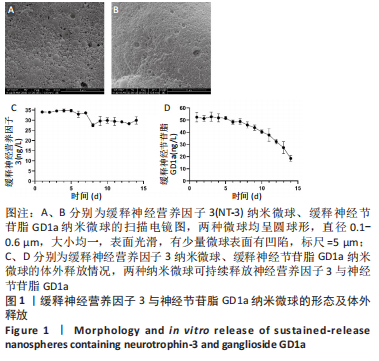
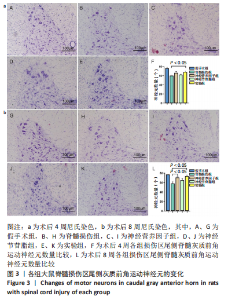
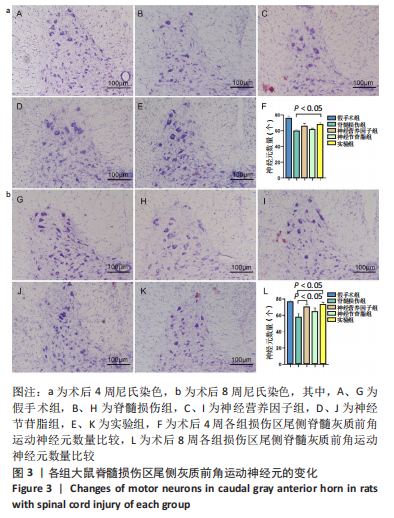
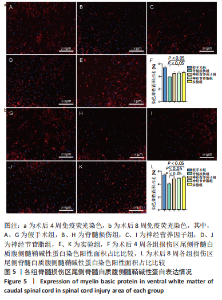
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (16): 2518-2524.doi: 10.12307/2023.168
Previous Articles Next Articles
Repair effect of poly(lactic-co-glycolic acid) nanospheres with sustained-release of neurotrophin-3 and ganglioside GD1a on spinal cord injury in rats
Xia Yu1, 2, Sun Jia2, Qi Zhengyan1, 2, Ma Lin1, 2, Ma Wenqian2, Niu Jianguo1, 2, Wen Yujun1, 2
- 1Department of Human Anatomy and Histology Embryology, School of Basic Medicine, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China; 2Ningxia Key Laboratory of Craniocerebral Diseases, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China
-
Received:2022-04-09Accepted:2022-06-08Online:2023-06-08Published:2022-11-10 -
Contact:Wen Yujun, Professor, Master’s supervisor, Department of Human Anatomy and Histology Embryology, School of Basic Medicine, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China; Ningxia Key Laboratory of Craniocerebral Diseases, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China -
About author:Xia Yu, Master candidate, Department of Human Anatomy and Histology Embryology, School of Basic Medicine, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China; Ningxia Key Laboratory of Craniocerebral Diseases, Ningxia Medical University, Yinchuan 750004, Ningxia Hui Autonomous Region, China -
Supported by:Ningxia Natural Science Foundation Project, No. 2020AAC03182 (to WYJ); National Natural Science Foundation of China, No. 81660216 (to WYJ); Ningxia Key Research & Development Program, No. 2021BEG03096 (to WYJ)
CLC Number:
Cite this article
Xia Yu, Sun Jia, Qi Zhengyan, Ma Lin, Ma Wenqian, Niu Jianguo, Wen Yujun. Repair effect of poly(lactic-co-glycolic acid) nanospheres with sustained-release of neurotrophin-3 and ganglioside GD1a on spinal cord injury in rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2518-2524.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
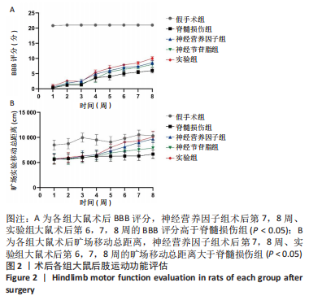
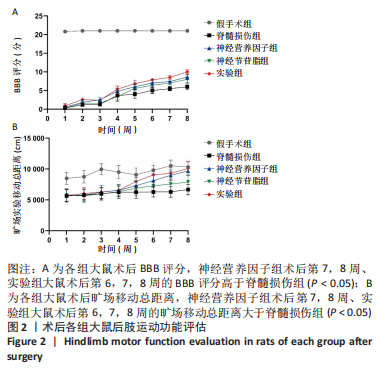
2.2 各组大鼠后肢运动功能恢复情况 所有大鼠术前BBB评分均为满分,术后无意外死亡。除假手术组外,其余4组大鼠术后1 d均呈截瘫状态,后肢运动功能丧失,靠前肢爬行,BBB评分为0分;从术后2周开始,大鼠后肢运动功能开始恢复,神经营养因子组、实验组大鼠术后第7,8周的BBB评分高于脊髓损伤组(P < 0.05),神经节苷脂组大鼠术后的BBB评分与脊髓损伤组比较差异无显著性意义(P > 0.05),见图2A。 旷场实验结果显示,神经营养因子组、实验组大鼠术后第7,8周的旷场移动总距离大于脊髓损伤组(P < 0.05),神经节苷脂组大鼠术后的旷场移动总距离与脊髓损伤组比较差异无显著性意义(P > 0.05),见图2B。"
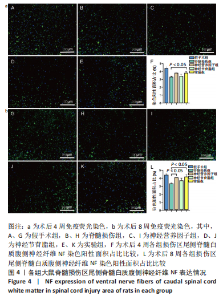
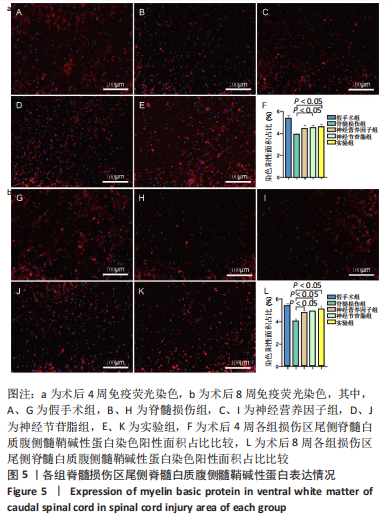
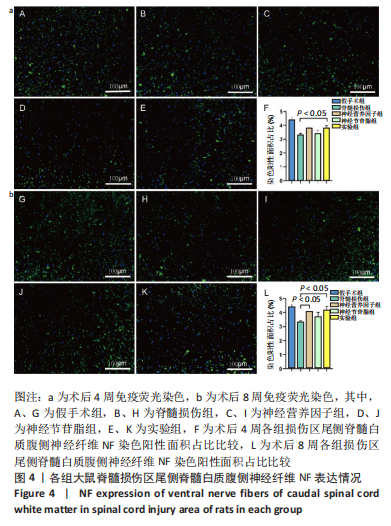
2.4 各组大鼠脊髓损伤区尾侧白质腹侧神经纤维NF表达 免疫荧光染色显示,与假手术组相比,其他4组大鼠脊髓损伤区尾侧白质腹侧神经纤维NF表达均不同程度地减少;术后4周,实验组大鼠脊髓损伤区尾侧脊髓白质腹侧神经纤维NF表达多于脊髓损伤组(P < 0.05),神经营养因子组、神经节苷脂组大鼠脊髓损伤区尾侧脊髓白质腹侧神经纤维NF表达与脊髓损伤组比较差异无显著性意义(P > 0.05)。术后8周,神经营养因子组、实验组大鼠脊髓损伤区尾侧脊髓白质腹侧神经纤维NF表达多于脊髓损伤组(P < 0.05),神经节苷脂组大鼠脊髓损伤区尾侧脊髓白质腹侧神经纤维NF表达与脊髓损伤组比较差异无显著性意义(P > 0.05),见图4。"
