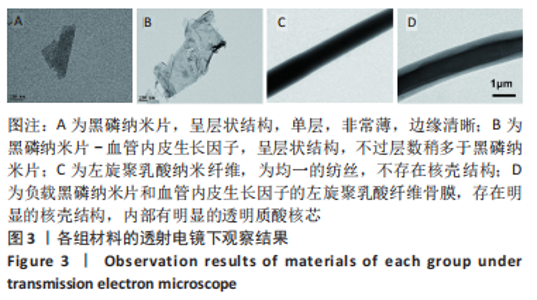
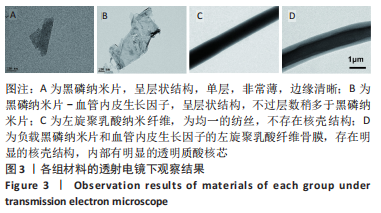
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (25): 4028-4037.doi: 10.12307/2023.163
Previous Articles Next Articles
In vitro experiment of composite nanofibrous periosteum to promote vascularization and osteogenic mineralization
Wang Wenbo, Xu Jingzhi, Wu Liang, Xi Kun, Xin Tianwen, Tang Jincheng, Gu Yong, Chen Liang
- Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China
-
Received:2022-02-16Accepted:2022-05-09Online:2023-09-08Published:2023-01-17 -
Contact:Chen Liang, Chief physician, Professor, Doctoral supervisor, Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China Gu Yong, Associate chief physician, Associate professor, Master’s supervisor, Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
About author:Wang Wenbo, Master candidate, Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China Xu Jingzhi, Master candidate, Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81772312, 81972078 (to CL)
CLC Number:
Cite this article
Wang Wenbo, Xu Jingzhi, Wu Liang, Xi Kun, Xin Tianwen, Tang Jincheng, Gu Yong, Chen Liang. In vitro experiment of composite nanofibrous periosteum to promote vascularization and osteogenic mineralization[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 4028-4037.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

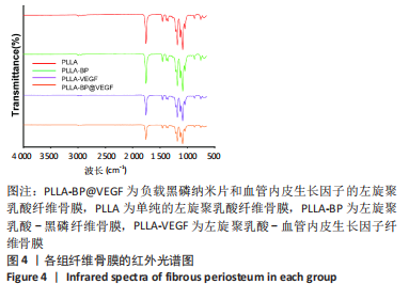
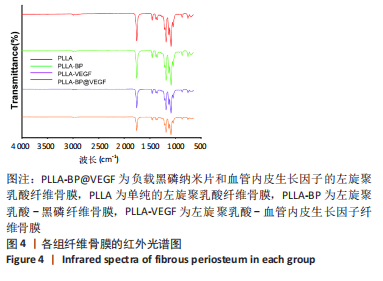
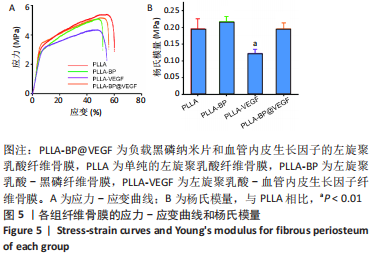
2.4 电纺丝纤维骨膜的力学拉伸 添加透明质酸、黑磷后,纤维骨膜因为内部元素变化,相应的力学性能会发生相应改变,对各组纤维骨膜进行了力学拉伸测试,绘制了应力-应变曲线,分析添加前后结果的变化,结果见图5A,所有纤维骨膜都表现出一定的抗拉强度,其中PLLA-BP、PLLA-VEGF和PLLA-BP@VEGF这3种微溶胶纤维的抗拉强度相近。与纯PLLA静电纺丝纤维相比,其他3组微溶胶纤维的最大抗拉强度略有下降,尤其是PLLA-VEGF支架,这可能与透明质酸核的植入有关;而黑磷的加入在一定程度上增加了纤维骨膜力学强度。PLLA-BP纤维骨膜的杨氏模量为(0.217±0.015) MPa,在4组中强度最大;PLLA纤维骨膜的杨氏模量为(0.196±0.029) MPa,PLLA-BP@VEGF纤维骨膜杨氏模量为(0.197±0.017) MPa,PLLA-VEGF纤维膜杨氏模量最小,为(0.123±0.011) MPa,见图5B。"
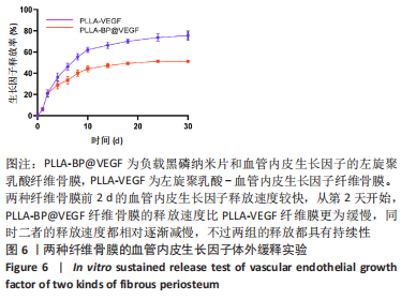
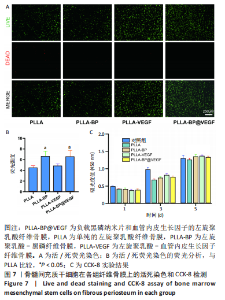
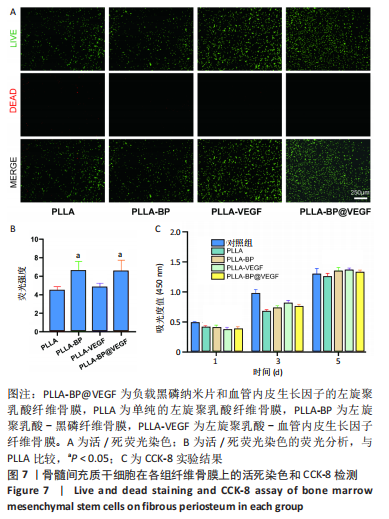
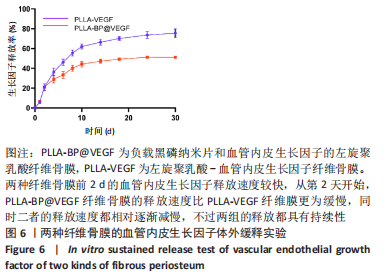
2.5 纤维骨膜中血管内皮生长因子的体外缓释 PLLA-VEGF纤维骨膜和PLLA-BP@VEGF纤维骨膜前2 d的血管内皮生长因子释放速度较快,总释放量分别为(20.99±2.81)%和(21.05±2.57)%;从第2天开始,PLLA-BP@VEGF纤维骨膜的释放速度比PLLA-VEGF纤维膜更为缓慢,同时二者的释放速度都相对逐渐减慢,不过两组的释放都具有持续性;至第14天,两者的总释放量分别为 (66.35±2.43)%和(47.19±1.64)%;最终释放时间达到30 d,累积释放量分别为(75.56±3.67)%和(51.16±0.86)%,见图6。至于PLLA-VEGF纤维骨膜释放的速度更为减缓,释放的总量也较少,这些可以归因于黑磷纳米片的吸附能力,直接影响了纤维内部生物因子的释放动力学。 2.6 电纺丝纤维骨膜的细胞生物相容性 "
