Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (30): 4896-4903.doi: 10.12307/2022.772
Previous Articles Next Articles
Mechanism and different transplantation approaches of mesenchymal stem cells in repairing chronic wounds
Chen Bei1, Zhang Li2, Liu Yi3, Sang Peng3, Jin Ying3, Lin Miaoyuan1, Xiao Leyao1, Xiong Huazhang3
- 1Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Discipline Inspection and Supervision Office, 3Department of Joint Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2021-08-30Accepted:2021-10-11Online:2022-10-28Published:2022-04-04 -
Contact:Zhang Li, Professor, Master’s supervisor, Discipline Inspection and Supervision Office, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China Liu Yi, Professor, Master’s supervisor, Department of Joint Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Chen Bei, Master candidate, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the Science and Technology Support Project of Zunyi City, No. (2020)218 (to SP); the Science and Technology Program of Guizhou Province, No. [2021] General 434 (to XHZ)
CLC Number:
Cite this article
Chen Bei, Zhang Li, Liu Yi, Sang Peng, Jin Ying, Lin Miaoyuan, Xiao Leyao, Xiong Huazhang. Mechanism and different transplantation approaches of mesenchymal stem cells in repairing chronic wounds[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4896-4903.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

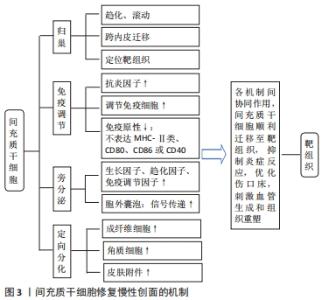
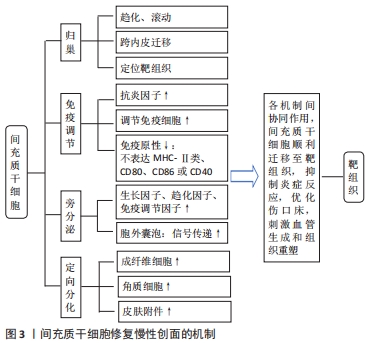
2.1.1 间充质干细胞的归巢能力与创面修复 间充质干细胞经全身给药后,能否成功归巢至靶组织是实现其治疗效果的关键因素。干细胞归巢是指体内循环干细胞或外源性干细胞定位并进入特定环境生态位的能力[22],作为一个多步骤的生理环节,其迁移过程主要包括滚动、趋化、跨内皮迁移以及定植入靶组织。目前间充质干细胞的归巢能力已经在伤口愈合中得到证明,其趋化性的实现主要通过间充质干细胞上表达的趋化因子受体和受损组织分泌的趋化因子相互介导,间充质干细胞向炎症微环境的迁移主要依赖于促炎刺激诱导的趋化因子12与在间充质干细胞表面表达的相应受体4的相互作用[23]。间充质干细胞拥有一个或多个功能性p-选择素配体,在迁移的下一阶段,间充质干细胞以P-选择素依赖的方式与内皮细胞相互作用,从而促进间充质干细胞募集[24]。然而间充质干细胞与内皮细胞的结合需要黏附分子,间充质干细胞的不同表面蛋白(如来自整合素家族的血管细胞黏附分子1)是跨内皮迁移阶段所必需的。迁移的最后阶段,当间充质干细胞锚定在内皮细胞上时,它们通过表达蛋白水解酶(如基质金属蛋白酶)穿过基底膜并进入靶组织[25]。 间充质干细胞归巢机制受到其他多种因素调控。有研究表明,局部和全身炎症信号影响间充质干细胞归巢至损伤部位[26]。组织损伤和炎症过程中释放的炎性细胞因子如肿瘤坏死因子α、白细胞介素1和干扰素γ等诱导细胞表面表达黏附分子,介导间充质干细胞向细胞外基质滚动和迁移[23],通过这些信号间的协同作用,将间充质干细胞导向损伤部位。这说明间充质干细胞的动员及其随后向损伤组织的归巢可能取决于全身和局部的炎症状态。另外有研究提到生长因子、骨桥蛋白也在间充质干细胞的迁移中起着重要作用[27]。有研究者在动物实验中验证了间充质干细胞在伤口中的归巢能力,SASAKI等[28]在小鼠皮内注射趋化因子(SLC/CCL21),证实了该趋化因子可以将循环的间充质干细胞募集到伤口部位,进而促进血管生成参与伤口修复。LIAO等[29]在动物伤口模型中经皮内注射间充质干细胞可使施用的细胞归巢至伤口,显著提高伤口愈合的速度和质量。综上,归巢机制能使间充质干细胞靶向迁移至炎症损伤部位,在间充质干细胞促进慢性创面修复过程中发挥重要作用,需进一步考虑影响间充质干细胞归巢的因素,提高归巢能力。 2.1.2 间充质干细胞的免疫调节与创面修复 炎症是机体对有害刺激的自我防御机制,适度的炎症反应有利于伤口愈合。然而在慢性伤口中,炎症反应常是过度、失调的,这常导致皮肤瘢痕形成及抑制创面再上皮化,降低伤口愈合质量[30]。间充质干细胞具有强大的免疫调节特性,但其免疫特性并非先天固有,炎症微环境是诱导其启动免疫功能的先决条件,炎症部位中的特定促炎因子如干扰素γ可激活间充质干细 胞[31],激活后的间充质干细胞通过分泌抗炎因子(如吲哚胺2,3-双加氧酶、一氧化氮及白细胞介素10)实现其免疫调节功能,减轻组织炎症反应,促进间充质干细胞和其他常驻细胞功能再生,参与重建组织微环境[32]。 间充质干细胞表达中度水平的组织相容性复合体MHC-Ⅰ类,但不表达MHC-Ⅱ类或参与体液及细胞介导免疫反应的共刺激分子CD80,CD86或CD40,这使间充质干细胞具有低免疫原性的特点[33],在自体和异体移植治疗方面成为最有吸引力的候选种子细胞之一。间充质干细胞和免疫细胞之间的直接接触也是其发挥免疫调节的重要原因,间充质干细胞分泌的可溶性因子可抑制宿主先天和适应性免疫细胞(B细胞、T细胞、自然杀伤细胞、树突状细胞和M1型巨噬细胞)的激活、成熟和增殖[34]。