| [1] GUO S, REDENSKI I, LANDAU S, et al. Prevascularized Scaffolds Bearing Human Dental Pulp Stem Cells for Treating Complete Spinal Cord Injury. Adv Healthc Mater. 2020;9(20):e2000974. [2] SHI B, DING J, LIU Y, et al. ERK1/2 pathway-mediated differentiation of IGF-1-transfected spinal cord-derived neural stem cells into oligodendrocytes. PloS One. 2014;9(8):e106038. [3] YANG B, ZHANG F, CHENG F, et al. Strategies and prospects of effective neural circuits reconstruction after spinal cord injury. Cell Death Dis. 2020;11(6):439. [4] GU Y, ZHANG R, JIANG B, et al. Repair of Spinal Cord Injury by Inhibition of PLK4 Expression Through Local Delivery of siRNA-Loaded Nanoparticles. J Mol Neurosci. 2021;72(3):544-554. [5] BADHIWALA JH, AHUJA CS, FEHLINGS MG. Time is spine: a review of translational advances in spinal cord injury. J Neurosurg Spine. 2019; 30(1):1-18. [6] YE J, XUE R, JI ZY, et al. Effect of NT-3 on repair of spinal cord injury through the MAPK signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(5):2165-2172. [7] MUHEREMU A, SHU L, LIANG J, et al. Sustained delivery of neurotrophic factors to treat spinal cord injury. Transl Neurosci. 2021;12(1):494-511. [8] HAN Q, XU X. Neurotrophin-3-mediated locomotor recovery: a novel therapeutic strategy targeting lumbar neural circuitry after spinal cord injury. Neural Regen Res. 2020;15(12):2241-2242. [9] XU Z, ZHANG L, ZHOU Y, et al. Histological and functional outcomes in a rat model of hemisected spinal cord with sustained VEGF/NT-3 release from tissue-engineered grafts. Artif Cells Nanomed Biotechnol. 2020;48(1):362-376. [10] CHINNOCK P, ROBERTS I. Gangliosides for acute spinal cord injury. Cochrane Database Syst Rev. 2005;2005(2):CD004444. [11] HE Y, LI J. Ganglioside GD1a inhibits LPS-induced inflammation in types of cells. Int Immunopharmacol. 2016;31:222. [12] AZZAZ F, YAHI N, DI SCALA C, et al. Ganglioside binding domains in proteins: Physiological and pathological mechanisms. Adv Protein Chem Struct Biol. 2022;128:289-324. [13] QIN J, SIKKEMA AH, VAN DER BIJ K, et al. GD1a Overcomes Inhibition of Myelination by Fibronectin via Activation of Protein Kinase A: Implications for Multiple Sclerosis. J Neurosci. 2017;37(41):9925-9938. [14] RAWJI KS, GONZALEZ MARTINEZ GA, SHARMA A, et al. The Role of Astrocytes in Remyelination. Trends Neurosci. 2020;43(8):596-607. [15] HUANG F, CHEN T, CHANG J, et al. A conductive dual-network hydrogel composed of oxidized dextran and hyaluronic-hydrazide as BDNF delivery systems for potential spinal cord injury repair. Int J Biol Macromol. 2021;167:434-445. [16] AHI ZB, ASSUNCAO-SILVA RC, SALGADO AJ, et al. A combinatorial approach for spinal cord injury repair using multifunctional collagen-based matrices: development, characterization and impact on cell adhesion and axonal growth. Biomed Mater. 2020;15(5):55024. [17] 卢仁培,邹志晨,赵丰年,等.聚乳酸-羟基乙酸共聚物复合支架在骨缺损修复再生中的作用与应用[J].中国组织工程研究,2022, 26(28):4525-4531. [18] YU S, YAO S, WEN Y, et al. Angiogenic microspheres promote neural regeneration and motor function recovery after spinal cord injury in rats. Sci Rep. 2016;6:33428. [19] JU R, WEN Y, GOU R, et al. The Experimental Therapy on Brain Ischemia by Improvement of Local Angiogenesis with Tissue Engineering in the Mouse. Cell Transplant. 