Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (35): 5721-5726.doi: 10.12307/2023.828
Previous Articles Next Articles
Traditional Chinese medicine regulates nuclear factor-kappa B signaling pathway against ischemia-reperfusion injury of skin flaps
Ma Suilu1, He Zhijun2, Liu Tao2, Li Jinpeng2, Li Yan2, He Bo1, Wei Xiaotao1, Wang Weiwei1
- 1Clinical College of Traditional Chinese Medicine, Gansu University of Chinese Medicine, Lanzhou 730030, Gansu Province, China; 2Department of Foot and Ankle Orthopedics, Gansu Hospital of Traditional Chinese Medicine, Lanzhou 730050, Gansu Province, China
-
Received:2022-10-31Accepted:2022-11-25Online:2023-12-18Published:2023-06-05 -
Contact:He Zhijun, Professor, Chief physician, Master’s supervisor, Department of Foot and Ankle Orthopedics, Gansu Hospital of Traditional Chinese Medicine, Lanzhou 730050, Gansu Province, China -
About author:Ma Suilu, Master candidate, Clinical College of Traditional Chinese Medicine, Gansu University of Chinese Medicine, Lanzhou 730030, Gansu Province, China -
Supported by:National Natural Science Foundation of China, No. 81660802 (to HZJ); Gansu Provincial Administration of Traditional Chinese Medicine Key Project, No. GZKZ-2020-2 (to HZJ); Gansu Provincial Key R&D Program Project, No. 21YF5FA021 (to HZJ); Gansu Provincial Youth Science and Technology Fund, No. 21JR11RA211 (to LT); Gansu Provincial Talent Innovation and Entrepreneurship Special Project, No. 2019-RC-63 (to HZJ)
CLC Number:
Cite this article
Ma Suilu, He Zhijun, Liu Tao, Li Jinpeng, Li Yan, He Bo, Wei Xiaotao, Wang Weiwei. Traditional Chinese medicine regulates nuclear factor-kappa B signaling pathway against ischemia-reperfusion injury of skin flaps[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5721-5726.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
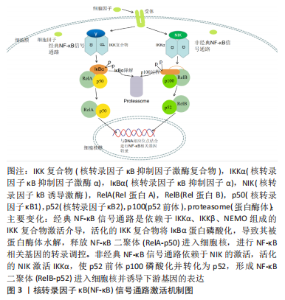
