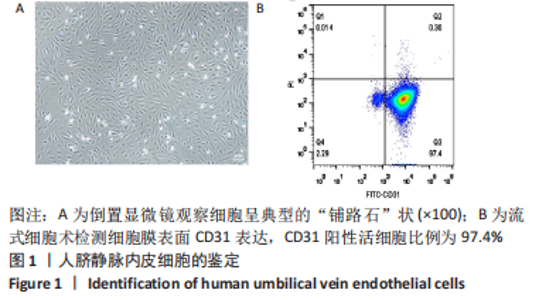
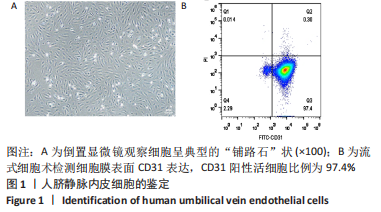
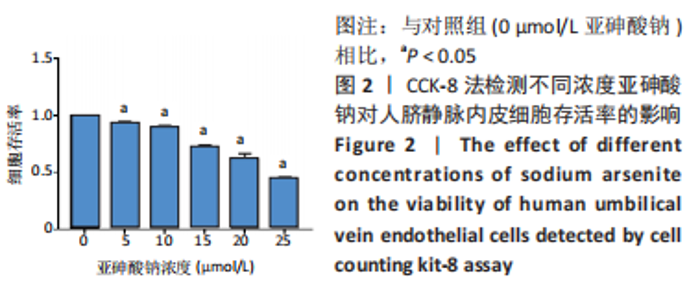
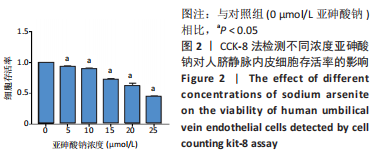
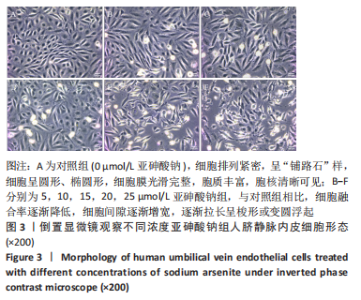
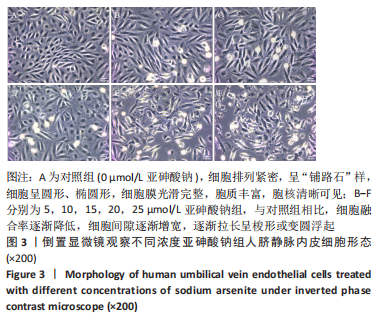
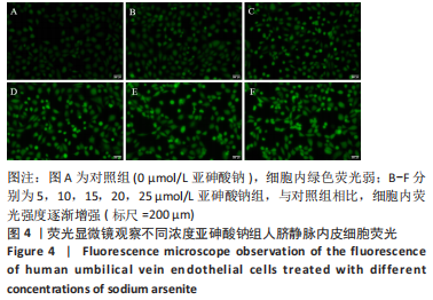
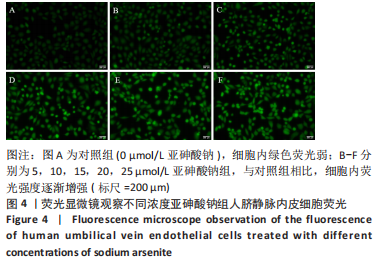
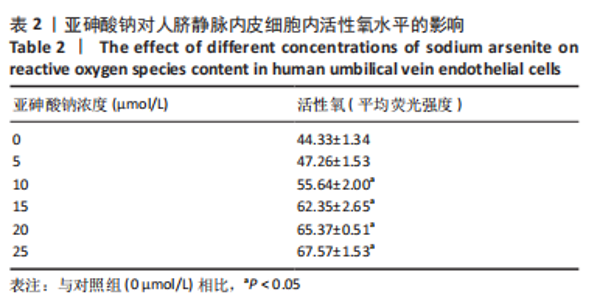
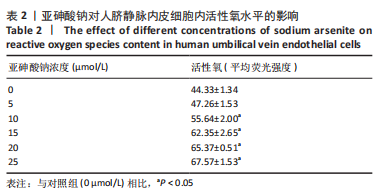
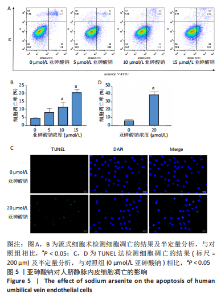
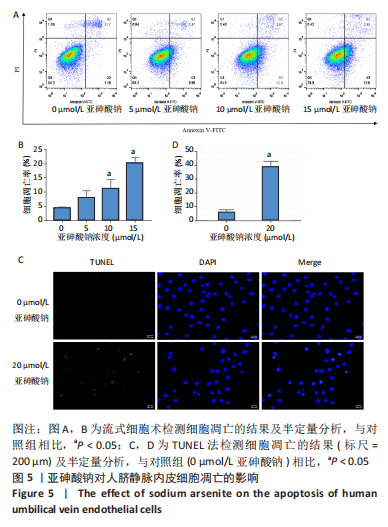
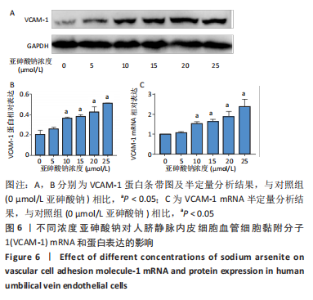
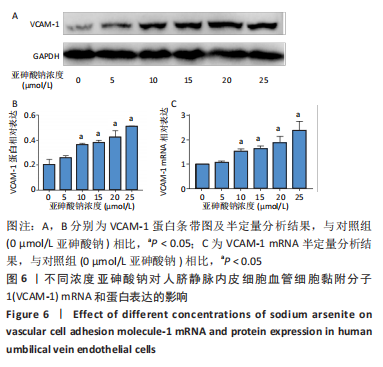
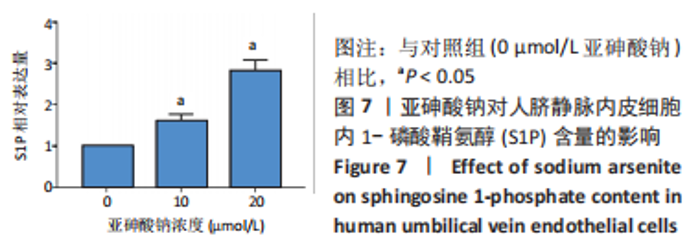
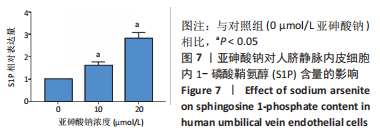
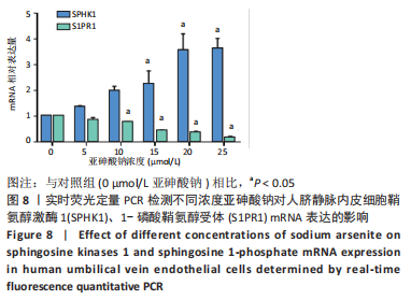
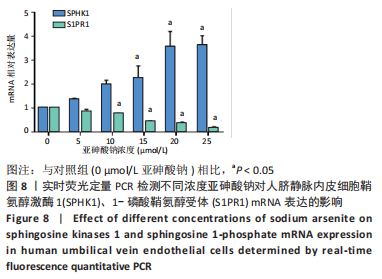
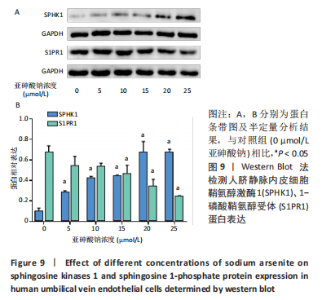
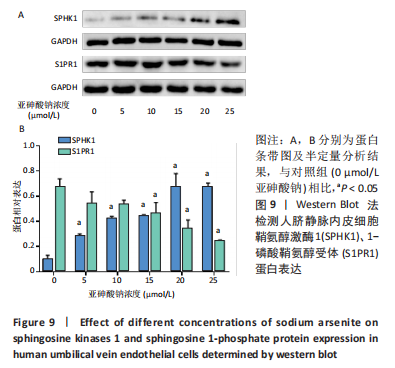
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (26): 4161-4167.doi: 10.12307/2023.121
Previous Articles Next Articles
Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis
Fang Xingyan1, Tian Zhenli1, Zhao Zheyi2, Wen Ping3, Xie Tingting1
- 1Center for Clinical Laboratory, the Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Sichuan Center for Disease Control and Prevention, Chengdu 614000, Sichuan Province, China; 3Department of Obstetrics and Gynecology, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2022-03-14Accepted:2022-05-13Online:2023-09-18Published:2023-01-28 -
Contact:Xie Tingting, MD, Associate professor, Master’s supervisor, Center for Clinical Laboratory, the Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Fang Xingyan, Master candidate, Center for Clinical Laboratory, the Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81560514 (to XTT)
CLC Number:
Cite this article
Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(26): 4161-4167.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] PALMA-LARA I, MARTINEZ-CASTILLO M, QUINTANA-PEREZ JC, et al. Arsenic exposure: A public health problem leading to several cancers. Regul Toxicol Pharmacol. 2020;110:104539. [2] SOBEL MH, SANCHEZ TR, JONES MR, et al. Rice Intake, Arsenic Exposure, and Subclinical Cardiovascular Disease Among US Adults in MESA. J Am Heart Assoc. 