在炎症条件下,间充质干细胞还可以影响树突状细胞、幼稚T细胞和效应T细胞以及自然杀伤细胞的细胞因子分泌模式,从而诱导更抗炎或耐受性的细胞表型[35]。当然,间充质干细胞发挥免疫调节的过程是动态平衡的,有研究发现,间充质干细胞可通过不同的受体(如Toll样受体)识别危险信号[36],间充质干细胞根据炎症信号类型或强度分泌细胞因子以促进或抑制免疫应答,维持免疫平衡,这说明间充质干细胞可在特定条件下向促炎和抗炎表型之间转换。由此可见间充质干细胞与免疫抑制因子和免疫细胞之间相互作用对伤口愈合至关重要。对于特殊老年人群,随着年龄的增加和身体功能的下降,其伤口与正常人慢性伤口相比更容易迁延不愈。 有研究发现,老年患者的慢性伤口即使没有感染,炎症细胞因子水平也比年轻人群高约4倍,而间充质干细胞在老年人伤口愈合的炎症期可抑制T细胞、肿瘤坏死因子α、干扰素γ的增殖,调节细胞因子的分泌,抑制过度炎症反应[37]。有研究在临床前动物皮肤损伤模型和临床烧伤患者中证实了间充质干细胞在创面修复中的免疫调节作用[38-39]。总而言之,间充质干细胞主要通过分泌特定因子来影响先天和适应性免疫系统,加之与免疫细胞直接接触这两者协同作用实现其免疫调节功能。 2.1.3 间充质干细胞旁分泌特性与创面修复 早期研究将间充质干细胞的治疗效果归因于其移植和分化能力。然而,有研究发现移植的间充质干细胞与宿主损伤部位的现有细胞混合比例很小,而且植入时间太短,在全身给药后仅不足1%的间充质干细胞存活超过1周[40],这表明间充质干细胞的治疗潜力并非是细胞本身发挥作用,可能是由其旁分泌机制介导[41]。后续研究证实了间充质干细胞的旁分泌作用是其发挥创面修复和再生功能的主要机制,其旁分泌功能是通过分泌可溶性因子和释放胞外囊泡(如外泌体和微泡)来实现的。间充质干细胞能够分泌多种自分泌和旁分泌因子,包括细胞因子、趋化因子、生长因子细胞外基质蛋白,以上这些是无细胞治疗的主要来源[42]。该类因子能促进受损组织血管生成,减少细胞凋亡和降低组织纤维化程度,增强神经元存活和分化,刺激细胞外基质重塑,减轻局部炎症,加速组织修复[43],具有血管生成、营养、神经调节、免疫调节和抗炎活性。慢性伤口由于长期迁延不愈,损伤部位缺乏功能性血管网络,细胞代谢废物清除不良,受损组织常伴随缺氧以及炎性浸润。 新生血管的形成是组织修复的关键步骤,体内的缺氧诱导因子1α被认为是间充质干细胞在缺氧条件下增强血管调节功能的主调节因子[44],该因子通过诱导血管内皮细胞生长因子促进血管生成[45]。还有研究发现当间充质干细胞在高浓度炎症细胞因子的条件下培养并被移植到炎症微环境中时,间充质干细胞开始产生抗血管生成分子,以阻止炎症组织中循环白细胞的迁移[41]。目前已知的细胞外囊泡主要包括凋亡小体、微泡和外泌体[46],它们是许多过程(如免疫反应、血管生成、增殖和分化)中细胞间通讯的重要介质之一。 间充质干细胞分泌的囊泡将其内容物(遗传物质、生长和免疫调节因子)传递到靶细胞,进而激活抗凋亡和促存活通路,促进受损组织修复和再生。有研究试图引入间充质干细胞衍生的外泌体作为治疗创面修复的一种更安全的选择[47-48],例如在糖尿病皮肤缺损动物模型中,不同来源间充质干细胞衍生的外泌体能有效促进血管再生和创面再上皮化,缩小创面受损面积。另外研究发现间充质干细胞与皮肤成纤维细胞相比,间充质干细胞条件培养基中具有更高水平促血管生成细胞因子(如血管内皮生长因子和胰岛素样生长因子1等),可增强伤口内皮细胞的增殖和新生血管形成[3]。利用间充质干细胞衍生的条件培养基治疗组织炎症作为无细胞治疗范畴,具有易于制备、储存和运输,易于给药等优点[49],在一定程度上克服了基于间充质干细胞治疗的许多限制,包括不必要的分化和同种异体免疫反应的潜在激活等。总之,间充质干细胞可以通过分泌生物活性可溶性因子并与不同类型的宿主细胞相互作用,从而建立有利于组织修复和体内平衡的再生微环境。 2.1.4 间充质干细胞的定向分化与创面修复 间充质干细胞的分化潜能早已受到广泛关注,其因具有分化为中胚层、外胚层和内胚层谱系的潜力而具有良好的可塑性。众多研究表明,在体外和体内扩增的间充质干细胞可以分化为驻留组织的细胞,修复受损组织并部分恢复其正常功能[50]。有研究发现,骨髓间充质干细胞可以直接分化和替代受损组织丢失成分[51],如成纤维细胞、角质细胞及皮肤附件。此外,有研究通过将骨髓间充质干细胞和脂肪间充质干细胞植入到大鼠组织扩张模型和皮肤损伤模型中,发现间充质干细胞能够直接分化为血管内皮细胞参与伤口愈合中的血管生成[52-53]。这一系列的研究证实,间充质干细胞通过分化为不同谱系在皮肤再生中起着关键作用。但由于自体间充质干细胞数量有限,在临床应用前必须通过体外扩增进行分离,这会严重降低间充质干细胞的增殖和分化潜能,甚至在培养扩增过程中迅速老化。因此,改善目前的培养条件以更好地保留间充质干细胞的原始特性已成为未来在实验室研究和临床治疗中的关键问题。 2.2 间充质干细胞在创面修复中的途径 "

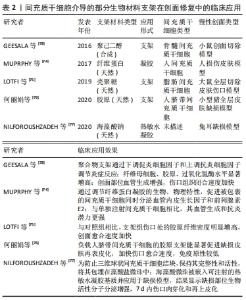
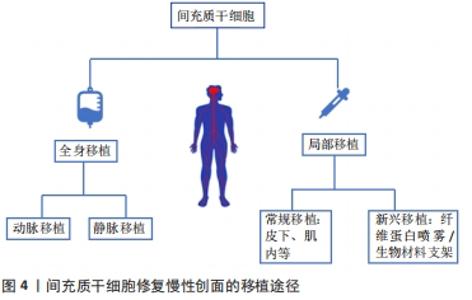
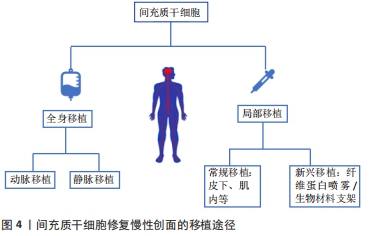
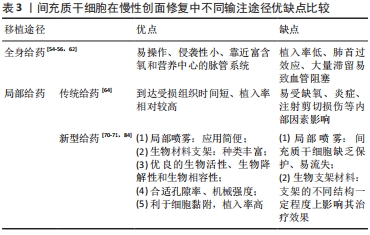
2.2.1 全身给药 间充质干细胞成功治疗慢性伤口的先决条件之一是明确最优输送方式。目前间充质干细胞治疗缺乏最佳给药途径,为最大限度地发挥间充质干细胞的愈合潜力,提高间充质干细胞修复创面的效率、质量及安全性,提高细胞植入率,探索最佳细胞输送途径显得尤为重要。全身给药是常用的输送方式之一,其主要通过血管途径实现,即静脉途径或动脉途径。静脉输注间充质干细胞治疗慢性创面是最常用的方法,其易于操作,侵袭性小,细胞迁移到目标组织后将保持靠近氧和营养中心的脉管系统[54],适用于创面部位无法直接接触或需要全身治疗的情况,利用间充质干细胞的迁移能力随血液循环到体内特定的炎症组织。但间充质干细胞能否通过静脉输注到达靶组织仍存在争议,有研究发现伤口处的间充质干细胞的植入效率较低,大多数间充质干细胞在全身输注后立即被截留在肺毛细血管中,很少向靶组织迁移,即肺首过效应,其中大量滞留于肺的间充质干细胞甚至会引起肺部血栓[55-56]。GAO等[57]分析了肺首过效应的可能原因,即细胞直径以及黏附因素,该研究观察到间充质干细胞在肺滞留的数量会随着血管扩张剂的给药而减少。上述研究说明,为提高细胞植入率和修复效果,在应用静脉途径输注间充质干细胞时应充分考虑细胞大小对修复效果的影响,必要时给予血管扩张剂的应用。相反,最近的其他研究发现,单核细胞通过吞噬作用从肺部清除间充质干细胞,清除后的间充质干细胞随血流迁移到其他部位并仍然提供治疗作用[58]。因此,关于静脉输注中的细胞植入率及归巢优化的相关问题有待进一步验证和探讨。 动脉输注给药是全身输送的另外一种常用方式,理论上动脉输送允许细胞至少绕过肺部一次,从而避免肺部首过效应。