Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (1): 42-48.doi: 10.12307/2022.982
Previous Articles Next Articles
Complications and death causes of peripheral blood stem cell transplantation in the treatment of thalassemia major
Huang Chuwen1, Jiang Hua1, Li Minqing2
- 1Department of Hematology and Oncology, 2Department of Preventive Health of Ministry of Medical Affairs, Guangzhou Women and Children Medical Center, Guangzhou 510623, Guangdong Province, China
-
Received:2021-12-31Accepted:2022-02-15Online:2023-01-08Published:2022-06-06 -
Contact:Li Minqing, Master, Associate chief physician, Department of Preventive Health of Ministry of Medical Affairs, Guangzhou Women and Children Medical Center, Guangzhou 510623, Guangdong Province, China -
About author:Huang Chuwen, Master, Physician, Department of Hematology and Oncology, Guangzhou Women and Children Medical Center, Guangzhou 510623, Guangdong Province, China
CLC Number:
Cite this article
Huang Chuwen, Jiang Hua, Li Minqing. Complications and death causes of peripheral blood stem cell transplantation in the treatment of thalassemia major[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 42-48.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
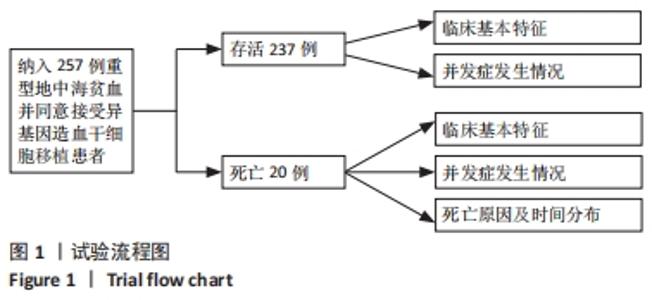
2.2 所有患者基本临床特征 该研究纳入257例患者,男172例,女85例;移植时中位年龄为6岁,年龄 < 4岁53例,4-8岁131例,> 8岁73例。移植前肝脏位于锁骨中线肋下< 2.5 cm 126例,2.5-4.0 cm 96例,> 4.0 cm 35例。移植前血清铁蛋白中位数为3 361 μg/L,血清铁蛋白< 3 000 μg/L 104例, 3 000-5 000 μg/L 94例,> 5 000 μg/L 59例。依据NF-08-TM患者危险分级,Ⅰ级23例,Ⅱ级210例,Ⅲ级24例。移植前左心射血分数≤60% 26例,余均> 60%。脾脏位于肋下1 cm 85例,1-3 cm 75例,> 3 cm 76例,脾切除21例。HLA全相合供者178例,不全相合供者59例。亲缘供者122例,无关供者115例。回输干细胞时单个核细胞 < 8.040×10?/kg 117例,≥ 8.040×10?/kg 120例。回输CD34+细胞< 5.930×106/kg 120例,≥ 5.930×106/kg 117例。全部患儿中性粒细胞及血小板均成功植入。中性粒细胞中位植入时间为+11 d(+11 d,+12 d),血小板中位植入时间为+12 d(+10 d,+13 d)。 2.3 死亡患者基本临床特征 移植后死亡20例,男13例,女7例。移植时中位年龄6岁,年龄 < 4岁2例,4-8岁9例,> 8岁9例。移植前肝脏位于锁骨中线肋下< 2.5 cm 9例,2.5-4.0 cm 7例,> 4.0 cm的4例。移植前血清铁蛋白中位数为3 570 μg/L,血清铁蛋白< 3 000 μg/L 9例,3 000-5 000 μg/L 4例,> 5 000 μg/L 7例。依据NF-08-TM患者危险分级,Ⅰ级0例,Ⅱ级16例,Ⅲ级4例。移植前左心射血分数≤60% 2例,> 60% 18例。脾脏位于肋下1 cm 6例,1-3 cm 3例,> 3 cm 10例,脾切除1例。HLA全相合供者14例,不全相合供者6例。亲缘供者10例,无关供者10例。回输干细胞时单个核细胞 < 8.040×10?/kg 8例,≥ 8.040×108/kg 12例。回输CD34+细胞 < 5.930×106/kg 8例,≥ 5.930×106/kg 12例。死亡组患者移植后中性粒细胞中位植入时间为+11.5 d(+11 d,+13 d),血小板中位植入时间为+12.5 d(+11 d,+14.75 d)。见表1。"

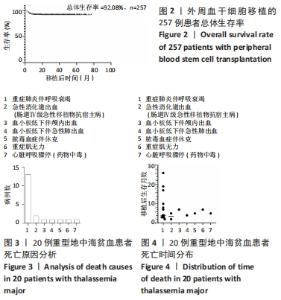
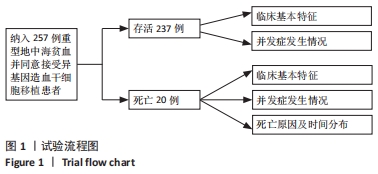
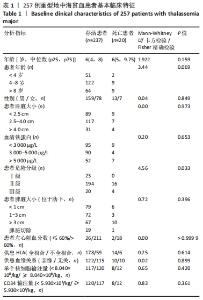
2.4 死亡原因及时间 随访至2021年2月,中位随访时间29个月,死亡20例,存活237例,累计总生存率为92.08%,见图2,累计无地中海贫血生存率为90.89%。死亡组中位生存时间5个月。有13例患者死于重症肺炎伴呼吸衰竭,其中仅1例病原明确为A型流感病毒,余均未获得明确的病原(纳入检测的病原包括肺炎支原体、肺炎衣原体、A型流感病毒、B型流感病毒、博卡病毒、鼻病毒、副流感病毒、腺病毒、呼吸道合胞病毒、肺炎链球菌、鲍曼不动杆菌、绿脓杆菌、念珠菌、毛霉菌等,均使用咽拭子PCR法或肺部灌洗液微生物高通量检测,检测阳性率低可能与早期留取标本时病菌载量低,后期已用抗生素或抗病毒药物等对症治疗,影响检测效果);2例患者发展为肠道Ⅳ度移植物抗宿主病,死于急性消化道出血;1例患者死于血小板低下致颅内出血;1例患者死于血小板低下伴急性肺出血;1例患者死于脓毒血症伴休克;1例患者移植后出现重症肌无力,逐步发展为依赖呼吸机辅助通气,家属自行放弃治疗;1例患者因自行误服大量硝苯地平后心脏呼吸骤停死亡,见图3。20例患者中,17例死于移植后1年以内,但仍有3例患者因重症肺炎死于移植后1年以上,见图4。"

