Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (32): 5209-5216.doi: 10.12307/2022.916
Previous Articles Next Articles
Signal peptide-CUB-epidermal growth factor-like domain protein family as coreceptors/ligands is involved in embryonic development and disease development
Wang Zijie1, Tian Zhihui2, 3, Wu Jiayuan1
- 1Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Department of Stomatology, Nanfang Hospital of Southern Medical University, Guangzhou 510000, Guangdong Province, China; 3School of Stomatology, Southern Medical University, Guangzhou 510000, Guangdong Province, China
-
Received:2021-11-26Accepted:2022-01-28Online:2022-11-18Published:2022-05-14 -
Contact:Wu Jiayuan, MD, Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Wang Zijie, Master candidate, Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:The National Natural Science Foundation of China, No. 81460102 (to WJY); the Foundation for Distinguished Young Talents in Technology Innovation of Zunyi, No. [2018]8 (to WJY); The Etiology and Prevention Team Project of Oral Infectious and Malignant Diseases in the Affiliated Stomatological Hospital of Zunyi Medical University, No. (2020)293 (to WJY); Guizhou Provincial Health Commission Science and Technology Fund Project, No. gzwjkj2020-1-163 (to WJY); General Project of Guangdong Provincial Natural Science Foundation, No. 2021A515011656 (to TZH); Southern Medical University Nanfang Hospital Dean’s General Project, No. 81772873 (to TZH).
CLC Number:
Cite this article
Wang Zijie, Tian Zhihui, Wu Jiayuan. Signal peptide-CUB-epidermal growth factor-like domain protein family as coreceptors/ligands is involved in embryonic development and disease development[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5209-5216.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
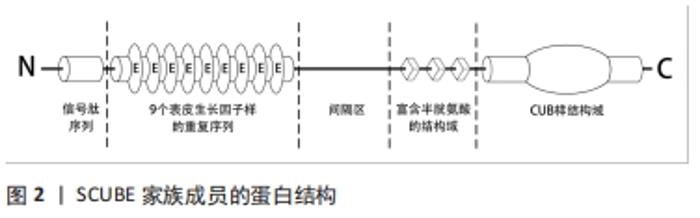
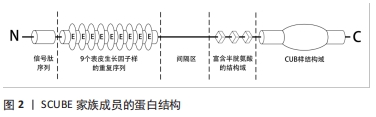
SCUBE家族基因所编码蛋白多肽的5个保守的结构域有着各自独特的功能:SCUBE蛋白氨基端的信号肽序列主要起到引导其进入分泌途径,介导蛋白质-蛋白质相互作用的功能[5];串联的表皮生长因子样重复序列是表皮生长因子家族成员的特征性结构,有研究者发现人类SCUBE1的表皮生长因子样重复序列可以通过调控肿瘤细胞之间或血小板之间的聚集与黏附功能,介导肿瘤和病理性血栓的形成[7-8]。FUCH等[9]发现SCUBE3的表皮生长因子样重复序列能以钙依赖方式介导成骨细胞亲和黏附功能。这些发现表明SCUBE家族的表皮生长因子样重复序列能增强细胞的聚集功能,可能在调控组织修复再生和免疫炎症方面有着举足轻重的作用;间隔区和3个富含半胱氨酸的重复序列可通过不同的膜结合机制将SCUBE锚定到细胞表面,其中间隔区同源性最差仅为46%-50%,间隔区有5个潜在的氨基端连接的糖基化位点,起到调节蛋白质转录、翻译和修饰的作用,3个富含半胱氨酸的结构域则起到参与调节配体受体结合,激活信号通路的功能[10-11];羧基端的CUB样结构域包含约110个氨基酸序列,是SCUBE蛋白中最为保守的区域,可通过连接受体调节蛋白质和蛋白质之间或蛋白质和蛋白多糖之间的相互作用,激活不同信号通路并引发相应的生物学效应[3,10,12]。 2.2 SCUBE调控的信号通路及其机制 "

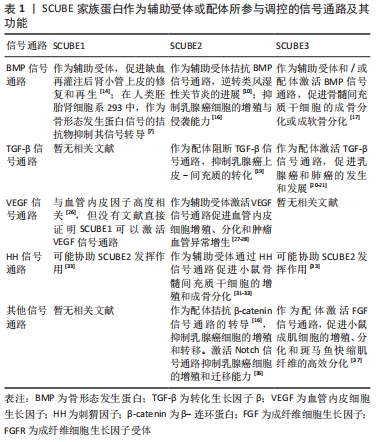
2.2.1 骨形态发生蛋白信号通路 骨形态发生蛋白是一组多功能同源二聚体蛋白,广泛存在于脊椎动物的胚胎和成体组织中,能诱导骨与软骨形成[13]。骨形态发生蛋白信号的转导大多需要与细胞膜上的受体结合才能将信号传递到胞内,SCUBE家族蛋白主要作为配体和/或辅助受体激活骨形态发生蛋白信号通路。 SCUBE1在骨形态发生蛋白信号通路中主要扮演辅助受体的角色,但在不同细胞中发挥的作用可能截然相反:在人肾小管上皮细胞中,SCUBE1可作为骨形态发生蛋白信号通路的辅助受体激活骨形态发生蛋白信号通路,增强信号转导效率,促进缺血再灌注后肾小管上皮的修复和再生[14];在斑马鱼胚胎发育过程中,SCUBE1也可作为辅助受体与骨形态发生蛋白和骨形态发生蛋白受体特异性结合形成受体配体复合物,增加骨形态发生蛋白信号的强度[15];然而,在人类胚胎肾细胞系293中,SCUBE1的CUB样结构域可结合骨形态发生蛋白2形成特异性复合物抑制骨形态发生蛋白2的活性,作为骨形态发生蛋白信号的拮抗物抑制其信号转导[7]。 