Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (16): 2618-2624.doi: 10.12307/2022.267
Application and existing problems of polyethylene glycol in the field of neurological reconstruction
Wei Jingjing1, Li Chenyang2, Wang Ping1, Li Yaxuan1, Ding Xiaofang1, Yang Lin1
- 1Zhuhai Campus of Zunyi Medical University, Zhuhai 519041, Guangdong Province, China; 2Nanchang University, Nanchang 330031, Jiangxi Province, China
-
Received:2021-03-23Revised:2021-03-25Accepted:2021-05-19Online:2022-06-08Published:2021-12-23 -
Contact:Yang Lin, PhD, Associate professor, Zhuhai Campus of Zunyi Medical University, Zhuhai 519041, Guangdong Province, China -
About author:Wei Jingjing, Master candidate, Zhuhai Campus of Zunyi Medical University, Zhuhai 519041, Guangdong Province, China Li Chenyang, Nanchang University, Nanchang 330031, Jiangxi Province, China Wei Jingjing and Li Chenyang contributed equally to this article. -
Supported by:National Natural Science Foundation of China, No. 81760416, 81960419, 81927804 (to YL); Ordinary Colleges and Universities Science and Technology Top Talent Support Program of Guizhou Province, No. Qianjiao He KY [2018] 056 (to YL)
CLC Number:
Cite this article
Wei Jingjing, Li Chenyang, Wang Ping, Li Yaxuan, Ding Xiaofang, Yang Lin. Application and existing problems of polyethylene glycol in the field of neurological reconstruction[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2618-2624.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
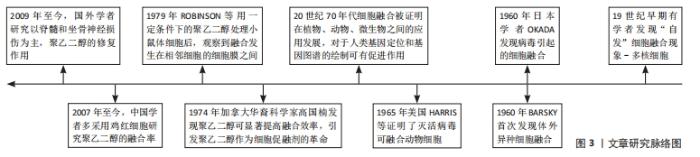
2.1 聚乙二醇的一般性状和促融合作用 2.1.1 一般性状 聚乙二醇是一类可降解、低毒性、味微苦的水溶性聚合物[12],相对分子质量为200-800时在常温下是液体,相对分子质量在800以上者逐渐变为半固体或固体状。低分子量聚乙二醇无色无臭,对人体毒害较大,随着分子质量增大其毒性降低。相对分子质量在4 000以上的聚乙二醇对人体安全,pH值呈中性,生物相容性良好,不易挥发,性质稳定。聚乙二醇应用于医药领域,多作为注射液配制剂、药物缓释剂、细胞融合诱导剂及干细胞移植等发挥作用[13]。 2.1.2 细胞融合 从19世纪开始,人们就发现细胞有自然融合的现象,到1960年Barsky首次发现体外异种细胞融合,Okada发现病毒引起的细胞融合,发展到1974年高国楠(Kao)团队发现聚乙二醇能够使细胞接触点质膜的磷脂分子发生疏散和重组,进而促进细胞融合或原生质体融合,融合效率高且实验成本低等,引起了聚乙二醇作为细胞促融剂的革命,见图3。"

中国研究人员采用相对分子质量为4 000的聚乙二醇,浓度为35%,细胞浓度为2×109 L-1,在35 ℃下反应40 min[14],在此基础上,调整弱碱性环境突出pH值约为7.8时[15],此时该细胞的融合效率可达最高,进一步提高溶液中Ca2+浓度为50 mol/L,Mg2+浓度在10-20 mmol/L,葡萄糖浓度在10-14 mmol/L时对细胞融合效应有一定的促进作用[16-18],其中聚乙二醇自身浓度的影响远大于其他反应条件的改变[19]。国外研究人员ROBINSON等[20]通过电子显微镜下观察到相对分子质量1 000,浓度为50%的聚乙二醇处理2×106个小鼠克隆体细胞后显示细胞最快在一两分钟内发生融合且对细胞骨架成分无明显不良反应,再通过Freeze-fracture技术呈现的图像观察聚乙二醇处理后,膜形态的热致性改变,得到在37 °C条件下,融合15 min膜改变达最大值,小鼠克隆体细胞表面之所以融合,是因为融合发生在伪足相互接触或与相邻细胞体接触的区域形成脑盘层结构进而发生融合。