Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (9): 1455-1461.doi: 10.12307/2023.240
Previous Articles Next Articles
3D printed porous tantalum metal in the treatment of developmental dysplasia of the hip: current status and application prospect
He Yinhao, Li Xiaosheng, Chen Hongwen, Chen Tiezhu
- Department of Bone, Joint and Sports Medicine, Hunan Provincial People’s Hospital (First Affiliated Hospital of Hunan Normal University), Changsha 410005, Hunan Province, China
-
Received:2022-01-29Accepted:2022-04-18Online:2023-03-28Published:2022-07-02 -
Contact:Chen Tiezhu, Master’s supervisor, Associate chief physician, Department of Bone, Joint and Sports Medicine, Hunan Provincial People’s Hospital (First Affiliated Hospital of Hunan Normal University), Changsha 410005, Hunan Province, China -
About author:He Yinhao, Master candidate, Department of Bone, Joint and Sports Medicine, Hunan Provincial People’s Hospital (First Affiliated Hospital of Hunan Normal University), Changsha 410005, Hunan Province, China -
Supported by:Project of Hunan Provincial Health Commission, No. 202204072627 (to CTZ); Project of Hunan Provincial Department of Education, No. 20C1162 (to CTZ)
CLC Number:
Cite this article
He Yinhao, Li Xiaosheng, Chen Hongwen, Chen Tiezhu. 3D printed porous tantalum metal in the treatment of developmental dysplasia of the hip: current status and application prospect[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1455-1461.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
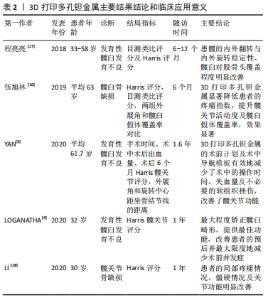
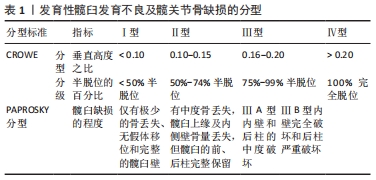
2.1.1 CROWE按股骨头上移程度分级 CROWE描述了股骨头和髋臼之间半脱位的百分比,按股骨头上移程度将发育性髋臼发育不良分为4级:Ⅰ级为股骨头直径< 50%半脱位,Ⅱ级为50%-74%半脱位,Ⅲ级为75%-99%半脱位,Ⅳ级为100%完全脱位[8]。 2.1.2 CROWE分型 CROWE等[8]按照股骨头下缘与真臼下缘的垂直距离与骨盆的垂直高度之比将发育性髋臼发育不良分Ⅲ型:CROWE Ⅰ型:比值< 0.10,CROWE Ⅱ型:0.10-0.15,CROWE Ⅲ型:0.16-0.20,CROWE Ⅳ型:> 0.20。大量文献报道,成人发育性髋臼发育不良常以CROWE分型为主[3-4,9-10]。 2.1.3 PAPROSKY分型 PAPROSKY等[11]按髋臼缺损的程度分型:Ⅰ型:仅有极少的骨丢失、无假体移位和完整的髋臼壁;Ⅱ型:有中度骨丢失,髋臼上缘及内侧壁骨量丢失,但髋臼的前、后柱完整保留。按位置将Ⅱ型区分为ⅡA (上侧)、ⅡB (外上侧)和ⅡC (内侧);Ⅲ型:髋臼边缘和支撑结构的严重破坏导致严重的骨质流失;ⅢA型:内壁和后柱的中度破坏;ⅢB型:内壁完全破坏和后柱严重破坏。 PAPROSKY分型虽通过X射线片直观的将髋臼缺损进行分型,经济便利,但随着3DPPTa精细个体化,为将全髋关节置换后并发症假体不稳定、脱落的风险降至最低,常常借助CT三维成像,最为全面地观测评估发育性髋臼发育不良及髋臼缺损的程度,定制出个体化的3DPPTa。 2.2 评估发育性髋臼发育不良生物力学的参数指标 正常髋臼关节面的曲率半径与股骨头表面相适应,几乎可以完全包盖股骨头,进而维持髋关节的稳定。而发育性髋臼发育不良由于髋臼覆盖股骨头面积减少,导致股骨头不稳定及向前外侧移位,使关节负重状态发生改变,引发髋关节疼痛、步态异常、力量下降等,从而继发髋关节炎[1]。目前,髋臼指数与中心边缘角是评价发育性髋臼发育不良最常用的参数指标[1,12]。 2.2.1 髋臼指数 又称T?nnis角,反映髋臼对于股骨头覆盖情况及测量承重区的重要参数,对于发育性髋臼发育不良的分析较为直接,也是术后评估手术预后的重要指标[13]。髋臼指数> 13°与发育性髋臼发育不良密切相关,其同时与患者年龄、股骨头大小、损伤程度等因素有关,髋臼指数常可区分不同髋臼的形态、覆盖率和方向[14],有助于评估前后髋臼覆盖,提升对无症状发育性髋臼发育不良人群的确诊率。也有报道指出,患有发育性髋臼发育不良的父母具有一定的遗传性[15],髋臼指数的测量可为发育性髋关节发育不良患者进行风险评估及为发育性髋关节发育不良患者后代提供临床预防诊疗措施,且可进行术前个体化3D打印建模与P-Ta形态大小的制定[16]。 2.2.2 中心边缘角 目前,大多数学者仍以中心边缘角作为评估发育性髋臼发育不良覆盖面积严重程度的测量标准,通过股骨头中心作身体纵轴的平行线,与股骨头中心和髋臼外缘连线的夹角来测量髋臼覆盖股骨头侧方面积,进而评估发育性髋臼发育不良的严重程度[17-18]。其侧方中心边缘角在20°-38°且无髋臼后倾现象,可视为正常;侧方中心边缘角< 20°为髋臼覆盖不足,被归类为发育性髋臼发育不良;侧方中心边缘角> 38°为过度覆盖[14]。