[1] HUANG W, NAGAO M, YONEMOTO N, et al. Evaluation of the efficacy and safety of romosozumab (evenity) for the treatment of osteoporotic vertebral compression fracture in postmenopausal women: A systematic review and meta-analysis of randomized controlled trials (CDM-J). Pharmacoepidemiol Drug Saf. 2023; 32(6):671-684.
[2] HERNANDEZ AV, PÉREZ-LÓPEZ FR, PISCOYA A, et al. Comparative efficacy of bone anabolic therapies in women with postmenopausal osteoporosis: A systematic review and network meta-analysis of randomized controlled trials. Maturitas. 2019; 129:12-22.
[3] HÄNDEL MN, CARDOSO I, VON BÜLOW C, et al. Fracture risk reduction and safety by osteoporosis treatment compared with placebo or active comparator in postmenopausal women: systematic review, network meta-analysis, and meta-regression analysis of randomised clinical trials. BMJ. 2023;381:e068033.
[4] LEWIECKI EM, BETAH D, HUMBERT L, et al. 3D-modeling from hip DXA shows improved bone structure with romosozumab followed by denosumab or alendronate. J Bone Miner Res. 2024;39(4):473-483.
[5] ZHIVODERNIKOV IV, KIRICHENKO TV, MARKINA YV, et al. Molecular and Cellular Mechanisms of Osteoporosis. Int J Mol Sci. 2023;24(21):15772.
[6] KOBAYAKAWA T, MIYAZAKI A, TAKAHASHI J, et al. Verification of efficacy and safety of ibandronate or denosumab for postmenopausal osteoporosis after 12-month treatment with romosozumab as sequential therapy: The prospective VICTOR study. Bone. 2022;162:116480.
[7] POUTOGLIDOU F, SAMOLADAS E, RAIKOS N, et al. Efficacy and safety of anti-sclerostin antibodies in the treatment of osteoporosis: A meta-analysis and systematic review. J Clin Densitom. 2022; 25(3):401-415.
[8] LEWIECKI EM, BLICHARSKI T, GOEMAERE S, et al. A Phase III Randomized Placebo-Controlled Trial to Evaluate Efficacy and Safety of Romosozumab in Men With Osteoporosis. J Clin Endocrinol Metab. 2018;103(9):3183-3193.
[9] LAU EMC, DINAVAHI R, WOO YC, et al. Romosozumab or alendronate for fracture prevention in East Asian patients: a subanalysis of the phase III, randomized ARCH study. Osteoporos Int. 2020;31(4):677-685.