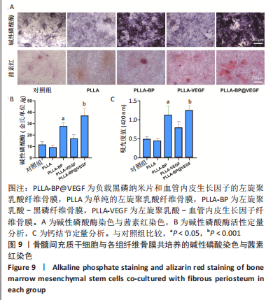
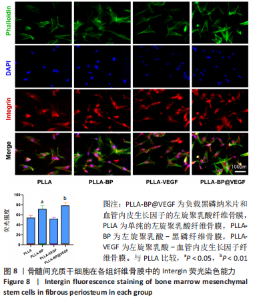
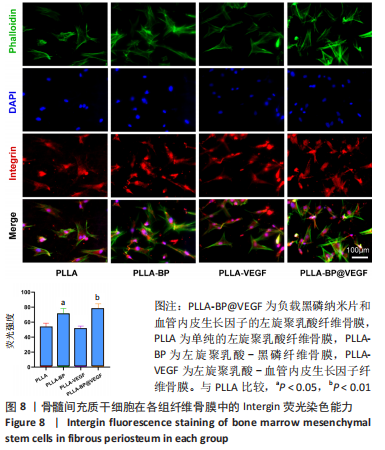
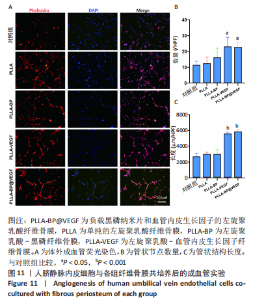
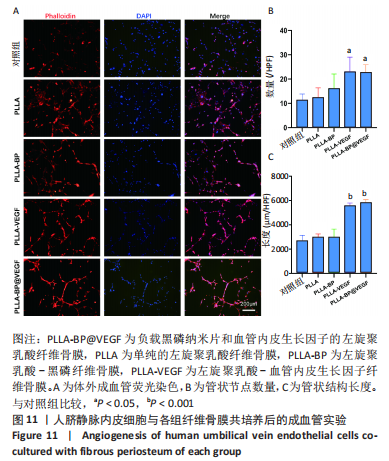
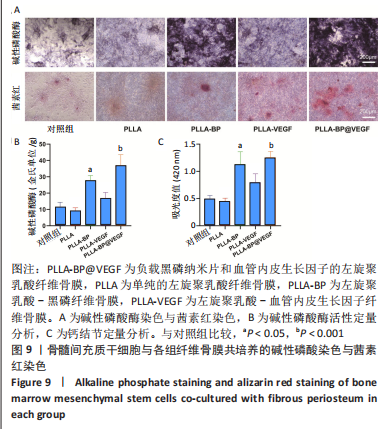
2.7 电纺丝纤维骨膜的体外成骨能力 碱性磷酸酶作为成骨分化早期的指标,可以反映出纤维骨膜对骨髓间充质干细胞早期成骨分化的影响。 图9A的碱性磷酸酶染色可见,各组纤维骨膜的成骨分化都存在上升趋势,因为分化程度表达不一,不同组别的染色颜色也由浅蓝到深蓝不等,其中对照组和纯PLLA组的染色颜色较浅,密度也较低;与PLLA-BP、PLLA-VEGF组相比,骨髓间充质干细胞在与PLLA-BP@VEGF复合纤维膜一起共培养后,染色颜色最深,体现出黑磷和血管内皮生长因子协同的促成骨作用。值得一提的是,PLLA-BP组的染色深度明显较PLLA-VEGF组更深,可能是因为黑磷募集成骨前驱细胞,较早启动细胞进行成骨分化。图9B中碱性磷酸酶活性的定量检测也印证了结果。 钙结节作为成骨分化的晚期标识物,经过茜素红染色后呈铁锈色,图9A茜素红染色显示,不同纤维骨膜组的钙结节形状各异,颜色深浅也各有不同。骨髓间充质干细胞在对照组中仅有极少红色沉淀;在PLLA组纤维骨膜上仅有极少量红染结构;在PLLA-VEGF组中可见散在的锈红色结节,分布分散;骨髓间充质干细胞在PLLA-BP和PLLA-BP@VEGF纤维骨膜表面都形成相对较深的铁锈色结节,说明黑磷良好的成骨能力;其中,PLLA-BP组可见一处明显深染铁锈色结节,而骨髓间充质干细胞在PLLA-BP@VEGF组纤维表面形成的铁锈色结节面积最大,表明黑磷和血管内皮生长因子的协同作用更好地促进了骨髓间充质干细胞的成骨矿化。用高氯酸溶解钙结节后测定溶解液420 nm的吸光度值,结果如图9C所示,PLLA-BP@VEGF组的吸光度值远远超出其他组,与茜素红染色结果基本一致。"
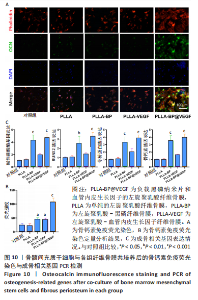
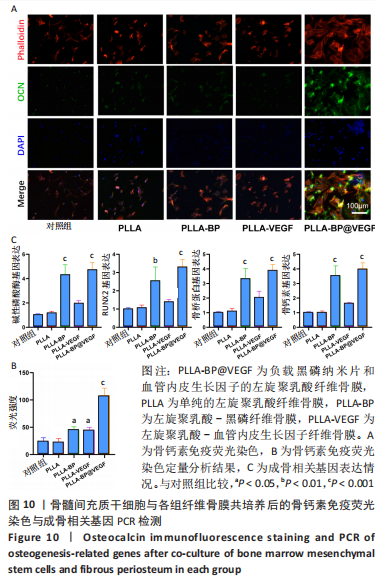
骨钙素也是观察晚期成骨分化的重要标志物。各组纤维骨膜骨钙素染色结果见图10A。当培养至21 d时进行,对细胞进行骨钙素免疫荧光染色,对照组的荧光比较微弱,PLLA组和PLLA-VEGF组的荧光强度也相对微弱;PLLA-BP组的荧光强度比上述3组明显增强;与其他组别相比,骨髓间充质干细胞在与PLLA-BP@VEGF复合纤维骨膜共培养后,骨钙素荧光最为明显,见图10B,这与碱性磷酸酶和钙结节测定所表达的强弱无明显差异。 图10C显示了骨髓间充质干细胞与纤维骨膜共培养7 d后的成骨相关基因表达情况PLLA-BP@VEGF组碱性磷酸酶、RUNX-2、骨桥蛋白、骨钙素的基因表达都要高于其他3组,具有显著优势;PLLA-BP组成骨基因表达水平相较PLLA组、PLLA-VEGF组也相对较高,这与碱性磷酸酶染色、茜素红染色和骨钙素染色结果无明显差异。"
| [1] AREALIS G, NIKOLAOU VS. Bone printing: new frontiers in the treatment of bone defects. Injury. 2015;46:S20-S22. [2] VERRIER S, ALINI M, ALSBERG E, et al. Tissue engineering and regenerative approaches to improving the healing of large bone defects. Eur Cell Mater. 2016;32:87-110. [3] ALONZO M, PRIMO FA, KUMAR SA, et al. Bone tissue engineering techniques, advances and scaffolds for treatment of bone defects. Curr Opin Biomed Eng. 2021;17:100248. [4] TANG D, TARE RS, YANG LY, et al. Biofabrication of bone tissue: approaches, challenges and translation for bone regeneration. Biomaterials. 2016;83:363-382. [5] BADYLAK SF, NEREM RM. Progress in tissue engineering and regenerative medicine. Proceed Nat Acad Sci. 2010;107(8):3285-3286. [6] YANG G, LIU H, CUI Y, et al. Bioinspired membrane provides periosteum-mimetic microenvironment for accelerating vascularized bone regeneration. Biomaterials. 