不同类型的细胞间存在差异性,因此促体细胞融合的作用条件不一定适用于神经细胞。神经细胞融合亦受聚乙二醇的浓度及相对分子质量等多因素的影响,若能得到有效促融合条件下的各因素指标,这样既可以提高融合效率又可以减少化学物质的毒副反应,可以逐步优化各项指标应用于前期临床[21]。 2.2.2 神经损伤和聚乙二醇融合修复 无论是从周围器官中接受到的信息还是高级控制中心发出的指令,都是依靠一系列电化学改变组成的快速神经冲动实现传播的。神经系统的功能单元被称作为神经元,神经元是一种由细胞核、含有细胞器的胞浆以及可以区分为轴突和树突的丝状突起组成的细胞[22]。轴突是从胞体或树突延伸分化而来,形成神经纤维,其作用是通过突触连结将神经冲动传导到其他神经元。周围神经中的神经纤维因创伤或其他原因与神经细胞的胞体离断后,其结构发生崩解和破坏,胞体肿胀,细胞核移向胞体一侧,尼氏体发生溶解消失,或固缩变形,此过程即为神经损伤,易引起神经冲动传导障碍,甚至神经元死亡。 在动物和人类的中枢神经损伤,即创伤性脑和脊髓损伤中,主要与轴突的压迫有关,并非脊髓的急性横断性损伤,压迫产生的损伤与长时间的轴突变性和功能缺失有关,创伤性脑损伤和脊髓损伤过程中,神经细胞的细胞膜损伤引起的一系列变化是导致功能丧失和残疾的重要原因,而膜破坏和活性氧的产生是导致神经元功能丧失、进行性退化和死亡的重要因素。有研究通过检测最具代表性乳酸脱氢酶,这种酶泄漏到细胞外空间,表明膜破坏,含量的大小表示细胞膜损伤程度[23]。其次由于神经元体细胞大小适中,通常有很长的轴突,而大多数中枢神经系统或外周神经系统的损伤均涉及轴突的切断或挤压损伤,导致完整的电信号和神经冲动无法传导,致使相应的靶器官和靶细胞无法进行正常生理活动,即在人类和动物整体性上表现为明显的运动和感觉障碍。 近年来聚乙二醇-融合特性被用于神经修复,可显著加速和增强神经细胞膜重封过程,通过恢复膜的完整性来实现断裂神经的有效再连接[21,25-27,29-30],见表2。LUO等[24]实验发现聚乙二醇在急性脊髓损伤中有修复神经元膜和抑制自由基产生的作用,将离体的脊髓在受压损伤后立即暴露于聚乙二醇3 min时,与损伤后未进行聚乙二醇治疗的对照组相比,实验组对相关分子的通透性降低,说明聚乙二醇对受损的细胞膜具有一定的修复作用,通过有效修复膜的完整性,从而减少机械性损伤引起的继发性损伤即减少活性氧的产生和对胞膜的损伤。BORGENS等[25]的实验证明将聚乙二醇应用于标准化的脊髓受压模型中,可以很快恢复神经冲动的传导。以修复轴突的连续性为切入点,有研究以大鼠坐骨神经挤压伤为模型[26-28],通过轴突内荧光染料扩散和动作电位传导,分别评估聚乙二醇融合的轴突其形态连续性和电生理连续性,再通过足故障不对称试验和足迹分析(坐骨神经指标)评估其短期和长期的大鼠下肢行为恢复情况,该实验证明了聚乙二醇不仅能有效修复轴突的切断和挤压损伤,还可以快速重建其丧失的运动行为。BAMBA等[29]用大鼠损伤模型通过设立3组不同时间段的聚乙二醇治疗模型,对比发现,聚乙二醇不仅可以迅速恢复轴突的连续性,对延迟修复的神经也有效。除此之外,GHERGHEREHCHI等[30]用实验证明了无盐的含Ca2+溶液会更有效地增加聚乙二醇的促融合效应,有效阻止或延缓远端神经细胞的瓦勒氏变性。由于细胞融合存在合适的作用条件,主要的影响条件为聚乙二醇自身的变化,常选用的相对分子质量为1 000,4 000,6 000,浓度为40%-75%的聚乙二醇进行诱导,效果良好、毒性较弱。其他如温度、pH值、时间、Ca2+、Mg2+及葡萄糖浓度等次要影响条件,可借鉴聚乙二醇促进体细胞融合时的浓度范围,进行分组实验来探索神经细胞适合的作用条件。此外RILEY等[31]研究发现,采用新型轴突融合装置,研制一种可控聚乙二醇递送量的装置,使得预期结果标准化,并且与对照组相比,聚乙二醇组最多可恢复50%实验动物下肢肢体运动行为的功能。"

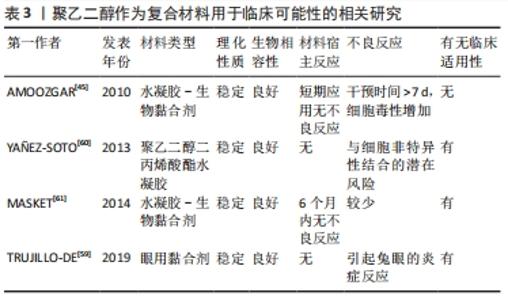
2.3 聚乙二醇介导神经再生机制的探索 受损的神经纤维会出现结构和功能的恢复过程,这一修复过程称为再生。再生的过程首先表现为胞体的尼氏体逐渐恢复正常形态,胞核回到胞体中央,继而与胞体相连的神经纤维的轴突向远侧段生出多条幼芽。这些幼芽穿过损伤处的组织间隙,沿着存活的许旺细胞索向远侧段生长,最后到达原来所分布的组织器官,周围神经的再生能力较强,但其再生速度也非常缓慢[32]。有研究揭示了神经系统是单个组成的神经元,它们通过细胞连接传递信息,与其他类型的细胞类似,具有通过促细胞膜融合,来达到神经细胞融合的潜力[33]。HOFFMAN等[34]提出用流式细胞术评估聚乙二醇促神经母细胞瘤细胞的融合效率,其结果提供了细胞水平上聚乙二醇融合的证据。 脊髓损伤后轴突生长的主要障碍包括损伤部位周围瘢痕的形成[35]、中枢神经系统髓鞘和成年哺乳动物中枢神经元生长能力的减弱[36],RODEMER等[37]提出轴突再密封性对脊髓损伤后神经元再生的作用。在七鳗鱼类中,神经元对轴突损伤的反应是由于神经元内在因素和环境因素之间复杂的相互作用所致,远距离的轴突切开术不太可能导致神经元死亡,由于脊髓损伤引起的轴突切开术导致脑内脊髓投射神经元的凋亡延迟,这种逆行凋亡与轴突的重新密封延迟有关,可以通过用聚乙二醇诱导快速的膜重新密封来加强凋亡延迟,因此识别神经元对轴突切开反应的潜在机制将潜在揭示新的治疗靶点,以增强脊髓损伤患者的再生和功能恢复。在分子水平上的机制,膜修复过程取决于细胞内级联反应,该过程涉及钙蛋白酶的局部活化,cAMP水平的增加和胞质氧化,最终导致囊泡回收蛋白,N-乙基马来酰亚胺敏感因子活化等。