石永言等[12]对1 494例4岁以后的健康人群骨盆正位片的中心边缘角进行系统对比分析:中心边缘角:4岁21.62°;9岁25.07°,成年时32.14°;> 18岁后维持在30°-34°;具体因种族、性别等因素而有所不同;目前,在评估发育性髋臼发育不良的严重程度仅仅只依靠中心边缘角测量股骨头外侧覆盖率存在一定争议。 2.2.3 弹性模量 传统的髋关节置换假体,因其高致密性,其弹性模量明显高于人体骨组织,易造成应力屏蔽和应力集中,导致假体不稳定,甚至脱落[19],严重时需进行二次翻修手术。由于3D打印个体化假体结构的高孔隙率及良好的生物兼容性,降低了弹性模量,增加了初始的稳定性,使骨长入固定[20];表明了多孔钽的弹性模量更接近人的骨性组织,骨结合性能更有优势,生物力学较多孔钛更为稳定[21]。 2.3 多孔钽金属的生物学特性 2.3.1 生物兼容性 发育性髋臼发育不良是先天性髋臼骨缺损而导致覆盖不足,将3DPPTa个体化假体锚定在髋臼发育不良处,因其具有高密度孔隙率及良好的机械强度,建立了良好的生物兼容性,促进骨再生及血管形成,与新生骨充分兼容,增加髋臼覆盖面积,在种植假体与骨骼之间保持较高的湿润性,从而为成骨细胞的黏附、生长和分化提供良好的生物环境[22];BALLA等[23]通过体外细胞培养实验证明了此观点,同时其也观察到在多孔钽表层的细胞密度是钛表面的6倍,促进了多孔钽金属的生物兼容性。目前,多孔钽金属被认为是医学界生物兼容性最好的硬组织植入材料[7,24]。 2.3.2 骨整合性 骨整合是指植入的假体直接与患者自体活骨相结合,牢固地将假体固定在植入部位。21世纪初,FERNANDEZ-FAIREN等[25]通过动物实验证实了多孔钽金属骨整合的功效,其团队表示多孔钽金属表面形成的氧化层静电是失活的,不会使蛋白质变性,它可以在没有纤维界面的情况下进行骨整合,随访一年显示骨整合发展完整;且骨整合的建立最早可在术后2周出现[22]。近年来,有学者认为:3DPPTa钽涂层减少了糖尿病患者成骨性能的障碍,改善了骨长入,促进了骨整合[22];但也有部分学者表明糖尿病仍是导致骨整合失败的危险因素之一。目前仍缺乏足够的临床试验及长期随访数据来证实。 2.3.3 骨诱导性 单独使用多孔钽作用于骨骼上时,为了使金属与骨骼相结合,必须在其表面形成一层磷灰石[25];而3D打印联合多孔钽金属使用水凝胶形式进行表面涂层修饰,很好地调控了高密度的孔隙率,提升了骨的诱导性,提高了渗透性,促进了营养物质互相交换,也加速了各种药物系统的速率,HUA等[7]表示良好的骨传导性为未来抗肿瘤进行靶向药物治疗时会起到重要作用。3DPPTa有良好的骨传导性,很好地避免应力屏蔽效应的发生,对维持生物学性能和力学性能有着很大的影响;进而能提高植入材料寿命[26]。 2.4 3DPPTa方案治疗发育性髋臼发育不良的研究进展 3DPPTa方案是将3D打印技术、天然金属材料-多孔钽金属联合运用方案,目前主要在发育性髋臼发育不良及髋关节骨缺损患者中评价这一方案的实效性。文章汇总了全球近3年高质量的5项试验研究[3-4,10,27-28],主要评估指标为生物学特性、生物力学特性、术前计划、术中导航模板、细胞生存率、Harris评分改善情况及假体不稳、脱落等风险减少率,见表2。 "


2.4.1 3D打印成型技术 目前国内外常用的3D打印成型技术:①选择性激光熔融[9,24,29];②熔融沉积成型技术[3,27];③化学气相沉积法[30-31]。 2.4.2 3D打印术前计划 由于骨盆复杂的解剖形状及操作空间有限,对髋关节置入假体手术具有很大的挑战性。为了减少术后并发症,3D打印通过术前的影像数据进行重建,采用计算机辅助虚拟手术,建立与实际覆盖不足或缺损尺寸相同的3D模型,更为直观地进行术前手术计划,能够在术中精准定位其相互空间距离、成角关系、方向及深度[32]。CHENG等[33]采用回顾性研究分析了60例发育性髋关节发育不良患者和55名健康人口通过3D打印进行建模模拟手术来制定术前计划,发育性髋臼发育不良组股骨头中心位置明显更靠前外上侧,进一步改善了3D术前计划,促进髋关节功能的恢复。ZENG等[34]通过前瞻性研究发现3D打印技术结合虚拟手术治疗10例髋臼骨折患者,有7例解剖复位至小于1.0 mm,其他3例复位1.0-2.0 mm,解剖复位令人满意,术后随访1周未发生感染、固定失败、形成深静脉血栓等严重并发症,为发育性髋关节发育不良患者提供更少侵入性、更精确、更可靠的个体化术前计划,从而减少手术时间,显著改善了髋臼骨折的预后。出色的术前计划会直接影响手术结果,而对于儿童发育性髋关节发育不良患者尤为重要[35]。 术前计划基于3D模型进行精确的解剖评估和术前训练,提高了术前诊断的准确性,对于避免术中障碍和充分解决患者特异性畸形至关重要[36]。有学者研究显示,3DPPTa的术前计划需要满足“四性原则”,即假体解剖匹配性原则、假体安全性原则、骨组织受力有效性原则及骨组织安全性原则[37];3D模拟结合CT扫描计算机辅助虚拟手术有助于更好的术前计划,对发育性髋臼发育不良、髋关节骨缺损是可行、准确、有效的改善方法,并为术前假体植入物的准备做好术前个体化规划。 2.4.3 术中3D打印导航模板 在传统全髋关节置换治疗发育性髋臼发育不良及骨缺损手术过程中常因解剖结构的广泛扭曲在识别和定位人体髋臼中而延长手术中时间,增加感染率[2]。而3D打印技术是在X射线、CT、MRI等影像学技术下进行光学扫描,快速创建出计算机设计的术中导航模板,通过个体化的导板在手术中引导钻头通过术前预先计划的轨迹钻孔,来确定病变部位及假体放置位置,准确的术中定位可以减少手术时间、组织损伤和手术方案的不确定性[38-39]。髋关节旋转中心主要评估置入传统假体的稳定性,其定位法常位于髋臼前后切迹线垂直线上方大约28 mm处,髋臼窝的扩孔的前倾角以(15±10)°和(40±10)°为标准进行研磨,而研磨尺寸根据髋臼大小及前后壁的骨量而定[40-41];然而,定位真髋臼及放置髋臼假体是术中最困难部分。TU等[42]通过12例患者随访平均72.42个月(38-135个月),从平均手术时间、平均失血量及平均住院时间等多个方面进行评估手术结果,表示良好的3D打印指导模板能快速定位真髋臼,根据术前计划对髋臼大小及植入假体进行理想匹配,避免了CT及X射线片所带来准确率的误差。 髋关节旋转中心异常也会增加全髋关节置换后肢体长度差异、外展肌无力等风险。有研究纳入22例发育性髋关节发育不良患者,分别对11例患者进行传统全髋关节置换处理,另外11例行3D打印导航模板处理,通过术中导航,精准地确定髋关节旋转中心而展开手术,分别记录3D打印组和对照组的手术时间及手术失血量,同时计算植入髋臼杯的理想外展角与实际角度之间的偏差及杯前倾角的偏差,3D打印组明显优于传统全髋关节置换组,且3D打印术后并发症的风险明显下降[38]。