2.1 核转录因子κB信号通路与皮瓣缺血再灌注损伤概述 炎性损伤是皮瓣缺血再灌注损伤发生的关键因素之一,也是引起皮瓣坏死的主要原因之一。因此,减少缺血再灌注皮瓣炎性损伤可作为防治皮瓣坏死的有利条件。核转录因子κB信号通路是调控机体炎症反应与免疫应答的关键性通路,在接收到来自机体内外应激源的刺激后,核转录因子κB信号通路通过两种途径被激活,即活化的核转录因子κB可通过介导炎症因子和黏附分子等细胞因子的转录引起组织细胞的炎性损伤。因此,通过研究核转录因子κB信号通路在皮瓣缺血再灌注损伤中的作用,可为抗皮瓣缺血再灌注损伤提供一个新的研究思路。 2.1.1 核转录因子κB信号通路概述 核转录因子κB是在B淋巴细胞中发现的一种能与免疫球蛋白κ轻链特异性结合的核蛋白因子[12],在调节人体免疫应答、细胞周期以及内环境稳态中发挥着重要作用。在脊椎动物中,核转录因子κB家族是由核转录因子κB蛋白[核转录因子κB2(p52)和核转录因子κB1(p50)]以及Rel蛋白[RelA(p65)、RelB和c-Rel]构成的二聚体[12-13]。在细胞生理状态下,核转录因子κB抑制因子(inhibitor of NF-κB,IκB)家族通过其C端特定的锚蛋白重复序列与核转录因子κB结合,并抑制其N端的核定位区域,抑制核转录因子κB的核易位与转录调控;细胞接受到刺激信号后,IκB 激酶(IκB Kinase,IKK)被激活,IκB蛋白会被活化的IKK磷酸化,最终被蛋白酶体降解,从而使核转录因子κB二聚体释放进入细胞核,进行转录调控,包括增殖、炎症和免疫等过程[14-15]。IKK复合物有IKKα(IKK1)、IKKβ(IKK2)以及IKKγ(NEMO,核转录因子κB必需调节剂)3个亚族[16-18],根据其介导激酶的不同,可将核转录因子κB的转录调控分为经典途径与非经典途径,见图3。 "

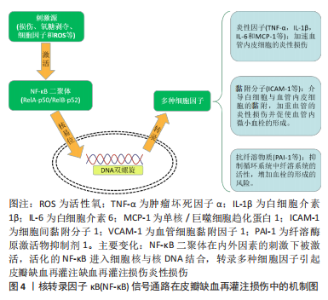
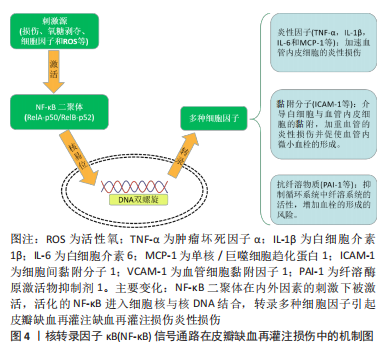
2.1.2 核转录因子κB信号通路在皮瓣缺血再灌注损伤中的机制 皮瓣缺血再灌注损伤过程中,核转录因子κB在损伤、氧糖剥夺、细胞因子以及活性氧等刺激下被激活,促进核转录因子κB相关基因的转录[19-20]。血管内皮细胞的增殖、分裂以及迁移是皮瓣血管再生的基础,对皮瓣创面的愈合至关重要。活化的核转录因子κB可调控血管内皮细胞分泌过多的肿瘤坏死因子α、白细胞介素1β、白细胞介素6以及单核/巨噬细胞趋化蛋白1(Monocyte/macrophage Chemotactic Protein-1,MCP-1)等炎性相关因子的释放[21],而肿瘤坏死因子α等炎性因子又可作为内源性刺激物进一步激活核转录因子κB[22],从而使组织内炎症持续存在并不断加重。此外,核转录因子κB还可促进细胞间黏附分子1(intercellular adhesion molecule-1,ICAM-1)和血管细胞黏附因子1(vascular cell adhesion molecule-1,VCAM-1)等黏附分子的表达,介导白细胞(如中心粒细胞和单核细胞)与血管内皮细胞的黏附,加重血管的炎性损伤并促使血管内微小血栓的形成[23]。新生血管血栓的形成对于皮瓣的存活是致命的,活化的核转录因子κB可促进纤溶酶原激活物抑制剂1(plasminogen activator inhibitor-1,PAI-1),抑制循环系统中纤溶系统的活性,增加血栓的形成的风险[24],见图4。此外,既往研究中发现,在皮瓣缺血再灌注损伤中,核转录因子κB的激活与p38丝裂原激活蛋白激酶(p38 mitogen-activated protein kinases,p38MAPK)以及Toll样受体4(Toll-like receptors,TLR4)的活化关系密切。"
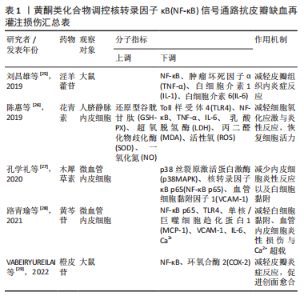
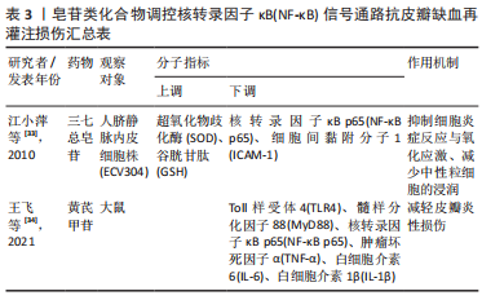
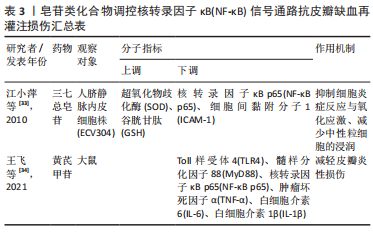