此外LIN等[38]提出迅速修复细胞骨架,是神经再生的关键因素,用微管切割蛋白增强细胞骨架重建,联合使用聚乙二醇,以提高轴突融合效率。实验数据显示聚乙二醇和微管切割蛋白的组合促进神经再生的效果优于单独使用聚乙二醇。脊髓损伤后持续功能障碍的主要原因是中枢神经系统功能连接的中断。因此,一个主要目标是促进轴突再生和可塑性。但目前有关聚乙二醇促进神经细胞修复的研究仍处于探索阶段,只能定性地认为其可通过加快细胞膜融合,有利于恢复轴突形态的完整性和功能重建,从而加速神经纤维的再生。 有研究采用神经母细胞瘤细胞[39],研究聚乙二醇介导立体神经细胞融合的优化方案发现:①用于体外神经细胞的融合研究聚乙二醇最佳浓度为70%;②加入30 μmol/L的亮蓝可以保护细胞免受聚乙二醇的破坏作用,而不会降低体外融合率;③少量腺苷5’-三磷酸(bzATP)激活嘌呤能(P2X7)受体可能会改善细胞融合,亮蓝和bzATP可作用于P2X7受体[40-41],激活离子型ATP门控受体,形成半通道蛋白(Pannexin)孔[42]。这种激活对切断的轴突融合具有重要意义,因为其涉及机体的炎症反应,并且受损的轴突充斥着钙,这会引起瓦勒变性[43]。亮蓝抑制P2X7受体,而bzATP激活此受体[44],低剂量5 μmol/L bzATP对聚乙二醇细胞融合有增强作用而不影响融合细胞的生存能力,具有促进神经细胞融合的作用。聚乙二醇促进神经纤维的修复研究目前处于探索阶段[45,61-63],见表3。"
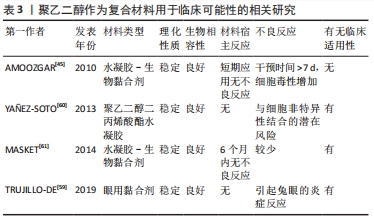

AMOOZGAR等[45]使用可光交联的4-叠氮基苯甲酸改性的壳聚糖(Az-C)和聚乙二醇的混合物作为新型生物黏合剂,用于吻合和稳定受损神经。一系列体内外实验表明,Az-C/聚乙二醇凝胶与神经组织和细胞相容,且通过测定神经细胞的电生理信号,发现Az-C/聚乙二醇凝胶的应用不会显著影响复合动作电位通过神经的传导,急性毒理学分析也未见明显的电位改变,间接说明含有聚乙二醇凝胶的此复合物可有效修复受损神经,并且能够持续递送聚乙二醇,是通过膜融合保护神经细胞的潜在辅助手段[46]。除此之外,聚乙二醇应用于生物工程组织补充材料,具有低毒性、低排斥性、低反应性和高韧性的特点被广泛应用[47-49]。 在体坐骨神经截断损伤模型研究结果显示,近端和远端神经末端之间在经典轴突生长诱导因子Netrins-1、Slit、Ephrin和Semaphorin等的指导下可形成新的轴突神经元连接损伤神经断端[50],有学者研究EphB信号通过Sox2依赖的许旺细胞分选指导周围神经再生[51],周围神经重新生长回其靶标的独特能力取决于其神经胶质细胞即许旺细胞的再生特性,在损伤部位进行荧光标记发现许旺细胞生长明显早于轴突,许旺细胞索可引导轴突穿过损伤部位,在神经修复中起重要作用[52]。在神经损伤后,通过从其他未受损的神经元发芽来补偿相邻路径的损失,或者在无脊椎动物中通过近端再生轴突可以直接接触其远端片段,重新建立连接并防止其退化,轴突再生增加的潜在机制之一似乎是基于cAMP水平。实际上,用膜通透性cAMP类似物(二丁酰cAMP;db-cAMP)预处理背根神经节神经元,经过预处理的神经元能够发出多个再生轴突分支,从而增加其内在再生长能力[53]。但由于轴突生长缓慢,再生轴突可能以与原轴突相似的细胞或相似的生长途径等方式,与其他邻近的神经元进行功能性连接,在轴突发生瓦勒变性之前进行有效连接,以此缩短生长周期[54]。基于以上原理,若能经实验发现联合使用聚乙二醇,并且有促进轴突生长的作用,则可有效证明聚乙二醇能加快神经断端连接速度,实现瓦勒变性之前轴突的有效连接,进一步证实了聚乙二醇的神经修复作用,进而为解决神经功能重建问题提供更加可靠的理论依据。"

| [1] Fitzharris M, Cripps RA, Lee BB. Estimating the global incidence of traumatic spinal cord injury. Spinal Cord. 2014;52(2):117-122. [2] Karsy M, Watkins R, Jensen MR, et al. Trends and cost analysis of upper extremity nerve Injury using the national (nationwide) inpatient sample. World Neurosurg. 2019;123:e488-e500. [3] Foster CH, Karsy M, Jensen MR, et al. Trends and cost-analysis of lower extremity nerve injury using the national inpatient sample. Neurosurg. 2019;85(2):250-256. [4] Palispis WA, Gupta R. Surgical repair in humans after traumatic nerve injury provides limited functional neural regeneration in adults. Exp Neurol. 