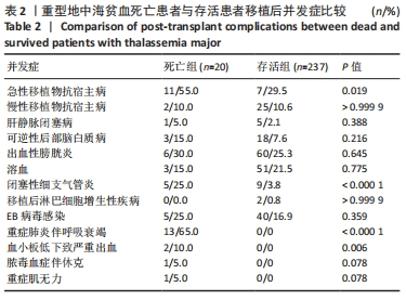
2.5 死亡组与存活组移植前基本资料比较 将移植前患者的危险分级、脾脏大小、左心室射血分数、性别、年龄、血清铁蛋白水平、肝脏大小、供患者的HLA相合程度、亲缘及无关供者、供者性别情况,移植时单个核细胞及CD34+细胞输注量等纳入危险因素。通过比较存活患者与死亡患者基本临床资料,单因素分析结果显示,两者在患者危险度分级方面存在统计学差异(P=0.033),生存分析统计显示:危险度分级为Ⅰ级、Ⅱ级、Ⅲ级的患儿总生存率分别为100%,92.26%,83.33%(P=0.032),危险度分级高的患者移植后死亡风险增加;此外,年龄似乎也存在差异趋势(P=0.069),生存分析统计显示年龄< 4岁、4-8岁、> 8岁患儿的总生存率分别为96.30%,93.16%,86.78%(P=0.065),移植时年龄> 8岁患者的死亡风险增加。 多因素分析(Cox回归分析)显示危险度分级是重型地中海贫血患者造血干细胞移植术后影响总生存率的独立危险因素(P=0.027),危险度分级为Ⅱ级的患者总死亡风险高于Ⅰ级,Ⅲ级总死亡风险高于Ⅱ级,危险比=3.002(可信区间:1.134-7.944)。 2.6 死亡组与存活组移植后并发症比较 将移植后中性粒细胞及血小板植入时间、常见并发症(急性移植物抗宿主病、慢性移植物抗宿主病、肝静脉闭塞病、可逆性后部脑白质病变、出血性膀胱炎、溶血、闭塞性细支气管炎、移植后淋巴细胞增生性疾病、EB病毒感染、重症肺炎伴呼吸衰竭、血小板低下伴严重出血、脓毒血症伴休克、重症肌无力)纳入分析。死亡组移植后中性粒细胞植入中位时间为11.5 d,存活组移植后中性粒细胞植入中位时间为11 d,两组间差异无显著性意义(P=0.401);死亡组移植后血小板植入中位时间为12.5 d,存活组移植后血小板植入中位时间为12 d,两组间差异无显著性意义(P=0.124)。 移植后出现急性移植物抗宿主病81例[Ⅰ、Ⅱ级63例,Ⅲ、Ⅳ级18例,中位发生时间+19 d(四分位距:+12 d,+37 d)],其中死亡组出现11例(Ⅰ度4例,Ⅱ度5例,Ⅲ度1例,Ⅳ度1例),发生率为55.0%,存活组出现70例(Ⅰ度20例,Ⅱ度34例,Ⅲ度11例,Ⅳ度5例),发生率为29.5%,两组间差异有显著性意义(P=0.019)。 移植后发生急性移植物抗宿主病患者的总生存率为86.8%,不发生急性移植物抗宿主病患者的总生存率为94.6%,两组间差异有显著性意义(P=0.023,危险比=2.67,可信区间为1.04-6.86)。 移植后出现慢性移植物抗宿主病27例[广泛型5例,22例为局限型,中位发生时间为+212 d(四分位距:+152 d,+260 d)],其中死亡组出现2例(1例广泛型,1例局限型),发生率为10.0%,存活组出现25例(4例广泛型,21例局限型),发生率为10.6%,两组间差异无显著性意义(P > 0.999 9)。 移植后出现肝静脉闭塞病6例,其中死亡组出现1例,发生率为5.0%,存活组出现5例,发生率为2.1%,两组间差异无显著性意义(P=0.388)。 移植后出现可逆性后部脑白质病21例,其中死亡组出现3例,发生率为15.0%,存活组出现18例,发生率为7.6%,两组间差异无显著性意义(P=0.216)。 移植后出现出血性膀胱炎66例,其中死亡组出现6例,发生率为30.0%,存活组出现60例,发生率为25.3%,两组间差异无显著性意义(P=0.645)。 移植后出现溶血54例,其中死亡组出现3例,发生率为15.0%,存活组出现51例,发生率为21.5%,两组间差异无显著性意义(P=0.775)。 移植后出现闭塞性细支气管炎14例,其中死亡组出现5例,发生率为25.0%,存活组出现9例,发生率为3.8%,两组间差异有显著性意义(P < 0.000 1)。移植后出现闭塞性细支气管炎患者总生存率为58.4%,不出现闭塞性细支气管炎患者总生存率为93.71%(P < 0.000 1,危险比=54.55,可信区间:7.592-392)。 移植后出现淋巴细胞增殖性疾病2例,其中死亡组出现0例,存活组出现2例,发生率为0.8%,两组间差异无显著性意义(P > 0.999)。 移植后出现EB病毒感染45例,其中死亡组出现5例,发生率为25.0%,存活组出现40例,发生率为16.9%,两组间差异无显著性意义(P=0.359)。 移植后出现重症肺炎伴呼吸衰竭13例,均为死亡组,发生率为65.0%,存活组0例,两组间差异有显著性意义(P < 0.000 1)。 移植后血小板低下致严重出血2例,均为死亡组,发生率为10.0%,存活组0例,两组间差异有显著性意义(P=0.006)。 移植后出现脓毒血症伴休克1例,为死亡病例,发生率为5.0%,存活组0例,两组间差异无显著性意义(P=0.078)。 移植后出现重症肌无力1例,为死亡病例,发生率为5.0%,存活组0例,两组间差异无显著性意义(P=0.078)。见表2。 "


多因素分析提示重症肺炎是重型地中海贫血患者造血干细胞移植后影响总生存率的独立危险因素(P=0.008);发生重症肺炎患者的总死亡风险高于移植后不发生重症肺炎的患者,危险比=3.983(可信区间:1.432-11.079)。急性移植物抗宿主病是重型地中海贫血患者造血干细胞移植后影响总生存率的独立危险因素(P=0.03);发生急性移植物抗宿主病患者的总死亡风险高于移植后不发生急性移植物抗宿主病的患者,危险比=2.66(可信区间:1.04-6.86)。闭塞性支气管炎是重型地中海贫血患者造血干细胞移植后影响总生存率的独立危险因素(P=0.001);发生闭塞性支气管炎患者的总死亡风险高于移植后不发生闭塞性支气管炎的患者,危险比= 5.99(可信区间:0.83-36.19)。 2.7 生物相容性 移植细胞过程中没有发生不良反应。 "