2.2 中药调控核转录因子κB信号通路抗皮瓣缺血再灌注损伤 中药是祖国传统医学的重要组成部分,也是现代医学研究热点,既往研究发现多种中药可有效抗皮瓣缺血再灌注损伤,从而提高皮瓣存活率。并且,随着研究的不断深入,许多中药抗缺血再灌注损伤的作用机制也被逐渐发现,核转录因子κB信号通路就是其中之一。此外,随着药理学的不断发展,中药治疗疾病的有效成分也被逐渐发现,并根据其化学特性分为不同的化合物类型。因此,文章总结调控核转录因子κB信号通路抗皮瓣缺血再灌注损伤的中药、中药有效成分以及中药复方,为中药防治皮瓣缺血再灌注损伤提供部分理论依据。 2.2.1 黄酮类化合物 淫羊藿苷是中药淫羊藿的主要活性成分,属于黄酮苷类化合物,具有抗炎、抗衰老和促进造血功能,改善血流动力学的药理作用。刘昌雄等[25]研究发现,淫羊藿苷可通过下调大鼠缺血再灌注损伤皮瓣组织中核转录因子κB和磷酸化核转录因子κB蛋白的表达,抑制核转录因子κB信号通路引起的炎症反应,促进缺血再灌注损伤皮瓣的存活。花青素是自然界广泛存在于植物中的水溶性类黄酮化合物,具有强大的抗自由基与抗氧化和抗炎能力。陈惠等[26]研究发现,花青素可抑制TLR4/核转录因子κB信号通路的激活,减轻脂多糖诱导的人脐静脉内皮细胞损伤,恢复人脐静脉内皮细胞的活力并减轻细胞的炎症反应与氧化应激。木犀草素是一种天然黄酮类化合物,存在于野菊花、金银花和紫苏等中草药中,具有抗炎、抗氧化、抗过敏以及抗纤维化等多种药理活性。孔学礼等[27]研究发现,木犀草素能够抑制微血管内皮细胞中p38MAPK/核转录因子κB p65的磷酸化,减少炎性细胞与炎症因子造成的微血管内皮细胞损伤。黄芩苷是来源于中药黄芩干燥根中一种黄酮类化合物,具有抑菌、抗血栓、抗氧化及抗炎的药理作用。在2021年,路青瑜等[28]研究发现,黄芩苷通过阻止核转录因子κB p65蛋白入核及核转录因子κB p65在细胞核内的转录活性,以此减少炎性因子与黏附分子等对微血管内皮细胞MEC的损伤。橙皮苷又称二氢黄酮甙,具有抗白细胞黏附、抗炎以及保护毛细血管的药理作用。VABEIRYUREILAI等[29]发现5%橙皮苷水凝胶可抑制核转录因子κB和环氧合酶2的基因表达,促进大鼠背部真皮深层创面的修复和再生,并显著创面缩短愈合时间。上述具体研究成果见表1。"
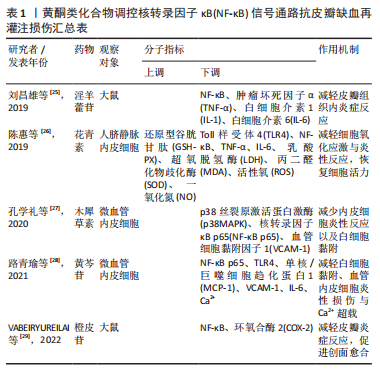
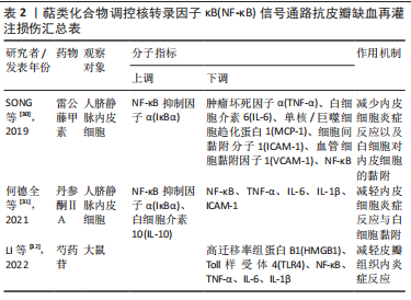
2.2.2 萜类化合物 雷公藤甲素是来源于中药雷公藤的一种环氧二萜内酯化合物,具有抗炎、抗肿瘤以及免疫抑制的药理作用。丹参酮ⅡA是来源于中药丹参的干燥根提取物之一,具有抗炎、抗氧化、抗凝、抗血小板凝集以及调节血脂的功效。SONG等[30]与何德全等[31]发现在脂多糖诱导的人脐静脉内皮细胞炎性损伤中,雷公藤甲素与丹参酮ⅡA预处理可通过抑制IκBα磷酸化并阻断核转录因子κB与核DNA的结合,抑制了核转录因子κB的转录激活,缓解人脐静脉内皮细胞的损伤。芍药苷是从中药赤芍、白芍中分离的一种蒎烷单萜苦味甙,具有抗氧化、抗凝、改善微循环以及免疫调节的功效。LI等[32]证实芍药苷可使大鼠缺血再灌注损伤皮瓣组织内高迁移率组蛋白B1表达下调,抑制TLR4/核转录因子κB信号通路的激活,缓解因炎症反应引起的缺血再灌注损伤皮瓣坏死。上述具体研究成果见表2。"
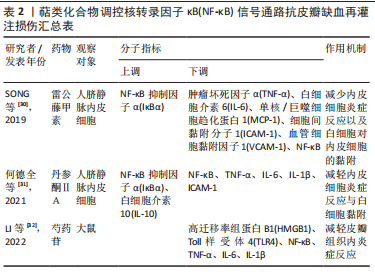
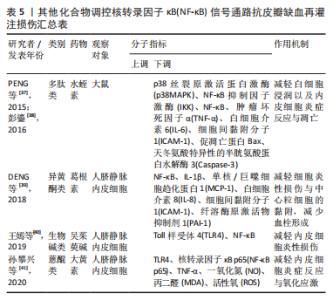
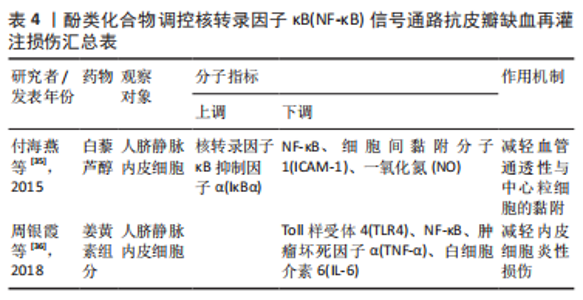
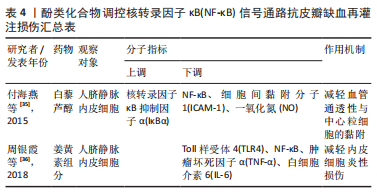
2.2.5 其他化合物 水蛭素是中药水蛭及其唾液腺中提取的一种小分子多肽,具有抗凝、抗血栓、抗炎、抑制血小板聚集、降血脂以及改善血液动力学的功效。PENG等[37]研究水蛭素在皮瓣缺血再灌注损伤中的抗炎作用时,证实了水蛭素可通过抑制ARs/p38MAPK/核转录因子κB信号通路减轻皮瓣炎症反应,促进皮瓣成活。在此基础上,彭鎏[38]研究发现,水蛭素不仅可通过抑制PARs/p38MAPK/IKK/核转录因子κB信号通路抗皮瓣缺血再灌注炎症损伤,还可通过该通路下调促凋亡蛋白的表达,发挥抗皮瓣凋亡的作用。葛根素是从中药葛根中分离出的异黄酮类衍生物,具有抗炎、扩血管以及改善血流动力学的功效。DENG等[39]研究发现,葛根素可阻断核转录因子κB p65的核易位,下调脂多糖诱导人脐静脉内皮细胞损伤后炎性因子、黏附分子以及抗纤溶物质的表达,缓解人脐静脉内皮细胞的损伤。吴茱萸碱是一种天然存在的吲哚生物碱,也是吴茱萸的主要活性成分,具有抗血小板聚集、抗血栓、抗炎及抗缺氧的药理作用。王嫣等[40]研究发现,吴茱萸碱可抑制TLR4/核转录因子κB信号通路,阻断核转录因子κB p65 mRNA的转录调控,保护脂多糖诱导的人脐静脉内皮细胞炎性损伤。大黄素是来源于中药虎杖以及大黄的蒽醌类化合物,具有抗炎、抗组胺、抗微生物生长及免疫抑制等功效。孙攀兴等[41]发现大黄素可通过下调TLR4、核转录因子κB p65及肿瘤坏死因子α蛋白的表达,抑制氧化物质与炎性因子的释放,减轻脂多糖诱导的人脐静脉内皮细胞炎性与氧化损伤。上述具体研究成果见表5。"