2014;23(1_suppl):83-95. [20] WEN Y, YU S, WU Y, et al. Spinal cord injury repair by implantation of structured hyaluronic acid scaffold with PLGA microspheres in the rat. Cell Tissue Res. 2016;364(1):17-28. [21] HAN Q, ORDAZ JD, LIU N, et al. Descending motor circuitry required for NT-3 mediated locomotor recovery after spinal cord injury in mice. Nat Commun. 2019;10(1):5815. [22] BASSO DM, BEATTIE MS, BRESNAHAN JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12(1):1-21. [23] 董瑞,毕燕琳,王明山.旷场实验在鼠行为学研究中的应用[J].国际麻醉学与复苏杂志,2020,41(5):535-540. [24] WANG J, LI D, LIANG C, et al. Scar Tissue‐Targeting Polymer Micelle for Spinal Cord Injury Treatment. Small. 2020;16(8):1906415. [25] BADHIWALA JH, WILSON JR, FEHLINGS MG. Global burden of traumatic brain and spinal cord injury. Lancet Neurol. 2019;18(1):24-25. [26] ZOU Y, MA D, SHEN H, et al. Aligned collagen scaffold combination with human spinal cord-derived neural stem cells to improve spinal cord injury repair. Biomater Sci. 2020;8(18):5145-5156. [27] MARQUARDT LM, DOULAMES VM, WANG AT, et al. Designer, injectable gels to prevent transplanted Schwann cell loss during spinal cord injury therapy. Sci Adv. 2020;6(14):z1039. [28] LIU D, SHEN H, SHEN Y, et al. Dual‐Cues Laden Scaffold Facilitates Neurovascular Regeneration and Motor Functional Recovery After Complete Spinal Cord Injury. Adv Healthc Mater. 2021;10(10):2100089. [29] KWIECIEN J. Barriers to axonal regeneration after spinal cord injury: a current perspective. Neural Regen Res. 2022;17(1):85-86. [30] LI G, ZHANG B, SUN JH, et al. An NT-3-releasing bioscaffold supports the formation of TrkC-modified neural stem cell-derived neural network tissue with efficacy in repairing spinal cord injury. Bioact Mater. 2021; 6(11):3766-3781. [31] SMITH DR, DUMONT CM, PARK J, et al. Polycistronic Delivery of IL-10 and NT-3 Promotes Oligodendrocyte Myelination and Functional Recovery in a Mouse Spinal Cord Injury Model. Tissue Eng Part A. 2020; 26(11-12):672-682. [32] SONG YH, AGRAWAL NK, GRIFFIN JM, et al. Recent advances in nanotherapeutic strategies for spinal cord injury repair. Adv Drug Deliv Rev. 2019;148:38-59. [33] ZERAATPISHEH Z, MIRZAEI E, NAMI M, et al. Local delivery of fingolimod through PLGA nanoparticles and PuraMatrix-embedded neural precursor cells promote motor function recovery and tissue repair in spinal cord injury. Eur J Neurosci. 2021;54(4):5620-5637. [34] SANTHOSH KT, ALIZADEH A, KARIMI-ABDOLREZAEE S. Design and optimization of PLGA microparticles for controlled and local delivery of Neuregulin-1 in traumatic spinal cord injury. J Control Release. 2017; 261:147-162. [35] LOWRY N, GODERIE SK, LEDERMAN P, et al. The effect of long-term release of Shh from implanted biodegradable microspheres on recovery from spinal cord injury in mice. Biomaterials. 