SCUBE2主要作为辅助受体拮抗骨形态发生蛋白信号通路:有研究者发现SCUBE2可以和SCUBE1结合形成蛋白复合物抑制骨形态发生蛋白2活性,通过拮抗骨形态发生蛋白信号通路逆转类风湿性关节炎的进展过程[10];此外,LIN等[16]发现在人乳腺癌细胞MCF-7中SCUBE2 羧基端的CUB样结构域可通过调控骨形态发生蛋白2活性拮抗骨形态发生蛋白信号通路的转导,从而抑制乳腺癌细胞的增殖与分化。 SCUBE3既可作为辅助受体也可作为配体调控骨形态发生蛋白信号通路[17]:SCUBE3作为辅助受体时,可结合骨形态发生蛋白2、骨形态发生蛋白4和骨形态发生蛋白受体Ⅰ,增强骨形态发生蛋白信号,促进骨形态发生蛋白信号通路依赖的成骨细胞分化;SCUBE3也可作为配体,直接与细胞膜脂筏上骨形态发生蛋白受体形成异质二聚体复合物,启动骨形态发生蛋白信号的传导并激发一系列的生物学效应。 2.2.2 转化生长因子β信号通路 转化生长因子β是一组具有多种生物学功能的细胞因子,在胚胎发育、组织修复与成体稳态平衡中发挥重要作用,转化生长因子β信号通路由转化生长因子β与转化生长因子β1型受体和转化生长因子β2型受体(丝氨酸/苏氨酸蛋白酶受体)及下游的效应因子组成[18]。在乳腺癌细胞中,SCUBE2 可以与钙黏蛋白形成复合物,通过阻断转化生长因子β信号通路从而抑制上皮-间充质转化过程和乳腺癌细胞的增殖、侵袭能力[19]。在乳腺癌的发生发展过程中,SCUBE3可激活转化生长因子β信号通路,促进乳腺癌的生长和发展,当乳腺癌细胞中的SCUBE3被敲低后,转化生长因子β1的表达也会下调,抑制乳腺癌的发生发展[20]。WU等[21]发现,在肺癌细胞中,SCUBE3可直接作为配体激活转化生长因子β信号通路。SCUBE3可以被基质金属蛋白酶2和基质金属蛋白酶9切割释放其C端的CUB样结构域,与转化生长因子β受体Ⅱ结合可使SMAD2/3的磷酸化,促进肺癌组织上皮-间充质转化、迁移、侵袭、细胞外基质沉积和血管生成。ZHAO等[22]发现,SCUBE3可通过激活转化生长因子β信号通路,促进肺癌组织血管生成。另外,SCUBE3对心肌的生长也至关重要[23],SCUBE3可通过其羧基端的CUB样结构域结合转化生长因子β受体后激活转化生长因子β信号通路,促进心肌的生长和重构。 2.2.3 血管内皮生长因子信号通路 血管内皮生长因子是调控生理性和病理性血管发生的主要调节因子,能在质膜上与跨膜的血管内皮生长因子受体酪氨酸激酶结合,诱导相应受体的聚合并激活膜近侧信号的复杂装配过程[24]。研究表明,在斑马鱼胚胎血管发育过程中,SCUBE1和SCUBE2可作为血管内皮生长因子的上游信号分子共同调控血管生成[25],单独抑制SCUBE1或SCUBE2并不会导致胚胎血管发育异常,只有同时抑制SCUBE1和SCUBE2时会显著抑制血管的生成,提示在血管生成过程中,SCUBE1和SCUBE2在细胞表面受体水平上具有冗余作用,两者共同调控斑马鱼胚胎血管化过程中的血管内皮生长因子信号传导。 在成体组织中,SCUBE家族蛋白是调控血管内皮生长因子信号的关键因子。FAVRE等[26]发现SCUBE1在小鼠肺泡毛细血管内皮细胞中高度富集,与血管内皮生长因子、血管内皮生长因子受体、血管生成素和血小板-内皮细胞黏附分子等血管生成因子的表达呈高度正相关,但尚未进一步研究SCUBE1和血管生成因子的相关性。此外,SCUBE2可作为血管生成因子受体2的辅助受体,在血管生成中增强血管生成因子诱导的信号传导,促进血管生成因子受体2介导的血管生成效率[27]。LIN等[28]发现,SCUBE2是血管内皮生长因子受体2介导的病理性血管生成所必需的辅助受体,在防止肿瘤生成领域有着潜在的应用价值。综上,SCUBE1和SCUBE2均可作为辅助受体参与激活血管内皮生长因子信号,调控血管生成,两者之间存在功能冗余,而与前两者结构相似的SCUBE3与血管内皮生长因子信号并未见研究报道。 2.2.4 刺猬因子信号通路 刺猬因子信号通路是一组在进化中高度保守的信号通路,在哺乳动物中刺猬因子基因家族包括音速刺猬因子(sonic hedgehog,SHH)、沙漠刺猬因子(desert hedgehog,DHH)和印度刺猬因子(indian hedgehog,IHH) [29]。刺猬因子信号通路则由刺猬因子配体、补丁受体、平滑受体、中间转导分子和下游效应因子组成,刺猬因子信号通路在胚胎发育、干细胞自稳态和组织极化等生理学过程起着重要的调控作用[30]。在斑马鱼胚胎发育过程中,SCUBE家族成员对刺猬因子信号转导是必不可少的,抑制SCUBE家族基因会导致刺猬因子信号转导能力的下降[31-32]。但3种SCUBE家族蛋白在转导刺猬因子信号的能力方面存在功能差异,JOHNSON等[33]发现,单独抑制SCUBE家族基因任何一员并不会完全抑制刺猬因子的信号传导,只有同时抑制SCUBE1和SCUBE2或SCUBE2和SCUBE3 时才能显著影响刺猬因子信号转导,表明SCUBE2可能是调控刺猬信号的关键成员。LIN等[34]发现,SCUBE2(-/-)小鼠会表现出软骨和骨形成缺陷的表型,体外实验也证明来源于SCUBE2(-/-)的小鼠骨髓间充质干细胞会出现刺猬因子所介导的成骨分化和增殖能力受损,在此过程中SCUBE2对音速刺猬因子信号通路也有一定作用,但作用没有印度刺猬因子显著。此外,在人胚胎肾细胞中,SCUBE2也可以通过促进音速刺猬因子配体的释放和溶解,促进慢缩肌纤维的正常发育[35]。 2.2.5 其他信号通路 除了上述的信号通路,SCUBE也参其激活其他多种信号通路:有研究者发现SCUBE2的表皮生长因子样重复序列能以钙依赖性方式与钙黏附蛋白相互作用,阻断β-连环蛋白信号通路的转导并抑制乳腺癌细胞的增殖和转移潜能[16];SCUBE2也能通过激活Notch信号通路调控三阴性乳腺癌细胞的生物学功能[36];在小鼠C2C12成肌细胞中,SCUBE3可作为成纤维细胞生长因子的辅助受体参与激活成纤维细胞生长因子/成纤维细胞生长因子受体信号通路,调控细胞的增殖和分化潜能。而在斑马鱼胚胎发育的过程中,SCUBE3也可激活成纤维细胞生长因子/成纤维细胞生长因子受体信号通路,促进快缩肌纤维的高效分化。进一步研究发现[37],SCUBE3激活成纤维细胞生长因子/成纤维细胞生长因子受体信号通路的具体机制是其通过其间隔区、CUB样结构域和成纤维细胞生长因子形成蛋白复合物所介导的。 在不同的生物学过程中,SCUBE家族蛋白可作为辅助受体和/或配体激活或拮抗多种信号通路,引发一系列相应的生物学效应,见表1。 "
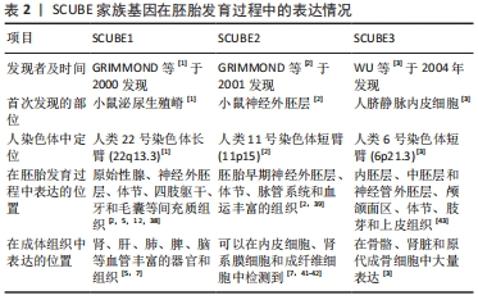
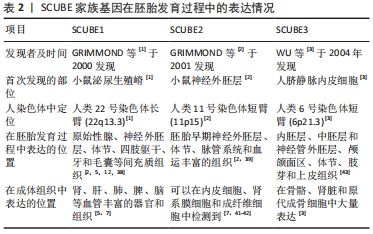