2020;9(4):e15658. [3] FARZAN SF, EUNUS HM, HAQUE SE, et al. Arsenic exposure from drinking water and endothelial dysfunction in Bangladeshi adolescents. Environ Res. 2022;208:112697. [4] XU S, ILYAS I, LITTLE PJ, et al. Endothelial Dysfunction in Atherosclerotic Cardiovascular Diseases and Beyond: From Mechanism to Pharmacotherapies. Pharmacol Rev. 2021;73(3):924-967. [5] GUO X, LIU X, WANG J, et al. Pigment epithelium-derived factor (PEDF) ameliorates arsenic-induced vascular endothelial dysfunction in rats and toxicity in endothelial EA.hy926 cells. Environ Res. 2020;186:109506. [6] LIU P, XUE Y, ZHENG B, et al. Crocetin attenuates the oxidative stress, inflammation and apoptosisin arsenic trioxide-induced nephrotoxic rats: Implication of PI3K/AKT pathway. Int Immunopharmacol. 2020;88:106959. [7] KORNER C, FROHLICH F. Compartmentation and functions of sphingolipids. Curr Opin Cell Biol. 2022;74:104-111. [8] CARTIER A, HLA T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy. Science. 2019;366(6463):eaar5551. [9] OGRETMEN B. Sphingolipid metabolism in cancer signalling and therapy. Nat Rev Cancer. 2018;18(1):33-50. [10] SATTAR R, SUMI MP, NIMISHA, et al. S1P signaling, its interactions and cross-talks with other partners and therapeutic importance in colorectal cancer. Cell Signal. 2021;86:110080. [11] ZOU J, MA X, ZHANG G, et al. Evaluation of the change in sphingolipids in the human multiple myeloma cell line U266 and gastric cancer cell line MGC-803 treated with arsenic trioxide. J Chromatogr B Analyt Technol Biomed Life Sci. 2015;1004:98-107. [12] LIU P, CHEN Z, MA X. Arsenic trioxide inhibits the activity of SphK1 by decreasing the level of phosphatidylserine and phosphatidic acid in the human gastric cancer cell line MGC-803. BIOCELL. 2022;46(3):737-743. [13] CRAMPTON SP, DAVIS J, HUGHES CC. Isolation of human umbilical vein endothelial cells (HUVEC). J Vis Exp. 2007;(3):183. [14] 陈小翠, 陈邦党, 杨毅宁,等. 人脐静脉内皮细胞分离培养和鉴定[J]. 细胞与分子免疫学杂志,2016,32(3):328-331. [15] ZHENG B, YANG Y, LI J, et al. Magnesium Isoglycyrrhizinate Alleviates Arsenic Trioxide-Induced Cardiotoxicity: Contribution of Nrf2 and TLR4/NF-kappaB Signaling Pathway. Drug Des Devel Ther. 2021;15:543-556. [16] HU Y, LI J, LOU B, et al. The Role of Reactive Oxygen Species in Arsenic Toxicity. Biomolecules. 2020;10(2):240. [17] HARIJITH A, PENDYALA S, REDDY NM, et al. Sphingosine kinase 1 deficiency confers protection against hyperoxia-induced bronchopulmonary dysplasia in a murine model: role of S1P signaling and Nox proteins. Am J Pathol. 