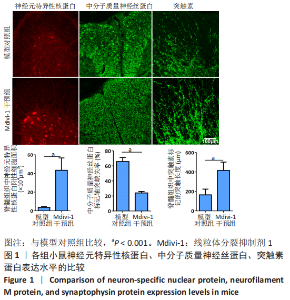
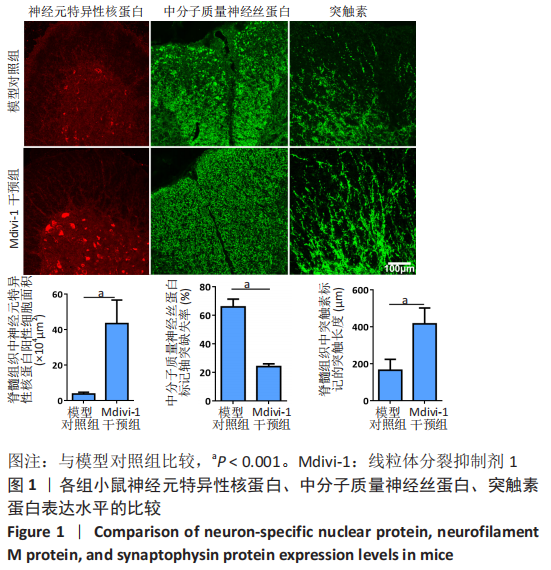
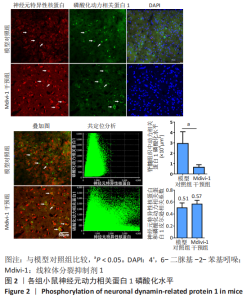
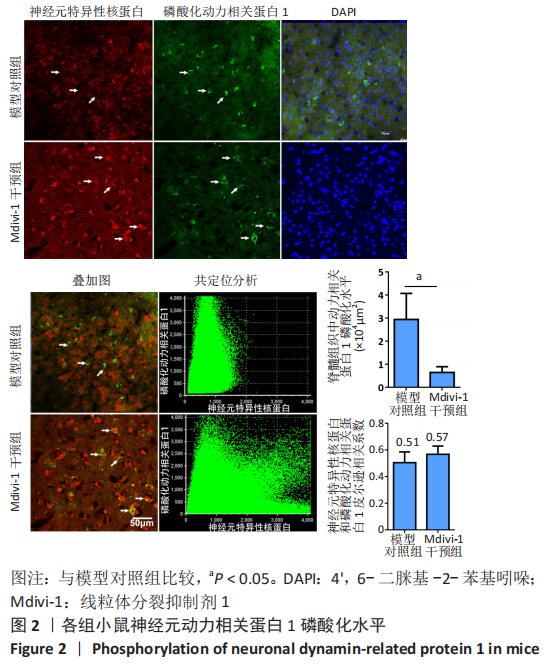
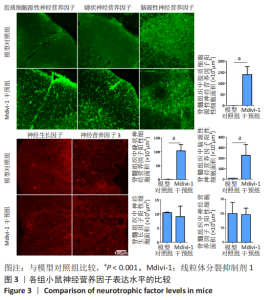
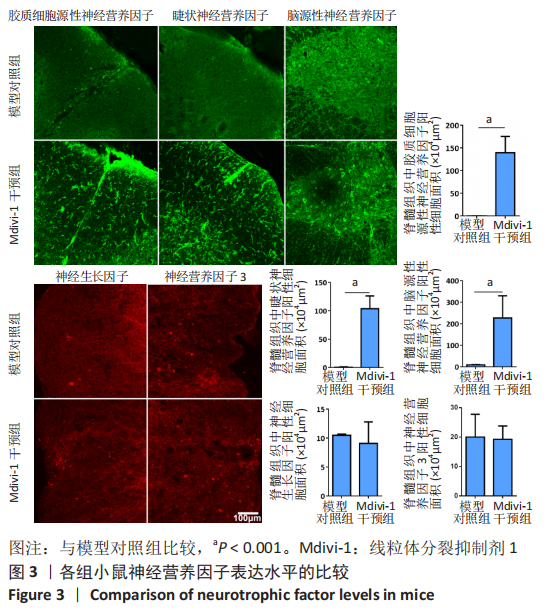
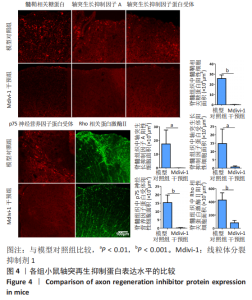
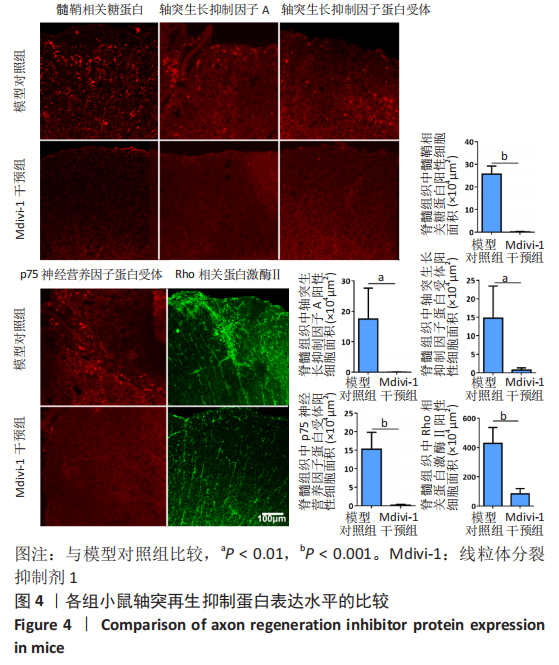
|
[1] MAKHANI N, TREMLETT H. The multiple sclerosis prodrome. Nat Rev Neurol. 2021;17(8):515-521.