有研究推测动脉内注射有助于更高水平的间充质干细胞移植到靶器官,增强细胞的归巢性和治疗效果[59-60]。虽然动脉输注途径在创面修复中取得一定效果[61],但关于动脉输注的安全性问题仍有争议,已经发现动脉内注射间充质干细胞引起血管阻塞的发生率和严重程度与细胞大小密切相 关[62]。无论静脉还是动脉注射,对间充质干细胞适当处理有助于提高注射的安全性,包括在移植前用高渗缓冲液处理间充质干细胞,优化细胞直径,减少注射细胞的数量和抗凝治疗等[63]。 2.2.2 局部给药 传统局部给药:相较于全身给药的高细胞损耗率和安全问题,局部给药作为伤口治疗的给药方式之一已逐渐被接受,其优点是细胞植入率相对较高,缩短到达受伤皮肤的时间,可直接到达目标组织[64]。DASH等[65]进行了一项随机对照研究,对实验组下肢慢性伤口患者采用肌肉和皮下途径注射间充质干细胞,结果显示注射间充质干细胞组患者的下肢伤口面积显著缩小、疼痛程度减轻、行走能力改善。后续研究进一步对比了不同局部给药途径在大鼠糖尿病溃疡模型中的应用效果,发现肌内移植与皮下移植相比,其组织中的血管内皮生长因子表达更高[66]。众多研究显示了间充质干细胞局部应用的有效性,但缺氧、炎症等内部因素以及注射操作等外部因素会影响间充质干细胞应用效果。据报道,在临床前模型中,伤口局部注射第11 天后的细胞植入率低至0%,其归因于注射过程中的剪切损伤[67]。此外有研究发现局部注射时,细胞流过注射器针头,机械力可诱发间充质干细胞细胞膜变形,导致活细胞计数降低[68],这提示研究者们在治疗过程中为减少间充质干细胞不必要的损坏,提高细胞植入率,控制注射的速度尤为重要。 新型局部给药:传统移植途径致使间充质干细胞在创面保留时间短、存活率低,限制了间充质干细胞的治疗效果。为提高间充质干细胞利用率,改善慢性伤口愈合质量,已经开发了许多替代的递送方式,主要包括应用局部喷雾和生物材料支架。有研究表明使用纤维蛋白喷雾局部递送自体间充质干细胞是可行的,其能加速伤口愈合[69]。局部喷雾易于给药,但由于间充质干细胞缺乏封装保护,细胞易在给药部位流失,应用率低。天然细胞外基质能为细胞附着和生长提供三维环境,生物材料支架的设计就模拟了细胞外基质的结构和功能。与大多数创面敷料不同,生物材料支架成功应用的最重要特性是具有优良的生物活性、生物降解性和生物相容性,利于细胞附着于细胞表面受体[70],这也是优于其他输注途径的关键,此外还具有用于运输营养物质和废物的合适孔隙率和机械强度[71],是慢性创伤治疗中间充质干细胞传递的理想载体。生物支架材料按其材料成分不同主要分为天然支架(如透明质酸、壳聚糖、海藻酸盐、胶原蛋白和弹性蛋白等)、合成支架(如聚酸酐和聚乙二醇等)和复合支架[72-73],支架为细胞输送提供了良好的微环境,有助于细胞黏附、增殖和分化,与远距离静脉注射相比,在一定程度上增强了间充质干细胞归巢到损伤部位的能力。当然,生物材料支架的类型不同,其为干细胞提供的生存、功能与治疗效果也不同,以下是间充质干细胞介导的部分生物材料支架在伤口愈合中的应用情况[74-78],见表2。"
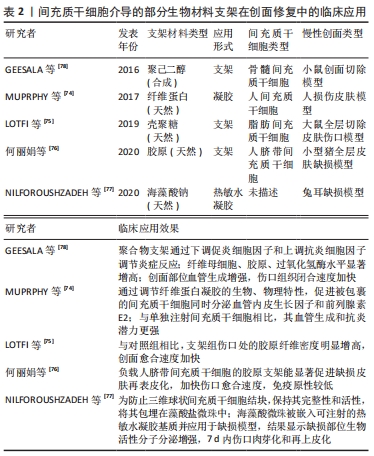
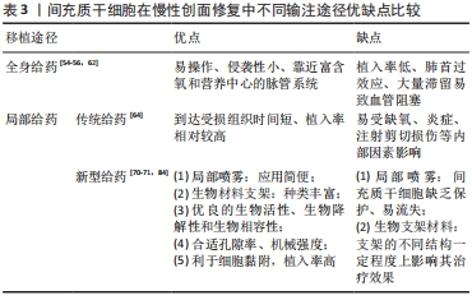
在以往研究中常采用胶原海绵、脱细胞真皮基质等片状生物支架运输间充质干细胞[79-80],但研究发现此类支架需要额外的细胞接种程序,且不能完美贴合慢性伤口不规则的几何形状。因此不同类型生物材料以水凝胶支架形式的应用成为优于片状支架的又一选择,其易于包埋间充质干细胞和填充不规则伤口,是能够吸收和维持大量水或生物液体的三维不溶性聚合物网络[81]。与标准条件下生长的间充质干细胞相比,水凝胶还能诱导间充质干细胞分泌血管生成细胞因子和表达与维持多能性和自我更新相关的转录因子[82]。除此之外,复合支架的应用也给间充质干细胞提供了有效载体,其因较高的稳定性和较强的力学性能而具有良好的可塑性和应用前景[83]。 关于支架的设计,支架的结构特征(支架的孔径、几何形状、分支以及通道)是物质运输的重要条件,在一定程度上影响了间充质干细胞的治疗效果。例如,与水凝胶和组织培养瓶的塑料表面相比,藻酸盐溶液的多孔支架允许间充质干细胞产生高浓度的细胞因子和生长因子[84]。还有新研究发现,间充质干细胞的代谢活性影响植入后细胞的存活、功能和命运,缺血环境中间充质干细胞存活的首要条件归因于葡萄糖而并非氧气[85]。考虑到代谢活性对细胞发挥功能的重要性,新一代含细胞支架的设计应考虑细胞代谢机制和影响因素,以期提高间充质干细胞的存活率。综上所述,与传统的移植方式相比,生物支架的应用增强了间充质干细胞的生存力,具有更强的可塑性,为慢性难愈性伤口治疗提供了新的治疗选择。 2.2.3 不同输注途径优缺点比较 不同给药途径的药物动力学和药效学大不相同,均会影响间充质干细胞在受者组织中的植入、保留和存活,进而限制其发挥创面修复作用。为探索合适的输注途径,提高间充质干细胞植入率和创面修复效果,现将不同给药途径优缺点加以总结,见表3。"
| [1] 黄琼蕾,金瑛.基于共词分析法探讨中国2010-2019年慢性伤口护理研究热点[J].护理管理杂志,2021,21(2):123-127. [2] FRYKBERG RG, BANKS J. Challenges in the treatment of chronic wounds. Adv Wound Care.2015;4(9):560-582. [3] 冯光,宋达疆,章一新,等.创面修复临床研究进展[J].中国美容整形外科杂志,2019,30(11):687-689. [4] NUSSBAUM SR, CARTER MJ, FIFE CE, et al. An economic evaluation of the impact, cost, and medicare policy implications of chronic nonhealing wounds. Value Health. 