2021;268:120561. [7] GUPTA S, TEOTIA AK, QAYOOM I, et al. Periosteum-Mimicking Tissue-Engineered Composite for Treating Periosteum Damage in Critical-Sized Bone Defects. Biomacromolecules. 2021;22(8):3237-3250. [8] FERNANDEZ DE GRADO G, KELLER L, IDOUX-GILLET Y, et al. Bone substitutes: a review of their characteristics, clinical use, and perspectives for large bone defects management. J Tissue Eng. 2018;9: 2041731418776819. [9] DIMITRIOU R, JONES E, MCGONAGLE D, et al. Bone regeneration: current concepts and future directions. BMC Med. 2011;9(1):66. [10] DWEK JR. The periosteum: what is it, where is it, and what mimics it in its absence? Skelet Radiol. 2010;39(4):319-323. [11] LI N, SONG J, ZHU G, et al. Periosteum tissue engineering-a review. Biomater Sci. 2016;4(11):1554-1561. [12] WANG Q, XU J, JIN H, et al. Artificial periosteum in bone defect repair-A review. Chin Chem Lett. 2017;28(9):1801-1807. [13] HE X, LI W, LIU K, et al. Anisotropic and robust hydrogels combined osteogenic and angiogenic activity as artificial periosteum. Composit Part B Eng. 2022:233. [14] QING Y, LI R, LI S, et al. Advanced Black Phosphorus Nanomaterials for Bone Regeneration. Int J Nanomedicine. 2020;15:2045-2058. [15] ZHOU Y, ZHANG MX, GUO ZN, et al. Recent advances in black phosphorus-based photonics, electronics, sensors and energy devices. Mater Horiz. 2017;4(6):997-1019. [16] LIU W, BI W, SUN Y, et al. Biomimetic organic-inorganic hybrid hydrogel electrospinning periosteum for accelerating bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110670. [17] DOGANOV RA, O’FARRELL EC, KOENIG SP, et al. Transport properties of pristine few-layer black phosphorus by van der Waals passivation in an inert atmosphere. Nat Commun. 2015;6(1):6647. [18] SUN Z, XIE H, TANG S, et al. Ultrasmall Black Phosphorus Quantum Dots: Synthesis and Use as Photothermal Agents. Angew Chem Int Ed Engl. 2015;54(39):11526-11530. [19] ZHU C, XU F, ZHANG L, et al. Ultrafast Preparation of Black Phosphorus Quantum Dots for Efficient Humidity Sensing. Chemistry. 2016;22(22): 7357-7362. [20] ZONG S, WANG L, YANG Z, et al. Black Phosphorus-Based Drug Nanocarrier for Targeted and Synergetic Chemophotothermal Therapy of Acute Lymphoblastic Leukemia. ACS Appl Mater Interfaces. 