[2] GLATIGNY S, BETTELLI E. Experimental Autoimmune Encephalomyelitis (EAE) as Animal Models of Multiple Sclerosis (MS). Cold Spring Harb Perspect Med. 2018;8(11):a028977.
[3] MCGINLEY MP, COHEN JA. Sphingosine 1-phosphate receptor modulators in multiple sclerosis and other conditions. Lancet. 2021; 398(10306):1184-1194.
[4] SUTIWISESAK R, BURNS TC, RODRIGUEZ M, et al. Remyelination therapies for multiple sclerosis: optimizing translation from animal models into clinical trials. Expert Opin Investig Drugs. 2021;30(8): 857-876.
[5] 黎新悦,罗富成.髓鞘可塑性与认知功能的研究进展[J].中国细胞生物学学报,2022,44(6):1202-1210.
[6] 林柳余,杭海伦,张纪红,等.中枢神经系统炎性脱髓鞘疾病相关抗体及其致病机制的研究进展[J].临床神经病学杂志,2022,35(3): 216-220.
[7] 左玉超. MARK2 通过调节微管相关蛋白介导 Nogo-66 抑制轴突生长[D]. 武汉:华中科技大学,2016.
[8] 王小梅,景会锋.有氧运动和谷氨酰胺对二型糖尿病大鼠氧化应激及相关因子表达的影响[J]. 中国应用生理学杂志,2019,35(2):150-154.
[9] NHU NT, LI Q, LIU Y, et al. Effects of Mdivi-1 on Neural Mitochondrial Dysfunction and Mitochondria-Mediated Apoptosis in Ischemia-Reperfusion Injury After Stroke: A Systematic Review of Preclinical Studies. Front Mol Neurosci. 2021;14:778569.
[10] LI YH, XU F, THOME R, et al. Mdivi-1, a mitochondrial fission inhibitor, modulates T helper cells and suppresses the development of experimental autoimmune encephalomyelitis. J Neuroinflammation. 2019;16(1):149.
[11] LIU X, ZHANG X, NIU X, et al. Mdivi-1 Modulates Macrophage/Microglial Polarization in Mice with EAE via the Inhibition of the TLR2/4-GSK3β-NF-κB Inflammatory Signaling Axis. Mol Neurobiol. 2022;59(1):1-16.
[12] BORDT EA, CLERC P, ROELOFS BA, et al. The Putative Drp1 Inhibitor mdivi-1 Is a Reversible Mitochondrial Complex I Inhibitor that Modulates Reactive Oxygen Species. Dev Cell. 2017;40(6):583-594.
[13] 项捷. 神经营养因子在神经发育及神经退行性疾病中作用机制研究[D].西安:中国人民解放军空军军医大学,2019.
[14] 何蓉. RhoA/ROCK信号通路在惊厥性脑损伤轴突再生中的作用研究[D].重庆:重庆医科大学,2016.
[15] 黄丹霞,黄赛娥.Nogo-A抑制信号通路在脑缺血后神经再生中的作用及研究进展[J].按摩与康复医学,2015,6(20):12-15.
[16] 宋岩.大鼠脑外伤后抑制Drp1在脑保护和行为学改善中的作用机制研究[D].济南:山东大学,2019.
[17] 李艳花,刘晓琴,牛春红,等.Mdivi-1对cuprizone诱导的脱髓鞘性病变小鼠少突胶质细胞的保护作用[J].中国病理生理杂志,2020, 36(6):1115-1121.
[18] LUO F, HERRUP K, QI X, et al Inhibition of Drp1 hyper-activation is protective in animal models of experimental multiple sclerosis. Exp Neurol. 2017;292:21-34.
[19] ANDERSON MB, DAS S, MILLER KE. Subcellular localization of neuronal nuclei (NeuN) antigen in size and calcitonin gene-related peptide (CGRP) populations of dorsal root ganglion (DRG) neurons during acute peripheral inflammation. Neurosci Lett. 2021;760:135974.