有研究通过17例(4男5髋,13女16髋)髋关节发育不良继发终末期骨关节炎患者在3D打印髋关节模型下进行术前模拟手术[43],平均随访时间为(18.35±6.86)个月,通过Harris髋关节评分进行评估,平均垂直骨盆片上髋关节旋转中心的水平距离分别恢复为(15.12±1.25) mm和(32.49±2.83) mm;3D打印导航模板对大龄发育性髋臼发育不良患儿在术中尤为重要,可以提高精准度、减少手术时间、减少辐射暴露、减少骨骺损伤,简化手术[44]。ZHENG等[35]对25例发育性髋关节发育不良患者进行对照分析,12例患者使用3D打印导航模板,而另13例接受传统无导航手术,结果表明3D打印导航组手术时间平均21.08 min,X射线曝光次数3.92次,无骨骺损伤;传统组手术时间平均46.92 min,X射线曝光次数6.69,骨骺损伤0.92次,3D打印导航组指标明显优于传统组。目前,3D打印导航模板可以通过术前计划,术中精确识别定位,恢复髋关节旋转中心正常功能,有效减少在术中因经验不足和技术误差而导致的假体松动、脱落[3];3D打印导航模板在发育性髋臼发育不良手术治疗中,将假体植入物精准地放置[45],减少手术时间及术中失血量,促进了髋关节功能恢复及预后。 2.4.4 3DPPTa方案治疗发育性髋臼发育不良的临床疗效 由于发育性髋关节发育不良患者常有一个浅髋臼、笔直狭窄的股骨管等解剖结构的广泛扭曲,尤其面对高度脱位的发育性髋关节发育不良患者,恢复其正常的髋关节髋关节旋转中心和维持良好的髋关节功能为术前计划的重要目标之一,目前尚存在一定的争议[46]。3DPPTa是使用CT的3D打印技术联合P-Ta而创建的骨模型,对评估发育性髋臼发育不良覆盖不足的面积个体化,精确地将假体作用于髋臼覆盖不足或缺损处,从而减少因不同的骨骼结构大小而导致植入假体的不稳定、甚至脱落的风险,减少术后并发症[47]。YAN等[3]近4年对25例患者进行随机对照试验,12例患者接受了3D打印技术的全髋关节置换,13例患者接受了传统的全髋关节置换,平均随访时间为1.6年,3D打印组手术时间、术中术后出血量及术后6个月Harris髋关节评分均优于传统髋关节置换组,未见感染和假体松动,外展角和髋关节旋转中心距坐骨结节线的距离有显着差异。 3D打印的术前计划及术中导航模板有效地减少了术中的操作时间、失血量及不必要的软组织挫伤,改善了髋关节功能。有研究表明,1例CROWS IV型的发育性髋臼发育不良的32岁女性患者在传统的全髋关节置换后,再次出现患髋的疼痛、间接性跛行,LOGANATHAN等[4]为患者施行了3DPPTa手术重建,通过术前建3D打印模型和植入物试验,准确评估髋臼缺陷情况,患者术后1周下床活动,术后14 d出院,随访1年,Harris髋关节评分为90分,最大程度矫正髋臼畸形,提供最佳功能,改善患者的预后并最大限度地减少术后并发症。程亮亮等[27]对8例(12髋)年龄33-58岁的发育性髋关节发育不良患者进行3DPPTa手术,术后随访6-12个月,平均8.2个月,术前目测类比评分(2.92±0.79)分,末次随访时目测类比评分为(0.83±0.72)分;术前Harris评分(69.67±4.6)分,末次随访升至(84.25±4.17)分,患髋的内外翻转与内外旋转稳定性、髋臼对股骨头覆盖程度得到明显改善。传统单一的多孔钽虽可有效修复骨缺损,但很难制定与缺损处完美匹配的假体去准确重建骨缺损[46]。FAN等[48]表示:3DPPTa的生物力学性能优于单一的多孔钛,在术后8周时,骨整合性更加明显,钽的抗压缩变形能力更接近猪骨支架,提高了细胞兼容性和骨整合。术后系统的康复训练有利于促进髋关节功能恢复,避免髋关节僵硬、压疮等术后并发症。GUO等[49]通过3DPPTa对新西兰兔的骨缺损进行研究分析,实验表明增加了骨向内生长和骨整合,使假体更为牢固,降低了假体松动、脱落等风险。LI等[28]描述了1例患有髋关节骨软骨瘤导致骨缺损的30岁女性,行3DPPTa术后24 h,使用被动装置增加活动范围,术后1个月开始拄拐行走,2个月开始佩戴支具行走,术后6个月,走路时无明显疼痛,术后1年可在没有外力帮助下正常行走,复查X射线片显示无假体松动和脱落现象。 软骨肉瘤所致的骨盆缺损常因缺损太大而给临床医师在治疗方案上带来了巨大挑战;因其较小时,患者自身常无明显症状及体征,而当患者出现疼痛等症状影响生活时,肿瘤已经相当大,且软骨肉瘤对化疗不敏感,手术才是最佳治疗方案,在软骨肉瘤切除后,临床医师所面对的骨盆的重建,是一项巨大挑战,而3D打印的个体化假体最大限度地恢复髋臼解剖结构,尤其是在没有模块化假体的特定区域,重建肿瘤切除术后导致的巨大骨缺损,最大限度地恢复髋臼结构及改善其功能等优势[36]。伍旭林等[10]研究表示:术后Harris评分由术前平均30分,术后改善最高可达98分,患者的局部疼痛情况、僵硬情况及关节功能明显改善。由于其高孔隙率促进细胞的营养物质交换与代谢,促进新骨的形成及血管、神经生成,有望提升患髋的生物力学性能,促进骨愈合。多孔钽金属良好的成骨特性,在联合3D打印后,能更好地对患者进行个体化使用,与患者自身骨组织进行很好地结合,更好地防止应力屏蔽及植入物假体的脱落。用3D打印导航模板进行处理,通过手术导航,精准确定髋关节旋转中心而开展的手术明显优于传统的全髋关节置换,3D打印组在术中时间、术中出血量、对骨骺的损伤及术后的随访满意度乃至患者术后的生活质量方面均有明显的优势,也进一步肯定了3D打印导航模板对于传统全髋关节置换的重要性及必需性。 随着3D打印电子计算机、分子生物学、材料学及组织工程学等技术的发展,3D打印能很好地和多孔钽进行结合,其在医学领域的应用也引起学者们的重视;3DPPTa通过联合三维CT创建的,精确地模拟患者髋部覆盖不足及缺损情况,可制定术前计划,模拟术中操作,将3DPPTa置入覆盖不足及缺损处,增加其覆盖面积,快速识别Harris窝及髋臼切迹的形态,确定真髋臼中心的具体位置,恢复髋关节髋关节旋转中心,从而减少传统置换术因术中暴露不充分、视野不足及不同的损伤情况及解剖变异而造成髋臼打磨不均匀、前倾角过大或过小等问题[50];且减少了因骨骼结构大小各异而导致植入假体的不稳定、甚至脱落的风险,进而缩短了手术时间、术中的失血量及术中对正常血管、神经的损伤,同时也减少了术后假体感染的风险;置入多孔3DPPTa后,由于其对自身孔隙率的调控较传统假体而言[26],其加速了细胞间的营养物质代谢及促进新骨、新血管及神经的生长,提高了假体与机体的生物兼容性,提升了骨整合的效率,促进了骨愈合进程[51];多孔钽金属的高熔点、良好的机械强度及强大的抗腐蚀能力[22],因其表面生成稳定的五氧化二钽(Ta2O5)保护膜[52],腐蚀敏感性是决定植入物使用寿命的主要因素,与3D打印联合,可有效防止髋关节因活动所导致骨质溶解,因其强耐受力,有望突破传统全髋关节置换的假体使用年限[53]。