2011;33(10):2892-2901. [36] ZERAATPISHEH Z, MIRZAEI E, NAMI M, et al. Local delivery of fingolimod through PLGA nanoparticles and PuraMatrix-embedded neural precursor cells promote motor function recovery and tissue repair in spinal cord injury. Eur J Neurosci. 2021;54(4):5620-5637. [37] CONG Y, WANG C, WANG J, et al. NT-3 Promotes Oligodendrocyte Proliferation and Nerve Function Recovery After Spinal Cord Injury by Inhibiting Autophagy Pathway. J Surg Res. 2020;247:128-135. [38] WANG Y, WU W, WU X, et al. Remodeling of lumbar motor circuitry remote to a thoracic spinal cord injury promotes locomotor recovery. Elife. 2018;7:e39016. [39] GALLEGUILLOS D, WANG Q, STEINBERG N, et al. Anti-inflammatory role of GM1 and other gangliosides on microglia. J Neuroinflammation. 2022;19(1):9. [40] LIN Y, LI C, LI J, et al. NEP1-40-modified human serum albumin nanoparticles enhance the therapeutic effect of methylprednisolone against spinal cord injury. J Nanobiotechnol. 2019;17(1):12. [41] DE MIRANDA AS, DE BARROS J, TEIXEIRA AL. Is neurotrophin-3 (NT-3): a potential therapeutic target for depression and anxiety? Expert Opin Ther Targets. 2020;24(12):1225-1238. [42] YU Y, LI Z, MA F, et al. Neurotrophin‐3 stimulates stem Leydig cell proliferation during regeneration in rats. J Cell Mol Med. 2020;24(23): 13679-13689. [43] ELLIOTT DI, TATOR CH, SHOICHET MS. Local Delivery of Neurotrophin-3 and Anti-NogoA Promotes Repair After Spinal Cord Injury. Tissue Eng Part A. 2016;22(9-10):733-741. [44] MORADIAN H, KESHVARI H, FASEHEE H, et al. Combining NT3-overexpressing MSCs and PLGA microcarriers for brain tissue engineering: A potential tool for treatment of Parkinson’s disease. Mater Sci Eng C Mater Biol Appl. 2017;76:934-943. [45] MAGISTRETTI PJ, GEISLER FH, SCHNEIDER JS, et al. Gangliosides: Treatment Avenues in Neurodegenerative Disease. Front Neurol. 2019; 10:859. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [3] | Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198. |
| [4] | Yang Jiujie, Li Zhi, Wang Shujie, Tian Ye, Zhao Wei. Intraoperative neurophysiological monitoring of functional changes following durotomy with decompression for acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1232-1236. |
| [5] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [6] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [7] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [8] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [9] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [10] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [11] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [12] | Hao Liufang, Duan Hongmei, Wang Zijue, Hao Fei, Hao Peng, Zhao Wen, Gao Yudan, Yang Zhaoyang, Li Xiaoguang. Spatiotemporal dynamic changes of ependymal cells after spinal cord injury in transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 883-889. |
| [13] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [14] | Zhang Qijian, Xu Ximing. Acquisition and application of ectodermal mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 928-934. |
| [15] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||