2017;290:106-114. [5] PASKAL AM, PASKAL W, PIETRUSKI P, et al. Polyethylene glycol: the future of posttraumatic nerve repair? systemic review. Int J Mol Sci. 2019;20(6): 1478. [6] ABDOU SA, Henderson PW. Fusogens: chemical agents that can rapidly restore function after nerve injury. J Surg Res. 2019;233:36-40. [7] Bittner GD, Sengelaub DR, Ghergherehchi CL. Conundrums and confusions regarding how polyethylene glycol-fusion produces excellent behavioral recovery after peripheral nerve injuries. Neural Regen Res. 2018; 13(1):53-57. [8] Bamba R, Waitayawinyu T, Nookala R, et al. A novel therapy to promote axonal fusion in human digital nerves. J Trauma Acute Care Surg. 2016;81(5 Suppl 2 Proceedings of the 2015 Military Health System Research Symposium):S177-S183. [9] Pasut G, Panisello A, Folch-Puy E, et al. Polyethylene glycols: an effective strategy for limiting liver ischemia reperfusion injury. World J Gastroenterol. 2016;22(28):6501-6508. [10] Mokarizadeh A, Mehrshad A, Mohammadi R. Local polyethylene glycol in combination with chitosan based hybrid nanofiber conduit accelerates transected peripheral nerve regeneration. J Invest Surg. 2016; 29(3):167-174. [11] 陈军,沈华.神经导管内支架修复周围神经缺损的研究应用与进展[J].中国组织工程研究,2017,21(8):1273-1279. [12] 肖立伟,戴富才,李政,等.聚乙二醇:绿色有机合成的新媒介[J].有机化学,2019,39(3):648-660. [13] Hunckler MD, Medina JD, Coronel MM, et al. Linkage groups within thiol-ene photoclickable peg hydrogels control in vivo stability. Adv Healthc Mater. 2019;8(14):e1900371. [14] 李玲,刘欢,贺亚玲,等.聚乙二醇诱导鸡红细胞融合实验方案的优化[J].农垦医学,2020,42(2):164-169. [15] 李秀兰,姜曰水.聚乙二醇诱导细胞融合影响因素探究 [J].菏泽学院学报,2007,29(5):79-82. [16] 仇燕,李朝炜,苗芳. 聚乙二醇诱导鸡红细胞融合条件的优化[J].生物技术,2011,21(3):50-53. [17] 吴绍函,许辰琪,温馨,等.Mg-(2+)浓度对细胞融合效果的影响[J].现代生物医学进展,2013,13(11):2037-2039,2099. [18] 杨明轩,游孟昊,王美文,等.葡萄糖浓度对细胞融合效果的影响[J].现代生物医学进展,2014,14(17):3236-3239. [19] 侯元,霍德胜,温得中. 提高细胞融合率的实验方法探讨[J].实验室研究与探索,2017,36(10):28-30, 95. [20] Robinson JM, Roos DS, Davidson RL, et al. Membrane alterations and other morphological features associated with polyethylene glycol-induced cell fusion. J Cell Sci. 1979;40:63-75. [21] Kwon BK, Roy J, Lee JH, et al. Magnesium chloride in a polyethylene glycol formulation as a neuro-protective therapy for acute spinal cord injury:preclinical refinement and optimization. J Neurotrauma. 2009;26:1379-1393. [22] Gafarov FM. Neural electrical activity and neural network growth. Neural Netw. 2018;101:15-24. [23] Koh JY, Choi DW. Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J Neurosci Methods.1987;20(1):83-90. [24] Luo J, Borgens R, Shi RY. Polyethylene glycol immediately repairs neuronal membranes and inhibits free radical production after acute spinal cord injury. J Neurochem. 