表面的生物光滑涂层可有效缓解多孔钽金属与骨组织黏膜的摩擦,提高预后,减少因假体松动而需再次翻修的概率[54]。总之,3DPPTa从疗效上看与传统手术效果相仿,但简化手术过程、降低手术难度,从术前、术中及术后的不良反应来看,3DPPTa具有传统手术不可比拟的优势,其在髋关节的置换中将有望逐步代替传统假体,延长传统假体使用年限,成为骨科相关领域的新型假体材料。 2.5 3DPPTa方案的不足及改正措施 2.5.1 临床研究质量较低 相关文献多为单中心非盲性研究,存在偏倚现象、尚无大量临床证据支持长期预后效果。随着生物工程及3D打印技术的逐渐成熟,3D打印技术在临床上的应用越来越广泛,3DPPTa近年在治疗发育性髋臼发育不良中逐渐成为研究热点,但3DPPTa多为动物实验研究及单中心非盲性研究,存在偏倚现象,尚无大量临床数据证实发育性髋关节发育不良患者在术后10-20年以上中长期预后效果,是否有显著假体移位、脱落等并发症,能否打破传统置换假体平均使用15年的限制,目前尚无定论。 2.5.2 钽熔点过高,导致表面存有少量未熔融的钽颗粒 由于钽金属熔点过高(2 996 ℃),加工过程中存在未熔融的钽颗粒,导致多孔钽金属表面较为粗糙;术后曾导致1例股外侧皮神经损伤,表现为大腿外侧皮肤感觉减退,基于营养神经对症治疗,术后6个月随访症状消失,有1例导致切口内脂肪液化,基于清创后痊愈[21]。 2.5.3 假体周围有感染风险[55] 有学者表示在表层涂一定量的Cu2+可增强其抗菌性,而在一定范围内,抗菌率随着Cu离子含量增加而不断增加[52],目前,还需更多的实验研究证实。 2.5.4 抗疲劳性强度较低 3DPPTa通过实验证明多孔结构材料虽有效降低种植体的弹性模量,但同时也降低了种植假体在体内抗疲劳强度,较传统的致密的假体更容易断裂[21];而且制备成型相对困难。 2.5.5 材料制备使用成本较高 多孔钽使用成本较其他植入物假体高[4],对生产设备要求较高,生产率低;媒体导入的相应格式有限,通过DICOM转载、计算机整合再通过3D打印的不同技术来进行个体化打印,生产周期长;难以应用于紧急治疗当中,限制了其广泛的发展及使用[56]。若能降低Ta粉原材料价格及质量问题,减少3DPPTa的生产成本及生产周期,研制更专业化的装备和工艺并提高熔融技术或表面涂层技术使假体表面更加光滑[56-57];且在假体的高孔隙率与其稳定性中找到平衡,那么必将加速3DPPTa在临床上的应用,且有更多更大的临床样本数据及更长的随访时间来证实其作为新型假体材料的优势。 "

| [1] LI Y, GUO Y, LI M, et al. Acetabular index is the best predictor of late residual acetabular dysplasia after closed reduction in developmental dysplasia of the hip. Int Orthop. 2018;42(3):631-640. [2] LUO R, LI G, LI B, et al. Comparison of the modified Smith-Petersen (S-P) and ilioinguinal (I-I) approaches for periacetabular osteotomy in adult developmental dysplasia of the hip: a retrospective study. J Orthop Surg Res. 2021;16(1):157. [3] YAN L, WANG P, Zhou H. 3D Printing navigation template used in total hip arthroplasty for developmental dysplasia of the hip. Indian J Orthop. 2020;54(6):856-862. [4] LOGANATHAN B, SHARMA V, KUMAR MR, et al. Acetabulum reconstruction with tantalumcup and augments in dysplastic hip type 4 using 3D printing technology. J Orthop Case Rep. 2020;10(7):18-21. [5] ERTVRK C, AYYILDIZ S, ERDOL C. Orthopedics and 3D technology in Turkey: a preliminary report. Joint Dis Relat Surg. 2021;32(2):279-289. [6] QIAN H, LEI T, LEI P, et al. Additively manufactured tantalum implants for repairing bone defects: a systematic review. Tissue Eng Part B Rev. 2021; 27(2):166-180. [7] HUA L, LEI T, QIAN H, et al. 3D-printed porous tantalum: recent application in various drug delivery systems to repair hard tissue defects. Expert Opin Drug Deliv. 2021;18(5):625-634. [8] CROWE JF, MANI VJ, RANAWAT CS. Total hip replacement in congenital dislocation and dysplasia of the hip. J Bone Joint Surg Am. 1979;61(1):15-23. [9] WANG H, SU K, SU L, et al. Comparison of 3D-printed porous tantalum and titanium scaffolds on osteointegration and osteogenesis. Mater Sci Eng C Mater Biol Appl. 2019;104:109908. [10] 伍旭林,邱冰,朱伟民,等.钽金属垫块翻修术联合3D打印技术在Paprosky 3型髋臼骨缺损中的疗效评价[J].中国老年学杂志,2019,39(20): 4992-4995. [11] PAPROSKY WG, PERONA PG, LAWRENCE JM. Acetabular defect classification and surgical reconstruction in revision arthroplasty. A 6-year follow-up evaluation. J Arthroplasty. 