2018;21(1):27-32. [5] KESHTKAR S, AZARPIRA N, GHAHREMANI MH. Mesenchymal stem cell-derived extracellular vesicles: novel frontiers in regenerative medicine. Stem Cell Res Ther. 2018;9(1):63. [6] LEE BC, KANG KS. Functional enhancement strategies for immunomodulation of mesenchymal stem cells and their therapeutic application. Stem Cell Res Ther. 2020;11(1):397. [7] CAO Y, GANG X, SUN C, et al. Mesenchymal Stem Cells Improve Healing of Diabetic Foot Ulcer. J Diabetes Res. 2017;2017:9328347. [8] KEAN TJ, LIN P, CAPLAN AI, et al. MSCs: delivery routes and engraftment, cell-targeting strategies, and immune modulation. Stem Cells Int. 2013;2013:732742. [9] FAKOYA AO. New delivery systems of stem cells for vascular regeneration in ischemia. Front Cardiovasc Med. 2017;4:7. [10] GAO L, XU W, LI T, et al. Stem cell therapy: a promising therapeutic method for intracerebral hemorrhage. Cell Transplant. 2018;27(12):1809-1824. [11] 谢江帆,杨思明,尚涛,等.间充质干细胞用于慢性创面治疗的作用途径及其相关机制[J].感染、炎症、修复,2014,15(4):248-250. [12] LEE DE, AYOUB N, AGRAWAL DK. Mesenchymal stem cells and cutaneous wound healing: novel methods to increase cell delivery and therapeutic efficacy. Stem Cell Res Ther. 2016;7:37. [13] MAQSOOD M, KANG M, WU X, et al. Adult mesenchymal stem cells and their exosomes: sources, characteristics, and application in regenerative medicine. Life Sci. 2020;256:118002. [14] FRIEDENSTEIN AJ, PIATETZKY-SHAPIRO II, PETRAKOVA KV. Osteogenesis in transplants of bone marrow cells. J Embryol Exp Morphol. 1966;16(3):381-390. [15] DEXTER TM, SPOONCER E. Growth and differentiation in the hemopoietic system. Annu Rev Cell Biol. 1987;3:423-441. [16] DEXTER TM, WRIGHT EG, KRIZSA F, et al. Regulation of haemopoietic stem cell proliferation in long term bone marrow cultures. Biomedicine. 1977;27(9-10): 344-349. [17] CAPLAN AI. Mesenchymal stem cells. J Orthop Res. 1991;9(5):641-650. [18] PITTENGER MF, MACKAY AM, BECK SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. [19] BADIAVAS EV, FALANGA V. Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol. 2003;139(4):510-516. [20] DENG W, HAN Q, LIAO L, et al. Engrafted bone marrow-derived flk-(1+) mesenchymal stem cells regenerate skin tissue. Tissue Eng. 2005;11(1-2):110-119. [21] LIU P, DENG Z, HAN S, et al. Tissue-engineered skin containing mesenchymal stem cells improves burn wounds. Artif Organs. 2008;32(12):925-931. [22] LIESVELD JL, SHARMA N, Aljitawi OS. Stem cell homing: from physiology to therapeutics. Stem Cells. 2020;38(10):1241-1253. [23] VOLAREVIC V, GAZDIC M, SIMOVIC MB, et al. Mesenchymal stem cell-derived factors: immuno-modulatory effects and therapeutic potential. Biofactors. 2017; 43(5):633-644. [24] RUSTER B, GOTTIG S, LUDWIG RJ, et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood. 2006; 108(12):3938-3944. [25] AN T, CHEN Y, TU Y, et al. Mesenchymal stromal cell-derived extracellular vesicles in the treatment of diabetic foot ulcers: application and challenges. Stem Cell Rev Rep. 2021;17(2):369-378. [26] RUSTAD KC, GURTNER GC. Mesenchymal stem cells home to sites of injury and inflammation. Adv Wound Care. 2012;1(4):147-152. [27] FU X, LIU G, HALIM A, et al. Mesenchymal stem cell migration and tissue repair. Cells. 2019,8(8):784. [28] SASAKI M, ABE R, FUJITA Y, et al. Mesenchymal stem cells are recruited into wounded skin and contribute to wound repair by transdifferentiation into multiple skin cell type. J Immunol. 