2002;83(2):471-480. [25] Borgens RB, Shi R. Immediate recovery from spinal cord injury through molecular repair of nerve membranes with polyethylene glycol. FASEB J. 2000;14(1):27-35. [26] Britt JM, Kane JR, Spaeth CS, et al. Polyethylene glycol rapidly restores axonal integrity and improves the rate of motor behavior recovery after sciatic nerve crush injury. J Neurophysiol. 2010;104(2):695-703. [27] Mikesh M, Ghergherehchi CL, Hastings RL, et al. Polyethylene glycol solutions rapidly restore and maintain axonal continuity, neuromuscular structures, and behaviors lost after sciatic nerve transections in female rats. J Neurosci Res 2018;96(7):1223-1242. [28] Spaeth CS, Robison T, Fan JD, et al. Cellular mechanisms of plasmalemmal sealing and axonal repair by polyethylene glycol and methylene blue. J Neurosci Res. 2012;90(5):955-966. [29] Bamba R, Riley DC, Kim JS, et al. Evaluation of a nerve fusion technique with polyethylene glycol in a delayed setting after nerve injury. J Hand Surg Am. 2018;43(1):82.e81-82.e87. [30] Ghergherehchi CL, Bittner GD, Hastings RL, et al. Effects of extracellular calcium and surgical techniques on restoration of axonal continuity by polyethylene glycol fusion following complete cut or crush severance of rat sciatic nerves. J Neurosci Res 2016;94(3):231-245. [31] Riley DC, Boyer RB, Deister CA, et al. Immediate enhancement of nerve function using a novel axonal fusion device after neurotmesis. Ann Plast Surg. 2017;79(6):590-599. [32] Panagopoulos GN, Megaloikonomos PD, Mavrogenis AF. The present and future for peripheral nerve regeneration. Orthopedics. 2017;40(1):e141-e156. [33] Giordano-Santini R, Linton C, Hilliard MA. Cell-cell fusion in the nervous system:alternative mechanisms of development, injury, and repair. Semin Cell Dev Biol. 2016;60:146-154. [34] Hoffman AN, Bamba R, Pollins AC, et al. Analysis of polyethylene glycol (PEG) fusion in cultured neuroblastoma cells via flow cytometry: techniques & optimization. J Clin Neurosci. 2017;36:125-128. [35] Filous AR, Silver J. “Targeting astrocytes in CNS injury and disease: a translational research approach”. Prog Neurobiol. 2016;144:173-187. [36] HE ZJ, JIN YS. Intrinsic control of axon regeneration. Neuron. 