1994;9(1):33-44. [12] 石永言,刘天婧,赵群,等.中国人髋关节中心边缘角正常值的测量[J].中华小儿外科杂志,2010,31(2):118-122. [13] ONAC O, ALPAY Y, YAPICI F, et al. Correlation of postoperative magnetic resonance image measurements with persisting acetabular dysplasia in open reduction of developmental hip dysplasia. Jt Dis Relat Surg. 2021; 32(2):461-467. [14] ANDERSON LA, ANDERSON MB, ERICKSON JA, et al. Acetabular wall indices help to distinguish acetabular coverage in asymptomatic adults with varying morphologies. Clin Orthop Relat Res. 2017;475(4):1027-1033. [15] AYANOGLU T, ATAOGLU MB, TOKGOZ N, et al. Assessing the risk of asymptomatic dysplasia in parents of children with developmental hip dysplasia. Acta Orthop Traumatol Turc. 2019,53(5):346-350. [16] OKUZU Y, GOTO K, KAWATA T, et al. The relationship between subluxation percentage of the femoroacetabular joint and acetabular width in asian women with developmental dysplasia of the hip. J Bone Joint Surg Am. 2017;99(7):e31. [17] WIBERG G. Shelf operation in congenital dysplasia of the acetabulum and in subluxation and dislocation of the hip. J Bone Joint Surg Am. 1953;35-A(1): 65-80. [18] VAUDREUIL NJ, MCCLINCY MP. Evaluation and treatment of borderline dysplasia: moving beyond the lateral center edge angle. Curr Rev Musculoskelet Med. 2020;13(1):28-37. [19] MIETTINEN HJ, MIETTINEN SS, Kettunen JS, et al. Revision hip arthroplasty using a porous tantalum acetabular component. Hip Int. 2021;31(6):782-788. [20] WANG F, CHEN H, YANG P, et al. Three-dimensional printed porous tantalum prosthesis for treating inflammation after total knee arthroplasty in one-stage surgery-a case report. J Int Med Res. 2020;48(3):1219690832. [21] SU KX, Ji P, WANG H, et al. In vivo study of 3D printed porous tantalum implant on osseointegration. Hua Xi Kou Qiang Yi Xue Za Zhi. 2018;36(3): 291-295. [22] WANG L, HU X, MA X, et al. Promotion of osteointegration under diabetic conditions by tantalum coating-based surface modification on 3-dimensional printed porous titanium implants. Colloids Surf B Biointerfaces. 2016; 148(16):440-452. [23] BALLA VK, BANERJEE S, BOSE S, et al. Direct laser processing of a tantalum coating on titanium for bone replacement structures. Acta Biomater. 2010; 6(6):2329-2334. [24] 吴先哲.骨科植入用多孔钽激光3D打印成形工艺及生物力学性能研究[D].北京:机械科学研究总院,2021. [25] FERNANDEZ-FAIREN M, MURCIA A, IGLESIAS R, et al. Osteointegration of porous tantalum stems implanted in avascular necrosis of the hip. Acta Ortop Mex. 2008;22(4):215-221. [26] RAN Q, YANG W, HU Y, et al. Osteogenesis of 3D printed porous Ti6Al4V implants with different pore sizes. J Mech Behav Biomed Mater. 2018; 84(18):1-11. [27] 程亮亮,赵德伟,杨磊,等. 3D打印多孔钽金属髋臼加强块在成人DDH髋关节重建术中的应用[J].中华骨科杂志,2018,38(11):650-657. [28] LI Z, CHEN G, XIANG Y, et al. Treatment of massive iliac chondrosarcoma with personalized three-dimensional printed tantalum implant: a case report and literature review. J Int Med Res. 2020;48(10):1220759060. [29] SING SL, WANG S, AGARWALA S, et al. Fabrication of titanium based biphasic scaffold using selective laser melting and collagen immersion. Int J Bioprint. 