematic review. Obes Rev. 2021;22(5):e13175. [29] BERVOETS L, VAN HOORENBEECK K, KORTLEVEN I, et al. Differences in gut microbiota composition between obese and lean children: a cross-sectional study. Gut Pathog. 2013;5(1):10. [30] YAN H, FEI N, WU G, et al. Regulated inflammation and lipid metabolism in colon mrna expressions of obese germfree mice responding to enterobacter cloacae b29 combined with the high fat diet. Front Microbiol. 2016;7(17):86-99. [31] LE ROY T, LéCUYER E, CHASSAING B, et al. The intestinal microbiota regulates host cholesterol homeostasis. BMC Biol. 2019;17(1):94. [32] BORGERAAS H, JOHNSON LK, SKATTEBU J, et al. Effects of probiotics on body weight, body mass index, fat mass and fat percentage in subjects with overweight or obesity: a systematic review and meta-analysis of randomized controlled trials. Obes Rev. 2018;19(2):219-232. [33] PATTERSON E, WALL R, LISAI S, et al. Bifidobacterium breve with α-linolenic acid alters the composition, distribution and transcription factor activity associated with metabolism and absorption of fat. Sci Rep. 2017;7(43):30-40. [34] KIM DH, JEONG D, KANG IB, et al. Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: direct reduction of cholesterol and upregulation of PPAR-α in adipose tissue. Mol Nutr Food Res. 2017;61(11):46-86. [35] ARAúJO J R, TAZI A, BURLEN-DEFRANOUX O, et al. Fermentation products of commensal bacteria alter enterocyte lipid metabolism. Cell Host Microbe. 2020;27(3):358-375.e7. [36] RODRIGUES V F, ELIAS-OLIVEIRA J, PEREIRA ÍS, et al. Akkermansia muciniphila and gut immune system: a good friendship that attenuates inflammatory bowel disease, obesity, and diabetes. Front Immunol. 2022;13(93):46-95. [37] BASOLO A, HOHENADEL M, ANG Q Y, et al. Effects of underfeeding and oral vancomycin on gut microbiome and nutrient absorption in humans. Nat Med. 2020;26(4):589-598. [38] LONG M, YANG S, LI P, et al. Combined use of C. butyricum Sx-01 and L. salivarius C-1-3 improves intestinal health and reduces the amount of lipids in serum via modulation of gut microbiota in mice. Nutrients. 2018;10(7):152-176. [39] CARVALHO BM, SAAD MJ. Influence of gut microbiota on subclinical inflammation and insulin resistance. Mediators Inflamm. 2013;2013(98):679-674. [40] DAVISON JM, LICKWAR CR, SONG L, et al. Microbiota regulate intestinal epithelial gene expression by suppressing the transcription factor Hepatocyte nuclear factor 4 alpha. Genome Res. 2017;27(7):1195-1206. [41] LAMICHHANE S, SEN P, ALVES MA, et al. Linking gut microbiome and lipid metabolism: moving beyond associations. Metabolites. 2021;11(1):55. [42] CUMMINGS JH, POMARE EW, BRANCH WJ, et al. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut. 1987;28(10):1221-1227. [43] MACFARLANE S, MACFARLANE GT. Regulation of short-chain fatty acid production. Proc Nutr Soc. 2003;62(1):67-72. [44] HE J, ZHANG P, SHEN L, et al. Short-chain fatty acids and their association with signalling pathways in inflammation, glucose and lipid metabolism. Int J Mol Sci. 2020;21(17):6356. [45] ZAMBELL KL, FITCH MD, FLEMING SE. Acetate and butyrate are the major substrates for de novo lipogenesis in rat colonic epithelial cells. J Nutr. 2003;133(11):3509-3515. [46] KINDT A, LIEBISCH G, CLAVEL T, et al. The gut microbiota promotes hepatic fatty acid desaturation and elongation in mice. Nat Commun. 2018;9(1):3760. [47] SHIMIZU H, MASUJIMA Y, USHIRODA C, et al. Dietary short-chain fatty acid intake improves the hepatic metabolic condition via FFAR3. Sci Rep. 2019;9(1):16574. [48] 邓邦利. 钙信号和短链脂肪酸调节糖代谢机制研究[D].天津:天津医科大学,2018. [49] DEN BESTEN G, BLEEKER A, GERDING A, et al. Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes. 2015;64(7):2398-2408. [50] OLANIYI KS, ATUMA CL, SABINARI IW, et al. Acetate-mediated-obestatin modulation attenuates adipose-hepatic dysmetabolism in high fat diet-induced obese rat model. Endocrine. 2022;76(3):558-569. [51] KIMURA I, OZAWA K, INOUE D, et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat Commun. 2013;4(18):29-42. [52] FROST G, SLEETH ML, SAHURI-ARISOYLU M, et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat Commun. 2014;5(36):11-29. [53] FORBES S, STAFFORD S, COOPE G, et al. Selective FFA2 agonism appears to act via intestinal PYY to reduce transit and food intake but does not improve glucose tolerance in mouse models. Diabetes. 2015;64(11):3763-3771. [54] PRIYADARSHINI M, LAYDEN BT. FFAR3 modulates insulin secretion and global gene expression in mouse islets. Islets. 