Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (30): 4834-4839.doi: 10.12307/2022.763
Previous Articles Next Articles
Transforming growth factor beta 3 in mediating chondrogenic differentiation of mesenchymal stem cells from different sources
Li Ruiyu, Shi Xu, Chen Qi, Zuo Hua, Li Kezhen
- Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China
-
Received:2021-08-30Accepted:2021-10-15Online:2022-10-28Published:2022-03-29 -
Contact:Chen Qi, MD, Associate chief physician, Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China -
About author:Li Ruiyu, Master candidate, Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China -
Supported by:Social Development Guiding Science and Technology Project of Zhenjiang City, No. FZ2020077 (to ZH); Student Innovation and Entrepreneurship Training Program of Jiangsu University, No. 3171401055 (to CQ)
CLC Number:
Cite this article
Li Ruiyu, Shi Xu, Chen Qi, Zuo Hua, Li Kezhen. Transforming growth factor beta 3 in mediating chondrogenic differentiation of mesenchymal stem cells from different sources[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4834-4839.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
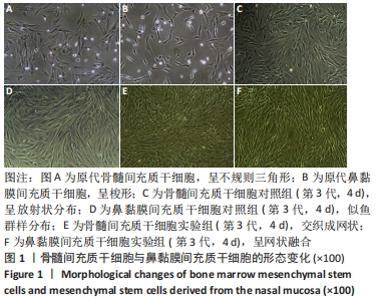
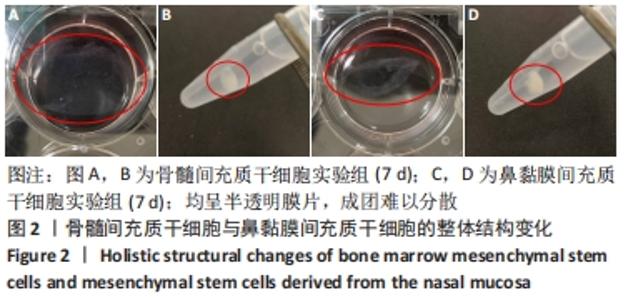
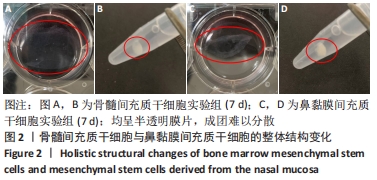
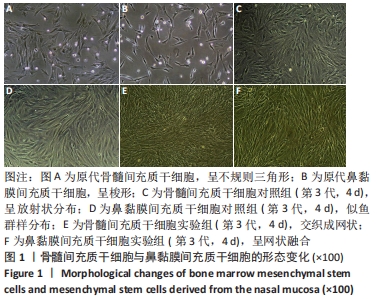
2.1 原代细胞与成软骨诱导分化过程中的镜下及整体结构改变 原代细胞接种后静置于培养箱2 d,行半换液去除部分悬浮细胞,可见细胞贴壁生长,形态不规则,10 d后镜下观察:骨髓间充质干细胞大部分呈不规则三角形,核呈椭圆形,可见双核细胞,见图1A;鼻黏膜间充质干细胞大部分呈梭形生长,核形态与骨髓间充质干细胞相似,见图1B。第3代骨髓间充质干细胞和鼻黏膜间充质干细胞在未含转化生长因子β3的成软骨诱导分化培养基中培养4 d镜下观察:骨髓间充质干细胞形成大小不一的集落,呈放射状紧密排列,多为长梭形,见图1C;鼻黏膜间充质干细胞贴壁似鱼群样分布,形态均一,生长速度较快,见图1D。第3代骨髓间充质干细胞和鼻黏膜间充质干细胞在含有转化生长因子β3的成软骨诱导分化培养基中培养4 d镜下观察:细胞交织成网状,难以区分细胞间隙,见图1E,F。"
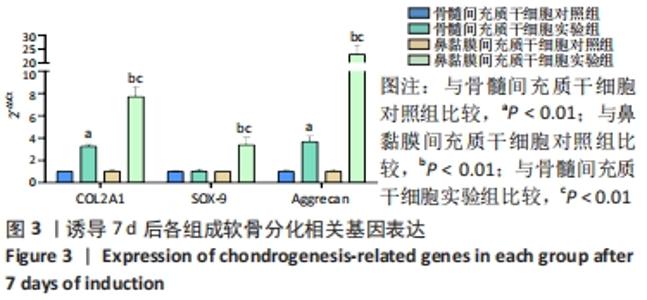
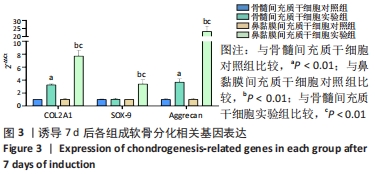
2.2 RT-PCR检测成软骨分化过程中的相关基因表达 干预7 d后,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的COL2A1、Aggrecan mRNA表达均较各自对照组显著上调(P < 0.01);鼻黏膜间充质干细胞实验组的COL2A1、Aggrecan mRNA表达较骨髓间充质干细胞实验组显著上调(P < 0.01);骨髓间充质干细胞实验组的SOX-9 mRNA表达与其对照组无显著差异(P > 0.05);鼻黏膜间充质干细胞实验组的SOX-9 mRNA表达较其对照组显著上调(P < 0.01);鼻黏膜间充质干细胞实验组的SOX-9 mRNA表达较骨髓间充质干细胞实验组显著上调(P < 0.01);骨髓间充质干细胞对照组和鼻黏膜间充质干细胞对照组在成软骨分化过程中COL2A1、SOX-9及Aggrecan mRNA表达无明显差异(P > 0.05),见图3。"
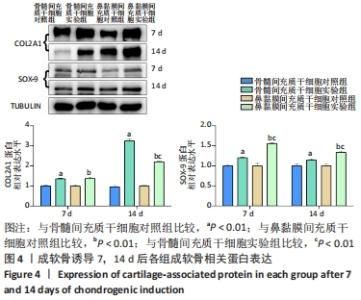
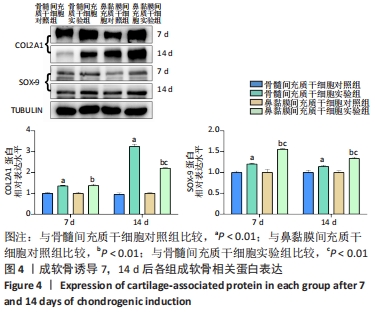
2.3 免疫印迹检测诱导软骨分化过程中相关蛋白表达水平 干预7,14 d后,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的COL2A1、SOX-9蛋白表达均高于各自对照组,差异均有显著性意义(P < 0.01);干预7 d时,两实验组的COL2A1蛋白表达差异无显著性意义(P > 0.05),然而干预14 d时,骨髓间充质干细胞实验组COL2A1蛋白表达高于鼻黏膜间充质干细胞实验组,差异有显著性意义(P < 0.01),干预7 d时,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的COL2A1蛋白表达均增加约1.4倍,而干预14 d时,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的COL2A1蛋白表达增加约3.2,2.2倍。干预7,14 d时,鼻黏膜间充质干细胞实验组的SOX-9蛋白表达高于骨髓间充质干细胞实验组,差异有显著性意义(P < 0.01),干预7 d时,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的SOX-9蛋白表达增加约1.2,1.6倍,而干预14 d时,骨髓间充质干细胞实验组和鼻黏膜间充质干细胞实验组的SOX-9蛋白表达增加约1.1,1.3倍;骨髓间充质干细胞对照组和鼻黏膜间充质干细胞对照组在成软骨分化过程中COL2A1、SOX-9蛋白表达无明显差异(P > 0.05),见图4。"