2.3 SCUBE家族在胚胎发育中的表达 2.3.1 SCUBE1 GRIMMOND等[1]于2000年通过cDNA文库在发育中小鼠的泌尿生殖嵴中首先发现SCUBE1基因。人类SCUBE1基因定位在22号染色体长臂(22q13.3)。研究者们利用整体原位杂交技术检测了小鼠胚胎发育过程中SCUBE1转录水平的表达,发现SCUBE1定位于小鼠的原始性腺、神经外胚层、体节、四肢躯干及毛囊间充质组织[2]。VANPOUCKE等[38]发现在大鼠胚胎发育过程中SCUBE1主要定位于原始性腺组织。XAVIER等[12]观察了SCUBE1在小鼠颅颌面部早期胚胎发育过程中的表达,发现SCUBE1在颌面部局限地表达于上下颌骨及神经嵴衍生牙间充质组织,表明SCUBE1可能在颌骨和牙本质的发育过程中起到一定的作用。在成体组织中,SCUBE1则定位于成人毛细血管内皮细胞、微小动静脉和血小板中[5,7],表明SCUBE1参与血管稳态的调控。 2.3.2 SCUBE2 SCUBE2是由GRIMMOND等[2]于2001年使用SCUBE1核苷酸和氨基酸序列通过BLAST在线数据库搜索预测发现的,与SCUB1在核苷酸水平上相似度高达81%。人类SCUBE2基因定位于11号染色体短臂(11p15)。在胚胎发育过程中,SCUBE2具有与SCUBE1相似的表达模式,早期即表达于神经外胚层、体节、脉管系统和血运丰富的组织[1,39]。XAVIER等[40]发现在小鼠胚胎发育过程中,SCUBE2显著富集于心脏和骨骼组织,特别是心室、心血管以及骨骼周围的软骨凝结区,表明SCUBE2可能在心脏和骨骼的正常发育过程中具有重要的作用。在成体组织中,SCUBE2特异性表达于血小板中,影响血小板的聚集和黏附功能[7]。与SCUBE1不同的是,SCUBE2广泛存在于人血管内皮细胞和一些非内皮细胞类型的细胞如成纤维细胞、肾间质细胞和乳腺导管上皮细胞中[41-42],而SCUBE1的表达则局限于如肝、肾、肺、脾和脑等一些高度血管化组织的内皮细胞中[43]。 2.3.3 SCUBE3 SCUBE3由WU等[3]最早发现于人脐静脉内皮细胞的基因,与家族其他成员有着60%的同质性和相似的结构域。人类SCUBE基因定位于6号染色体短臂(6p21.3)。在小鼠胚胎发育过程中,SCUBE3最初在胚胎8.5 d 神经外胚层的腹侧菱脑和尾侧神经孔中表达,随后拓展至内胚层、中胚层和外胚层的神经管、颅颌面区、体节、肢芽和牙源性上皮,在胚胎发育后期,SCUBE3则主要定位于肋骨、四肢和椎骨周围的软骨凝结物中及肾脏肾小球[43]。在成体组织,SCUBE3则主要富集于骨骼、牙齿等硬组织,在心脏和血管内皮中表达降低[3]。 SCUBE家族在脊椎动物胚胎发育过程中呈现出时空动态的表达模式,是调控多种组织和器官正常发育的重要基因,见表2。 "

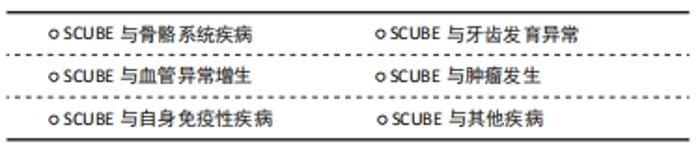
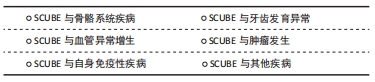
2.4.1 SCUBE与骨骼系统疾病 SCUBE1在胚胎发生早期即表达、分泌并锚定在细胞膜表面,TU等[7]研究发现,与野生型小鼠相比,SCUBE1(Δcub/Δcub)基因敲除鼠出现胚胎颅骨和脑组织缺失的表型,阿尔新蓝染色和茜素红染色结果显示除了颅骨的缺失,基因敲除鼠还会出现全身性骨和软骨发育不良。 与SCUBE1相似,通过基因消融技术构建的基因敲除小鼠SCUBE2(-/-)表现出明显的骨和软骨发育缺陷表型,体外研究也证明,抑制SCUBE2表达将会降低成骨细胞和成软骨细胞增殖和分化能力,表明SCUBE2对骨和软骨的分化至关重要[34]。 SCUBE3在肱骨和股骨等组织中表达高度富集,是骨组织发育和生长的主要调控因子。成骨细胞可通过自分泌、旁分泌或内分泌的形式使SCUBE3作用于局部或者远处的细胞,发挥一定的生物学效应[3,44]。FUCHS等[9]建立了第一个SCUBE3(N294K/N294K)条件基因敲除小鼠,发现SCUBE3的缺失会导致小鼠骨形态异常、骨代谢障碍、听力障碍,类似于人类的佩吉特骨病。LIN等[17]发现SCUBE3双等位基因失活变异会导致人类个体发育的综合征性障碍,出现诸如生长减缓、身材矮小、独特的颅面外观和骨骼发育缺陷,该研究所构建的条件基因敲除小鼠SCUBE3(-/-)表现出与人类相似的表型,出现骨骼和颌面发育异常、体型缩小、骨和软骨发育异常等缺陷。体外实验证明重组SCUBE3蛋白可以促进骨髓间充质干细胞成骨分化,而SCUBE3基因敲除小鼠骨髓间充质干细胞的增殖和成骨分化被显著抑制。 2.4.2 SCUBE与牙齿发育异常 牙源性上皮和间充质之间的交互式作用对牙齿的形成至关重要,SCUBE家族成员在牙齿形成过程中有着时空动态的表达模式[12,43,45],见图3。 "


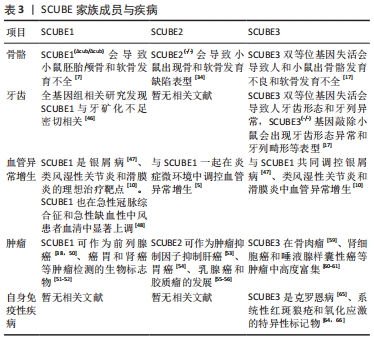
KUHNISCH等[46]通过全基因组关联研究发现,人磨牙切牙矿化不足与SCUBE1基因密切相关。LIN等[17]的研究也发现SCUBE3双等位基因失活会导致人牙齿形态和排列异常,该研究所构建的条件基因敲除小鼠SCUBE3(-/-)会出现与人类相似的牙齿形态异常和牙列畸形等表型。虽然目前关于SCUBE与牙齿相关疾病的研究较少,但鉴于SCUBE家族成员在牙胚中开始表达的时间与牙齿开始形成的时间一致,SCUBE1和SCUBE3在牙胚上皮间充质之间的表达为互补模式,SCUBE1和SCUBE3基因缺失导致的牙齿形态异常和牙齿矿化异常,可以推测SCUBE家族对牙的发育至关重要,SCUBE家族成员特别是SCUBE1和SCUBE3可能是调控组织工程牙再生的关键信号分子。 2.4.3 SCUBE与血管异常增生 血管增生是指由现有的血管网生出新的微血管,它对组织的再生、发育、修复来说都至关重要。血管异常增生指的是本来静止的血管内皮细胞在炎症因子、缺氧微环境、肿瘤微环境等刺激下发生异常增生,滋生出的不规则的毛细血管网,SCUBE家族成员均参与了血管异常增生:YANG等[10]对滑膜炎的病理机制提出假说,认为SCUBE家族3个成员都是滑膜关节炎中病理性血管生成的关键因子,SCUBE可以促进类风湿滑关节炎病理性血管的生成。血管异常增生也是银屑病的重要发病机制,CAPKIN等[47]证实银屑病患者血液中SCUBE1、SCUBE3和血管内皮生长因子含量显著增高,提出SCUBE1和SCUBE3可作为血管内皮生长因子的上游因子,在银屑病病理性血管生成中发挥重要作用。YANG等[5]发现血管内皮细胞所表达的SCUBE1和SCUBE2与炎症因子呈显著正相关,炎症微环境可以促进血管异常增生。缺氧微环境也对血管异常增生有着显著的影响:临床试验表明,SCUBE1在一些缺氧相关疾病包括急性冠脉综合征和急性缺血性中风患者血清中显著上调[48]。体外实验证明缺氧微环境会诱导内皮细胞增殖,并且新生的内皮细胞中SCUBE1的mRNA 和蛋白表达显著上调[49],提示缺氧微环境与SCUBE1介导的血管异常增生密切相关。上述结果表明了SCUBE家族成员特别是SCUBE1可作为血管异常增生诊断与组织工程血管生成的关键调控因子。 2.4.4 SCUBE与肿瘤发生 SCUBE1在人前列腺癌细胞中高度富集,与前列腺癌组织中成纤维细胞的活性密切相关[38,50]。MENTESE等[51]发现胃癌术后复发组织中SCUBE1显著上调,表明SCUBE1可作为筛选胃癌患者治疗后复发的生物标志物。KARAGUZEL等[52]发现SCUBE1是肾癌预后评估的生物标志物,但SCUBE1与肾癌类型、分级、分期等病理参数并无显著相关性。以上研究表明,SCUBE1与肿瘤密切相关,可作为早期肿瘤筛查的生物标志物。 SCUBE2与肝癌、胃癌、乳腺癌和神经胶质瘤等肿瘤密切相关[53-56]。