Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (25): 4084-4092.doi: 10.12307/2022.418
Previous Articles Next Articles
Stem cell transplantation in the treatment of premature ovarian failure: a meta-analysis based on 13 animal studies
Zhao Shuying1, Guo Guangling2, Liu Chenchen1, Zhang Chao3, Dong Sirui2, Gong Qinqin2, Ji Luwei2
- 1Postgraduate Training Base, Taihe Hospital, Jinzhou Medical University, Shiyan 442000, Hubei Province, China; 2Taihe Hospital, China Anti-aging Center of Affiliated Hospital of Hubei University of Medicine, Shiyan 442000, Hubei Province, China; 3Center for Evidence-Based Medicine and Clinical Research, Affiliated Taihe Hospital of Hubei University of Medicine, Shiyan 442000, Hubei Province, China
-
Received:2020-11-21Accepted:2021-01-07Online:2022-09-08Published:2022-01-26 -
Contact:Guo Guangling Master, Associate chief physician, Associate professor, Master’s supervisor, Taihe Hospital, China Anti-aging Center of Affiliated Hospital of Hubei University of Medicine, Shiyan 442000, Hubei Province, China -
About author:Zhao Shuying, Master candidate, Postgraduate Training Base, Taihe Hospital, Jinzhou Medical University, Shiyan 442000, Hubei Province, China -
Supported by:the Hubei Provincial Department of Education Project, No. B2013108 (to GGL)
CLC Number:
Cite this article
Zhao Shuying, Guo Guangling, Liu Chenchen, Zhang Chao, Dong Sirui, Gong Qinqin, Ji Luwei. Stem cell transplantation in the treatment of premature ovarian failure: a meta-analysis based on 13 animal studies[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4084-4092.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
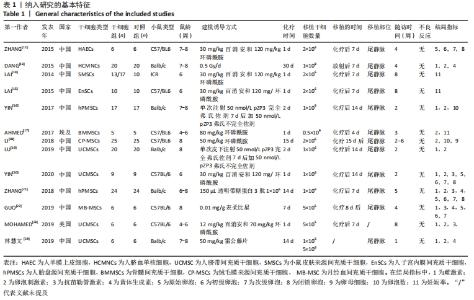
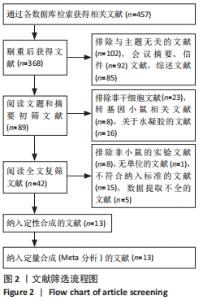
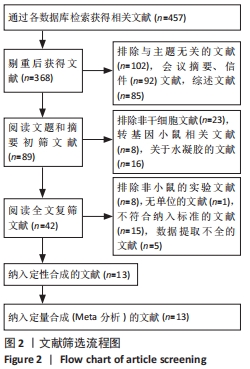
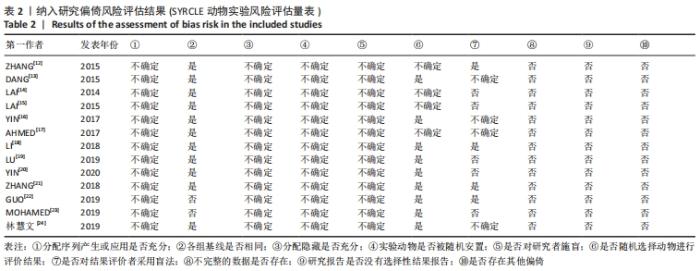
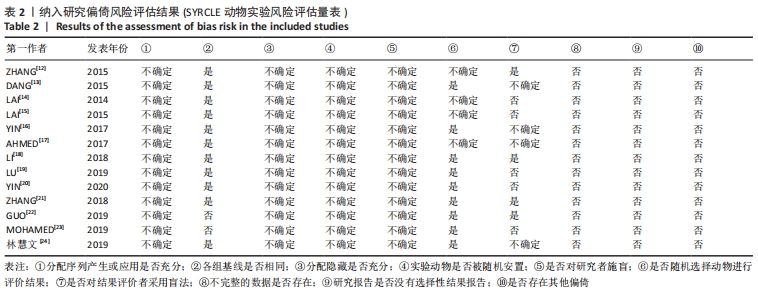
