Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (20): 3243-3249.doi: 10.12307/2022.628
Previous Articles Next Articles
Role and regulatory mechanism of glycophagy in glycogen metabolism
Lyu Yuhu1, Cheng Lin1, Lin Wentao2, Peng Fenglin1
- 1College of Sport and Health, Guangxi Normal University, Guilin 541006, Guangxi Zhuang Autonomous Region, China; 2School of Physical Education and Sports Science, Zhuhai College of Science and Technology, Zhuhai 519041, Guangdong Province, China
-
Received:2021-09-14Accepted:2021-10-15Online:2022-07-18Published:2022-01-20 -
Contact:Peng Fenglin, DS, Professor, Doctoral supervisor, College of Sport and Health, Guangxi Normal University, Guilin 541006, Guangxi Zhuang Autonomous Region, China -
About author:Lyu Yuhu, PhD candidate, Lecturer, College of Sport and Health, Guangxi Normal University, Guilin 541006, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 31560291 (to PFL)
CLC Number:
Cite this article
Lyu Yuhu, Cheng Lin, Lin Wentao, Peng Fenglin. Role and regulatory mechanism of glycophagy in glycogen metabolism[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3243-3249.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
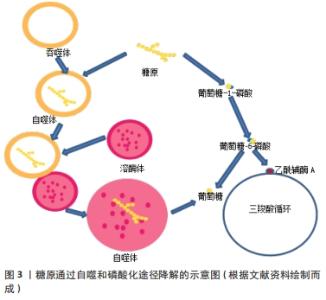
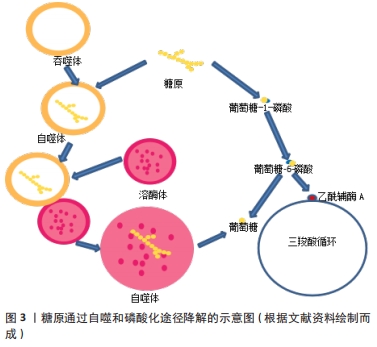
2.1 糖噬底物的形态、功能及降解途径 2.1.1 糖噬底物的形态、功能 糖原是糖噬的底物,也是细胞内的支链多糖和能源储存形式[10],空间上主要贮存在胞浆和自噬体内[11]。糖原的贮存形态有α颗粒和β颗粒两种,其中α颗粒糖原体积比低,常见于肝脏之中,β颗粒糖原体积比高,常见于肌肉之中[12];也有研究认为还存在γ颗粒,但研究证实γ颗粒为β颗粒上富含蛋白质的亚基[13]。由于肝糖原、肌糖原的功能存在差异,因此颗粒贮存的特点可能反映了不同组织所需的糖原分解与释放的不同。肝糖原的主要作用之一是维持体内血糖的相对稳定,对肝细胞的研究已经确定了含淀粉结合域蛋白1(starch-binding domain-containing protein 1,STBD1)和GABA(A)受体相关蛋白1(GABA type a receptor associated protein like 1,GABARAPL1)为调节这一过程的两个关键蛋白[7]。不同于肝脏和骨骼肌,心肌组织中α和β两种颗粒均含有,可能与心肌特殊的供能需求有关[14-15],但是心肌糖原的贮存、结构和功能之间的确切关系尚不清楚。 2.1.2 糖噬底物的降解途径 ①磷酸化降解途径:糖原可被磷酸化为葡萄糖-1-磷酸(glucose-1-phosphate,G1P或G-1-P),之后G1P转变为葡萄糖-6-磷酸(glucose-6-phosphate,G6P或G-6-P),G6P可以生成葡萄糖,或生成乙酰辅酶A进入三羧酸循环。②糖噬降解途径:早在1971年ORCI等[16]就发现了富含糖原的溶酶体,称为“糖原溶酶体”,糖原进入溶酶体可被酸性α-葡萄糖苷酶(acid α-glucosidase,GAA)水解[17],之后许多研究一直使用“糖原溶酶体”这一名称[18-19],2000年之后相关研究将此过程称为糖原的自噬[20],认为属于非选择性大自噬的范畴。心肌内也充满自噬体[21],且糖原自噬衍生的非磷酸化葡萄糖协助糖原降解衍生的G6P抵抗产后低血糖症,确保新生儿期葡萄糖稳态的微调[22]。需要指出的是有研究者认为内质网、糖原颗粒和吞噬体可以形成三角作为糖原分解和糖噬的中心[23]。糖原的降解途径如图3所示。"
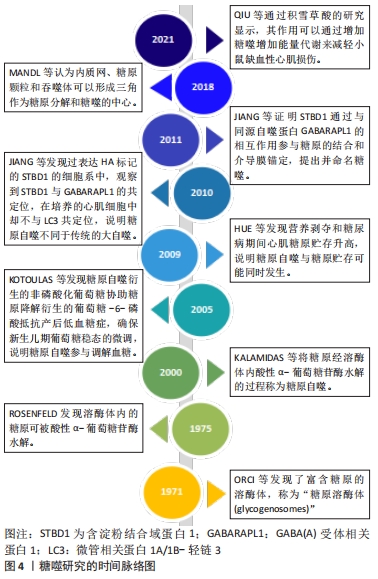
动物模型发现自噬相关基因(autophagy-related gene,ATG)缺陷的ATG5和ATG7小鼠[24-25],出生后第1天就会死亡,可能与自噬在能量应激时表现出的积极作用相关。尽管胰岛素抵抗增加,但抑制自噬会降低由遗传和饮食引起的肥胖症的血糖水平[26],也可能是由于自噬介导的糖原降解受损所致,表明自噬降解并释放葡萄糖与肝糖原水解并非同一过程。电子显微镜图像显示新生大鼠心肌细胞中糖原是双膜结构[27],营养剥夺和糖尿病期间心肌糖原贮存升高[28],这与营养剥夺形成了鲜明的对比,一方面心肌因营养剥夺或糖尿病引起的糖吸收障碍而迫切需要能量供应;另一方面心肌的糖原贮存却在不断增加,明显与大自噬相关研究认为的营养剥夺期间自噬上调有相悖之处[29]。微管相关蛋白1A/1B-轻链3(MAP1LC3,LC3)由LC3Ⅰ和LC3Ⅱ组成,通常作为反映大自噬程度的标志性因子,LC3Ⅱ/LC3Ⅰ更是作为自噬通量的标志[30],在过表达HA标记的STBD1的细胞系中,观察到STBD1与GABARAPL1的共定位,在培养的心肌细胞中却不与LC3共定位[31],表明心肌细胞中的糖原自噬与大自噬有所不同。直到2011年,JIANG等[3]证明STBD1通过与GABARAPL1相互作用参与糖原的结合和介导膜锚定,提出并命名糖噬,认为糖噬是选择性自噬而不是非选择自噬,且得到了证明[15]。研究显示,ATG7缺乏的成年小鼠禁食期间肝糖原加速耗竭,但进食时则不然[32],说明因自噬降解释放的葡萄糖下降增加了肝糖原应激以释放更多的葡萄糖来维持血糖的相对稳定,而进食时因饮食摄糖而不同,证明糖噬可能参与了血糖平衡过程。后经研究现发糖噬是由STBD1与糖原结合,然后与吞噬体上的GABARAPL1结合被溶酶体中的GAA降解为非磷酸化的葡萄糖的过程[33],STBD1和GABARAPL1为糖噬的标记蛋白。糖噬研究的时间脉络如图4所示。"

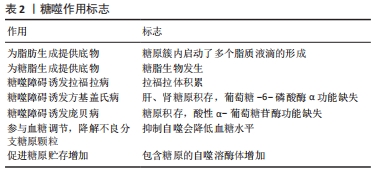