SHENG等[57]研究发现表没食子儿茶素没食子酸酯可以通过降低DNA甲基转移酶的表达和活性,显著抑制SCUBE2甲基化状态,减缓了乳腺癌的进展。YANG等[58]发现SCUBE2在非小细胞型肺癌细胞系中低水平表达,SCUBE2的过表达会显著抑制非小细胞型肺癌细胞的增殖、迁移和侵袭。SCUBE2在结直肠癌组织中的转录和翻译水平降低,与临床分期、肿瘤浸润深度、淋巴结转移、远处转移和组织学分级呈负相关,在抑制结直肠癌进展和预后方面起着重要作用[42]。与SCUBE1和SCUBE3相反,过表达SCUBE2可以抑制肿瘤的发生发展,SCUBE2可能成为一种新的肿瘤潜在治疗靶点。 SCUBE3在骨肉瘤、肾癌和唾液腺样囊性癌等肿瘤中高度富集[59-61]。YANG等[20] 报道了SCUBE3在乳腺癌中显著升高,SCUBE3可通过激活转化生长因子β1促进乳腺癌细胞的增殖、侵袭以及转移。XUE等[62]发现SCUBE3被基质金属蛋白酶2和基质金属蛋白酶9切割后释放的片段可以与转化生长因子β受体Ⅱ结合,激活转化生长因子β信号通路,促进神经胶质瘤的进展。骆傲然[63]发现肝癌细胞中SCUBE3的表达显著高于正常细胞,SCUBE3可以通过结合转化生长因子β受体Ⅱ激活磷脂酰肌醇3激酶/蛋白激酶B信号通路,促进糖原合成酶激酶3β的磷酸化进而引起细胞周期蛋白E1的积累,从而促进肝癌细胞的增殖和迁移。 2.4.5 SCUBE与自身免疫性疾病 自身免疫性疾病是指机体对自身抗原发生免疫反应而导致自身组织损害所引起的一类疾病。SCUBE与自身免疫性疾病关系密切:QI等[64]发现系统性红斑狼疮患者全血中SCUBE3 mRNA表达低于健康人群对照组,SCUBE3有可能是系统性红斑狼疮的一个遗传易感性因子;克罗恩病是一种原因不明的肠道炎症性疾病,CARVALHO等[65]研究发现,克罗恩病患者血清SCUBE3与维生素D含量显著降低,推测SCUBE3变化可能是克罗恩病的重要致病因素;LIU等[66]发现SCUBE3能通过抑制氧化应激调控胃肠道的炎症反应,参与消化道细菌防御、免疫调节、内肽酶活性调节等反应,与消化道健康和功能紧密相关。以上研究表明SCUBE3可作为自身免疫性疾病的一个潜在治疗靶点,但自身免疫疾病的发生是由复杂的信号网络调控的,还需要更多的研究深入探索SCUBE家族与自身免疫疾病的关系。 2.4.6 SCUBE与其他疾病 SCUBE家族还与神经和肌肉的形成密切相关[31,37,67]:SCUBE2基因缺陷会导致斑马鱼肌肉和神经管发育缺陷,TU等[37]分别在体外C2C12成肌细胞模型和体内斑马鱼模型上证明了SCUBE3是调控肌肉生成的关键上游调节因子,SCUBE3异常表达会导致成肌细胞分化异常和斑马鱼快缩纤维发育异常。此外,SCUBE可能与毛囊形成密切相关[44]:在毛囊发育过程中,SCUBE3最早富集于胚胎12.5 d的毛囊基板处,胚胎14.5 d表达逐渐变强,随着时间的推移,胚胎16.5 d毛囊外围更多区域包括毛囊外根鞘和毛发基质中也可以检测到更多SCUBE3的表达,SCUBE3的动态时空表达模式暗示着其可能在毛囊发生中扮演着重要的角色。 SCUBE家族基因和蛋白在硬组织疾病、血管异常增生性疾病、自身免疫疾病和肿瘤等多种疾病发生过程中扮演着重要角色,见表3。研究SCUBE在不同疾病中的作用,有利于探讨SCUBE家族成员对不同疾病作用机制及未来在临床应用中提供治疗策略。 "
| [1] GRIMMOND S, LARDER R, VAN HATEREN N, et al. Cloning, mapping, and expression analysis of a gene encoding a novel mammalian EGF-related protein (SCUBE1). Genomics. 2000;70(1):74-81. [2] GRIMMOND S, LARDER R, VAN HATEREN N, et al. Expression of a novel mammalian epidermal growth factor-related gene during mouse neural development. Mech Dev. 2001;102(1-2):209-211. [3] WU BT, SU YH, TSAI MT, et al. A novel secreted, cell-surface glycoprotein containing multiple epidermal growth factor-like repeats and one CUB domain is highly expressed in primary osteoblasts and bones. J Biol Chem. 2004;279(36):37485-37490. [4] SABBAH DA, HAJJO R, SWEIDAN K. Review on Epidermal Growth Factor Receptor (EGFR) Structure, Signaling Pathways, Interactions, and Recent Updates of EGFR Inhibitors. Curr Top Med Chem. 2020;20(10):815-834. [5] YANG RB, NG CK, WASSERMAN SM, et al. Identification of a novel family of cell-surface proteins expressed in human vascular endothelium. J Biol Chem. 2002;277(48):46364-46373. [6] ZENG F, HARRIS RC. Epidermal growth factor, from gene organization to bedside. Semin Cell Dev Biol. 2014;28:2-11. [7] TU CF, YAN YT, WU SY, et al. Domain and functional analysis of a novel platelet-endothelial cell surface protein, SCUBE1. J Biol Chem. 2008; 283(18):12478-12488. [8] TU CF, SU YH, HUANG YN, et al. Localization and characterization of a novel secreted protein SCUBE1 in human platelets. Cardiovasc Res. 2006;71(3):486-495. [9] FUCHS H, SABRAUTZKI S, PREZMECK GK, et al. The First Scube3 Mutant Mouse Line with Pleiotropic Phenotypic Alterations. G3 (Bethesda). 