[20] LEE Y, LEE BH, YIP W, et al. Neurofilament Proteins as Prognostic Biomarkers in Neurological Disorders. Curr Pharm Des. 2020;25(43): 4560-4569.
[21] THIEL G. Synapsin I, synapsin II, and synaptophysin: marker proteins of synaptic vesicles. Brain Pathol. 1993;3(1):87-95.
[22] BIDO S, SORIA FN, FAN RZ, et al. Mitochondrial division inhibitor-1 is neuroprotective in the A53T-α-synuclein rat model of Parkinson’s disease. Sci Rep. 2017;7(1):7495.
[23] ZHANG Y, RUI T, LUO C, et al. Mdivi-1 alleviates brain damage and synaptic dysfunction after intracerebral hemorrhage in mice. Exp Brain Res. 2021;239(5):1581-1593.
[24] 师一民,李明,王蕾.神经营养因子在多发性硬化神经再生修复中的作用研究进展[J].医学综述,2016,22(24):4785-4788.
[25] 季晓飞,董惠洁.脑源性神经营养因子基因多态性与散发性多发性硬化的关联研究[J].大连医科大学学报,2012,34(5):481-483.
[26] GAETANI L, SALVADORI N, CHIPI E, et al. Cognitive impairment in multiple sclerosis: lessons from cerebrospinal fluid biomarkers. Neural Regen Res. 2021;16(1):36-42.
[27] NASRNEZHAD R, HALALKHOR S, SADEGHI F, et al. Piperine Improves Experimental Autoimmune Encephalomyelitis (EAE) in Lewis Rats Through its Neuroprotective, Anti-inflammatory, and Antioxidant Effects. Mol Neurobiol. 2021;58(11):5473-5493.
[28] BRAUN D, MADRIGAL JL, FEINSTEIN DL. Noradrenergic regulation of glial activation: molecular mechanisms and therapeutic implications. Curr Neuropharmacol. 2014;12(4):342-352.
[29] LEE JY, KIM MJ, THOMAS S, et al. Limiting Neuronal Nogo Receptor 1 Signaling during Experimental Autoimmune Encephalomyelitis Preserves Axonal Transport and Abrogates Inflammatory Demyelination. J Neurosci. 2019;39(28):5562-5580.
[30] THEOTOKIS P, GRIGORIADIS N. p75NTR and TROY: Uncharted Roles of Nogo Receptor Complex in Experimental Autoimmune Encephalomyelitis. Mol Neurobiol. 2018;55(8):6329-6336.
[31] KAN QC, ZHANG HJ, ZHANG Y, et al. Matrine Treatment Blocks NogoA-Induced Neural Inhibitory Signaling Pathway in Ongoing Experimental Autoimmune Encephalomyelitis. Mol Neurobiol. 2017;54(10):8404-8418.
[32] MAVADDATIYAN L, KHEZRI S, ABTAHI FROUSHANI SM. Molecular effects of curcumin on the experimental autoimmune encephalomyelitis. Vet Res Forum. 2021;12(1):47-52.
[33] CHANG HM, WU HC, SUN ZG, et al. Neurotrophins and glial cell line-derived neurotrophic factor in the ovary: physiological and pathophysiological implications. Hum Reprod Update. 2019;25(2): 224-242.
[34] LI-NA Z, DENG C, DA X, et al. Mesencephalic astrocyte-derived neurotrophic factor and its role in nervous system disease. Neurol Sci. 2017;38(10):1741-1746.
[35] ROSICH K, HANNA BF, IBRAHIM RK, et al. The Effects of Glial Cell Line-Derived Neurotrophic Factor after Spinal Cord Injury. J Neurotrauma. 2017;34(24):3311-3325.
[36] LIU H, TANG X, GONG L. Mesencephalic astrocyte-derived neurotrophic factor and cerebral dopamine neurotrophic factor: New endoplasmic reticulum stress response proteins. Eur J Pharmacol. 2015;750:118-122.
[37] CASTRÉN E, RANTAMÄKI T. Role of brain-derived neurotrophic factor in the aetiology of depression: implications for pharmacological treatment. CNS Drugs. 2010;24(1):1-7.
[38] EREMIN DV, ILCHIBAEVA TV, TSYBKO AS. Cerebral Dopamine Neurotrophic Factor (CDNF): Structure, Functions, and Therapeutic Potential. Biochemistry (Mosc). 2021;86(7):852-866.
[39] THOENEN H, ZAFRA F, HENGERER B, et al. The synthesis of nerve growth factor and brain-derived neurotrophic factor in hippocampal and cortical neurons is regulated by specific transmitter systems. Ann N Y Acad Sci. 1991;640:86-90.
[40] ALBERT K, AIRAVAARA M. Neuroprotective and reparative effects of endoplasmic reticulum luminal proteins - mesencephalic astrocyte-derived neurotrophic factor and cerebral dopamine neurotrophic factor. Croat Med J. 2019;60(2):99-108.
|