2.2.2 糖噬障碍可诱发相关疾病 糖噬功能障碍可能会引起很多相关疾病如遗传性肌病甚至癌症的发生[36-38]。糖噬障碍可引起拉福拉病(Lafora disease)而导致拉福拉体积累[5-6],由用来编码laforin磷酸酶的EPM2A基因和编码malin泛素连接酶的E3NHLRC1基因功能缺失突变所引起[39],糖原的磷酸化可能是将其螯合到自噬体中的先决条件,laforin或malin缺失导致糖原聚集及其有针对性的靶向作用,加上自噬受损进而导致拉福拉体的生成[8]。STBD1的过表达可能有助于改善拉福拉病的病情[40],拉福拉-马林复合物(laforin-malin complex)可调节葡萄糖的摄取,但不调节糖原降解,过高的细胞葡萄糖水平可能是导致拉福拉病中糖原异常增高的诱因[41]。糖噬障碍还可能引起方基盖氏病(von Gierke disease),是Ia型糖原贮积病,由葡萄糖-6-磷酸酶α的功能丧失引起G6P转化为葡萄糖障碍[7]。研究表明Ia型糖原贮积病肝脏内自噬通量下降[42],肾脏自噬受损,内质网应激增加。用雷帕霉素治疗1周,可以逆转葡萄糖-6-磷酸酶α敲除小鼠肾脏的内质网应激[43]。有研究认为自噬是Ia型糖原贮积病防御能力的一种[44],受葡萄糖-6-磷酸酶α表达活性的介导,且葡萄糖-6-磷酸酶α的表达活性是否达到3%可能是肝脏自噬能力的分界线,达到或超过3%自噬正常,小于3%自噬受损[45]。糖噬障碍还可能引起庞贝病,是Ⅱ型常染色体隐性糖原贮积病,由染色体上编码GAA的基因突变引起[8]。重组人酸α-葡萄糖苷酶替代疗法是较好的治疗庞贝病的方法,可以恢复心脏功能而显著延长婴儿患者的寿命,但研究发现酶替代疗法对骨骼肌的效果不强,治疗年龄越小效果可能越好[9]。庞贝病患者和GAA基因敲除小鼠中,肌肉中生产性自噬的失败对疾病病理贡献很大,通过充当重组酶的槽并阻止其有效传递至溶酶体来干扰酶替代疗法[46]。抑制mTORC1可能有利于庞贝病患者糖原在肌肉中积累[47]。GAA活性完全缺乏的患者中,甚至在分娩之前就已经发生了糖原的积累[48],当出生时肌肉高度损害、溶酶体破裂、糖原大量沉积却没有自噬结构的积累,表明这种机制可能在胎儿期很少被修改,也可能是由于营养物质通过脐带以补偿酶缺乏的作用[49],如表2所示。 2.2.3 糖噬参与血糖调节和糖原贮存 糖噬增加还伴随大自噬标记物表达的增加[15],说明糖噬和大自噬可能存在协同效应,且研究显示抑制自噬会降低由遗传和饮食引起的肥胖症的血糖水平[26]。当肝脏和肌肉通过禁食耗尽其糖原储存时,心肌却在能量应激时积累糖原并上调糖原自噬,与细胞质中糖原的磷酸化代谢途径不同,糖噬以其相对独立的代谢途径来应对饥饿应激[15]。骨骼肌的研究显示,糖噬通过动态调节来响应细胞或环境变化,参与降解不良分支糖原颗粒和能量代谢过程[50]。积雪草酸的研究显示,其作用通过增加糖噬增加能量代谢来减轻小鼠缺血性心肌损伤[51]。脊索瘤细胞的研究认为,其内存在嵌入糖原的溶酶体异质超结构似乎也是通过自噬动员了糖原的贮存[7],说明糖原积累和糖噬增加可能同时发生。另有研究显示糖原被吞噬体(phagosome)吞噬后并没有发生降解,而是贮存在吞噬体内,认为吞噬体内糖原贮存可能是预防严重心脏缺血等应激时的一种保护机制,而过量储存可能是病理的,心肌糖原的过度积累与严重的功能缺陷相关[52],说明一定范围内的糖原贮存或糖噬都对心肌缺血或能量应激起到了保护作用,但过量积累甚至是在能量应激期间仍表现为过量积累可能对机体有害,同时也说明糖噬是糖原降解固有的且不可或缺的途径,并非糖原降解的冗余通路,此过程存在障碍将引起糖原代谢异常,可能会危及健康甚至生命。饥饿诱导心肌糖原贮存可能存在物种差异[53],针对不同性别的研究发现禁食后仅雌鼠心脏内心肌糖原蓄积增加,且缺血再灌注后女性心脏的缺血后恢复面积更大,梗死面积也相对较小[15]。电子显微镜研究显示,能量应激后女性心肌贮存的β颗粒糖原比例升高,而男性心肌贮存的糖原却未见显著性差异[54-55],说明糖原的贮存还可能存在性别的差异且更偏向β颗粒,可能是女性容易患糖尿病性心肌病的原因之一[56]。如表2所示。 2.3 糖噬调节的信号通路"

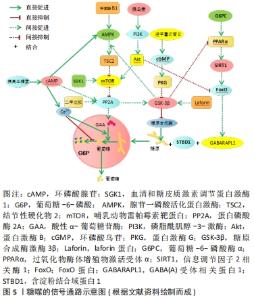
2.3.1 糖噬的抑制通路 PI3K/mTOR信号通路:磷酸肌醇-3-激酶(phosphoinositide-3-Kinase,PI3K)是脂质磷酸激酶,催化肌醇第3位的磷酸化。新生动物肝细胞的研究表明,磷酸肌醇/雷帕霉素靶蛋白途径参与了糖原自噬降解的控制,通过作用于靶标蛋白磷酸酶2A(PP2A)调节糖原自噬[43]。FESCHENKO等[57]研究表明,糖原自噬由出生后低血糖期分泌的胰高血糖素诱导,并受胰岛素和胃肠外葡萄糖抑制,通过PI3K/mTOR通路起到抑制作用。另有研究也表明哺乳动物雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)可抑制肝糖原和心糖原的自噬[27,58]。如图5所示。"


Akt/FoxO信号通路:研究发现细胞外葡萄糖和胰岛素可介导糖噬和心肌细胞糖原含量,在高糖浓度下胰岛素引起糖原积累和STBD1升高;当血糖浓度在5 mmol/L时,胰岛素引起蛋白激酶B磷酸化增多[59]。胰岛素[59]、Fox蛋白和micro-RNAs等均参与STBD1和GABARAPL1的调节[60-62],且胰岛素可以激活Akt进而抑制FoxO1和FoxO3与GABARAPL1启动子结合而抑制GABARAPL1的转录[63],从而抑制糖原进入溶酶体而限制糖噬,起到降低血糖的作用,与胰岛素降血糖的宏观作用相吻合。如图5所示。 SGK1/mTOR信号通路:拉福拉病中,血清和糖皮质激素调节蛋白激酶1(serum/glucocorticoid-induced kinase 1,SGK1)的激活可能与糖原水平升高、mTOR激活和自噬缺陷有关[64],抑制SGK1会降低质膜结合的葡萄糖转运蛋白而引起葡萄糖摄取及糖原积累降低,SGK1激活mTOR、抑制糖原分解和加剧糖原贮存,其调节途径也可能通过激活mTOR抑制蛋白磷酸酶2A而起作用。如图5所示。 2.3.2 糖噬的激活通路 cAMP/PKA信号通路:大鼠近端肾小管上皮细胞的研究证实环磷酸腺苷(cyclic adenosine monophosphate,cAMP)通过非直接激活蛋白磷酸酶2A调节糖原自噬[57]。新生动物肝细胞的研究也表明cAMP/cAMP依赖性蛋白激酶(protein kinase A,PKA)参与了糖原自噬降解的控制,与PI3K/mTOR一起汇集于同一靶标蛋白磷酸酶2A调节糖原自噬[43]。另有研究显示,cAMP通路也参与了心脏糖原自噬的调控[27]。FESCHENKO等[57]研究表明cAMP/PKA为糖噬的诱导途径。mTOR激活可引起自噬受损,与腺苷酸活化蛋白激酶(adenosine monophosphate-activated protein kinase,AMPK)信号下调有关[42],而cAMP增多尤其是cAMP/ATP上升可以激活AMPK。通过结节性硬化物(TSC)敲除的研究显示,庞贝病小鼠整个肌肉中mTOR的重新激活导致萎缩的逆转和自噬的显著消除,同样说明mTOR介导了糖噬的过程[65]。如图5所示。 G6PC/SIRT1/FoxO信号通路:研究显示,肝脏葡萄糖-6-磷酸酶α(G6PC)缺乏抑制过氧化物酶体增殖物激活受体α的表达,使肝脏沉默信息调节因子2相关酶1(silencing information regulatory factor 2 related enzyme 1,SIRT1)信号下调而增加ATG乙酰化,降低了ATG12-ATG5结合的同时下调诱导自噬基因的FoxO信号[66]。