2016;6(12):4035-4046. [10] YANG M, GUO M, HU Y, et al. Scube regulates synovial angiogenesis-related signaling. Med Hypotheses. 2013;81(5):948-953. [11] TSAI MT, CHENG CJ, LIN YC, et al. Isolation and characterization of a secreted, cell-surface glycoprotein SCUBE2 from humans. Biochem J. 2009;422(1):119-128. [12] XAVIER GM, SHARPE PT, COBOURNE MT. Scube1 is expressed during facial development in the mouse. J Exp Zool B Mol Dev Evol. 2009; 312B(5):518-524. [13] GILLMAN CE, JAYASURIYA AC. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater Sci Eng C Mater Biol Appl. 2021;130:112466. [14] LIAO WJ, LIN H, CHENG CF, et al. SCUBE1-enhanced bone morphogenetic protein signaling protects against renal ischemia-reperfusion injury. Biochim Biophys Acta Mol Basis Dis. 2019;1865(2): 329-338. [15] TSAO KC, TU CF, LEE SJ, et al. Zebrafish scube1 (signal peptide-CUB (complement protein C1r/C1s, Uegf, and Bmp1)-EGF (epidermal growth factor) domain-containing protein 1) is involved in primitive hematopoiesis. J Biol Chem. 2013;288(7):5017-5026. [16] LIN YC, CHEN CC, CHENG CJ, et al. Domain and functional analysis of a novel breast tumor suppressor protein, SCUBE2. J Biol Chem. 2011;286(30):27039-27047. [17] LIN YC, NICETA M, MUTO V, et al. SCUBE3 loss-of-function causes a recognizable recessive developmental disorder due to defective bone morphogenetic protein signaling. Am J Hum Genet. 2021;108(1):115-133. [18] ZOU ML, CHEN ZH, TENG YY, et al. The Smad Dependent TGF-beta and BMP Signaling Pathway in Bone Remodeling and Therapies. Front Mol Biosci. 2021;8:593310. [19] LIN YC, LEE YC, LI LH, et al. Tumor suppressor SCUBE2 inhibits breast-cancer cell migration and invasion through the reversal of epithelial-mesenchymal transition. J Cell Sci. 2014;127(Pt 1):85-100. [20] YANG X, HU J, SHI C, et al. Activation of TGF-beta1 Pathway by SCUBE3 Regulates TWIST1 Expression and Promotes Breast Cancer Progression. Cancer Biother Radiopharm. 2020;35(2):120-128. [21] WU YY, PECK K, CHANG YL, et al. SCUBE3 is an endogenous TGF-beta receptor ligand and regulates the epithelial-mesenchymal transition in lung cancer. Oncogene. 2011;30(34):3682-3693. [22] ZHAO C, QIN Q, WANG Q, et al. SCUBE3 overexpression predicts poor prognosis in non-small cell lung cancer. Biosci Trends. 2013;7(6):264-269. [23] YANG HY, CHENG CF, DJOKO B, et al. Transgenic overexpression of the secreted, extracellular EGF-CUB domain-containing protein SCUBE3 induces cardiac hypertrophy in mice. Cardiovasc Res. 2007;75(1):139-147. [24] KAUFMAN NEM, DHINGRA S, JOIS SD, et al. Molecular Targeting of Epidermal Growth Factor Receptor (EGFR) and Vascular Endothelial Growth Factor Receptor (VEGFR). Molecules. 2021;26(4):1076. [25] TSAO KC, LIN YC, CHEN YT, et al. Zebrafish scube1 and scube2 cooperate in promoting Vegfa signaling during embryonic vascularization. Cardiovasc Res. 2021:cvab125. doi: 10.1093/cvr/cvab125. [26] FAVRE CJ, MANCUSO M, MAAS K, et al. Expression of genes involved in vascular development and angiogenesis in endothelial cells of adult lung. Am J Physiol Heart Circ Physiol. 