| [1] RATTANANON P, ANURATHAPAN U, BHUKHAI K, et al. The Future of Gene Therapy for Transfusion-Dependent Beta-Thalassemia: The Power of the Lentiviral Vector for Genetically Modified Hematopoietic Stem Cells. Front Pharmacol. 2021;12:730873. [2] GLUBA-BRZÓZKA A, FRANCZYK B, RYSZ-GÓRZYŃSKA M, et al. Pathomechanisms of Immunological Disturbances in β-Thalassemia. Int J Mol Sci. 2021;22(18):9677. [3] 杨花梅,孙菲,马燕琳,等. β-地中海贫血治疗的研究进展[J].实用医学杂志,2021,37(6):816-820. [4] OIKONOMOPOULOU C, GOUSSETIS E. HSCT remains the only cure for patients with transfusion-dependent thalassemia until gene therapy strategies are proven to be safe. Bone Marrow Transplant. 2021;56(12): 2882-2888. [5] MATHEWS V, SAVANI BN. Conditioning regimens in allo-SCT for thalassemia major. Bone Marrow Transplant. 2014;49(5):607-610. [6] HE Y, JIANG H, LI C, et al. Long-term results of the NF-08-TM protocol in stem cell transplant for patients with thalassemia major: A multi-center large-sample study. Am J Hematol. 2020;95(11):E297-E299. [7] HUANG C, QU Y, LIU S, et al. Hematopoietic stem cell transplantation for thalassemia major using HLA fully-matched and mismatched donor grafts. Transl Pediatr. 2021;10(6):1552-1565. [8] LI C, MATHEWS V, KIM S, et al. Related and unrelated donor transplantation for beta-thalassemia major: results of an international survey. Blood Adv. 2019;3(17):2562-2570. [9] RAJASEKAR R, MATHEWS V, LAKSHMI KM, et al. Cellular immune reconstitution and its impact on clinical outcome in children with beta thalassemia major undergoing a matched related myeloablative allogeneic bone marrow transplant. Biol Blood Marrow Transplant. 2009;15(5):597-609. [10] VELLAICHAMY SWAMINATHAN V, RAVICHANDRAN N, RAMANAN KM, et al. Augmented immunosuppression and PTCY-based haploidentical hematopoietic stem cell transplantation for thalassemia major. Pediatr Transplant. 2021;25(2):e13893. [11] QIN F, SHI L, LI Q, et al. Immune recovery after in vivo T-cell depletion myeloablative conditioning hematopoietic stem cell transplantation in severe beta-thalassemia children. Eur J Haematol. 2019;103(4):342-350. [12] SANPAKIT K, NARKBUNNAM N, BUABOONNAM J, et al. Impact of splenectomy on outcomes of hematopoietic stem cell transplantation in pediatric patients with transfusion-dependent thalassemia. Pediatr Blood Cancer. 2020;67(9):e28483. [13] LERTKOVIT O, ANURATHAPAN U, HONGENG S, et al. Chronic graft-versus-host disease in children and adolescents with thalassemia after hematopoietic stem cell transplantation. Int J Hematol. 2021;113(4): 556-565. [14] JANOWIAK-MAJERANOWSKA A, MAJERANOWSKI A. Regarding chronic graft-versus-host disease in children and adolescents with thalassemia after hematopoietic stem cell transplantation. Int J Hematol. 