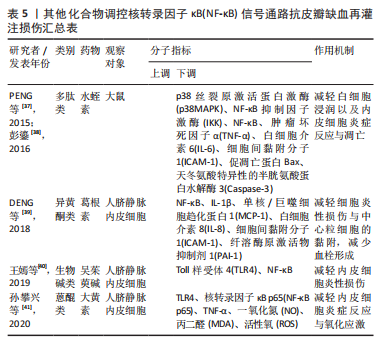
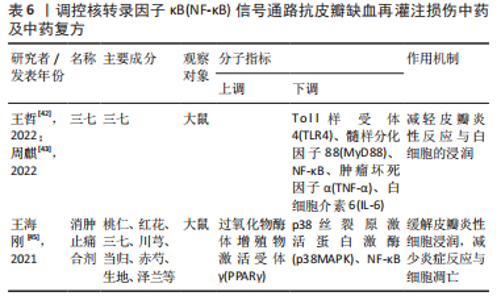
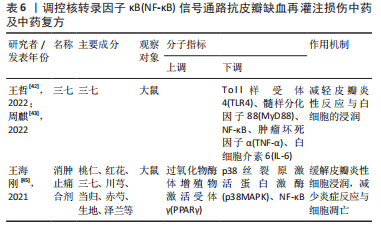
2.2.6 中药及其复方 三七味甘、微苦,性温,归肝、胃经,具有散瘀止、消肿定痛的功效。王哲[42]通过网络药理学研究三七对大鼠皮瓣缺血再灌注损伤的作用机制中发现,三七可缓解缺血再灌注损伤皮瓣组织内中心粒细胞浸润对皮瓣组织结构及其新生血管的破坏,减少血清内的肿瘤坏死因子α、白细胞介素6的表达。在此基础上,周麒[43]进一步研究发现,三七抗皮瓣缺血再灌注损伤炎性损伤是通过抑制TLR4、MyD88和核转录因子κB mRNA的表达而发生的。 消肿止痛合剂(主要成分为桃仁、红花、三七、川芎、当归、赤芍、生地和泽兰等)是甘肃省中医院院内制剂,具有活血祛瘀生新的功效。过氧化物酶体增殖物激活受体γ(peroxisome proliferators-activated receptor γ,PPARγ)是一种由配体激活的核转录因子,在缺血再灌注损伤组织中广泛表达,具有一定的抗炎作用[44]。王海刚[45]研究发现,消肿止痛合剂可通过抑制p38MAPK-PPARγ/核转录因子κB信号通路明显缓解缺血再灌注损伤皮瓣中炎性细胞的浸润,减少炎症反应与细胞凋亡,促进大鼠皮瓣成活,在这一过程中PPARγ蛋白表达上调,而p38MAPK、核转录因子κB表达下调。上述具体研究成果见表6。"
| [1] WU XR, WEI PJ, ZHAO YH, et al. Effects of ilioinguinal composite tissue flaps in repairing skin and soft tissue defects on hand or foot. Zhonghua Shao Shang Za Zhi. 2020;36(8):722-725. [2] YUAN K, ZHANG F, LINEAWEAVER WC, et al. The coverage of soft-tissue defects around the foot and ankle using free or local flaps: a comparative cohort study. Ann Plast Surg. 2021;86(6):668-673. [3] WANG L, SONG D, SONG A, et al. Application of modified designed bilobed latissimus dorsi myocutaneous flap in chest wall reconstruction of locally advanced breast cancer patients. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2021;35(9):1172-1176. [4] CERVENKA B, BEWLEY AF. Free flap monitoring: a review of the recent literature. Curr Opin Otolaryngol Head Neck Surg. 2015;23(5):393-398. [5] BALLESTÍN A, CASADO JG, ABELLÁN E, et al. Ischemia-reperfusion injury in a rat microvascular skin free flap model: a histological, genetic, and blood flow study. PLoS One. 2018;13(12):e0209624. [6] 章盖,耿乐乐,方勇.缺血再灌注对小鼠胸部皮瓣的影响及机制研究[J].中华损伤与修复杂志(电子版),2019,14(6):416-425. [7] 王海刚,何志军.皮瓣缺血再灌注损伤的相关机制研究进展[J].