2003;285(5):H1917-H1938. [27] LIN YC, CHAO TY, YEH CT, et al. Endothelial SCUBE2 Interacts With VEGFR2 and Regulates VEGF-Induced Angiogenesis. Arterioscler Thromb Vasc Biol. 2017;37(1):144-155. [28] LIN YC, LIU CY, KANNAGI R, et al. Inhibition of Endothelial SCUBE2 (Signal Peptide-CUB-EGF Domain-Containing Protein 2), a Novel VEGFR2 (Vascular Endothelial Growth Factor Receptor 2) Coreceptor, Suppresses Tumor Angiogenesis. Arterioscler Thromb Vasc Biol. 2018; 38(5):1202-1215. [29] SKODA AM, SIMOVIC D, KARIN V, et al. The role of the Hedgehog signaling pathway in cancer: A comprehensive review. Bosn J Basic Med Sci. 2018;18(1):8-20. [30] LEE RT, ZHAO Z, INGHAM PW. Hedgehog signalling. Development. 2016;143(3):367-372. [31] WOODS IG, TALBOT WS. The you gene encodes an EGF-CUB protein essential for Hedgehog signaling in zebrafish. PLoS Biol. 2005;3(3):e66. [32] JAKOBS P, EXNER S, SCHURMANN S, et al. Scube2 enhances proteolytic Shh processing from the surface of Shh-producing cells. J Cell Sci. 2014; 127(Pt 8):1726-1737. [33] JOHNSON JL, HALL TE, DYSON JM, et al. Scube activity is necessary for Hedgehog signal transduction in vivo. Dev Biol. 2012;368(2):193-202. [34] LIN YC, ROFFLER SR, YAN YT, et al. Disruption of Scube2 Impairs Endochondral Bone Formation. J Bone Miner Res. 2015;30(7): 1255-1267. [35] CREANGA A, GLENN TD, MANN RK, et al. Scube/You activity mediates release of dually lipid-modified Hedgehog signal in soluble form. Genes Dev. 2012;26(12):1312-1325. [36] CHEN JH, KUO KT, BAMODU OA, et al. Upregulated SCUBE2 expression in breast cancer stem cells enhances triple negative breast cancer aggression through modulation of notch signaling and epithelial-to-mesenchymal transition. Exp Cell Res. 2018;370(2):444-453. [37] TU CF, TSAO KC, LEE SJ, et al. SCUBE3 (signal peptide-CUB-EGF domain-containing protein 3) modulates fibroblast growth factor signaling during fast muscle development. J Biol Chem. 2014;289(27):18928-18942. [38] VANPOUCKE G, ORR B, GRACE OC, et al. Transcriptional profiling of inductive mesenchyme to identify molecules involved in prostate development and disease. Genome Biol. 2007;8(10):R213. [39] HOLLWAY GE, MAULE J, GAUTIER P, et al. Scube2 mediates Hedgehog signalling in the zebrafish embryo. Dev Biol. 2006;294(1):104-118. [40] XAVIER GM, ECONOMOU A, SENNA GUIMARÃES AL, et al. Characterization of a mouse Scube3 reporter line. Genesis. 2010; 48(12):684-692. [41] CHENG CJ, LIN YC, TSAI MT, et al. SCUBE2 suppresses breast tumor cell proliferation and confers a favorable prognosis in invasive breast cancer. Cancer Res. 2009;69(8):3634-3641. [42] SONG Q, LI C, FENG X, et al. Decreased expression of SCUBE2 is associated with progression and prognosis in colorectal cancer. Oncol Rep. 2015;33(4):1956-1964. [43] HAWORTH K, SMITH F, ZOUPA M, et al. Expression of the Scube3 epidermal growth factor-related gene during early embryonic development in the mouse. Gene Expr Patterns. 