2019;11(6):5896-5902. [21] UTT KL, RIVERO P, MEHBOUDI M, et al. Intrinsic Defects, Fluctuations of the Local Shape, and the Photo-Oxidation of Black Phosphorus. ACS Cent Sci. 2015;1(6):320-327. [22] WU N, WANG XM, DAS CM, et al. Bioengineering applications of black phosphorus and their toxicity assessment. Environ Sci-Nano. 2021;8(12):3452-3477. [23] ZHANG Y, MA C, XIE J, et al. Black Phosphorus/Polymers: Status and Challenges. Adv Mater. 2021;33(37):e2100113. [24] DEVESCOVI V, LEONARDI E, CIAPETTI G, et al. Growth factors in bone repair. Chir Organi Mov. 2008;92(3):161-168. [25] APTE RS, CHEN DS, FERRARA N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell. 2019;176(6):1248-1264. [26] SARAN U, GEMINI PIPERNI S, CHATTERJEE S. Role of angiogenesis in bone repair. Arch Biochem Biophys. 2014;561:109-117. [27] LEE SJ, KIM ME, NAH H, et al. Vascular endothelial growth factor immobilized on mussel-inspired three-dimensional bilayered scaffold for artificial vascular graft application: In vitro and in vivo evaluations. J Colloid Interface Sci. 2019;537:333-344. [28] WANG XX, YU GF, ZHANG J, et al. Conductive polymer ultrafine fibers via electrospinning: Preparation, physical properties and applications. Prog Mater Sci. 2021:115. [29] RAHMATI M, MILLS DK, URBANSKA AM, et al. Electrospinning for tissue engineering applications. Prog Mater Sci. 2021:117. [30] ZHANG C, LI Y, WANG P, et al. Electrospinning of nanofibers: Potentials and perspectives for active food packaging. Compr Rev Food Sci Food Saf. 2020;19(2):479-502. [31] YU DG, WANG M, LI X, et al. Multifluid electrospinning for the generation of complex nanostructures. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020;12(3):e1601. [32] HU X, LIU S, ZHOU G, et al. Electrospinning of polymeric nanofibers for drug delivery applications. J Control Release. 2014;185:12-21. [33] YAZDANPANAH A, TAHMASBI M, AMOABEDINY G, et al. Fabrication and characterization of electrospun poly- L -lactide/gelatin graded tubular scaffolds: Toward a new design for performance enhancement in vascular tissue engineering. Prog Nat Sci Mater Int. 2015;25(5):405-413. [34] LURAGHI A, PERI F, MORONI L. Electrospinning for drug delivery applications: A review. J Control Release. 