Design
A retrospective cohort study.
Time and setting
Investigations were conducted in the Military Institute of Organ Transplantation, Changzheng Hospital, China from January 2010 to December 2012.
Subjects
A total of 152 patients who had transplanted for more than one year and progressed to chronic renal allograft dysfunction were recruited for this study. Kidney transplant patients who developed proteinuria and hypertension, with a simultaneous or delayed rise in serum creatinine level over months were evaluated and treated in the outpatient department. These patients met the following criteria were included in this study. Prior written and informed consent was obtained from each patient and the study was approved by the Ethics Committee of Changzheng Hospital (Shanghai, China).
Inclusion criteria[12]: (1) Patients whose renal function deteriorated progressively and serum creatinine increased 25% within six months; (2) Patients whose renal function did not improve through adjusting the immunosuppressive therapy; (3) Ruled out the possibility that could lead to chronic renal damage, such as acute rejection, calcineurin inhibitor nephrotoxicity, urinary obstruction, renal artery stenosis and infection; (4) No acute rejection occurred within six months prior to the study.
Exclusion criteria: (1) patients with hepatitis B virus or hepatitis C virus infection and impaired liver function; (2) patients with reversible factors that can cause renal function deterioration, such as insufficient blood volume, acute stress and poor blood pressure control.
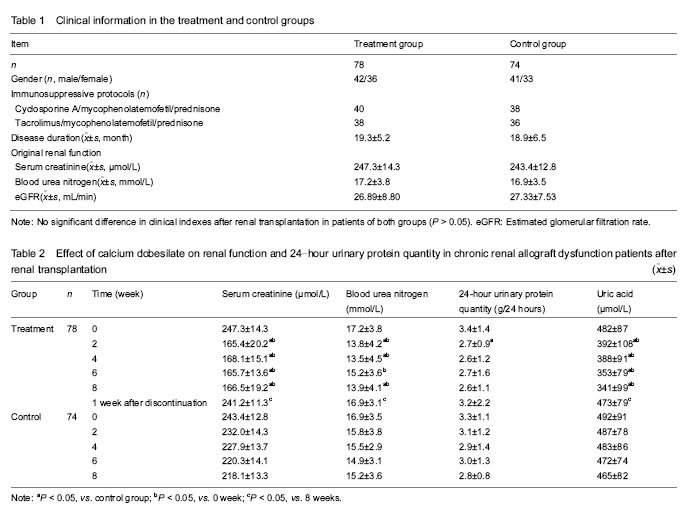
The recruited patients were randomly divided into two groups: a treatment group (n=78), including 42 males and 36 females with an average age of (43.79±13.40) years; and a control group (n=74), including 41 males and 33 females with an average age of (45.41±11.99) years. All patients in both groups were treated with calcineurin inhibitor-based triple immunosuppressive protocols. Cyclosporine A, mycophenolatemofetil and prednisone were used in 40 cases of the treatment group and 38 cases of the control group. Tacrolimus, mycophenolatemofetil and prednisone were used in 38 cases of the treatment group and 36 cases of the control group.
Methods
Treatment with calcium dobesilate after renal transplantation
Comprehensive therapies were applied on all patients, including food restriction (low salt, low fat and high-quality protein diet), homeostasis (maintaining the balance of fluid, electrolytes and acid-base, supplement of iron, folic acid and erythropoietin to improve anemia), blood pressure control and removal of intestinal toxins. Patients in the treatment group received 500 mg of calcium dobesilate (Kangya Pharmaceutical Co., Ltd, Ningxia Province, China) three times daily for eight weeks. None of patients received angiotensin-converting enzyme inhibitor and/or angiotensin receptor blocker drugs.
Follow-up observation
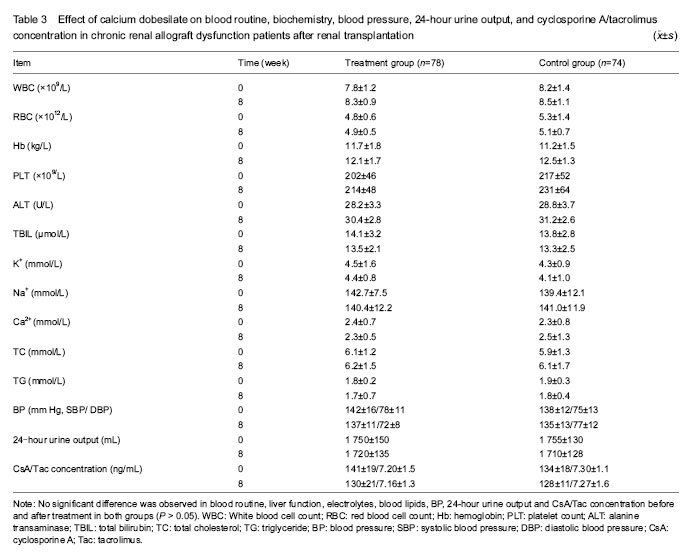
Medical histories of all patients were recorded in details and they were followed up every two weeks by outpatient clinical visits or telephone calls. Blood pressure, 24-hour urine output, blood cell count, liver and kidney function, 24-hour urinary protein quantity, blood lipids, electrolytes and cyclosporine A/tacrolimus concentrations were all measured prior to and following treatment. (1) Every two weeks during the treatment, venous blood was taken between 7:00 and 8:00 a.m. after fasting for 10 hours and the venous blood serum was separated for the detection of alanine aminotransferase, total bilirubin, blood urea nitrogen, serum creatinine, uric acid, total cholesterol, triglyceride, electrolytes and cyclosporine A/tacrolimus concentrations. (2) The morning resting state blood pressure and average 24-hour urine output were recorded before and eight weeks after the treatment. (3) Adverse reactions were determined and treated symptomatically based on symptoms and laboratory tests.
Main outcome measures
(1) Effect of calcium dobesilate on renal function and 24-hour urinary protein quantity in chronic renal allograft dysfunction patients; (2) influence of calcium dobesilate on blood pressure and the other indicators of blood test in chronic renal allograft dysfunction patients; (3) interaction of calcium dobesilate and immunosuppressant; (4) adverse reactions of calcium dobesilate in chronic renal allograft dysfunction patients.
Statistical analysis
Continuous variables were expressed as mean ± SD. Independent sample t-test was performed to evaluate the data between the treatment group and the control group, and one-way repeated measurements analysis of variance was performed to analyze the data before and after calcium dobesilate treatment in each group. P-value of less than 0.05 was considered statistically significant. All statistical calculations were performed with the SPSS 13.0 software.