甘肃科技,2022,38(20):129-134. [8] EISENHARDT SU, SCHMIDT Y, KARAXHA G, et al. Monitoring molecular changes induced by ischemia/reperfusion in human free muscle flap tissue samples. Ann Plast Surg. 2012;68(2):202-208. [9] LEE JH, YOU HJ, LEE TY, et al. Current status of experimental animal skin flap models: ischemic preconditioning and molecular factors. Int J Mol Sci. 2022;23(9):5234. [10] DACHO A, HANUSCH C, LYUTENSKI S, et al. Pyrrolidine dithiocarbamate as an effector and trigger in ischemia-reperfusion injury in adipocutaneous flaps in rats. J Otolaryngol Head Neck Surg. 2012;41(3):176-182. [11] WENG H, HE L, LIU X, et al. Natural lactucopicrin alleviates importin-α3-mediated NF-κB activation in inflammated endothelial cells and improves sepsis in mice. Biochem Pharmacol. 2021;186:114501. [12] KAILEH M, SEN R. NF-κB function in B lymphocytes. Immunol Rev. 2012; 246(1):254-271. [13] PATEL H, ZAGHLOUL N, LIN K, et al. Hypoxia-induced activation of specific members of the NF-kB family and its relevance to pulmonary vascular remodeling. Int J Biochem Cell Biol. 2017;92:141-147. [14] WILLIAMS LM, GILMORE TD. Looking down on NF-κB. Mol Cell Biol. 2020; 40(15):e00104-20. [15] THOMS HC, STARK LA. The NF-κB nucleolar stress response pathway. Biomedicines. 2021;9(9):1082. [16] DURAND JK, BALDWIN AS. Targeting IKK and NF-κB for therapy. Adv Protein Chem Struct Biol. 2017;107:77-115. [17] MULERO MC, HUXFORD T, GHOSH G. NF-κB, IκB, and IKK: integral components of immune system signaling. Adv Exp Med Biol. 2019;1172:207-226. [18] MCCORKELL KA, MAY MJ. Noncanonical NF-κB activation and SDF-1 expression in human endothelial cells. Methods Mol Biol. 2015;1280:155-180. [19] RAH DK, MIN HJ, KIM YW, et al. Effect of platelet-rich plasma on ischemia-reperfusion injury in a skin flap mouse model. Int J Med Sci. 2017;14(9):829-839. [20] MAO X, LIU L, CHENG L, et al. Adhesive nanoparticles with inflammation regulation for promoting skin flap regeneration. J Control Release. 