2007;7(5):630-634. [44] WANG L, ZHANG L, YAN H, et al. Genome-wide association studies identify the loci for 5 exterior traits in a Large White × Minzhu pig population. PLoS One. 2014;9(8):e103766. [45] XAVIER GM, PANOUSOPOULOS L, COBOURNE MT. Scube3 is expressed in multiple tissues during development but is dispensable for embryonic survival in the mouse. PLoS One. 2013;8(1):e55274. [46] KUHNISCH J, THIERING E, HEITMULLER D, et al. Genome-wide association study (GWAS) for molar-incisor hypomineralization (MIH). Clin Oral Investig. 2014;18(2):677-682. [47] CAPKIN AA, DEMIR S, MENTESE A, et al. Can signal peptide-CUB-EGF domain-containing protein (SCUBE) levels be a marker of angiogenesis in patients with psoriasis? Arch Dermatol Res. 2017;309(3):203-207. [48] DAI DF, THAJEB P, TU CF, et al. Plasma concentration of SCUBE1, a novel platelet protein, is elevated in patients with acute coronary syndrome and ischemic stroke. J Am Coll Cardiol. 2008;51(22):2173-2180. [49] ZHUANG J, DEANE JA, YANG RB, et al. SCUBE1, a novel developmental gene involved in renal regeneration and repair. Nephrol Dial Transplant. 2010;25(5):1421-1428. [50] ORR B, GRACE OC, BROWN P, et al. Reduction of pro-tumorigenic activity of human prostate cancer-associated fibroblasts using Dlk1 or SCUBE1. Dis Model Mech. 2013;6(2):530-536. [51] MENTESE A, FIDAN E, SUMER AU, et al. Is SCUBE 1 a new biomarker for gastric cancer? Cancer Biomark. 2012;11(5):191-195. [52] KARAGUZEL E, MENTESE A, KAZAZ IO, et al. SCUBE1: a promising biomarker in renal cell cancer. Int Braz J Urol. 2017;43(4):638-643. [53] ZHEN L, NING G, WU L, et al. Prognostic value of aberrantly expressed methylation genes in human hepatocellular carcinoma. Biosci Rep. 2020;40(10):BSR20192593. [54] WANG X, ZHONG RY, XIANG XJ. Reduced expression of SCUBE2 predicts poor prognosis in gastric cancer patients. Int J Clin Exp Pathol. 2018;11(2):972-980. [55] FEI H, CHEN S, XU C. RNA-sequencing and microarray data mining revealing: the aberrantly expressed mRNAs were related with a poor outcome in the triple negative breast cancer patients. Ann Transl Med. 2020;8(6):363. [56] GUO E, LIU H, LIU X. Overexpression of SCUBE2 Inhibits Proliferation, Migration, and Invasion in Glioma Cells. Oncol Res. 2017;25(3):437-444. [57] SHENG J, SHI W, GUO H, et al. The Inhibitory Effect of (-)-Epigallocatechin-3-Gallate on Breast Cancer Progression via Reducing SCUBE2 Methylation and DNMT Activity. Molecules. 2019; 24(16):2899. [58] YANG B, MIAO S, LI Y. SCUBE2 inhibits the proliferation, migration and invasion of human non-small cell lung cancer cells through regulation of the sonic hedgehog signaling pathway. Gene. 2018;672:143-149. [59] LIANG W, YANG C, PENG J, et al. The Expression of HSPD1, SCUBE3, CXCL14 and Its Relations with the Prognosis in Osteosarcoma. Cell Biochem Biophys. 2015;73(3):763-768. [60] JOOSTEN SC, ODEH SNO, KOCH A, et al. Development of a prognostic risk model for clear cell renal cell carcinoma by systematic evaluation of DNA methylation markers. Clin Epigenetics. 