2013;183(4):1169-1182. [18] HUANG LS, SUDHADEVI T, FU P, et al. Sphingosine Kinase 1/S1P Signaling Contributes to Pulmonary Fibrosis by Activating Hippo/YAP Pathway and Mitochondrial Reactive Oxygen Species in Lung Fibroblasts. Int J Mol Sci. 2020;21(6):2064. [19] COOPER KL, VOLK LB, DOMINGUEZ DR, et al. Contribution of NADPH oxidase to the retention of UVR-induced DNA damage by arsenic. Toxicol Appl Pharmacol. 2022;434:115799. [20] TSAI CL, TSAI CW, CHANG WS, et al. Protective Effects of Baicalin on Arsenic Trioxide-induced Oxidative Damage and Apoptosis in Human Umbilical Vein Endothelial Cells. In Vivo. 2021;35(1):155-162. [21] TSAI CL, TSAI CW, CHANG WS, et al. Protective Effects of Crocetin on Arsenic Trioxide-induced Oxidative Stress in Human Umbilical Vein Endothelial Cells. In Vivo. 2021;35(6):3157-3163. [22] JOZEFCZUK E, GUZIK T J, SIEDLINSKI M. Significance of sphingosine-1-phosphate in cardiovascular physiology and pathology. Pharmacol Res. 2020;156:104793. [23] DAVAILLE J, GALLOIS C, HABIB A, et al. Antiproliferative properties of sphingosine 1-phosphate in human hepatic myofibroblasts. A cyclooxygenase-2 mediated pathway. J Biol Chem. 2000;275(44):34628-34633. [24] DAVAILLE J, LI L, MALLAT A, et al. Sphingosine 1-phosphate triggers both apoptotic and survival signals for human hepatic myofibroblasts. J Biol Chem. 2002;277(40):37323-37330. [25] YANG CC, HSIAO LD, SHIH YF, et al. Sphingosine 1-Phosphate-Upregulated COX-2/PGE2 System Contributes to Human Cardiac Fibroblast Apoptosis: Involvement of MMP-9-Dependent Transactivation of EGFR Cascade. Oxid Med Cell Longev. 2022;2022: 7664290. [26] YANG CC, HSIAO LD, SU MH, et al. Sphingosine 1-Phosphate Induces Cyclooxygenase-2/Prostaglandin E2 Expression via PKC alpha-dependent Mitogen-Activated Protein Kinases and NF-kappaB Cascade in Human Cardiac Fibroblasts. Front Pharmacol. 2020;11:569802. [27] NATARAJAN V, HA AW, DONG Y, et al. Expression profiling of genes regulated by sphingosine kinase1 signaling in a murine model of hyperoxia induced neonatal bronchopulmonary dysplasia. BMC Genomics. 2017;18(1):664. [28] BUNDERSON M, COFFIN JD, BEALL HD. Arsenic induces peroxynitrite generation and cyclooxygenase-2 protein expression in aortic endothelial cells: possible role in atherosclerosis. Toxicol Appl Pharmacol. 