2021;334:463-484. [35] HE Y, LI X, MA J, et al. Programmable Codelivery of Doxorubicin and Apatinib Using an Implantable Hierarchical-Structured Fiber Device for Overcoming Cancer Multidrug Resistance. Small. 2019;15(8):e1804397. [36] ASADI H, GHAEE A, NOURMOHAMMADI J, et al. Electrospun zein/graphene oxide nanosheet composite nanofibers with controlled drug release as antibacterial wound dressing. Int J Polym Mater. 2019;69(3): 173-185. [37] ZHOU L, ZHU C, EDMONDS L, et al. Microsol-electrospinning for controlled loading and release of water-soluble drugs in microfibrous membranes. RSC Adv. 2014;4(81):43220-43226. [38] WU L, GU Y, LIU L, et al. Hierarchical micro/nanofibrous membranes of sustained releasing VEGF for periosteal regeneration. Biomaterials. 2020;227:119555. [39] HU K, OLSEN BR. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J Clin Invest. 2016;126(2):509-526. [40] STREET J, BAO M, DEGUZMAN L, et al. Vascular endothelial growth factor stimulates bone repair by promoting angiogenesis and bone turnover. Proc Natl Acad Sci U S A. 2002;99(15):9656. [41] SCHMITZ JP, HOLLINGER JO, MILAM SB. Reconstruction of bone using calcium phosphate bone cements: A critical review. J Oral Maxillofac Surg. 1999;57(9):1122-1126. [42] JEONG J, KIM JH, SHIM JH, et al. Bioactive calcium phosphate materials and applications in bone regeneration. Biomater Res. 2019;23:4. [43] LEVINGSTONE TJ, HERBAJ S, DUNNE NJ. Calcium Phosphate Nanoparticles for Therapeutic Applications in Bone Regeneration. Nanomaterials (Basel). 2019;9(11):1570. [44] BOHNER M, GBURECK U, BARRALET JE. Technological issues for the development of more efficient calcium phosphate bone cements: A critical assessment. Biomaterials. 2005;26(33):6423-6429. [45] WU S, HE F, XIE G, et al. Black Phosphorus: Degradation Favors Lubrication. Nano Lett. 2018;18(9):5618-5627. [46] HUANG K, WU J, GU Z. Black Phosphorus Hydrogel Scaffolds Enhance Bone Regeneration via a Sustained Supply of Calcium-Free Phosphorus. ACS Appl Mater Interfaces. 2019;11(3):2908-2916. [47] LEE YB, SONG SJ, SHIN YC, et al. Ternary nanofiber matrices composed of PCL/black phosphorus/collagen to enhance osteodifferentiation. J Ind Eng Chem. 2019;80:802-810. [48] Zhao W, Xue Z, Wang J, et al. Large-Scale, Highly Efficient, and Green Liquid-Exfoliation of Black Phosphorus in Ionic Liquids. ACS Appl Mater Interfaces. 