2015;7(2):e1045182. [55] SMITH PM, HOWITT MR, PANIKOV N, et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science. 2013;341(6145):569-573. [56] OHIRA H, FUJIOKA Y, KATAGIRI C, et al. Butyrate attenuates inflammation and lipolysis generated by the interaction of adipocytes and macrophages. J Atheroscler Thromb. 2013;20(5):425-442. [57] KIMURA I, INOUE D, HIRANO K, et al. The SCFA receptor GPR43 and energy metabolism. front endocrinol (Lausanne). 2014;5(85):66-79. [58] ZHENG M, YANG X, WU Q, et al. Butyrate attenuates hepatic steatosis induced by a high-fat and fiber-deficient diet via the hepatic GPR41/43-CaMKII/HDAC1-CREB pathway. Mol Nutr Food Res. 2022;e2200597. [59] LI Z, YI CX, KATIRAEI S, et al. Butyrate reduces appetite and activates brown adipose tissue via the gut-brain neural circuit. Gut. 2018;67(7):1269-1279. [60] CHáVEZ-TALAVERA O, HAAS J, GRZYCH G, et al. Bile acid alterations in nonalcoholic fatty liver disease, obesity, insulin resistance and type 2 diabetes: what do the human studies tell? Curr Opin Lipidol. 2019;30(3):244-254. [61] TRAUNER M, CLAUDEL T, FICKERT P, et al. Bile acids as regulators of hepatic lipid and glucose metabolism. Digestive Diseases. 2010;28(1):220-224. [62] BHATNAGAR S, DAMRON HA, HILLGARTNER FB. Fibroblast growth factor-19, a novel factor that inhibits hepatic fatty acid synthesis. J Biol Chem. 2009;284(15): 10023-10033. [63] NEAL MD, LEAPHART C, LEVY R, et al. Enterocyte TLR4 mediates phagocytosis and translocation of bacteria across the intestinal barrier. J Immunol. 2006;176(5):3070-3079. [64] VIJAY-KUMAR M, AITKEN JD, CARVALHO FA, et al. Metabolic syndrome and altered gut microbiota in mice lacking Toll-like receptor 5. Science. 2010;328(5975):228-231. [65] CANI PD, AMAR J, IGLESIAS MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761-1772. [66] WEISBERG SP, MCCANN D, DESAI M, et al. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796-1808. [67] ABDALLAH ISMAIL N, RAGAB SH, ABD ELBAKY A, et al. Frequency of Firmicutes and Bacteroidetes in gut microbiota in obese and normal weight Egyptian children and adults. Arch Med Sci. 2011;7(3):501-507. [68] GOMES AC, HOFFMANN C, MOTA JF. The human gut microbiota: metabolism and perspective in obesity. Gut Microbes. 2018;9(4):308-325. [69] MASUI R, SASAKI M, FUNAKI Y, et al. G protein-coupled receptor 43 moderates gut inflammation through cytokine regulation from mononuclear cells. Inflamm Bowel Dis. 2013;19(13):2848-2856. [70] VINOLO MA, RODRIGUES HG, HATANAKA E, et al. Suppressive effect of short-chain fatty acids on production of proinflammatory mediators by neutrophils. J Nutr Biochem. 2011;22(9):849-855. [71] 李亚梦,魏秋阳,刘明宇,等.肠道菌群及其代谢产物氧化三甲胺与冠心病的关系及相关技术方法的研究进展[J].中国微生态学杂志,2022,34(10):1222-1227. [72] LIU J, LAI L, LIN J, et al. Ranitidine and finasteride inhibit the synthesis and release of trimethylamine N-oxide and mitigates its cardiovascular and renal damage through modulating gut microbiota. Int J Biol Sci. 2020;16(5):790-802. [73] SHIH DM, WANG Z, LEE R, et al. Flavin containing monooxygenase 3 exerts broad effects on glucose and lipid metabolism and atherosclerosis. J Lipid Res. 2015;56(1):22-37. [74] KOETH R A, LAM-GALVEZ BR, KIRSOP J, et al. l-Carnitine in omnivorous diets induces an atherogenic gut microbial pathway in humans. J Clin Invest. 2019; 129(1):373-387. [75] GAO X, LIU X, XU J, et al. Dietary trimethylamine N-oxide exacerbates impaired glucose tolerance in mice fed a high fat diet. J Biosci Bioeng. 2014;118(4):476-481. [76] LIANG X, ZHANG Z, LV Y, et al. Reduction of intestinal trimethylamine by probiotics ameliorated lipid metabolic disorders associated with atherosclerosis. Nutrition. 2020;79-80(11):941-955. [77] REN W, WU Q, DENG L, et al. Simultaneous medium chain fatty acids production and process carbon emissions reduction in a continuous-flow reactor: re-understanding of carbon flow distribution. Environ Res. 2022;212(Pt B):113294. [78] ZHAO J, HU J, MA X. Sodium caprylate improves intestinal mucosal barrier function and antioxidant capacity by altering gut microbial metabolism. Food Funct. 2021;12(20):9750-9762. [79] GREGOR A, AUERNIGG-HASELMAIER S, TRAJANOSKI S, et al. Colonic medium-chain fatty acids act as a source of energy and for colon maintenance but are not utilized to acylate Ghrelin. Nutrients. 