| [1] JIANG Y, TUAN RS. Origin and function of cartilage stem/progenitor cells in osteoarthritis. Nat Rev Rheumatol. 2015;11(4):206-212. [2] NEOGI T. The epidemiology and impact of pain in osteoarthritis. Osteoarthritis Cartilage. 2013;21(9):1145-1153. [3] DA SILVA MEIRELLES L, CHAGASTELLES PC, NARDI NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119(Pt 11):2204-2213. [4] KIM SW, LEE IK, YUN KI, et al. Adult stem cells derived from human maxillary sinus membrane and their osteogenic differentiation. Int J Oral Maxillofac Implants. 2009;24(6):991-998. [5] HWANG SH, PARK SH, CHOI J, et al. Characteristics of mesenchymal stem cells originating from the bilateral inferior turbinate in humans with nasal septal deviation. PLoS One. 2014;9(6):e100219. [6] HAN C, WANG YJ, WANG YC, et al. Caveolin-1 downregulation promotes the dopaminergic neuron-like differentiation of human adipose-derived mesenchymal stem cells. Neural Regen Res. 2021; 16(4):714-720. [7] PESTKA JM, BODE G, SALZMANN G, et al. Clinical outcome of autologous chondrocyte implantation for failed microfracture treatment of full-thickness cartilage defects of the knee joint. Am J Sports Med. 2012;40(2):325-331. [8] CHEN FH, TUAN RS. Mesenchymal stem cells in arthritic diseases. Arthritis Res Ther. 2008;10(5):223. [9] PITTENGER MF, MACKAY AM, BECK SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. [10] IM GI, SHIN YW, LEE KB. Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthritis Cartilage. 2005;13(10):845-853. [11] LINDSAY SL, MCCANNEY GA, WILLISON AG, et al. Multi-target approaches to CNS repair: olfactory mucosa-derived cells and heparan sulfates. Nat Rev Neurol. 2020;16(4):229-240. [12] ZHOU F, MA D, OUYANG M, et al. Repair mechanism of mesenchymal stem cells derived from nasal mucosa in orbital fracture. Am J Transl Res. 2018;10(6):1722-1729. [13] SHAFIEE A, KABIRI M, AHMADBEIGI N, et al. Nasal septum-derived multipotent progenitors: a potent source for stem cell-based regenerative medicine. Stem Cells Dev. 2011;20(12):2077-2091. [14] GIRARD SD, DEVÉZE A, NIVET E, et al. Isolating nasal olfactory stem cells from rodents or humans. J Vis Exp. 2011;(54):2762. [15] KARIMI S, BAGHER Z, NAJMODDIN N, et al. Alginate-magnetic short nanofibers 3D composite hydrogel enhances the encapsulated human olfactory mucosa stem cells bioactivity for potential nerve regeneration application. Int J Biol Macromol. 2021;167:796-806. [16] 吕德民,史文涛,陆浩,等.组织型转谷氨酰胺酶2在鼻黏膜间充质干细胞分化过程中的表达[J].