2002;184(1):11-18. [29] WANG L, HITRON JA, WISE JT, et al. Ethanol enhances arsenic-induced cyclooxygenase-2 expression via both NFAT and NF-kappaB signalings in colorectal cancer cells. Toxicol Appl Pharmacol. 2015;288(2):232-239. [30] OOKI A, BEGUM A, MARCHIONNI L, et al. Arsenic promotes the COX2/PGE2-SOX2 axis to increase the malignant stemness properties of urothelial cells. Int J Cancer. 2018;143(1):113-126. [31] 王甜, 赵哲仪, 穆银贵, 等. 亚砷酸钠所致L-02人肝细胞损伤与p14ARF表达下调及MDM2、p53表达增加有关[J]. 细胞与分子免疫学杂志,2020,36(6):507-512. [32] 赵哲仪, 王正蓉, 方兴艳, 等. 亚砷酸钠对AML12肝细胞氧化应激、凋亡损伤及Hippo信号通路的影响[J]. 中国组织工程研究, 2022,26(26):4186-4191. [33] ZHONG G, HU T, TANG L, et al. Arsenic causes mitochondrial biogenesis obstacles by inhibiting the AMPK/PGC-1alpha signaling pathway and also induces apoptosis and dysregulated mitophagy in the duck liver. Ecotoxicol Environ Saf. 2021;230:113117. [34] TRONCOSO M F, ORTIZ-QUINTERO J, GARRIDO-MORENO V, et al. VCAM-1 as a predictor biomarker in cardiovascular disease. Biochim Biophys Acta Mol Basis Dis. 2021;1867(9):166170. [35] OBINATA H, HLA T. Sphingosine 1-phosphate and inflammation. Int Immunol. 2019;31(9):617-625. [36] ALVAREZ SE, HARIKUMAR KB, HAIT NC, et al. Sphingosine-1-phosphate is a missing cofactor for the E3 ubiquitin ligase TRAF2. Nature. 2010; 465(7301):1084-1088. [37] XIA P, WANG L, MORETTI PA, et al. Sphingosine kinase interacts with TRAF2 and dissects tumor necrosis factor-alpha signaling. J Biol Chem. 2002;277(10):7996-8003. [38] WANG L, XING X P, HOLMES A, et al. Activation of the sphingosine kinase-signaling pathway by high glucose mediates the proinflammatory phenotype of endothelial cells. Circ Res. 2005;97(9):891-899. [39] ROSEN H, GONZALEZ-CABRERA PJ, SANNA MG, et al. Sphingosine 1-phosphate receptor signaling. Annu Rev Biochem. 2009;78:743-768. [40] REN K, LU YJ, MO ZC, et al. ApoA-I/SR-BI modulates S1P/S1PR2-mediated inflammation through the PI3K/Akt signaling pathway in HUVECs. J Physiol Biochem. 2017;73(2):287-296. [41] YANG Y, WANG Q, WANG W, et al. Semaphorin 4A antibody alleviates arsenic-induced hepatotoxicity in mice via inhibition of AKT2/NF-kappaB inflammatory signaling.Toxicol Appl Pharmacol. 2021;410: 115364. [42] ZHANG C, FERRARI R, BEEZHOLD K, et al. Arsenic Promotes NF-Kappab-Mediated Fibroblast Dysfunction and Matrix Remodeling to Impair Muscle Stem Cell Function. Stem Cells. 