2021; 114(1):147-148. [15] YESILIPEK MA, UYGUN V, KUPESIZ A, et al. Thalassemia-free and graft-versus-host-free survival: outcomes of hematopoietic stem cell transplantation for thalassemia major, Turkish experience. Bone Marrow Transplant. 2022 Feb 24. doi: 10.1038/s41409-022-01613-w. Online ahead of print. [16] ROSTAMI T, KASAEIAN A, MALEKI N, et al. The effect of bone marrow-derived mesenchymal stem cell co-transplantation with hematopoietic stem cells on liver fibrosis alleviation and survival in patients with class III β-thalassemia major. Stem Cell Res Ther. 2021;12(1):213. [17] ROSTAMI T, MALEKI N, KASAEIAN A, et al. Co-transplantation of bone marrow-derived mesenchymal stem cells with hematopoietic stem cells does not improve transplantation outcome in class III beta-thalassemia major: A prospective cohort study with long-term follow-up. Pediatr Transplant. 2021;25(3):e13905. [18] GAUTHIER A, BLEYZAC N, GARNIER N, et al. Goal-Oriented Monitoring of Cyclosporine Is Effective for Graft-versus-Host Disease Prevention after Hematopoietic Stem Cell Transplantation in Sickle Cell Disease and Thalassemia Major. Biol Blood Marrow Transplant. 2020;26(12):2285-2291. [19] LI Q, LUO J, ZHANG Z, et al. G-CSF-Mobilized Blood and Bone Marrow Grafts as the Source of Stem Cells for HLA-Identical Sibling Transplantation in Patients with Thalassemia Major. Biol Blood Marrow Transplant. 2019;25(10):2040-2044. [20] FOUZIA NA, EDISON ES, LAKSHMI KM, et al. Long-term outcome of mixed chimerism after stem cell transplantation for thalassemia major conditioned with busulfan and cyclophosphamide. Bone Marrow Transplant. 2018;53(2):169-174. [21] CHEN H, LI XY, ZHAN LP, et al. Prediction, management, and prognosis of mixed chimerism after hematopoietic stem cell transplantation in transfusion-dependent pediatric thalassemia patients. Pediatr Transplant. 2020;24(8):e13876. [22] MULAS O, MOLA B, CAOCCI G, et al. Conditioning Regimens in Patients with β-Thalassemia Who Underwent Hematopoietic Stem Cell Transplantation: A Scoping Review. J Clin Med. 2022;11(4):907. [23] SUN L, WANG N, CHEN Y, et al. Unrelated Donor Peripheral Blood Stem Cell Transplantation for Patients with beta-Thalassemia Major Based on a Novel Conditioning Regimen. Biol Blood Marrow Transplant. 2019; 25(8):1592-1596. [24] FENG J, LEE V, LEUNG AWK, et al. Double-unit unrelated cord blood transplantation for thalassemia major: Comparison with HLA-identical sibling bone marrow transplantation. Pediatr Transplant. 2021;25(3):e13901. [25] MUSTAFA M, QATAWNEH M, AL JAZAZI M, et al. Hematopoietic Stem Cell Transplantation in Thalassemia Patients: a Jordanian Single Centre Experience. Mater Sociomed. 