2.2 纳入文献的基本特征以及质量评价 共纳入13篇文献[12-24],共348只小鼠,干细胞组的小鼠184只,对照组的小鼠164只。在干细胞类型方面,其中4篇是脐带间充质干细胞[19-20,23-24],2篇是人胎盘源间充质干细胞[16, 22],1篇是人羊膜上皮细胞[12],1篇是人脐血单核细胞[13],1篇是皮肤来源的间充质干细胞[14],1篇是人子宫内膜间充质干细胞[15],1篇是骨髓来源的间充质干细胞[17],1篇是绒毛膜来源间充质干细胞[18],1篇是人月经血间充质干细胞[23]。共包括7个研究对象为C57BL/6的雌性小鼠[12, 15,17-18,20,22-23],5个研究对象为BALB/c的雌性小鼠[13,16,19,21,24],1个研究对照为ICR的雌性小鼠[14]。纳入研究的基线特征见表1。根据SYRCLE动物实验偏倚风险评估工具进行质量评价纳入研究,总体为中等质量研究,见表2。"
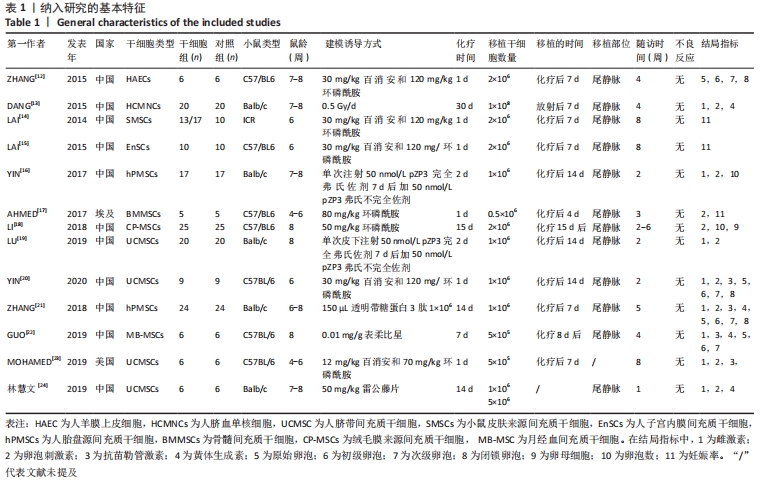
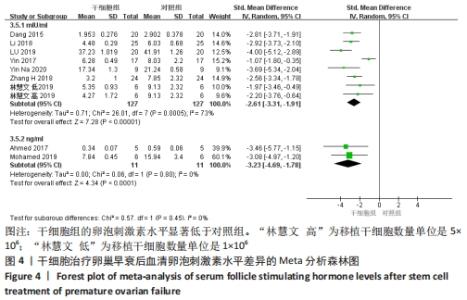
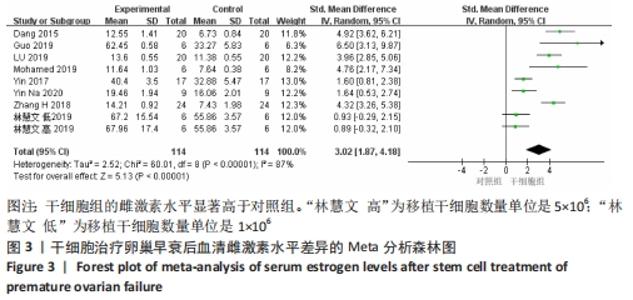
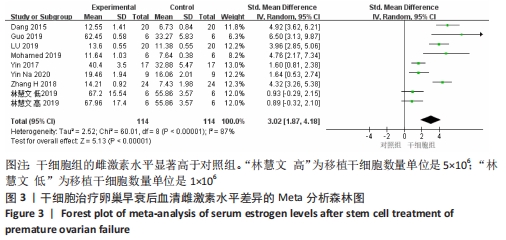
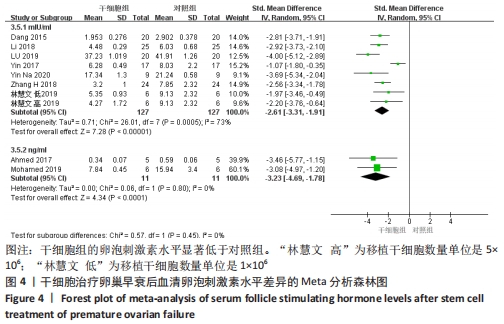
2.3.2 各组血清卵泡刺激素水平差异 共9篇文献报道了诱导小鼠卵巢早衰后干细胞对卵泡刺激素修复能力的影响[13,16-21,23-24]。因其中1篇研究包括脐带间充质干细胞高浓度和脐带间充质干细胞低浓度的2个剂量水平的研究[24],因此对卵泡刺激素的分析包括了10个研究,共纳入干细胞组138只小鼠,对照组138小鼠。基于单位mIU/mL亚组的各研究结果之间存在异质性(I2 > 50%),因此采用随机效应模型进行Meta分析。结果提示,干细胞组的卵泡刺激素水平显著低于对照组(SMD=-2.61,95%CI:-3.31至-1.91,P < 0.000 01)。单位ng/mL亚组各研究结果不存在异质性(I2 < 50%),结果提示,干细胞组的卵泡刺激素水平同样显著低于对照组(SMD=-3.23,95%CI:-4.69至-1.78,P < 0.00 01),见图4。"
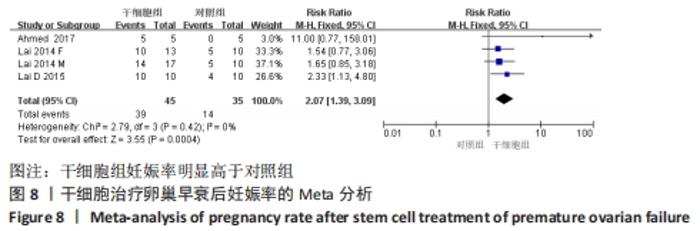
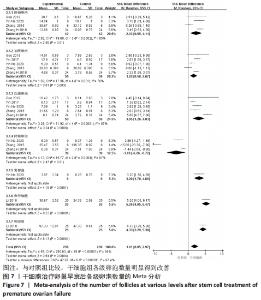
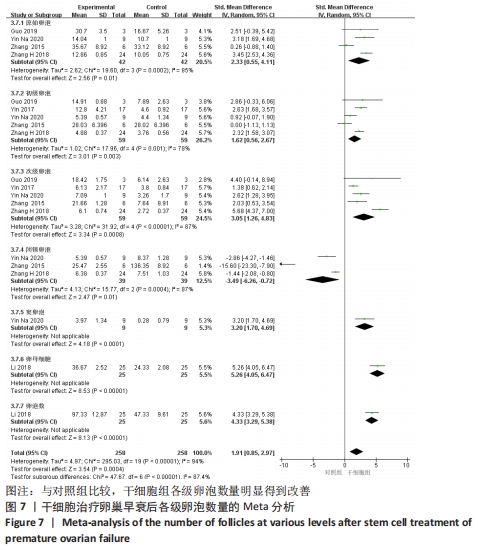
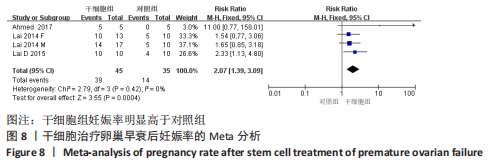
初级卵泡:共纳入5篇文献报道干细胞治疗对小鼠卵巢早衰初级卵泡的影响[12,16,20-22]。各研究间有存在异质性(I2 > 50%)。采用随机效应模型,结果提示两组比较差异有显著性意义(SMD=1.62,95%CI:0.56-2.67,P=0.003),见图7。 次级卵泡:共纳入5篇文献报道干细胞治疗对小鼠卵巢早衰次级卵泡的影响[12,16,20-22]。干细胞组59只小鼠,对照组59只小鼠。各组研究间存在异质性(I2 > 50%),采用随机效应模型。结果提示两组比较差异有显著性意义(SMD=3.05,95%CI:1.26-4.83,P= 0.000 8),见图7。 闭锁卵泡:共纳入3篇文献报道干细胞治疗对小鼠卵巢早衰闭锁卵泡的影响[12,20-21]。各研究间存在异质性(I2 > 50%),采用随机效应模型,结果提示两组比较差异有显著性意义(SMD=-3.49,95%CI:-6.26至-0.72,P=0.01),见图7。 窦卵泡:纳入1篇文献报道干细胞治疗对小鼠卵巢早衰对窦卵泡的影响[20]。因单篇研究无异质性,结果提示两组比较差异有显著性意义(SMD=3.20,95%CI:1.70-4.69,P < 0.000 1),见图7。 卵母细胞:纳入1篇研究报道干细胞治疗对小鼠卵巢早衰卵母细胞的影响[18]。干细胞组25只小鼠,对照组25只小鼠。因单篇研究无异质性,结果提示两组比较差异有显著性意义(SMD=5.26,95%CI:4.05-6.47,P < 0.000 01),见图7。 卵泡数:纳入1篇文献报道干细胞治疗对小鼠卵巢早衰对卵泡数量的影响[18]。