2016;34(3):732-742. [43] ANWAR M, MEHTA D. Post-translational modifications of S1PR1 and endothelial barrier regulation. Biochim Biophys Acta Mol Cell Biol Lipids. 2020;1865(9):158760. [44] GAENGEL K, NIAUDET C, HAGIKURA K, et al. The sphingosine-1-phosphate receptor S1PR1 restricts sprouting angiogenesis by regulating the interplay between VE-cadherin and VEGFR2. Dev Cell. 2012;23(3):587-599. [45] GALVANI S, SANSON M, BLAHO VA, et al. HDL-bound sphingosine 1-phosphate acts as a biased agonist for the endothelial cell receptor S1P1 to limit vascular inflammation. Sci Signal. 2015;8(389):a79. [46] CAI Z, ZHANG Y, ZHANG Y, et al. Use of a Mouse Model and Human Umbilical Vein Endothelial Cells to Investigate the Effect of Arsenic Exposure on Vascular Endothelial Function and the Associated Role of Calpains. Environ Health Perspect. 2019;127(7):77003. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Tang Liang, Li Xiheng, Niu Ruijuan, Li Xinyue, Zou Xinying, Mao Tianjiao, Li Jiang. Naringin regulates the function of RAW264.7 macrophages to affect the osteogenic differentiation of MC-3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1205-1210. |
| [5] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [6] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [7] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [8] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [9] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [10] | Cui Lianxu, Jiang Wenkang, Lu Dahong, Xu Junrong, Liu Xiaocui, Wang Bingyun. Clinical-grade human umbilical cord mesenchymal stem cells affect the improvement of neurological function in rats with traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 835-839. |
| [11] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [12] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [13] | Zhang Qing, Gao Chunlan, Yu Feifei, Zhang Zhenghao, Ma Fang, Gao Yuan, Li Guizhong, Jiang Yideng, Ma Shengchao. Ephrin A receptor 2 DNA methylation increases in pancreatic beta cell apoptosis induced by homocysteine [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 714-719. |
| [14] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [15] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||