2019;297: 91-101. [21] LIU X, SHAO Y, TU J, et al. Trimethylamine-N-oxide-stimulated hepatocyte-derived exosomes promote inflammation and endothelial dysfunction through nuclear factor-kappa B signaling. Ann Transl Med. 2021;9(22):1670. [22] BISWAS R, BAGCHI A. Inhibition of TRAF6-Ubc13 interaction in NFkB inflammatory pathway by analyzing the hotspot amino acid residues and protein-protein interactions using molecular docking simulations. Comput Biol Chem. 2017;70:116-124. [23] LI F, LIANG H, YOU H, et al. Targeting HECTD3-IKKα axis inhibits inflammation-related metastasis. Signal Transduct Target Ther. 2022;7(1):264. [24] CHEN Y, ZHENG Y, LIU L, et al. Adiponectin inhibits TNF-α-activated PAI-1 expression via the cAMP-PKA-AMPK-NF-κB axis in human umbilical vein endothelial cells. Cell Physiol Biochem. 2017;42(6):2342-2352. [25] 刘昌雄,黄雄杰,肖湘君,等.淫羊藿苷对大鼠皮瓣缺血再灌注损伤后炎症反应的抑制作用[J].中国临床药理学杂志,2019,35(6):532-535. [26] 陈惠,胡勇.花青素通过TLR4/NF-κB通路对脂多糖诱导的血管内皮损伤的保护作用[J].实用医药杂志,2019,36(1):59-62. [27] 孔学礼,霍桂桃,李佳,等.木犀草素通过抑制p65 NF-κB、促进p85 PI3K调节微血管内皮细胞VCAM-1表达[J].生物化学与生物物理进展, 2020,47(8):675-684. [28] 路青瑜,郭丽,张启云,等.黄芩苷对脂多糖致内皮细胞炎症反应的保护作用及机制研究[J].中国药理学通报,2021,37(2):251-257. [29] VABEIRYUREILAI M, LALRINZUALI K, JAGETIA GC. NF-κB and COX-2 repression with topical application of hesperidin and naringin hydrogels augments repair and regeneration of deep dermal wounds. Burns. 2022;48(1):132-145. [30] SONG C, WANG Y, CUI L, et al. Triptolide attenuates lipopolysaccharide-induced inflammatory responses in human endothelial cells: involvement of NF-κB pathway. BMC Complement Altern Med. 2019;19(1):198. [31] 何德全,陈友权,王世祥.丹参酮ⅡA通过NF-κB通路抑制脂多糖诱导人脐静脉血管内皮细胞炎性反应的作用机制[J].中西医结合心脑血管病杂志,2021,19(3):428-432. [32] LI WJ, LIU YY, HE JB, et al. Effect of paeoniflorin on distal survival of random flaps. Int Immunopharmacol. 2022;105:108562. [33] 江小萍,李海全,潘少霞,等.三七总皂苷对缺氧再给氧血管内皮细胞NF-κB及ICAM-1表达的影响[J].中西医结合心脑血管病杂志,2010,8(9): 1086-1088. [34] 王飞,田阳,徐骁然,等.黄芪甲苷通过调控TLR-4/NF-κB信号通路对大鼠皮瓣缺血再灌注损伤的影响[J].中国皮肤性病学杂志,2021,35(5):497-503. [35] 付海燕,胡占升,杜红阳,等.