因单篇研究无异质性,结果提示两组比较差异有显著性意义(SMD=4.33,95%CI:3.29-5.38,P < 0.000 01),见图7。 2.3.6 各组妊娠率差异 共纳入3篇研究干细胞治疗对小鼠卵巢早衰妊娠率的影响[14-15,17]。干细胞组小鼠45只,对照小鼠35只。其中1篇研究包括雌性皮肤间充质干细胞移植和雄性皮肤间充质干细胞2个干细胞类型的研究[14],因此对妊娠率的分析包括了4个研究。各研究间不存在异质性(I2=0%),选择固定效应模型。两组差异有显著性意义(RR=2.07,95%CI:1.39-3.09,P=0.000 4),见图8。"
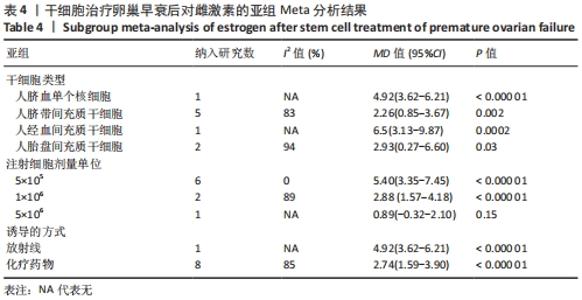
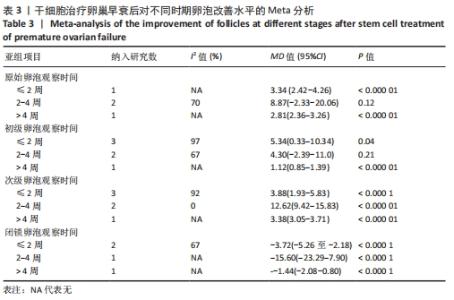
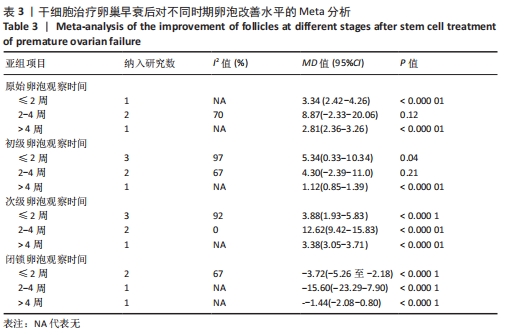
初级卵泡:基于不同时间段,与对照组相比,干细胞组移植后≤ 2周(MD=5.34,95%CI:0.33-10.34,P=0.04)与移植后> 4周(MD=1.12,95%CI:0.85-1.39,P < 0.000 01)均高于对照组。而移植后2-4周干细胞组与对照组比较差异无显著性意义(MD=4.30,95%CI:-2.39-11.0,P=0.21),见表3。 次级卵泡:基于不同时间段,与对照组相比,干细胞组移植后≤2周(MD=3.88,95%CI:1.93-5.83,P < 0.001)、移植后2-4周(MD=12.62,95%CI:9.42-15.83,P < 0.000 01)、移植后> 4周(MD=3.38,95%CI:3.05-3.71,P < 0.000 01)均高于对照组,见表3。 闭锁卵泡:基于不同时间段,与对照组相比,在移植后≤ 2周(MD=-3.72,95%CI:-5.26至-2.18,P < 0.000 1)、移植后2-4周(MD=-15,95%CI:-23.29至-7.90,P < 0.000 1)、移植后> 4周(MD=-1.44,95%CI:-2.08至-0.80,P < 0.000 1)均高于对照组,见表3。 2.3.8 不同细胞来源、细胞剂量、动物模型建立方式的亚组Meta分析结果 干细胞类型:基于不同来源的干细胞,与对照组相比,干细胞组人脐血单个核细胞组的雌激素水平明显升高(MD=4.92,95%CI:3.62-6.21,P < 0.000 01)、 干细胞组人脐带间充质干细胞组的雌激素水平明显升高(MD=2.26,95%CI:0.85-3.67,P=0.002)、干细胞组人经血间充质干细胞组的雌激素水平明显升高(MD=6.5,95%CI:3.13-9.87,P=0.000 2)、干细胞组人胎盘间充质干细胞组的雌激素水平明显升高(MD=2.93,95%CI:0.27-6.60,P=0.03)均高于对照组,见表4。"
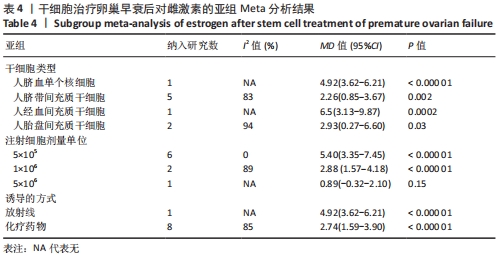
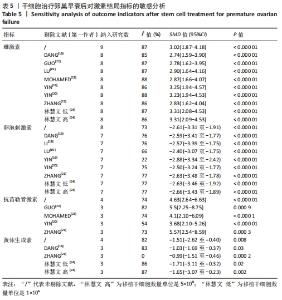
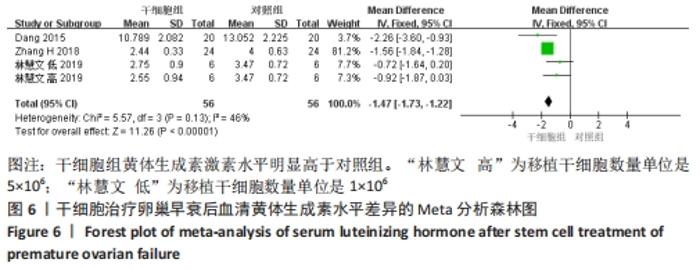
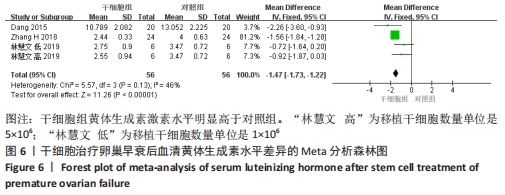
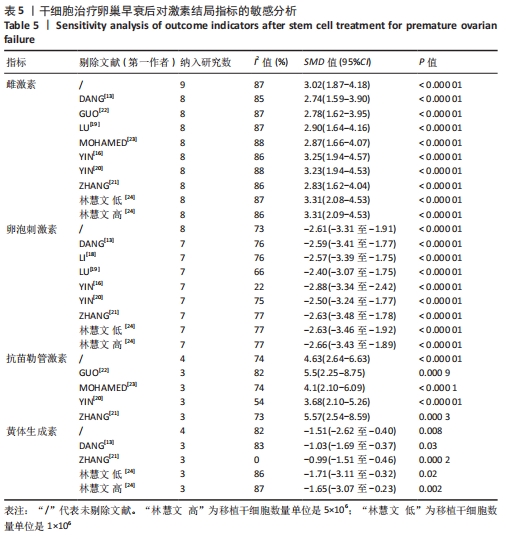
注射细胞剂量:基于不同注射细胞剂量,与对照组相比,干细胞组注射剂量5×105 (MD=5.40,95%CI:3.35-7.45,P < 0.000 01)与注射剂量1×106 (MD=2.88,95%CI:1.57-4.18,P < 0.000 01)均高于对照组。而注射剂量5×106干细胞组与对照组比较差异无显著性意义(MD=0.89,95%CI:-0.32-2.10,P=0.15),见表4。 诱导方式:基于不同诱导方式,与对照组相比,干细胞组放射线(MD=4.92,95%CI:3.62-6.21,P < 0.000 01)与化疗药物(MD=2.74,95%CI:1.59-3.90,P < 0.000 01)均高于对照组,见表4。 