ATG12-ATG5可促进自噬泡延伸,所以葡萄糖-6-磷酸酶α的缺乏或功能受损将使自噬泡延伸障碍、自噬小体形成受损而降低吞噬糖原的能力。GJORGJIEVA等[44]认为SIRT1信号下调是独立于mTOR途径的肝自噬受损的基础;而通过基因转移恢复肝脏葡萄糖-6-磷酸酶α表达可以使有缺陷的自噬正常化,恢复SIRT1、FoxO3a、AMPK和过氧化物酶体增殖物激活受体α信号,并纠正与葡萄糖-6-磷酸酶α缺乏相关的代谢异常[67]。以上分析表明,葡萄糖-6-磷酸酶α缺乏促使SIRT1下调是独立于mTOR通路的糖噬的又一通路。如图5所示。 Ca2+信号通路:缺血心肌胞浆Ca2+增加情况下会增强GAA水解糖原的活性,加速糖噬过程[68]。遗传性肌病和心肌病中发现有自噬体和糖原积累[69]。先天缺乏GAA时,即使在能量应激时糖原仍然会在骨骼肌、呼吸肌和心肌中积累[70-72],由于细胞质内糖原没有通过磷酸化途径降解而积累,原因可能是糖噬有识别特殊的糖原结构促使其进入溶酶体内贮存的作用。有研究也表明溶酶体中钙的积累会影响糖原自噬[43]。而关于新生大鼠的研究发现胰高血糖素可能增加了大鼠肝或心脏GAA的酶活性[73]。如图5所示。 cGMP/PKG/GSK-3β信号通路:胰岛素可促进糖原合成,而糖原合成酶激酶3β(glycogen synthase kinase-3β,GSK-3β)可磷酸化糖原合成酶而抑制其糖原合成。laforin可使GSK-3β在ser9处去磷酸化而激活和减弱mTOR的作用[74-75],进而起到激活自噬的作用。缺血期间GSK-3β的激活刺激自噬,而再灌注期间的失活抑制自噬[76],GSK-3β可通过AMPK/mTOR途径激活自噬而改善肝脏缺血再灌注损伤[77]。AMPK不仅通过激活结节性硬化物2抑制mTOR,而且通过直接磷酸化mTORC1或ULK1(哺乳动物ATG1同源基因)来刺激自噬[76]。SIRT1和AMPK正调控自噬[78-79],肝激酶B1通过AMPK-a亚基的磷酸化激活AMPK而促进自噬[80]。由于GSK-3β去磷酸化激活自噬同时抑制糖原合成酶而使糖原合成减少,所以自噬的增加可能伴随着糖原合成的减少,这可能与能量应激有关,如图5所示。 研究指出二甲双胍可增加心脏自噬活性[81],经二甲双胍处理的寄生虫糖原含量会下降[82],认为发生了糖噬现象,可能与二甲双胍促进蛋白磷酸酶2A激活GSK-3β有关[83],而增加GSK-3β活性还可抑制过氧化物酶体增殖物激活受体α进而抑制糖原的贮存[84]。另有研究表明,淫羊藿次苷Ⅱ不仅使GSK-3β失活,还恢复了环磷酸鸟苷(cyclic guanosic monophosphate,cGMP)/蛋白激酶G(protein kinase G,PKG)途径,且GSK-3β是PKG的下游和自噬相关蛋白的上游也被证实,通过抑制过度自噬减轻脑缺血/再灌注损伤[85]。而糖皮质激素戒断引起的糖皮质激素或肾上腺功能不全会抑制糖原合成酶的酶活性和/或激活糖噬[86],可能也与GSK-3β相关。除了以上的调节通路外,最近的研究显示Wdfy3编码基因也参与了糖噬的调控[87],但其调节的信号通路是否与以上的通路重合仍有待进一步研究。"

| [1] ZHAO H, TANG MZ, LIU MQ, et al. Glycophagy: An emerging target in pathology. Clin Chim Acta. 2018;484:298-303. [2] CONDELLO M, PELLEGRINI E, CARAGLIA M, et al. Targeting Autophagy to Overcome Human Diseases. Int J Mol Sci. 2019;20(3):725. [3] JIANG SX, WELLS CD, ROACH PJ. Starch-binding domain-containing protein 1 (Stbd1) and glycogen metabolism: Identification of the Atg8 family interacting motif (AIM) in Stbd1 required for interaction with GABARAPL1. Biochem Biophys Res Commun. 2011;413(3):420-425. [4] KOLB-LENZ D, FUCHS R, LOHBERGER B, et al. Characterization of the endolysosomal system in human chordoma cell lines: is there a role of lysosomes in chemoresistance of this rare bone tumor? Histochem Cell Biol. 2018;150:83-92. [5] SINHA P, VERMA B, GANESH S. Trehalose Ameliorates Seizure Susceptibility in Lafora Disease Mouse Models by Suppressing Neuroinflammation and Endoplasmic Reticulum Stress. Mol Neurobiol. 2021;58:1088-1101. [6] PURI R, GANESH S. Laforin in autophagy: A possible link between carbohydrate and protein in Lafora disease? . Autophagy. 2010;6(8): 1229-1231. [7] CHOU JY, JUN HS, MANSFIELD BC. Glycogen storage disease type I and G6Pase-β deficiency: etiology and therapy. Nat Rev Endocrinol. 2010;6(12):676-688. [8] PASCARELLA A, TERRACCIANO C, FARINA O, et al. Vacuolated PAS-positive lymphocytes as an hallmark of Pompe disease and other myopathies related to impaired autophagy. J Cell Physiol. 2018;233(8): 5829-5837. [9] KISHNANI PS, CORZO D, NICOLINO M, et al. Recombinant human acid α-glucosidase: major clinical benefits in infantile-onset Pompe disease. Neurology. 2007;68(20):99-109. [10] GREENBERG CC, JURCZAK MJ, DANOS AM, et al. Glycogen branches out: new perspectives on the role of glycogen metabolism in the integration of metabolic pathways. Am J Physiol Endocrinol Metab. 