2016;90(3): 437-451. [37] Rodemer W, Selzer ME. Role of axon resealing in retrograde neuronal death and regeneration after spinal cord injury. Neural Regen Res. 2019; 14(3):399-404. [38] Lin YF, Xie Z, Zhou J, et al. Effect of exogenous spastin combined with polyethylene glycol on sciatic nerve injury. Neural Regen Res. 2019;14(7): 1271-1279. [39] Messineo E, Pollins A, Thayer W. Optimization and evaluation of an in vitro model of PEG-mediated fusion of nerve cell bodies. J Clin Neurosci. 2019;63:189-195. [40] Vázquez-Cuevas FG, Martínez-Ramírez AS, Robles-Martínez L, et al. Paracrine stimulation of P2X7 receptor by ATP activates a proliferative pathway in ovarian carcinoma cells. J Cell Biochem. 2014;115(11):1955-1966. [41] Rodriguez-Feo CL, Sexton KW, Boyer RB, et al. Blocking the P2X7 receptor improves outcomes after axonal fusion. J Surg Res. 2013;184(1): 705-713. [42] Iglesias R, Locovei S, Roque A, et al. P2X7 receptor-Pannexin1 complex: pharmacology and signaling. Am J Physiol Cell Physiol. 2008; 295(3):C752-C760. [43] Pannuzzo M, De Jong DH, Raudino A, et al. Simulation of polyethylene glycol and calcium-mediated membrane fusion. J Chem Phys. 2014;140(12):124905. [44] Arbeloa J, Pérez-Samartín A, Gottlieb M, et al. P2X7 receptor blockade prevents ATP excitotoxicity in neurons and reduces brain damage after ischemia. Neurobiol Dis. 2012;45(3):954-961. [45] Amoozgar Z, Rickett T, Park J, et al. Semi-interpenetrating network of polyethylene glycol and photocrosslinkable chitosan as an in-situ-forming nerve adhesive. Acta Biomater. 2012;8(5):1849-1858. [46] Scott R, Marquardt L, Willits RK. Characterization of poly (ethylene glycol) gels with added collagen for neural tissue engineering. J Biomed Mater Res A. 2010;93(3):817-823. [47] Oveissi F, Naficy S, Lee A, et al. Materials and manufacturing perspectives in engineering heart valves:a review. Mater Today Bio. 2020;5:100038. [48] Wang JZ, You ML, Ding ZQ, et al. A review of emerging bone tissue engineering via PEG conjugated biodegradable amphiphilic copolymers. Mater Sci Eng C Mater Biol Appl. 2019;97:1021-1035. [49] Im GI. Application of kartogenin for musculoskeletal regeneration. J Biomed Mater Res A. 2018;106(4):1141-1148. [50] Cattin AL, Burden JJ, Van Emmenis L, et al. Macrophage-induced blood vessels guide schwann cell-mediated regeneration of peripheral nerves. Cell. 2015;162(5):1127-1139. [51] Parrinello S, Napoli I, Ribeiro S, et al. EphB signaling directs peripheral nerve regeneration through Sox2-dependent Schwann cell sorting. Cell. 2010;143(1):145-155. [52] Dadsetan M, Knight AM, Lu L, et al. Stimulation of neurite outgrowth using positively charged hydrogels. Biomaterials. 