2017;3(1):7. [30] BEAUREGARD N, AL-FURAIJI M, Dias G, et al. Enhancing iCVD modification of electrospun membranes for membrane distillation using a 3D printed scaffold. Polymers (Basel). 2020;12(9):2074. [31] CHENG C, GUPTA M. Surface functionalization of 3D-printed plastics via initiated chemical vapor deposition. Beilstein J Nanotechnol. 2017;8:1629-1636. [32] LU S, XIN X, HUANG W, et al. Progress in clinical application of 3D printed navigational template in orthopedic surgery. Nan Fang Yi Ke Da Xue Xue Bao. 2020;40(8):1220-1224. [33] CHENG R, ZHANG H, Kernkamp WA, et al. Relations between the Crowe classification and the 3D femoral head displacement in patients with developmental dysplasia of the hip. BMC Musculoskelet Disord. 2019; 20(1):530. [34] ZENG C, XING W, WU Z, et al. A combination of three-dimensional printing and computer-assisted virtual surgical procedure for preoperative planning of acetabular fracture reduction. Injury. 2016;47(10):2223-2227. [35] ZHENG P, XU P, YAO Q, et al. 3D-printed navigation template in proximal femoral osteotomy for older children with developmental dysplasia of the hip. Sci Rep. 2017;7(1):44993. [36] LIANG S, XIE J, WANG F, et al. Application of three-dimensional printing technology in peripheral hip diseases. Bioengineered. 2021;12(1):5883-5891. [37] 刘洋,吴先哲,马幸双,等.个性化3D打印多孔钽植入假体设计的基本原则[J].重庆医学,2021,50(16):2822-2825. [38] ZHANG YZ, CHEN B, LU S, et al. Preliminary application of computer-assisted patient-specific acetabular navigational template for total hip arthroplasty in adult single development dysplasia of the hip. Int J Med Robot. 2011;7(4): 469-474. [39] FENG ZH, LI XB, PHAN K, et al. Design of a 3D navigation template to guide the screw trajectory in spine: a step-by-step approach using Mimics and 3-Matic software. J Spine Surg. 2018;4(3):645-653. [40] ZHANG H, ZHOU J, GUAN J, et al. How to restore rotation center in total hip arthroplasty for developmental dysplasia of the hip by recognizing the pathomorphology of acetabulum and Harris fossa? J Orthop Surg Res. 2019;14(1):339. [41] JOHNSTON RC, BRAND RA, CROWNINSHIELD RD. Reconstruction of the hip. A mathematical approach to determine optimum geometric relationships. J Bone Joint Surg Am. 1979;61(5):639-652. [42] TU Q, DING HW, CHEN H, et al. Preliminary application of 3D-printed individualised guiding templates for total hip arthroplasty in Crowe type IV developmental dysplasia of the hip. Hip Int. 2020. doi:10.1177/1120700020948006. [43] ZHANG H, GUAN JZ, ZHANG Z, et al. Restoring rotation center in total hip arthroplasty for developmental dysplasia of the hip with the assistance of three dimensional printing technology: a pilot study. J Orthop Surg. 2022;14(1):119-128. [44] ZHENG