2006;291(1):E1-E8. [11] IWAMASA T, TSURU T, HAMADDA T, et al. Physicochemical and Ultrastructural Studies on Glycogenosomes in Newborn Rat Hepatocytes. Pathol Res Pract. 1980;167(2):363-373. [12] RYU JH, DRAIN J, KIM JH, et al. Comparative structural analyses of purified glycogen particles from rat liver, human skeletal muscle and commercial preparations. Int J Biol Macromol. 2009;45(5):478-482. [13] PRATS C, GRAHAM TE, SHEARER J. The dynamic life of the glycogen granule. J Biol Chem. 2018;293(19):7089-7098. [14] BESFORD QA, SULLIVAN MA, ZHENG L, et a. The structure of cardiac glycogen in healthy mice. Int J Biol Macromol. 2012;51(5):887-891. [15] REICHELT ME, MELLOR KM, CURL CL, et al. Myocardial glycophagy-a specific glycogen handling response to metabolic stress is accentuated in the female heart. J Mol Cell Cardiol. 2013;65:67-75. [16] ORCI L, STAUFFACHER W. Glycogenosomes in renal tubular cells of diabetic animals. J Ultrastruct Res. 1971;36(3-4):499-503. [17] ROSENFELD EL. Alpha-glucosidases (gamma-amylases) in human and animal organisms. Pathol Biol (Paris). 1975;23(1):71-84. [18] CAVANAGH JB, JONES HB. Glycogenosomes in the aging rat brain: their occurrence in the visual pathways. Acta Neuropathol. 2000;99(5):496-502. [19] REYES-LUGO M, SÁNCHEZ T, FINOL HJ, et al. Neurotoxic activity and ultrastructural changes in muscles caused by the brown widow spider Latrodectus geometricus venom. Rev Inst Med Trop Sao Paulo. 2009;51(2):95-101. [20] KALAMIDAS SA, KOTOULAS OB. Glycogen autophagy in newborn rat hepatocytes. Histol Histopathol. 2000;15(4):1011-1018. [21] KOTOULAS OB, KALAMIDAS SA, KONDOMERKOS DJ. Glycogen Autophagy. Microsc Res Tech. 2004;64(1):10-20. [22] KOTOULAS OB, KALAMIDAS SA, KONDOMERKOS DJ. Glycogen autophagy in glucose homeostasis. Pathol Res Pract. 2006;202:631-638. [23] MANDL J, BANHEGYI G. The ER - Glycogen Particle - Phagophore Triangle: A Hub Connecting Glycogenolysis and Glycophagy? Pathol Oncol Res. 2018;24(4):821-826. [24] KUMA A, HATANO M, MATSUI M, et al. The role of autophagy during the early neonatal starvation period. Nature. 2004;432(7020):1032-1036. [25] KOMATSU M, WAGURI S, UENO T, et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J Cell Biol. 2005;169(3):425-434. [26] YANG L, LI P, FU SN. Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance. Cell Meta. 2010;11(6):467-478. [27] KONDOMERKOS DJ, KALAMIDAS SA, KOTOULAS OB, et al. Glycogen autophagy in the liver and heart of newborn rats. The effects of glucagon, adrenalin or rapamycin. Histol Histopathol. 2005;20(3):689-696. [28] HUE L, TAEGTMEYER H. The Randle cycle revisited: a new head for an old hat. Am J Physiol Endocrinol Metab. 2009;297(3):E578-E591. [29] MASOUD WGT, CLANACHAN AS, LOPASCHUK GD. The Failing Heart: Is It an Inefficient Engine or an Engine Out of Fuel? . Cardiac Remodeling. 2013;5:65-84. [30] ZHANG XN, YAN HJ, YUAN Y, et al. Cerebral ischemia-reperfusion-induced autophagy protects against neuronal injury by mitochondrial clearance. Autophagy. 2013;9(9):1321-1333. [31] JIANG SX, HELLER B, TAGLIABRACCI VS, et al. Starch binding domain-containing protein 1/genethonin 1 is a novel participant in glycogen metabolism. J Biol Chem. 2010;285(45):34960-34971. [32] KARSLI-UZUNBAS G, GUO JY, PRICE S, et al. Autophagy is Required for Glucose Homeostasis and Lung Tumor Maintenance. Cancer Discov. 