白藜芦醇衍生物TMS对脂多糖诱导人脐静脉内皮细胞中一氧化氮、血管细胞间黏附分子1及核转录因子κB表达的影响[J].中国生物制品学杂志,2015,28(8):808-813. [36] 周银霞,张崇,傅海珍,等.姜黄素类组分通过下调TLR4/NF-κB通路抑制LPS诱导的血管内皮细胞损伤[J].中南药学,2018,16(11):1521-1525. [37] PENG L, PAN X, YIN G. Natural hirudin increases rat flap viability by anti-inflammation via PARs/p38/NF-κB pathway. Biomed Res Int. 2015;2015: 597264. [38] 彭鎏. 水蛭素通过p38 MAPK/IKK/NF-κB通路调节大鼠血运障碍皮瓣炎症反应及细胞凋亡相关因子表达的研究[D].南宁:广西医科大学,2016. [39] DENG HF, WANG S, LI L, et al. Puerarin prevents vascular endothelial injury through suppression of NF-κB activation in LPS-challenged human umbilical vein endothelial cells. Biomed Pharmacother. 2018;104:261-267. [40] 王嫣,杨卫平,彭芳,等.吴茱萸碱通过TLR4/NF-κB通路抑制脂多糖诱导的人脐静脉内皮细胞损伤[J].细胞与分子免疫学杂志,2019,35(12): 1088-1093. [41] 孙攀兴,邱春光.大黄素通过调控TLR4/NF-κB通路对脂多糖诱导血管内皮细胞氧化损伤的保护作用研究[J].药物评价研究,2020,43(6):1040-1045. [42] 王哲.基于网络药理学探讨三七对大鼠皮瓣缺血再灌注损伤的影响及作用机制[D].长沙:湖南中医药大学,2022. [43] 周麒.三七调控大鼠腹壁穿支皮瓣缺血再灌注TLR4-MyD88-NF-kB信号通路的实验研究[D].长沙:湖南中医药大学,2022. [44] XIANG S, CHEN K, XU L, et al. Bergenin exerts hepatoprotective effects by inhibiting the release of inflammatory factors, apoptosis and autophagy via the PPAR-γ pathway. Drug Des Devel Ther. 2020;14:129-143. [45] 王海刚.基于p38MAPK-PPARγ/NF-κB信号通路探讨消肿止痛合剂对皮瓣缺血再灌注损伤的干预机制研究[D].兰州:甘肃中医药大学,2021. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [3] | Liu Xin, Hu Man, Zhao Wenjie, Zhang Yu, Meng Bo, Yang Sheng, Peng Qing, Zhang Liang, Wang Jingcheng. Cadmium promotes senescence of annulus fibrosus cells via activation of PI3K/Akt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1217-1222. |
| [4] | Wei Juan, Li Ting, Huan Mengting, Xie Ying, Xie Zhouyu, Wei Qingbo, Wu Yunchuan. Mechanism by which static exercise improves insulin resistance in skeletal muscle of type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1271-1276. |
| [5] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [6] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [7] | Yang Yifeng, Huang Jian, Ye Nan, Wang Lin. Ischemia-reperfusion injury in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 955-960. |