P, YAO Q, XU P, et al. Application of computer-aided design and 3D-printed navigation template in Locking Compression Pediatric Hip Plate (TauMu) placement for pediatric hip disease. Int J Comput Assist Radiol Surg. 2017;12(5):865-871. [45] TOMAZEVIC M, KRISTAN A, KAMATH AF, et al. 3D printing of implants for patient-specific acetabular fracture fixation: an experimental study. Eur J Trauma Emerg Surg. 2021;47(5):1297-1305. [46] ZHANG H, LIU Y, DONG Q, et al. Novel 3D printed integral customized acetabular prosthesis for anatomical rotation center restoration in hip arthroplasty for developmental dysplasia of the hip crowe type III: a case report. Medicine (Baltimore). 2020;99(40):e22578. [47] 黎庆钿,林博夫,陈学潘,等.3D技术辅助钽金属块植入修复严重髋臼骨缺损的早期疗效[J].中华骨科杂志,2020,40(3):129-137. [48] FAN H, DENG S, TANG W, et al. Highly porous 3D printed tantalum scaffolds have better biomechanical and microstructural properties than titanium scaffolds. Biomed Res Int. 2021;2021:2899043. [49] GUO Y, XIE K, JIANG W, et al. In Vitro and in Vivo Study of 3D-printed porous tantalum scaffolds for repairing bone defects. ACS Biomater Sci Eng. 2019;5(2):1123-1133. [50] 陈检文,董立明,蒋科,等.髋臼假体安装位置与无菌性松动的相关分析[J]中国矫形外科杂志,2022,30(1):1-5. [51] DOU X, WEI X, LIU G, et al. Effect of porous tantalum on promoting the osteogenic differentiation of bone marrow mesenchymal stem cells in vitro through the MAPK/ERK signal pathway. J Orthop Translat. 2019;19:81-93. [52] DING Z, WANG Y, ZHOU Q, et al. Microstructure, wettability, corrosion resistance and antibacterial property of Cu-MTa2O5 multilayer composite coatings with different cu incorporation contents. Biomolecules. 2019; 10(1):28. [53] SOWA M, SIMKA W. Electrochemical impedance and polarization corrosion studies of tantalum surface modified by DC plasma electrolytic oxidation. Materials (Basel). 2018;11(4):545. [54] MA L, CHENG S, Ji X, et al. Immobilizing magnesium ions on 3D printed porous tantalum scaffolds with polydopamine for improved vascularization and osteogenesis. Mater Sci Eng C Mater Biol Appl. 2020;117:111303. [55] 刘旭.金属加强块在严重髋臼骨缺损重建中的临床应用[D].长春:吉林大学,2020. [56] 杨柳,王富友.医学3D打印多孔钽在骨科的应用[J].第三军医大学学报,2019,41(19):1859-1866. [57] 吴昊,郭征. 3D打印多孔钛与钛合金及多孔钽骨科植入物的性能比较及应用展望[J].中华创伤骨科杂志,2020,22(10):916-920. [58] ZHOU Y, KANG X, LI C, et al. Application of a 3-dimensional printed navigation template in Bernese periacetabular osteotomies: a cadaveric study. Medicine (Baltimore). 2016;95(50):e5557. |
| [1] | Wang Yanjin, Zhou Yingjie, Chai Xubin, Zhuo Hanjie. Meta-analysis of the efficacy and safety of 3D printed porous titanium alloy fusion cage in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1434-1440. |
| [2] | Peng Zhixin, Yan Wengang, Wang Kun, Zhang Zhenjiang. Finite element analysis and structural optimization design of 3D printed forearm braces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1340-1345. |