2014;4(8):914-927. [33] DELBRIDGE LM, MELLOR KM, TAYLOR DJ, et al. Myocardial autophagic energy stress responses-macroautophagy, mitophagy, and glycophagy. Am J Physiol Heart Circ Physiol. 2015;308(10):H1194-H1204. [34] MAYEUF-LOUCHART A, LANCEL S, Sebti Y, et al. Glycogen Dynamics Drives Lipid Droplet Biogenesis during Brown Adipocyte Differentiation. Cell Rep. 2019;29(6):1410-1418.e6. [35] MAYEUF-LOUCHART A. Uncovering the Role of Glycogen in Brown Adipose Tissue. Pharm Res. 2021;38:9-14. [36] PALHEGYI AM, SERANOVA E, DIMOVA S,et al. Biomedical Implications of Autophagy in Macromolecule Storage Disorders. Front Cell Dev Biol. 2019;7:179. [37] KAUR J, DEBNATH J. Autophagy at the crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol. 2015;16(8):461-472. [38] ZHANG WZ, HAN Z, XUE Y, et al. iCAL: a new pipeline to investigate autophagy selectivity and cancer. Autophagy. 2021;17(7):1799-1801. [39] PARIHAR R, RAI A, GANESH S, et al. Lafora disease: from genotype to phenotype. J Genet. 2018;97(3):611-624. [40] CONWAY O, AKPINAR HA, ROGOV VV, et al. Selective Autophagy Receptors in Neuronal Health and Disease. J Mol Biol. 2020;432(8): 2483-2509. [41] SINGH PK, SINGH S, GANESH S. The Laforin-Malin Complex Negatively Regulates Glycogen Synthesis by Modulating Cellular Glucose Uptake via Glucose Transporters. Mol Cell Biol. 2012;32(3):652-663. [42] FARAH BL, LANDAU DJ, SINHA RA, et al. Induction of autophagy improves hepatic lipid metabolism in glucose-6-phosphatase deficiency. J Hepatol. 2016;64:(2):370-379. [43] FARAH BL, LANDAU DJ, WU YJ, et al. Renal Endoplasmic Reticulum Stress is Coupled to Impaired Autophagy in a Mouse Model of GSD Ia. Mol Genet Metab. 2017;122(3):95-98. [44] GJORGJIEVA M, CALDERARO J, MONTEILLET L, et al. Dietary exacerbation of metabolic stress leads to accelerated hepatic carcinogenesis in glycogen storage disease type Ia. J Hepatol. 2018; 69(5):1074-1087. [45] ZHANG LS, LEE C, ARNAOUTOVA I, et al. Gene therapy using a novel G6PC-S298C variant enhances the longterm efficacy for treating glycogen storage disease type Ia. Biochem Biophys Res Commun. 2020; 527(3):824-830. [46] SHEA L, RABEN N. Autophagy in skeletal muscle: implications for Pompe disease. Int J Clin Pharmacol Ther. 2009;47(Suppl 1):S42-S47. [47] ASHE KM, TAYLOR KM, CHU QM, et al. Inhibition of glycogen biosynthesis via mTORC1 suppression as an adjunct therapy for Pompe disease. Mol Genet Metab. 2010;100(4):310-315. [48] PHUPONG V, SHUANGSHOTI S, SUTTHIRUANGWONG P, et al. Prenatal diagnosis of Pompe disease by electron microscopy. Arch Gynecol Obstet. 2005;271(3):259-261. [49] RODRÍGUEZ-ARRIBAS M, BRAVO-SAN PEDRO JM, Gómez-Sánchez R, et al. Pompe Disease and Autophagy: Partners in Crime, or Cause and Consequence? Curr Med Chem. 2016;23(21):2275-2285. [50] HEDEN TD, CHOW LS, HUGHEY CC, et al. Regulation and role of glycophagy in skeletal muscle energy metabolism. Autophagy. 2021;1-12. doi: 10.1080/15548627.2021.1969633. [51] QIU F, YUAN Y, LUO W, et al. Asiatic acid alleviates ischemic myocardial injury in mice by modulating mitophagy- and glycophagy-based energy metabolism. Acta Pharmacol Sin. 2021; doi: 10.1038/s41401-021-00763-9. [52] CHANDRAMOULI C, VARMA U, STEVENS EM, et al. Myocardial glycogen dynamics: New perspectives on disease mechanisms. Clin Exp Pharmacol Physiol. 