2008;180(4):2581-2587. [29] LIAO Y, ITOH M, YANG A, et al. Human cord blood-derived unrestricted somatic stem cells promote wound healing and have therapeutic potential for patients with recessive dystrophic epidermolysis bullosa. Cell Transplant. 2014;23(3):303-317. [30] LANDEN NX, LI D, STAHLE M. Transition from inflammation to proliferation: a critical step during wound healing. Cell Mol Life Sci. 2016;73(20):3861-3885. [31] BERNARDO ME, FIBBE WE. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell. 2013;13(4):392-402. [32] GAZDIC M, VOLAREVIC V, ARSENIJEVIC N, et al. Mesenchymal stem cells: a friend or foe in immune-mediated diseases. Stem Cell Rev Rep. 2015;11(2):280-287. [33] KIM KH, BLASCO-MORENTE G, CUENDE N, et al. Mesenchymal stromal cells: properties and role in management of cutaneous diseases. J Eur Acad Dermatol Venereol. 2017;31(3):414-423. [34] REGMI S, PATHAK S, KIM JO, et al. Mesenchymal stem cell therapy for the treatment of inflammatory diseases: challenges, opportunities, and future perspectives. Eur J Cell Biol. 2019;98(5-8):151041. [35] YAGI H, SOTO-GUTIERREZ A, PAREKKADAN B, et al. Mesenchymal stem cells: Mechanisms of immunomodulation and homing. Cell Transplant. 2010;19(6):667-679. [36] JIANG W, XU J. Immune modulation by mesenchymal stem cells. Cell Prolif. 2020; 53(1):e12712. [37] ELLOSO M, KAMBLI A, AIJAZ A, et al. Burns in the elderly: potential role of stem cells. Int J Mol Sci. 2020;21(13):4604. [38] LIU L, YU Y, HOU Y, et al. Human umbilical cord mesenchymal stem cells transplantation promotes cutaneous wound healing of severe burned rats. PLoS One. 2014;9(2):e88348. [39] RASULOV MF, VASILENKO VT, ZAIDENOV VA, et al. Cell transplantation inhibits inflammatory reaction and stimulates repair processes in burn wound. Bull Exp Biol Med. 2006;142(1):112-115. [40] XING X, LI Z, YANG X, et al. Adipose-derived mesenchymal stem cells-derived exosome-mediated microRNA-342-5p protects endothelial cells against atherosclerosis. Aging (Albany NY). 2020;12(4):3880-3898. [41] VIZOSO FJ, EIRO N, CID S, et al. Mesenchymal stem cell secretome: toward cell-free therapeutic strategies in regenerative medicine. Int J Mol Sci. 2017;18(9): 3880-3898. [42] GUNAWARDENA T, RAHMAN MT, ABDULLAH B, et al. Conditioned media derived from mesenchymal stem cell cultures: the next generation for regenerative medicine. J Tissue Eng Regen Med. 2019;13(4):569-586. [43] BOULESTERAU J, MAUMUS M, ROZIER P, et al. Mesenchymal stem cell derived extracellular vesicles in aging. Front Cell Dev Biol. 2020;8:107. [44] JAKOVLJEVIC J, HARRELL CR, FELLABAUM C, et al. Modulation of autophagy as new approach in mesenchymal stem cell-based therapy. Biomed Pharmacother. 2018;104:404-410. [45] ZHANG Y, HAO Z, WANG P, et al. Exosomes from human umbilical cord mesenchymal stem cells enhance fracture healing through HIF-1α-mediated promotion of angiogenesis in a rat model of stabilized fracture. Cell Prolif. 