2020;32(4):277-282. [26] NAKAMURA M, MEGURI Y, IKEGAWA S, et al. Reduced dose of PTCy followed by adjuvant α-galactosylceramide enhances GVL effect without sacrificing GVHD suppression. Sci Rep. 2021;11(1):13125. [27] XU X, YANG J, CAI Y, et al. Low dose anti-thymocyte globulin with low dose posttransplant cyclophosphamide (low dose ATG/PTCy) can reduce the risk of graft-versus-host disease as compared with standard-dose anti-thymocyte globulin in haploidentical peripheral hematopoietic stem cell transplantation combined with unrelated cord blood. Bone Marrow Transplant. 2021;56(3):705-708. [28] ANURATHAPAN U, HONGENG S, PAKAKASAMA S, et al. Hematopoietic Stem Cell Transplantation for Severe Thalassemia Patients from Haploidentical Donors Using a Novel Conditioning Regimen. Biol Blood Marrow Transplant. 2020;26(6):1106-1112. [29] WANG X, ZHANG X, YU U, et al. Co-Transplantation of Haploidentical Stem Cells and a Dose of Unrelated Cord Blood in Pediatric Patients with Thalassemia Major. Cell Transplant. 2021;30:963689721994808. [30] KIM KH, LEE J, KIM HJ, et al. Efficacy and safety of high-dose budesonide/formoterol in patients with bronchiolitis obliterans syndrome after allogeneic hematopoietic stem cell transplant. J Thorac Dis. 2020;12(8):4183-4195. [31] KWOK WC, LIANG BM, LUI MMS, et al. Rapid versus gradual lung function decline in bronchiolitis obliterans syndrome after haematopoietic stem cell transplantation is associated with survival outcome. Respirology. 2019;24(5):459-466. [32] WALTHER S, RETTINGER E, MAURER HM, et al. Long-term pulmonary function testing in pediatric bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Pediatr Pulmonol. 2020; 55(7):1725-1735. |
| [1] | Zhou Jie, Pei Xibo, Wan Qianbing. Advances and biological application of asymmetric dressings [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 434-440. |
| [2] | Wang Kaiyu, Hu Peng, Wei Zairong, Huang Guangtao, Zhou Jian, He Guijia, Nie Kaiyu. Use of expanders and implants in breast reconstruction complicated with infection [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 461-469. |
| [3] | Han Tao, Hao Jianqiang, Li Wenbo, Shi Jie, Gao Qiuming. Advantages and problems of antibiotic-loaded bone cements for bone and joint infections [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 470-477. |
| [4] | Fu Chunmei, Zhang Pu, Wang Yang, Li Xiaolin, Xue Yan, Fu Jie, Zhang Cixian, Yang Yujuan, Duan Yaya, Feng Kai. Allogeneic hematopoietic stem cell transplantation in the treatment of 24 patients with severe aplastic anemia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 15-20. |