2015;42(4):415-425. [53] MENAHAN LA, SOBOCINSKI KA. Comparison of carbohydrate and lipid metabolism in mice and rats during fasting. Comp Biochem Physiol B. 1983;74(4):859-864. [54] BERNASOCHI GB, BOON WC, DELBRIDGE LMD, et al. The myocardium and sex steroid hormone influences. Curr Opin Physiol. 2018;6:1-9. [55] REICHELT M, MELLOR K, STAPLETON D, et al. Glycogen Responses During Metabolic Stress are Accentuated in Female Hearts: A Role for Myocardial Glycophagy. Heart Lung Circulation. 2013;22:S63-S63. [56] REICHELT ME, MELLOR KM, BELL JR, et al. Sex, sex steroids, and diabetic cardiomyopathy: making the case for experimenta focus. Am J Physiol Heart Circ Physiol. 2013;305(6):H779-H792. [57] FESCHENKO MS, STEVENSON E, NAIRN AC, et al. A Novel cAMP-Stimulated Pathway in Protein Phosphatase 2A Activation. J Pharmacol Exp Ther. 2002;302(1):111-118. [58] KALAMIDAS SA, KONDOMERKOS DJ, KOTOULAS OB,et al. Electron Microscopic and Biochemical Study of the Effects of Rapamycin on Glycogen Autophagy in the Newborn Rat Liver. Microsc Res Tech. 2004; 63:215-219. [59] MELLOR KM, VARMA U, STEPLETON DI, et al. Cardiomyocyte glycophagy is regulated by insulin and exposure to high extracellular glucose. Am J Physiol Heart Circ Physiol. 2014;306(8):H1240-H1245. [60] PAULA-GOMES S, GONCALVES DA, BAVIERA AM, et al. Insulin suppresses atrophy‐and autophagy‐related genes in heart tissue and cardiomyocytes through AKT/FOXO signaling. Horm Metab Res. 2013; 45(12):849-855. [61] MO JW, ZHANG DF, YANG RZ. MicroRNA-195 regulates proliferation, migration, angiogenesis and autophagy of endothelial progenitor cells by targeting GABARAPL1. Biosci Rep. 2016;36(5):e00396. [62] DELBRIDGE LMD, MELLOR KM, TAYLOR DJ, et al. Myocardial stress and autophagy: mechanisms and potential therapies. Nat Rev Cardiol. 2017;14(7):412-425. [63] SENGUPTA A, MOLKENTIN JD, YUTZEY KE. FoxO transcription factors promote autophagy in cardiomyocytes. J Biol Chem. 2009;284(41): 28319-28331. [64] SINGH PK, SINGH S, GANESH S. Activation of serum/glucocorticoid-induced kinase 1 (SGK1) underlies increased glycogen levels, mTOR activation, and autophagy defects in Lafora disease. Mol Biol Cell. 2013;24(24):3776-3786. [65] LIM JA, LI LS, SHIRIHAI OS,et al. Modulation of mTOR signaling as a strategy for the treatment of Pompe disease. EMBO Mol Med. 2017; 9(3):353-370. [66] CHO JH, KIM GY, PAN CJ, et al. Downregulation of SIRT1 signaling underlies hepatic autophagy impairment in glycogen storage disease type Ia. PLoS Genet. 2017;13(5):e1006819. [67] GAUTAM S, ZHANG L, ARNAOUTOVA I, et al. The signaling pathways implicated in impairment of hepatic autophagy in glycogen storage disease type Ia. Hum Mol Genet. 2020;29(5):834-844. [68] KALAMIDAS SA, KOTOULAS OB, HANN AC. Studies on Glycogen Autophagy: Effects of Phorbol Myristate Acetate, Ionophore A23187, or Phentolamine. Microsc Res Tech. 2002;57(6):507-511. [69] NISHINO I, FU J, TANJI K, et al. Primary LAMP-2 deficiency causes X-linked vacuolar cardiomyopathy and myopathy (Danon disease). Nature. 2000; 406(6798):906-910. [70] 张娜.