| [3] | Chai Hao, Yang Deyong, Zhang Lei, Shu Li. 3D printing personalized osteotomy guide technology versus conventional total knee arthroplasty on the accuracy of lower limb force alignment: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 646-654. |
| [4] | Zhai Hongjie, Han Guanda, Li Lei, Dong Xiaohui, Jiang Zhiquan, Lou Feiyun. 3D printed polyetheretherketone material for skull defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 380-384. |
| [5] | Jiang Haifang, Liu Rong, Hu Peng, Chen Wei, Wei Zairong, Yang Chenglan, Nie Kaiyu. Application of 3D printing technology in the precise and personalized treatment of cleft lip and palate [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 413-419. |
| [6] | Chen Jingqiao, Li Ying, Meng Maohua, Xu Xingxing, Wang Qinying, Wang Huan, Lu Jing, Shu Jiayu, Dong Qiang. Research progress in platelet-rich fibrin in stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 441-446. |
| [7] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [8] | Song Yuxin, Zhang Tongtong, Niu Jianxiong, Wang Zengping, Wen Jie, Zhang Qunli, Xue Wen, Liu Lin. Precise screw placement of 3D printing model and orthopedic robot in spinal deformity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 904-907. |
| [9] | Wang Ruanbin, Cheng Liqian, Chen Kai. Application and value of polymer materials in three-dimensional printing biological bones and scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 610-616. |
| [10] | Huang Shibo, Xie Hui, Wang Zongpu, Wang Weidan, Qin Kairong, Zhao Dewei. Application of degradable high-purity magnesium screw in the treatment of developmental dysplasia of the hip [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 493-498. |
| [11] | Yang Yi, Cao Guangru, Wang Chong, Yuan Hao, Cai Yuqiang. 3D-printed thoracolumbar spine tuberculosis model and guide plate to guide the accuracy and safety of surgery [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5798-5806. |
| [12] | He Guowen, Hu Baijun, Gao Dawei, Chen Liang. Treatment of Schatzker type V and VI tibial plateau fractures with 3D printing preoperative planning combined with double reverse traction device [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5764-5769. |
| [13] | Liu Chang, Xu Ling. Modified coaxial 3D-printed akermanite scaffold with hollow-pipe structure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5425-5429. |
| [14] | Ma Ning, Wang Cong, Liu Ningqiang, Xie Tong, Yang Chaojian, Guo Chongjun, Niu Dongsheng, Liang Yuqi. Stability of 3D-printed titanium trabecular metal socket cups during total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5278-5282. |
| [15] | Sheng Xiaolei, Liu Su, Wang Jin, Zhao Lei, Zhu Yi, Zhang Wei, Gu Qi, Yuan Feng, Tian Shoujin, Ge Jianfei. Femoral neck fractures in middle-aged and young adults using femoral neck system assisted by 3D printed guide plate [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5290-5296. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||