2019; 52(2):e12570. [46] PHAN J, KUMAR P, HAO D, et al. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. J Extracell Vesicles. 2018; 7(1):1522236. [47] FUI LW, LOK M, GOVINDASAMY V, et al. Understanding the multifaceted mechanisms of diabetic wound healing and therapeutic application of stem cells conditioned medium in the healing process. J Tissue Eng Regen Med. 2019; 13(12):2218-2233. [48] QIU H, LIU S, WU K, et al. Prospective application of exosomes derived from adipose-derived stem cells in skin wound healing: a review. J Cosmet Dermatol. 2020;19(3):574-581. [49] HU P, YANG Q, WANG Q, et al. Mesenchymal stromal cells-exosomes: a promising cell-free therapeutic tool for wound healing and cutaneous regeneration. Burns Trauma. 2019;7:38. [50] LI H, FU X. Mechanisms of action of mesenchymal stem cells in cutaneous wound repair and regeneration. Cell Tissue Res. 2012;348(3):371-377. [51] MARANDA EL, RODRIGUEZ-MENOCAL L, BADIAVAS EV. Role of mesenchymal stem cells in dermal repair in burns and diabetic wounds. Curr Stem Cell Res Ther. 2017;12(1):61-70. [52] ZHOU SB, CHIANG CA, LIU K, et al. Intravenous transplantation of bone marrow mesenchymal stem cells could effectively promote vascularization and skin regeneration in mechanically stretched skin. Br J Dermatol. 2015;172(5):1278-1285. [53] NIE C, YANG D, XU J, et al. Locally administered adipose-derived stem cells accelerate wound healing through differentiation and vasculogenesis. Cell Transplant. 2011;20(2):205-216. [54] DE BECKER A, RIET IV. Homing and migration of mesenchymal stromal cells: how to improve the efficacy of cell therapy? World J Stem Cells. 2016;8(3):73-87. [55] EVANS CE, PALAZON A, SIM J, et al. Modelling pulmonary microthrombosis coupled to metastasis: distinct effects of thrombogenesis on tumorigenesis. Biol Open. 2017;6(5):688-697. [56] BRAID LR, WOOD CA, WIESE DM, et al. Intramuscular administration potentiates extended dwell time of mesenchymal stromal cells compared to other routes. Cytotherapy. 2018;20(2):232-244. [57] GAO J, DENNIS JE, MUZIC RF, et al. The dynamic in vivo distribution of bone marrow-derived mesenchymal stem cells after infusion. Cells Tissues Organs. 2001;169(1):12-20. [58] DE WITTE S, LUK F, SIERRA PJ, et al. Immunomodulation by therapeutic mesenchymal stromal cells (MSC) is triggered through phagocytosis of msc by monocytic cells. Stem Cells. 2018;36(4):602-615. [59] JANOWSKI M, LYCZEK A, ENGELS C, et al. Cell size and velocity of injection are major determinants of the safety of intracarotid stem cell transplantation. J Cereb Blood Flow Metab. 