糖噬在晚发型Pompe病(LOPD)病程转归和致病机制中的作用[D].济南:山东大学,2019. [71] LIU Y, YANG Y, WANG BB, et al. Infantile Pompe disease: A case report and review of the Chinese literature. Exp Ther Med. 2016;11(1):235-238. [72] RABEN N, WONG A, RALSTON E, et al. Autophagy and mitochondria in Pompe disease: Nothing is so new as what has long been forgotten. Am J Med Genet C Semin Med Genet. 2012;160C (1):13-21. [73] KONDOMERKOS DJ, KALAMIDAS SA, KOTOULAS OB. An electron microscopic and biochemical study of the effects of glucagon on glycogen autophagy in the liver and heart of newborn rats. Microsc Res Tech. 2004;63(2):87-93. [74] LIU Y, WANG Y, WU C, et al. Dimerization of Laforin Is Required for Its Optimal Phosphatase Activity, Regulation of GSK3 Phosphorylation, and Wnt Signaling. J Biol Chem. 2006;281(46):34768-34774. [75] GIORGI FS, BIAGIONI F, LENZI P, et al. The role of autophagy in epileptogenesis and in epilepsy-induced neuronal alterations. J Neural Transm (Vienna). 2015;122(6):849-862. [76] ZHAI PY, SADOSHIMA J. Glycogen synthase kinase-3 beta controls autophagy during myocardial ischemia and reperfusion. Autophagy. 2012;8(1):138-139. [77] KONG DF, HUA XW, QIN T, et al. Inhibition of glycogen synthase kinase 3 beta protects liver against ischemia/reperfusion injury by activating 5 ‘ adenosine monophosphate-activated protein kinase-mediated autophagy. Hepatol Res. 2019;49(4):462-472. [78] GALLUZZI L, PIETROCOLA F, LEVINE B, et al. Metabolic Control of Autophagy. Cell. 2014;159:1263-1276. [79] SMITH BK, MARCINKO K, DESJARDINS EM, et al. Treatment of nonalcoholic fatty liver disease: role of AMPK. Am J Physiol Endocrinol Metab. 2016;311:E730-E740. [80] SHAW RJ, LAMIA KA, VASQUEZ D, et al. The Kinase LKB1 Mediates Glucose Homeostasis in Liver and Therapeutic Effects of Metformin. Science. 2005;310(5754):1642-1646. [81] XIE ZL, HE CY, ZOU MH. AMP-activated protein kinase modulates cardiac autophagy in diabetic cardiomyopathy. Autophagy. 2011;7(10):1254-1255. [82] LOOS JA, NICOLAO MC, CUMINO AC. Metformin promotes autophagy in Echinococcus granulosus larval stage. Mol Biochem Parasitol. 2018;224:61-70. [83] ELGENDY M, CIRÒ M, HOSSEINI A, et al. Combination of Hypoglycemia and Metformin Impairs Tumor Metabolic Plasticity and Growth by Modulating the PP2A-GSK3b-MCL-1 Axis. Cancer Cell. 2019;35(5):798-815.e5. [84] REN F, ZHANG L, ZHANG X, et al. Inhibition of glycogen synthase kinase 3β promotes autophagy to protect mice from acute liver failure mediated by peroxisome proliferator-activated receptor α. Cell Death Dis. 2016;7:e2151. [85] GAO JM, LONG L, XU F, et al. Icariside II, a phosphodiesterase 5 inhibitor, attenuates cerebral ischaemia/reperfusion injury by inhibiting glycogen synthase kinase-3 beta-mediated activation of autophagy. Br J Pharmacol. 2020;177(6):1434-1452. [86] DIVARI S, De LUCIA F, BERIO E, et al. Dexamethasone and prednisolone treatment in beef cattle: influence on glycogen deposition and gene expression in the liver. Domest Anim Endocrinol. 2020;72:106444. [87] NAPOLI E, PANOUTSOPOULOS AA, KYSAR P, et al. Wdfy3 regulates glycophagy, mitophagy, and synaptic plasticity. J Cereb Blood Flow Metab. 2021;271678X211027384. doi: 10.1177/0271678X211027384. |