中国组织工程研究,2020,24(19): 2959-2964. [17] QI C, XIAOFENG X, XIAOGUANG W. Effects of toll-like receptors 3 and 4 in the osteogenesis of stem cells. Stem Cells Int. 2014;2014:917168. [18] HWANG SH, PARK SH, CHOI J, et al. Age-related characteristics of multipotent human nasal inferior turbinate-derived mesenchymal stem cells. PLoS One. 2013;8(9):e74330. [19] GE L, JIANG M, DUAN D, et al. Secretome of Olfactory Mucosa Mesenchymal Stem Cell, a Multiple Potential Stem Cell. Stem Cells Int. 2016;2016:1243659. [20] JOHNSTONE B, HERING TM, CAPLAN AI, et al. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238(1):265-272. [21] WANG W, RIGUEUR D, LYONS KM. TGFbeta signaling in cartilage development and maintenance. Birth Defects Res C Embryo Today. 2014;102(1):37-51. [22] DUSFOUR G, MAUMUS M, CAÑADAS P, et al. Mesenchymal stem cells-derived cartilage micropellets: A relevant in vitro model for biomechanical and mechanobiological studies of cartilage growth. Mater Sci Eng C Mater Biol Appl. 2020;112:110808. [23] SHEN H, LIN H, SUN AX, et al. Acceleration of chondrogenic differentiation of human mesenchymal stem cells by sustained growth factor release in 3D graphene oxide incorporated hydrogels. Acta Biomater. 2020;105:44-55. [24] ZHANG H, MA X, ZHANG L, et al. The ability to form cartilage of NPMSC and BMSC in SD rats. Int J Clin Exp Med. 2015;8(4):4989-4996. [25] AKIYAMA H, CHABOISSIER MC, MARTIN JF, et al. The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev. 2002;16(21):2813-2828. [26] JAYASURIYA CT, CHEN Y, LIU W, et al. The influence of tissue microenvironment on stem cell-based cartilage repair. Ann N Y Acad Sci. 2016;1383(1):21-33. [27] LEFEBVRE V, DVIR-GINZBERG M. SOX9 and the many facets of its regulation in the chondrocyte lineage. Connect Tissue Res. 2017;58(1): 2-14. [28] AUGELLO A, DE BARI C. The regulation of differentiation in mesenchymal stem cells. Hum Gene Ther. 2010;21(10):1226-1238. [29] IKEGAMI D, AKIYAMA H, SUZUKI A, et al. Sox9 sustains chondrocyte survival and hypertrophy in part through Pik3ca-Akt pathways. Development. 2011;138(8):1507-1519. [30] HATA K, TAKAHATA Y, MURAKAMI T, et al. Transcriptional Network Controlling Endochondral Ossification. J Bone Metab. 2017;24(2):75-82. [31] BUHRMANN C, BUSCH F, SHAYAN P, et al. Sirtuin-1 (SIRT1) is required for promoting chondrogenic differentiation of mesenchymal stem cells. J Biol Chem. 2014 ;289(32):22048-22062. [32] GONZALEZ-FERNANDEZ T, TIERNEY EG, CUNNIFFE GM, et al. Gene Delivery of TGF-β3 and BMP2 in an MSC-Laden Alginate Hydrogel for Articular Cartilage and Endochondral Bone Tissue Engineering. Tissue Eng Part A. 2016;22(9-10):776-787. [33] CASANOVA MR, ALVES DA SILVA M, COSTA-PINTO AR, et al. Chondrogenesis-inductive nanofibrous substrate using both biological fluids and mesenchymal stem cells from an autologous source. Mater Sci Eng C Mater Biol Appl. 2019;98:1169-1178. [34] BHARDWAJ N, DEVI D, MANDAL BB. Tissue-engineered cartilage: the crossroads of biomaterials, cells and stimulating factors. Macromol Biosci. 