2009;30(23-24):3874-3881. [53] Hilliard MA. Axonal degeneration and regeneration:a mechanistic tug-of-war. J Neurochem. 2009;108(1):23-32. [54] Tuszynski MH, Steward O. Concepts and methods for the study of axonal regeneration in the CNS. Neuron. 2012;74(5):777-791. [55] Bittner GD, Sengelaub DR, Trevino RC, et al. The curious ability of polyethylene glycol fusion technologies to restore lost behaviors after nerve severance. J Neurosci Res. 2016;94(3):207-230. [56] Vargas SA, Bittner GD. Natural mechanisms and artificial PEG-induced mechanism that repair traumatic damage to the plasmalemma in eukaryotes. Curr Top Membr. 2019;84:129-167. [57] Lu X, Perera TH, Aria AB, et al. Polyethylene glycol in spinal cord injury repair:a critical review. J Exp Pharmacol. 2018;10:37-49. [58] Ghergherehchi CL, Mikesh M, Sengelaub DR, et al. Polyethylene glycol (PEG) and other bioactive solutions with neurorrhaphy for rapid and dramatic repair of peripheral nerve lesions by PEG-fusion. J Neurosci Methods. 2019;314:1-12. [59] Trujillo-de Santiago G, Sharifi R, Yue K, et al. Ocular adhesives: design, chemistry, crosslinking mechanisms, and applications. Biomaterials. 2019;197:345-367. [60] Yañez-Soto B, Liliensiek SJ, Murphy CJ, et al. Biochemically and topographically engineered poly(ethylene glycol) diacrylate hydrogels with biomimetic characteristics as substrates for human corneal epithelial cells. J Biomed Mater Res A. 2013;101(4):1184-1194. [61] Masket S, Hovanesian JA, Levenson J, et al. Hydrogel sealant versus sutures to prevent fluid egress after cataract surgery. J Cataract Refract Surg. 2014;40(12):2057-2066. |
| [1] | Kong Yamin, Yan Juntao, Ma Bingxiang, Li Huawei. Massage vibration intervenes with MyoD expression and proliferation and differentiation of muscle satellite cells in rats with sciatic nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1160-1166. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Zhang Xiaoyun, Li Huanan, Chen Feng, Chai Yuan, Gan Bin, Li Song, Chen Dingpeng. Potential molecular mechanism of Guizhi Shaoyao Zhimu Decoction in the treatment of gouty arthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 245-252. |