| [5] | Cui Xing, Sun Xiaoqi, Zheng Wei, Ma Dexin. Huangqin Decoction regulates autophagy to intervene with intestinal acute graft-versus-host disease in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1057-1062. |
| [6] | Jiang Xu, Cao Fuyang, Xiong Ao, Yang Meng, Tan Jun, Yu Yang, Zhang Shaokun, Xu Jianzhong. Distribution, drug resistance, and clinical characteristics of pathogenic bacteria in 102 cases of periprosthetic joint infection [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5837-5843. |
| [7] | Pan Jianke, Zhao Di, Jin Xiao, Yang Weiyi, Luo Minghui, Liu Jun, Han Yanhong, Cao Houran. Implant-related errors and complications in medial open-wedge high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5849-5856. |
| [8] | Cao Fei, Hui Min, Dong Xiling, Wang Le, Wang Zuxu, Zhang Min, Zhang Xiaoming, Liu Tongbin. Preparation of silver-loaded nanohydroxyapatite/polycaprolactone composite nanofiber scaffold and its osteogenic and antibacterial properties [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5461-5467. |
| [9] | Kong Dai, Chen Xiangli, Pei Xiaohang, Niu Xiaona, Chen Yuqing, Zhu Zunmin, Lei Pingchong, Sun Kai, Liu Zhongwen. Omission of day 11 methotrexate in regimen for the prophylaxis of graft-versus-host disease after haploid hematopoietic stem cell transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5026-5031. |
| [10] | Wen Jianyun, Miao Lili, Guan Di, Liu Xuan, Chen Libai, Feng Xiaoqin, Xu Xiaoxiao, Liu Qiujun, Wu Xuedong, He Yuelin. Correlation of the level of plasma biomarkers with acute graft-versus-host disease in children [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5032-5039. |
| [11] | Yu Manya, Kong Fansheng, Zhang Jie, Xu Jie, Cui Xing. Pathogenesis of intestinal acute graft-versus-host disease after haploid hematopoietic stem cell transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4806-4811. |
| [12] | Ci Wentao, Zhang Xinlong, Yan Shi, Wang Zhao. Reducing the recurrence of infection after the application of Masquelet technique for osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4546-4552. |
| [13] | Xu Ran, Chen Xingyu, Li Zhiqiang. Antibacterial agents loaded on hydroxyapatite scaffolds: action mechanism between the drug and the scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4411-4416. |
| [14] | Chen Dabing, Yang Ting. Immune reconstruction and anti-cytomegalovirus immunity of mesenchymal stem cells after transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4052-4057. |
| [15] | Zhang Huabin, Zhang Muchen, Liu Chang, Xu Manman, Yin Qichuan, Zhang Aijun. Viability of different fat derivatives in vitro and the outcome after transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3779-3784. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||