2021;13(1):103. [61] HAN N, LU H, ZHANG Z, et al. Comprehensive and in-depth analysis of microRNA and mRNA expression profile in salivary adenoid cystic carcinoma. Gene. 2018;678:349-360. [62] XUE YY, LU YY, SUN GQ, et al. CN-3 induces mitochondrial apoptosis in glioma via Ros-mediated PI3K/AKT pathway. Pharmazie. 2021;76(5): 208-214. [63] 骆傲然.SCUBE3通过TGF β信号通路促进肝癌细胞增殖的分子机制研究[D].重庆:重庆医科大学,2019. [64] QI YY, ZHAO YF, ZHAI YL, et al. SCUBE3 Is Likely a Susceptibility Gene for Systemic Lupus Erythematosus for Chinese Populations. J Immunol Res. 2020;2020:8897936. [65] CARVALHO AY, BISHOP KS, HAN DY, et al. The role of Vitamin D level and related single nucleotide polymorphisms in Crohn’s disease. Nutrients. 2013;5(10):3898-3909. [66] LIU R, LIU J, ZHAO G, et al. Relevance of the intestinal health-related pathways to broiler residual feed intake revealed by duodenal transcriptome profiling. Poult Sci. 2019;98(3):1102-1110. [67] KAWAKAMI A, NOJIMA Y, TOYODA A, et al. The zebrafish-secreted matrix protein you/scube2 is implicated in long-range regulation of hedgehog signaling. Curr Biol. 2005;15(5):480-488. [68] ULUSOY S, OZKAN G, MENTESE A, et al. Signal peptide-CUB-EGF domain-containing protein 1 (SCUBE1) level in hemodialysis patients and parameters affecting that level. Clin Biochem. 2012;45(16-17): 1444-1449. |
| [1] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [2] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [3] | Yang Zhishan, Tang Zhenglong. YAP/TAZ, a core factor of the Hippo signaling pathway, is involved in bone formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1264-1271. |
| [4] | Song Hehua, Wei Zairong. Diabetic peripheral neuropathy: research and therapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1278-1285. |
| [5] | Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198. |
| [6] | Tang Liang, Li Xiheng, Niu Ruijuan, Li Xinyue, Zou Xinying, Mao Tianjiao, Li Jiang. Naringin regulates the function of RAW264.7 macrophages to affect the osteogenic differentiation of MC-3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1205-1210. |
| [7] | Zhang Yongqiang, Pan Feng, Sun Peng, Tan Minghui, Xuan Liuming, Wang Qinzhang. Effect of Gja1 gene recombinant lentivirus on Cx43 protein and mRNA expression in a diabetic guinea pig bladder model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1161-1165. |
| [8] | Jia Shengqi, Luo Wenlong, Tian Dingyuan, Zhang Xinhui, Cui Qian, Wang Chao, Pei Hanjun. Expression of mitochondrial sirtuin 3 in mice with acute renal ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1172-1178. |
| [9] | Zhao Lu, Zhao Yifei, Gao Da, Liu Yanfang, Fu Tingting, Xu Jiangyan. Expression of suppressor of Zeste 12 in kidney tissues of rats with diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1179-1186. |
| [10] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [11] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [12] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [13] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [14] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [15] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||