2013;33(6):921-927. [60] CUI LL, KERKELA E, BAKREEN A, et al. The cerebral embolism evoked by intra-arterial delivery of allogeneic bone marrow mesenchymal stem cells in rats is related to cell dose and infusion velocity. Stem Cell Res Ther. 2015;6(1):11. [61] ZHAO QS, XIA N, ZHAO N, et al. Localization of human mesenchymal stem cells from umbilical cord blood and their role in repair of diabetic foot ulcers in rats. Int J Biol Sci. 2013;10(1):80-89. [62] GE J, GUO L, WANG S, et al. The size of mesenchymal stem cells is a significant cause of vascular obstructions and stroke. Stem Cell Rev Rep. 2014;10(2):295-303. [63] POLTAVTSEVA RA, POLTAVTSEV AV, LUTSENKO GV, et al. Myths, reality and future of mesenchymal stem cell therapy. Cell Tissue Res. 2019;375(3):563-574. [64] HUSELSTEIN C, RAHOUADJ R, DE ISLA N, et al. Mechanobiology of mesenchymal stem cells: Which interest for cell-based treatment? Biomed Mater Eng. 2017;28(s1):S47-S56. [65] DASH NR, DASH SN, ROUTRAY P, et al. Targeting nonhealing ulcers of lower extremity in human through autologous bone marrow-derived mesenchymal stem cells. Rejuvenation Res. 2009;12(5):359-366. [66] WAN J, CAI Q, LIU Y. Effect of different transplantations with bone-marrow derived mesenchymal stem cells on diabetic foot ulcers in rats. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2013;38(4):347-355. [67] GARG RK, RENNERT RC, DUSCHER D, et al. Capillary force seeding of hydrogels for adipose-derived stem cell delivery in wounds. Stem Cells Transl Med. 2014; 3(9):1079-1089. [68] ZHANG M, METHOT D, POPPA V, et al. Cardiomyocyte grafting for cardiac repair: graft cell death and anti-death strategies. J Mol Cell Cardiol. 2001;33(5):907-921. [69] FALANGA V, IWAMOTO S, CHARTIER M, et al. Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng. 2007;13(6):1299-1312. [70] KOHANE DS, LANGER R. Polymeric biomaterials in tissue engineering. Pediatr Res. 2008;63(5):487-491. [71] GUO B, MA PX. Conducting polymers for tissue engineering. Biomacromolecules. 2018;19(6):1764-1782. [72] TURNER NJ, BADYLAK SF. The use of biologic scaffolds in the treatment of chronic nonhealing wounds. Adv Wound Care (New Rochelle). 2015;4(8):490-500. [73] DASH BC, Xu Z, LIN L, et al. Stem Cells and Engineered Scaffolds for Regenerative Wound Healing. Bioengineering (Basel). 2018;5(1):23. [74] MURPHY KC, WHITEHEAD J. Engineering fibrin hydrogels to promote the wound healing potential of mesenchymal stem cell spheroids. Acta Biomater. 2017;64: 176-186. [75] LOTFI M, NADERI-MESHKIN H, MAHDIPOUR E, et al. Adipose tissue-derived mesenchymal stem cells and keratinocytes co-culture on gelatin/chitosan/β-glycerol phosphate nanoscaffold in skin regeneration. Cell Biol Int. 2019. doi: 10.1002/cbin.11119. [76] 何丽娟,张博文,贾雅丽,等.人脐带间充质干细胞复合胶原支架治疗小型猪皮肤缺损的实验研究[J].军事医学,2020,44(3):192-197. [77] NILFOROUSHZADEH MA, KHODADADI YM, BARADARAN GS, et al. Mesenchymal stem cell spheroids embedded in an injectable thermosensitive hydrogel: an in situ drug formation platform for accelerated wound healing. ACS Biomater Sci Eng. 2020;6(9):5096-5109. [78] GEESALA R, BAR N, DHOKE NR, et al. Porous polymer scaffold for on-site delivery of stem cells--Protects from oxidative stress and potentiates wound tissue repair. Biomaterials. 