2.4 敏感性分析结果 通过逐篇剔除每项研究后,对4个结局指标血清雌激素、卵泡刺激素、抗苗勒管激素和黄体生成素水平分别进行敏感性分析显示,在雌激素结局指标中逐篇剔除每项研究,较总体异质性没有明显降低,结果稳定;在卵泡刺激素结局指标中,剔除YIN 等[16]的研究后异质性降低(I2=22%,P < 0.000 01);在抗苗勒管激素结局指标中,剔除MOHAMED等[23]的研究后异质性降低(I2=54%,P < 0.000 01);在黄体生成素结局指标中,剔除ZHANG等 [21]的研究后异质性降低(I2=0%,P < 0.000 2),见表5;据以上分析现在异质性来源不确切,不同的干细胞类型、放化疗药物、放化疗时间、取血清的时机等都可能是异质性来源,需要更大的样本量加以证实。"
| [1] TUCKER EJ, GROVER SR, BACHELOT A, et al. Premature ovarian insufficiency: new perspectives on genetic cause and phenotypic spectrum. Endocr Rev. 2016;37(6):609-635. [2] PODFIGURNA-STOPA A, CZYZYK A, GRYMOWICZ M, et al. Premature ovarian insufficiency: the context of long-term effects. J Endocrinol Invest. 2016;39(9):983-990. [3] ZHANG C. The roles of different stem cells in premature ovarian failure. Curr Stem Cell Res Ther. 2020;15(6):473-481. [4] NELSON LM. Clinical practice. Primary ovarian insufficiency. N Engl J Med. 2009;360(6):606-614. [5] VABRE P, GATIMEL N, MOREAU J, et al. Environmental pollutants, a possible etiology for premature ovarian insufficiency: a narrative review of animal and human data. Environ Health. 2017;16(1):37. [6] 刘荣霞,杨炳,余丽梅,等.不同来源间充质干细胞治疗卵巢早衰的作用及机制研究进展[J].山东医药,2018,58(40):106-110. [7] 梁颖施,陈烨.卵巢功能早衰致不孕症的治疗现状及进展新研究[J].中医临床研究,2018,10(12):90-91. [8] NAJI A, EITOKU M, FAVIER B, et al. Biological functions of mesenchymal stem cells and clinical implications. Cell Mol Life Sci. 2019;76(17):3323-3348. [9] CHAVAKIS E, URBICH C, DIMMELER S. Homing and engraftment of progenitor cells: a prerequisite for cell therapy. J Mol Cell Cardiol. 2008;45(4):514-522. [10] HOOIJMANS CR, ROVERS MM, DE VRIES RB, et al. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14:43. [11] 陈茜.针灸联合补肾活血中药干预卵巢储备功能低下的Meta分析[D].