[10] MCCOLM J, HU L, WOMACK T, et al. Single- and multiple-dose randomized studies of blosozumab, a monoclonal antibody against sclerostin, in healthy postmenopausal women. J Bone Miner Res. 2014;29:935-43.
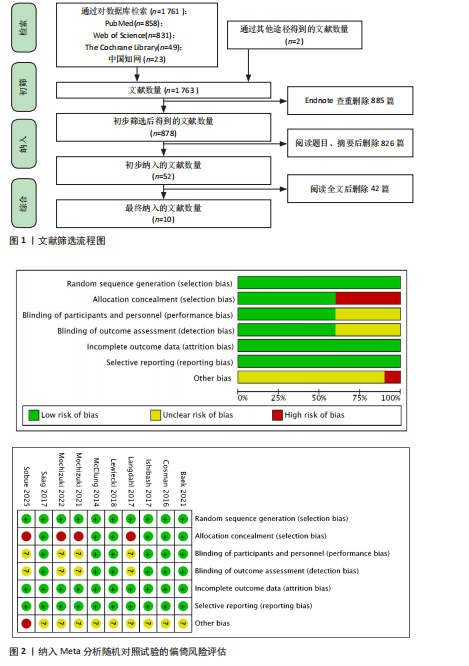
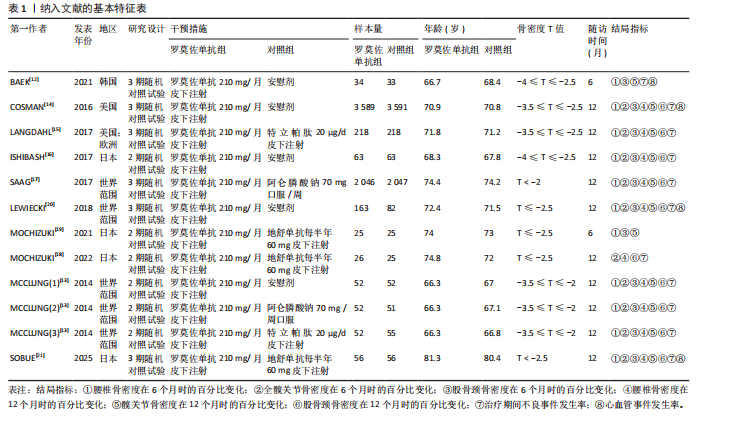
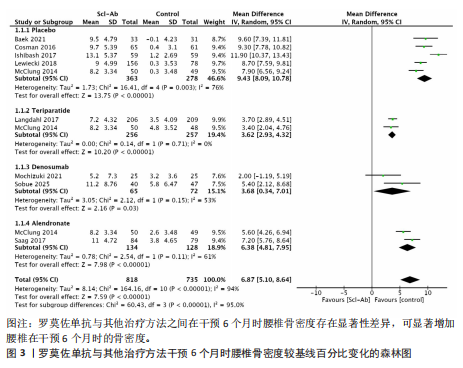
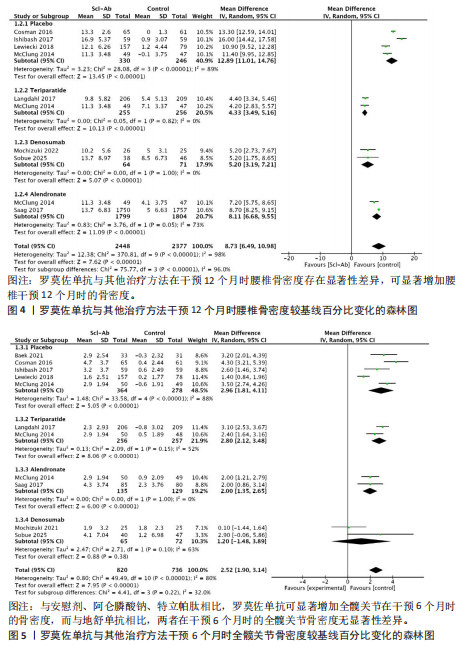
[11] MOHER D, LIBERATI A, TETZLAFF J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010; 8:336-341.
[12] BAEK KH, CHUNG YS, KOH JM, et al. Romosozumab in Postmenopausal Korean Women with Osteoporosis: A Randomized, Double-Blind, Placebo-Controlled Efficacy and Safety Study. Endocrinol Metab (Seoul). 2021;36(1):60-69.
[13] MCCLUNG MR, GRAUER A, BOONEN S, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014;370(5):412-420.
[14] COSMAN F, CRITTENDEN DB, ADACHI JD, et al. Romosozumab Treatment in Postmenopausal Women with Osteoporosis. N Engl J Med. 2016; 375(16):1532-1543.
[15] LANGDAHL BL, LIBANATI C, CRITTENDEN DB, et al. Romosozumab (sclerostin monoclonal antibody) versus teriparatide in postmenopausal women with osteoporosis transitioning from oral bisphosphonate therapy: a randomised, open-label, phase 3 trial. Lancet. 2017;390(10102): 1585-1594.
[16] ISHIBASHI H, CRITTENDEN DB, MIYAUCHI A, et al. Romosozumab increases bone mineral density in postmenopausal Japanese women with osteoporosis: A phase 2 study. Bone. 2017; 103:209-215.
[17] SAAG KG, PETERSEN J, BRANDI ML, et al. Romosozumab or Alendronate for Fracture Prevention in Women with Osteoporosis. N Engl J Med. 2017;377(15):1417-1427.
[18] MOCHIZUKI T, YANO K, IKARI K, et al. Comparison of romosozumab versus denosumab treatment on bone mineral density after 1 year in rheumatoid arthritis patients with severe osteoporosis: A randomized clinical pilot study. Mod Rheumatol. 2023;33(3):490-495.
[19] MOCHIZUKI T, YANO K, IKARI K, et al. Effects of romosozumab or denosumab treatment on the bone mineral density and disease activity for 6 months in patients with rheumatoid arthritis with severe osteoporosis: An open-label, randomized, pilot study. Osteoporos Sarcopenia. 2021;7(3): 110-114.