2016;77:1-13. [79] KIM CH, LEE JH, WON JH, et al. Mesenchymal stem cells improve wound healing in vivo via early activation of matrix metalloproteinase-9 and vascular endothelial growth factor. J Korean Med Sci. 2011;26(6):726-733. [80] NIE C, ZHANG G, YANG D, et al. Targeted delivery of adipose-derived stem cells via acellular dermal matrix enhances wound repair in diabetic rats. J Tissue Eng Regen Med. 2015;9(3):224-235. [81] CHEN Z, ZHANG B, SHU J, et al. Human decellularized adipose matrix derived hydrogel assists mesenchymal stem cells delivery and accelerates chronic wound healing. J Biomed Mater Res A. 2021;109(8):1418-1428. [82] RUSTAD KC, WONG VW, SORKIN M, et al. Enhancement of mesenchymal stem cell angiogenic capacity and stemness by a biomimetic hydrogel scaffold. Biomaterials. 2012;33(1):80-90. [83] FU J, ZHANG Y, CHU J, et al. Reduced graphene oxide incorporated acellular dermal composite scaffold enables efficient local delivery of mesenchymal stem cells for accelerating diabetic wound healing. ACS Biomater Sci Eng. 2019;5(8):4054-4066. [84] QAZI TH, MOONEY DJ, DUDA GN, et al. Biomaterials that promote cell-cell interactions enhance the paracrine function of MSCs. Biomaterials. 2017;140:103-114. [85] SALAZAR-NORATTO GE, LUO G, DENOEUD C, et al. Understanding and leveraging cell metabolism to enhance mesenchymal stem cell transplantation survival in tissue engineering and regenerative medicine applications. Stem Cells. 2020;38(1):22-33. |
| [1] | Wang Jianping, Zhang Xiaohui, Yu Jinwei, Wei Shaoliang, Zhang Xinmin, Xu Xingxin, Qu Haijun. Application of knee joint motion analysis in machanism based on three-dimensional image registration and coordinate transformation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-5. |
| [2] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [3] | Xiang Xinjian, Liu Fang, Wu Liangliang, Jia Daping, Tao Yue, Zhao Zhengnan, Zhao Yu. High-dose vitamin C promotes the survival of autologous fat transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1242-1246. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Tang Wenjing, Wu Siyuan, Yang Chen, Tao Xi. Inflammatory responses in post-stroke depression [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1278-1285. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Xuan Juanjuan, Bai Hongtai, Zhang Jixiang, Wang Yaoquan, Chen Guoyong, Wei Sidong. Role of regulatory T cell subsets in liver transplantation and progress in clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1143-1148. |
| [10] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [11] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [12] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [13] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [14] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [15] | Zhang Yujie, Yang Jiandong, Cai Jun, Zhu Shoulei, Tian Yuan. Mechanism by which allicin inhibits proliferation and promotes apoptosis of rat vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1084. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||