武汉:湖北中医药大学,2019. [12] ZHANG Q, XU M, YAO X, et al. Human amniotic epithelial cells inhibit granulosa cell apoptosis induced by chemotherapy and restore the fertility. Stem Cell Res Ther. 2015;6(1):152. [13] DANG J, JIN Z, LIU X, et al. Human cord blood mononuclear cell transplantation for the treatment of premature ovarian failure in nude mice. Int J Clin Exp Med. 2015;8(3):4122-4127. [14] LAI D, WANG F, DONG Z, et al. Skin-derived mesenchymal stem cells help restore function to ovaries in a premature ovarian failure mouse model. PLoS One. 2014;9(5):e98749. [15] LAI D, WANG F, YAO X, et al. Human endometrial mesenchymal stem cells restore ovarian function through improving the renewal of germline stem cells in a mouse model of premature ovarian failure. J Transl Med. 2015;13:155. [16] YIN N, ZHAO W, LUO Q, et al. Restoring ovarian function with human placenta-derived mesenchymal stem cells in autoimmune-induced premature ovarian failure mice mediated by treg cells and associated cytokines. Rep Sci. 2018;25(7):1073-1082. [17] BADAWY A, SOBH MA, AHDY M, et al. Bone marrow mesenchymal stem cell repair of cyclophosphamide-induced ovarian insufficiency in a mouse model. Int J Womens Health. 2017;9:441-447. [18] LI J, YU Q, HUANG H, et al. Human chorionic plate-derived mesenchymal stem cells transplantation restores ovarian function in a chemotherapy-induced mouse model of premature ovarian failure. Stem Cell Res Ther. 2018;9(1):81. [19] LU X, CUI J, CUI L, et al. The effects of human umbilical cord-derived mesenchymal stem cell transplantation on endometrial receptivity are associated with Th1/Th2 balance change and uNK cell expression of uterine in autoimmune premature ovarian failure mice. Stem Cell Res Ther. 2019;10(1):214. [20] YIN N, WU C, QIU J, et al. Protective properties of heme oxygenase-1 expressed in umbilical cord mesenchymal stem cells help restore the ovarian function of premature ovarian failure mice through activating the JNK/Bcl-2 signal pathway-regulated autophagy and upregulating the circulating of CD8+CD28- T