2015;7(50):27608-27612. [49] ZILETTI A, CARVALHO A, TREVISANUTTO PE, et al. Phosphorene oxides: Bandgap engineering of phosphorene by oxidation. Phys Rev B. 2015;91(8):085407. [50] BHARDWAJ N, KUNDU SC. Electrospinning: A fascinating fiber fabrication technique. Biotechnol Adv. 2010;28(3):325-347. [51] DONG S, SUN J, LI Y, et al. Electrospun nanofibrous scaffolds of poly (l-lactic acid)-dicalcium silicate composite via ultrasonic-aging technique for bone regeneration. Mater Sci Eng C. 2014;35:426-433. [52] EREN BONCU T, USKUDAR GUCLU A, CATMA MF, et al. In vitro and in vivo evaluation of linezolid loaded electrospun PLGA and PLGA/PCL fiber mats for prophylaxis and treatment of MRSA induced prosthetic infections. Int J Pharm. 2020;573:118758. |
| [1] | Li Zikai, Zhang Chengcheng, Xiong Jiaying, Yang Xirui, Yang Jing, Shi Haishan. Potential effects of ornidazole on intracanal vascularization in endodontic regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-7. |
| [2] | Sun Yuting, Wu Jiayuan, Zhang Jian. Physical factors and action mechanisms affecting osteogenic/odontogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1531-1540. |
| [3] | Lou Guo, Zhang Min, Fu Changxi. Exercise preconditioning for eight weeks enhances therapeutic effect of adipose-derived stem cells in rats with myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1363-1370. |
| [4] | Aikepaer · Aierken, Chen Xiaotao, Wufanbieke · Baheti. Osteogenesis-induced exosomes derived from human periodontal ligament stem cells promote osteogenic differentiation of human periodontal ligament stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1388-1394. |
| [5] | De Ji, Suo Langda, Wei Yuchen, Wang Bin, Awangcuoji, Renqingcuomu, Cui Jiuzeng, Zhang Lei, Ba Gui. Comprehensive analysis of genes related to endometrial receptivity and alternative splicing events in northwest Tibetan cashmere goats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1429-1436. |
| [6] | Chen Yuning, Jiang Ying, Liao Xiangyu, Chen Qiongjun, Xiong Liang, Liu Yue, Liu Tong. Buqi Huoxue Compounds intervene with the expression of related factors and autophagy related proteins in a rat model of cerebral ischemia/reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1152-1158. |