2021;13(11):3807. [80] SAM QH, LING H, YEW WS, et al. The divergent immunomodulatory effects of short chain fatty acids and medium chain fatty acids. Int J Mol Sci. 2021;22(12):6453. [81] VAN DE HEIJNING JM, OOSTING A, KEGLER D, et al. An increased dietary supply of medium-chain fatty acids during early weaning in rodents prevents excessive fat accumulation in adulthood. Nutrients. 2017;9(6):631. [82] SZTERK A, OFIARA K, STRUS B, et al. Content of health-promoting fatty acids in commercial sheep, cow and goat cheeses. Foods. 2022;11(8):1116. [83] KITAHARA T, KOYAMA N, MATSUDA J, et al. Antimicrobial activity of saturated fatty acids and fatty amines against methicillin-resistant Staphylococcus aureus. Biol Pharm Bull. 2004;27(9):1321-1326. [84] KRISHNAN S, DING Y, SAEDI N, et al. Gut microbiota-derived tryptophan metabolites modulate inflammatory response in hepatocytes and macrophages. Cell Rep. 2018;23(4):1099-1111. [85] SHESTOPALOV AV, SHATOVA OP, GAPONOV AM, et al. The study of tryptophan metabolite concentrations in blood serum and fecal extracts from obese children. Biomed Khim. 2020;66(6):494-501. [86] OSADCHIY V, LABUS JS, GUPTA A, et al. Correlation of tryptophan metabolites with connectivity of extended central reward network in healthy subjects. PLoS One. 2018;13(8):e0201772. [87] HENAO-MEJIA J, WILLIAMS A, GOFF LA, et al. The microRNA miR-181 is a critical cellular metabolic rheostat essential for NKT cell ontogenesis and lymphocyte development and homeostasis. Immunity. 2013;38(5):984-997. [88] VIRTUE AT, MCCRIGHT SJ, WRIGHT JM, et al. The gut microbiota regulates white adipose tissue inflammation and obesity via a family of microRNAs. Sci Transl Med. 2019;11 (496):eaav1892. |
| [1] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [2] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [3] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [4] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [5] | Qin Yixiong, Yuan Zijian, Xie Shanzhou, Zhu Yingwen, Chen Minghui, Han Biao, Guo Yong. Effect of tea polyphenols on proliferation and differentiation of osteoblasts stimulated by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5665-5669. |
| [6] | Zhang Huiyu, Yu Jingwen, Bai Zhenjun, Li Liang, Mu Bingtao, Zhang Jinfeng, Xie Jiawei. Triptolide protects damaged neurons by regulating microglial polarization [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5342-5347. |
| [7] | Leng Siyi, Pu Rui, Chen Ziyang, Yang Qihang, Song Yongjing, Liu Hui. Roles of exercise intervention in intestinal flora in autoimmune diseases [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5219-5226. |
| [8] | Du Chendan, Du Bingli, Ning Xiao, Zong Mingrui, Li Bing, Wu Xiuping. Mechanism of clock gene Bmal1 in temporomandibular joint osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5227-5231. |
| [9] | Liu Guanjuan, Song Na, Huo Hua, Luo Shanshan, Cheng Yuting, Xiong Yue, Hong Wei, Liao Jian. Zoledronic acid inhibits lipopolysaccharide-induced osteoclast differentiation by regulating NLRP3 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4677-4683. |
| [10] | Sun Fangyuan, Meng Jialei, Ma Yuhui, Geng Huan, Zhang Tao. Effects of ligustrazine on inflammation and oxidative stress in rat cardiomyocytes induced by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3253-3258. |
| [11] | Cui Zhenhua, Lin Xiafei, Chen Yongmin, Lin Ye, Li Guanyu, Song Zhenhua. Acupuncture and moxibustion combined with rehabilitation therapy improve neurological function and intestinal flora following cerebral ischemia in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2692-2698. |
| [12] | Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun. Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5583-5588. |
| [13] | Sheng Weibei, Xiong Ao, Liu Su, Deng Jiapeng, Weng Jian, Yu Fei, Chen Yingqi, Zeng Hui. Effect of phosphocreatine modified chitosan hydrogel on polarization and inflammatory factor expression in rat bone marrow-derived macrophages [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5040-5046. |
| [14] | Zheng Meijie, Yang Weizhe, Liu Jialin, Han Xiangzhen, Zhou Qiqi, He Huiyu. Apoptosis of mouse bone marrow-derived macrophages induced by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4962-4966. |
| [15] | Wu Juhua, Yang Yanan, Weng Xiquan, Zhao Fangfang, Xu Guoqin, Lin Wenta. Effects of hypoxic exercise on skeletal muscle energy metabolism of rats with alimentary obesity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4598-4604. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