cells. Stem Cell Res Ther. 2020;11(1):49. [21] ZHANG H, LUO Q, LU X, et al. Effects of hPMSCs on granulosa cell apoptosis and AMH expression and their role in the restoration of ovary function in premature ovarian failure mice. Stem Cell Res Ther. 2018;9(1):20. [22] GUO F, XIA T, ZHANG Y, et al. Menstrual blood derived mesenchymal stem cells combined with Bushen Tiaochong recipe improved chemotherapy-induced premature ovarian failure in mice by inhibiting GADD45b expression in the cell cycle pathway. Reprod Biol Endocrinol. 2019;17(1):56. [23] MOHAMED SA, SHALABY S, BRAKTA S, et al. Umbilical cord blood mesenchymal stem cells as an infertility treatment for chemotherapy induced premature ovarian insufficiency. Biomedicines. 2019;7(1):7. [24] 林慧文,梁志健,吴哲,等.脐带间充质干细胞治疗雷公藤多苷诱导卵巢早衰模型小鼠的机制研究[J].生物医学工程与临床,2019,23(4):387-393. [25] HOOIJMANS CR, INTHOUT J, RITSKES-HOITINGA M, et al. Meta-analyses of animal studies: an introduction of a valuable instrument to further improve healthcare. ILAR J. 2014;55(3):418-426. [26] AUSTIN PC, MERLO J. Intermediate and advanced topics in multilevel logistic regression analysis. Stat Med. 2017;36(20):3257-3277. [27] HIGGINS JP, THOMPSON SG, DEEKS JJ, et al. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560. [28] 吴洁,陈蓉.早发性卵巢功能不全的激素补充治疗专家共识[J].中华妇产科杂志, 2016,51(12):881-886. [29] SUN L, LI D, SONG K, et al. Exosomes derived from human umbilical cord mesenchymal stem cells protect against cisplatin-induced ovarian granulosa cell stress and apoptosis in vitro. Sci Rep. 2017;7(1):2552. [30] SONG D, ZHONG Y, QIAN C, et al. Human umbilical cord mesenchymal stem cells therapy in cyclophosphamide-induced premature ovarian failure rat model. Biomed Res Int. 2016;2016:2517514. [31] ABD-ALLAH SH, SHALABY SM, PASHA HF, et al. Mechanistic action of mesenchymal stem cell injection in the treatment of chemically induced ovarian failure in rabbits. Cytotherapy. 