2015;15(2):153-182. [35] THORP H, KIM K, KONDO M, et al. Fabrication of hyaline-like cartilage constructs using mesenchymal stem cell sheets. Sci Rep. 2020;10(1): 20869. [36] PELTTARI K, PIPPENGER B, MUMME M, et al. Adult human neural crest-derived cells for articular cartilage repair. Sci Transl Med. 2014; 6(251):251ra119. [37] MUMME M, BARBERO A, MIOT S, et al. Nasal chondrocyte-based engineered autologous cartilage tissue for repair of articular cartilage defects: an observational first-in-human trial. Lancet. 2016; 388(10055):1985-1994. |
| [1] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [2] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [3] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [4] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [5] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [6] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [7] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [8] | Wu Chengcong, Wang Fang, Wan Jianshan, Wu Zheng, Sun Rong, Huang Hefei, Qian Xuankun, Ou Hua, Ren Jing. Adenovirus-mediated bone morphogenetic protein 2 induces osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4757-4761. |
| [9] | Dong Yi, Shan Shuai, Liu Jialin, Han Xiangzhen, He Huiyu. Circular RNA mmu_circ_0001775 knockdown improves the osteogenic ability of mouse bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4767-4772. |
| [10] | Shen Enpu, Huang Ba, Liu Danping, Qi Hui, Wu Zhiwen, Li Beibei. Exosomes derived from melatonin-modified bone marrow mesenchymal stem cells promote osteogenesis of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4800-4805. |
| [11] | Huang Gao, Xu Jun, Chen Wenge. Implantation of bone marrow mesenchymal stem cells-loaded platelet-rich plasma combined with extracorporeal shock wave in the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4812-4818. |
| [12] | Jia Qiyu, Huang Xiaoxia, Guo Jian, Huang Jinyong, Guo Xiaobin, Abdussalam·Alimujiang, Wu Tong, Ma Chuang. Integrin-targeted peptide promotes proliferation of bone marrow mesenchymal stem cells in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4780-4786. |
| [13] | Zhan Yuanbo, Liu Xinpeng, Xu Wenxia, Miao Nan, Mu Sen, Zhang Ruimin, Li Ying. Expression of histone deacetylase 9 in bone marrow mesenchymal stem cells during senescence [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4762-4766. |
| [14] | Wu Xiaosong, Gong Chao, Lou Pan, Wang Wei. Possibility of forskolin and 3-isobutyl-1-methylxanthine in inducing bone marrow mesenchymal stem cells to differentiate into Schwann cells-like phenotype [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4787-4792. |
| [15] | Gao Zilong, Li Ting, Lyu Zheng, Shen Huarui. Psoralen combined with transforming growth factor beta 1 induces the differentiation of bone marrow mesenchymal stem cells into chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4884-4888. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||