| [7] | Han Haihui, Meng Xiaohu, Xu Bo, Ran Le, Shi Qi, Xiao Lianbo. Effect of fibroblast growth factor receptor 1 inhibitor on bone destruction in rats with collagen-induced arthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 968-977. |
| [8] | Zhao Shuai, Li Dongyao, Wei Suiyan, Cao Yijing, Xu Yan, Xu Guoqiang. Biocompatibility of poly(vinylidene fluoride) piezoelectric bionic periosteum prepared by electrospinning [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 730-737. |
| [9] | Wu Chen, Jiang Jiahui, Su Dou, Liu Chen, Ci Chao. Decellularized skin matrix/polyurethane blended fibrous scaffolds promote repair of skin defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 745-751. |
| [10] | Sun Xianjuan, Wang Qiuhua, Zhang Jinyi, Yang Yangyang, Wang Wenshuang, Zhang Xiaoqing. Adhesion, proliferation, and vascular smooth muscle differentiation of bone marrow mesenchymal stem cells on different electrospinning membranes [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 661-669. |
| [11] | Liu Haoyang, Xie Qiang, Shen Mengran, Ren Yansong, Ma Jinhui, Wang Bailiang, Yue Debo, Wang Weiguo . Application, research hotspots, and shortcomings of degradable zinc-based alloys in bone defect repair and reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 839-845. |
| [12] | Zhang Yu, Xu Ruian, Fang Lei, Li Longfei, Liu Shuyan, Ding Lingxue, Wang Yuexi, Guo Ziyan, Tian Feng, Xue Jiajia. Gradient artificial bone repair scaffold regulates skeletal system tissue repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 846-855. |
| [13] | Peng Yong, Hu Jiangping, Zhu Huan. Effects of low-load blood flow restriction exercise and high-intensity resistance exercise on the thigh microcirculation function of athletic young men [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(2): 393-401. |
| [14] | An Jiangru, Zhang Jinyi, Wang Qiuhua, Yang Yangyang, Wang Wenshuang, Zhang Xiaoqing. Mesenchymal stem cells combined with polycaprolactone-hyaluronic acid electrospinning membrane in repair of endometrial injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3369-3379. |
| [15] | Tian Jiayu, Li Duohua, Zhang Feng, Feng Hu, Sun Wei. Construction and performance evaluation of polycaprolactone/nanodiamond-phospholipid composite materials [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3380-3387. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||