2013;15(1):64-75. [32] HE Y, CHEN D, YANG L, et al. The therapeutic potential of bone marrow mesenchymal stem cells in premature ovarian failure. Stem Cell Res Ther. 2018;9(1):263. [33] SHEN J, CAO D, SUN JL. Ability of human umbilical cord mesenchymal stem cells to repair chemotherapy-induced premature ovarian failure. World J Stem Cells. 2020;12(4):277-287. [34] SHEIKHANSARI G, AGHEBATI-MALEKI L, NOURI M, et al. Current approaches for the treatment of premature ovarian failure with stem cell therapy. Biomed Pharmacother. 2018;102:254-262. [35] JAMIL Z, FATIMA SS, AHMED K, et al. Anti-mullerian hormone: above and beyond conventional ovarian reserve markers. Dis Markers. 2016;2016:5246217. [36] VOLAREVIC V, GAZDIC M, SIMOVIC MARKOVIC B, et al. Mesenchymal stem cell-derived factors: immuno-modulatory effects and therapeutic potential. Biofactors. 2017;43(5):633-644. [37] DING C, ZOU Q, WANG F, et al. Human amniotic mesenchymal stem cells improve ovarian function in natural aging through secreting hepatocyte growth factor and epidermal growth factor. Stem Cell Res Ther. 2018;9(1):55. [38] WANG S, YU L, SUN M, et al. The therapeutic potential of umbilical cord mesenchymal stem cells in mice premature ovarian failure. Biomed Res Int. 2013;2013:690491. [39] ZHENG Q, FU X, JIANG J, et al. Umbilical cord mesenchymal stem cell transplantation prevents chemotherapy-induced ovarian failure via the NGF/TrkA pathway in rats. Biomed Res Int. 2019;2019:6539294. [40] FU X, HE Y, XIE C, et al. Bone marrow mesenchymal stem cell transplantation improves ovarian function and structure in rats with chemotherapy-induced ovarian damage. Cytotherapy. 2008;10(4):353-363. [41] WANG Z, WEI Q, WANG H, et al. Mesenchymal stem cell therapy using human umbilical cord in a rat model of autoimmune-induced premature ovarian failure. Stem Cells Int. 2020;2020:3249495. [42] LU LL, LIU YJ, YANG SG, et al. Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica. 2006;91(8):1017-1026. |
| [1] | Jing Jinpeng, Zhang Yue, Liu Xiaomin, Liu Yi. Traditional Chinese medicine injection for promoting blood circulation in prevention of deep vein thrombosis after orthopedic surgery: network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1467-1476. |
| [2] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [3] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [4] | Liu Gang, Ma Chao, Wang Le, Zeng Jie, Jiao Yong, Zhao Yi, Ren Jingpei, Hu Chuanyu, Xu Lin, Mu Xiaohong. Ankle-foot orthoses improve motor function of children with cerebral palsy: a Meta-analysis based on 12 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1299-1304. |
| [5] | Wu Min, Zhang Yeting, Wang Lu, Wang Junwei, Jin Yu, Shan Jixin, Bai Bingyi, Yuan Qiongjia. Effect of concurrent training sequences on body composition and hormone response: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1305-1312. |
| [6] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [7] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [8] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [9] | Xiong Tinglin, Ying Menghui, Zhang Lisha, Zhang Xiaogang, Yang Yan. Electrophysiological characteristics of cardiomyocytes differentiated from induced pluripotent stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1063-1067. |
| [10] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| [11] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [12] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [13] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [14] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [15] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||