| [8] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [9] | Hu Guangzhi, Lu Hongyan. Changes in pulmonary pericytes and tube formation of pulmonary vascular endothelial cells in mouse models of broncho-pulmonary dysplasia [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 522-527. |
| [10] | Wei Yuanxun, Chen Feng, Lin Zonghan, Zhang Chi, Pan Chengzhen, Wei Zongbo. The mechanism of Notch signaling pathway in osteoporosis and its prevention and treatment with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 587-593. |
| [11] | Yang Qipei, Chen Feng, Cui Wei, Zhang Chi, Wu Ruiqi, Song Zhenheng, Meng Xin. Signaling pathways related to kaempferol active monomers in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4242-4249. |
| [12] | Liu Ying, Liu Yalei, Liu Yu. Screening and analysis of differentially expressed long non-coding RNAs in adriamycin-induced myocardial injury antagonized with daisy leaf gentianone [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4121-4128. |
| [13] | Gao Simiao, Wu Xiaoguang, Han Xue, Xu Shiqi, Li Kuihua, Peng Yong. Establishment of a rat model of traumatic brain injury using the modified Feeney’s free-fall method [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4164-4169. |
| [14] | Li Jiale, Luo Dasheng, Zheng Liujie, Liu Wei, Yao Yunfeng. Human osteoarthritic chondrocytes up-regulate the expression of osteoprotegerin in osteoblasts via the Indian hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4194-4201. |
| [15] | Liu Gang, Zeng Jie, Zhao Yalin, Deng Bowen, Jiang Shengyuan, Zhang Yaqi, Zhao Yi, Ren Jingpei, Hu Chuanyu, Xu Lin, Mu Xiaohong. Shujin Jiannao Prescription alleviates inflammation in the cerebral cortex of rats with hypoxic-ischemic cerebral palsy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3674-3679. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||