| [1] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [2] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [3] | Cui Xing, Sun Xiaoqi, Zheng Wei, Ma Dexin. Huangqin Decoction regulates autophagy to intervene with intestinal acute graft-versus-host disease in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1057-1062. |
| [4] | Mo Weibin, Huang Tianchang, Zeng Zhiwei, Yan Linbo. Effects of Pueraria lobata flavonoids on expressions of beta-catenin and glycogen synthase kinase 3beta in the brain of rats undergoing exhaustive exercise after long endurance exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 736-741. |
| [5] | Mi Jianguo, Qiao Rongqin, Liu Shaojin. Bushen Jianpi Huoxue Recipe improves bone metabolism, oxidative stress, and autophagy in osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4147-4152. |
| [6] | Zeng Chunrong, Liu Menglan, Xie Yang, Li Zuoxiao. Preventive and therapeutic effects of naringin on experimental autoimmune encephalomyelitis via regulating microglial polarization [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4127-4135. |
| [7] | Zhang Gaofei, Wang Di, Li Jiamei, Lou Hanxiao, Zeng Yueqin, Liu Wenjun. Mesenchymal stem cells promote wound healing by regulating the autophagy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4058-4063. |
| [8] | Jiang Jie, Zhao Baixiao, Chen Libin, Wen Li, Zhang Shanshan, Ma Jie, Zhao Hua. Effect of moxibustion pretreatment on autophagy and NLRP3 inflammasome expression in cerebral ischemia-reperfusion model rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3615-3619. |
| [9] | Mei Hong, Qin Song, Li Kang, Chen Miao. Salbutamol attenuates hyperoxia-induced acute lung injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3657-3663. |
| [10] | Fan Shuai, Wu Chunfei, Liang Zujian, Xu Zhaohui, Xie Pingjin, Zhu Genfu. Bushen Tiaogan Prescription containing serum in the treatment of osteoarthritis by promoting chondrocyte autophagy in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3178-3183. |
| [11] | Cai Zhiguo, Du Shasha, Yang Kun, Zhao Na, Liu Qi. Lipopolysaccharides mediate autophagy of mouse insulinoma βtc6 cells in high glucose state [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3127-3132. |
| [12] | Wang Sihan, Chen Junji, Mo Weibin. Effects of Dendrobium officinale flavonoid on oxidative stress and autophagy in the liver of an exhaustive exercise rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3212-3219. |
| [13] | Sun Youqiang, Ma Chao, Liang Mengmeng, Xin Pengfei, Zhang Hua, Xiang Xiaobing. The pivotal role of autophagy in bone cells: bone-related cell activity and bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 276-282. |
| [14] | You Kun, Liu Yuanxin. Protective effect of exercise preconditioning on lung injury induced by hypobaric hypoxia in rats based on the nuclear factor E2-related factor 2/antioxidant response element signal pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2702-2707. |
| [15] | Cao Shuxing, Song Yongzhou, Li Ming, Zhang Hongliang, Zhao Lihui, Ma Wei. Regulatory role of curcumin in hydrogen peroxide-induced autophagy in chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2161-2166. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||