[20] LEWIECKI EM, BLICHARSKI T, GOEMAERE S, et al. A Phase III Randomized Placebo-Controlled Trial to Evaluate Efficacy and Safety of Romosozumab in Men With Osteoporosis. J Clin Endocrinol Metab. 2018;103(9):3183-3193.
[21] SOBUE Y, KOSUGIYAMA H, ASAI S, et al. A randomized controlled trial comparing romosozumab and denosumab in elderly women with primary osteoporosis and knee osteoarthritis. Sci Rep. 2025;15(1):22441.
[22] RECKNOR CP, RECKER RR, BENSON CT, et al. The Effect of Discontinuing Treatment With Blosozumab: Follow-up Results of a Phase 2 Randomized Clinical Trial in Postmenopausal Women With Low Bone Mineral Density. J Bone Miner Res. 2015;30:1717-1725.
[23] WEN F, DU H, DING L, et al. Clinical efficacy and safety of drug interventions for primary and secondary prevention of osteoporotic fractures in postmenopausal women: Network meta-analysis followed by factor and cluster analysis. PLoS One. 2020;15(6):e0234123.
[24] BHANDARI M, SCHEMITSCH EH, KARACHALIOS T, et al. Romosozumab in Skeletally Mature Adults with a Fresh Unilateral Tibial Diaphyseal Fracture: A Randomized Phase-2 Study. J Bone Joint Surg Am. 2020;102(16):1416-1426.
[25] SCHEMITSCH EH, MICLAU T, KARACHALIOS T, et al. A Randomized, Placebo-Controlled Study of Romosozumab for the Treatment of Hip Fractures. J Bone Joint Surg Am. 2020;102(8):693-702.
[26] PADHI D, ALLISON M, KIVITZ AJ, et al. Multiple doses of sclerostin antibody romosozumab in healthy men and postmenopausal women with low bone mass: a randomized, double-blind, placebo-controlled study. J Clin Pharmacol. 2014; 54(2):168-178.
[27] TAKADA J, DINAVAHI R, MIYAUCHI A, et al. Relationship between P1NP, a biochemical marker of bone turnover, and bone mineral density in patients transitioned from alendronate to romosozumab or teriparatide: a post hoc analysis of the STRUCTURE trial. J Bone Miner Metab. 2020;38(3):310-315.
[28] HU M, ZHANG Y, GUO J, et al. Meta-analysis of the effects of denosumab and romosozumab on bone mineral density and turnover markers in patients with osteoporosis. Front Endocrinol (Lausanne). 2023;14:1188969.
[29] KEAVENY TM, CRITTENDEN DB, BOLOGNESE MA, et al. Greater Gains in Spine and Hip Strength for Romosozumab Compared With Teriparatide in Postmenopausal Women With Low Bone Mass. J Bone Miner Res. 2017;32(9):1956-1962.
[30] GENANT HK, ENGELKE K, BOLOGNESE MA, et al. Effects of Romosozumab Compared With Teriparatide on Bone Density and Mass at the Spine and Hip in Postmenopausal Women With Low Bone Mass. J Bone Miner Res. 2017; 32(1):181-187.
[31] CHENG SH, CHU W, CHOU WH, et al. Cardiovascular Safety of Romosozumab Compared to Commonly Used Anti-osteoporosis Medications in Postmenopausal Osteoporosis: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials. Drug Saf. 2025;48(1):7-23.
[32] GOLLEDGE J, THANIGAIMANI S. Role of Sclerostin in Cardiovascular Disease. Arterioscler Thromb Vasc Biol. 2022;42(7):e187-e202.
[33] BOVIJN J, KREBS K, CHEN CY, et al. Evaluating the cardiovascular safety of sclerostin inhibition using evidence from meta-analysis of clinical trials and human genetics. Sci Transl Med. 2020; 12(549):eaay6570.
[34] MIGLIORINI F, COLAROSSI G, BARONCINI A, et al. Pharmacological Management of Postmenopausal Osteoporosis: a Level I Evidence Based-Expert Opinion. Expert Rev Clin Pharmacol. 2021;14(1): 105-119.
[35] ALTMAN DG, BLAND JM. Absence of evidence is not evidence of absence. BMJ. 1995; 311(7003):485.
[36] SINGH S, DUTTA S, KHASBAGE S, et al. A systematic review and meta-analysis of efficacy and safety of Romosozumab in postmenopausal osteoporosis. Osteoporos Int. 2022;33(1):1-12.
|