| [4] | Yang Xinghua, Zhang Jing, Chen Daiyun, Xiong Shijiang. Selection of conditions for fabricated porous scaffolds in bone tissue engineering by silk fibroin protein [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2545-2550. |
| [5] | Cai Xiaoxuan, Lü Yingnian, Qi Yi. Long-circulating lipsomes: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2613-2617. |
| [6] | Hu Taotao, Chang Shusen, Wei Zairong. Macrophage polarization effectively promotes nerve regeneration after peripheral nerve injury through the M2 phenotypic regulation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2285-2290. |
| [7] | Zheng Youfei, Shi Wentao, Liu Xiaogu, Zhuang Qin, Lü Demin, Zhang Jiayin, Yuan Yiwen, Zhang Zhijian, Xu Xiaofeng. Biocompatibility of plant-derived nerve conduits to nasal ectomesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2040-2044. |
| [8] | Shan Wen, Shi Wei. Expression and function of colony-stimulating factor 1 in differentiation of neural stem cells induced by activated astrocyte conditioned medium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2069-2074. |
| [9] | Sun Jianwei, Yang Xinming, An Xiaogang. Effects of various methods on improving bone marrow mesenchymal stem cell transplantation for spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2075-2080. |
| [10] | Gong Chao, Zhang Yuqiang, Wang Wei. Role and mechanism of cell therapy in repair of peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2114-2119. |
| [11] | Zhang Xuelei, Luo Gan, Yu Shenghui, Gu Zuchao, Peng Xu, He Xueling, Liu Yan, Zhang Xiaomei. Acellular nerve scaffold combined with bone marrow mesenchymal stem cells and platelet-rich gel for femoral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 27-32. |
| [12] | Zhao Laihe, Xia Bing, Ma Teng, Gao Jianbo, Li Shengyou, Gao Xue, Zheng Yi, Hu Guangwen, Luo Zhuojing, Huang Jinghui. Extracellular matrix of Schwann-like cells induced by bone marrow mesenchymal stem cells promotes axonal regeneration after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 33-39. |
| [13] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [14] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [15] | Song Kaikai, Zhang Kai, Jia Long. Microenvironment and repair methods of peripheral nervous system injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 651-656. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

