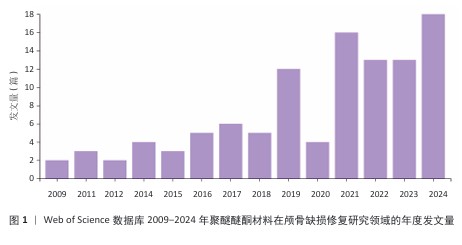
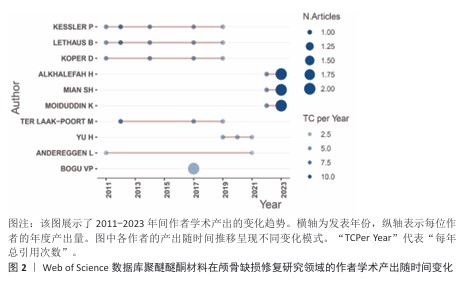
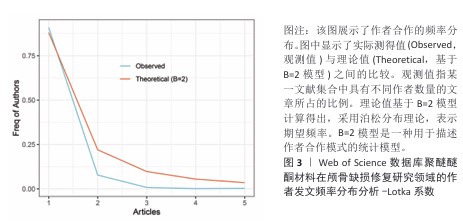
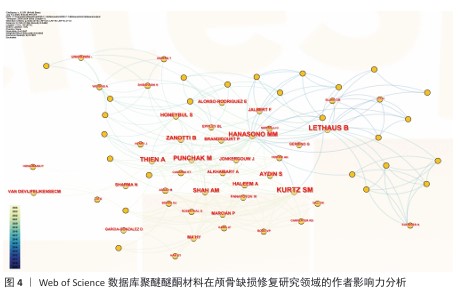
[1] SUN Y, LIU J, CHEN K, et al. Polydopamine grafting polyether ether ketone to stabilize growth factor for efficient osteonecrosis repair. Sci Rep. 2025;15(1):3697.
[2] ZHANG J, SU Y, RAO X, et al. Additively manufactured polyether ether ketone (PEEK) skull implant as an alternative to titanium mesh in cranioplasty. Int J Bioprint. 2022;9(1):634.
[3] CHEN J, CAO G, LI L, et al. Modification of polyether ether ketone for the repairing of bone defects. Biomed Mater. 2022;17(4). doi: 10.1088/1748-605X/ac65cd.
[4] 陈智谦,穆雄铮.颅骨缺损修补材料应用的Meta分析[J].中国组织工程研究,2018,22(30): 4913-4920.
[5] BAI Z, ZHAO Y, ZHANG W, et al. Mussel-inspired bifunctional chimeric peptides macromolecules functionalize 3D-printed porous scaffolds for enhanced antimicrobial and osseointegration properties in bone defect repair. Int J Biol Macromol. 2025;308(Pt 4):142668.
[6] FRASSANITO P, FRASCHETTI F, BIANCHI F, et al. Management and prevention of cranioplasty infections. Childs Nerv Syst. 2019;35(9): 1499-1506.
[7] DENG Y, GAO X, SHI XL, et al. Graphene Oxide and Adiponectin-Functionalized Sulfonated Poly(etheretherketone) with Effective Osteogenicity and Remotely Repeatable Photodisinfection. Chem Mater. 2020;32(5): 2180-2193.
[8] HONIGMANN P, SHARMA N, OKOLO B, et al. Patient-Specific Surgical Implants Made of 3D Printed PEEK: Material, Technology, and Scope of Surgical Application. Biomed Res Int. 2018; 2018:4520636.
[9] LETHAUS B, BLOEBAUM M, KOPER D, et al. Interval cranioplasty with patient-specific implants and autogenous bone grafts--success and cost analysis. J Craniomaxillofac Surg. 2014;42(8):1948-1951.
[10] LETHAUS B, SAFI Y, TER LAAK-POORT M, et al. Cranioplasty with customized titanium and PEEK implants in a mechanical stress model. J Neurotrauma. 2012;29(6):1077-1083.
[11] KOPER D, TER LAAK-POORT M, LETHAUS B, et al. Cranioplasty with patient-specific implants in repeatedly reconstructed cases. J Craniomaxillofac Surg. 2019;47(5):709-714.
[12] LETHAUS B, TER LAAK MP, LAEVEN P, et al. A treatment algorithm for patients with large skull bone defects and first results. J Craniomaxillofac Surg. 2011;39(6):435-440.
[13] ZEGERS T, TER LAAK-POORT M, KOPER D, et al. The therapeutic effect of patient-specific implants in cranioplasty. J Craniomaxillofac Surg. 2017;45(1):82-86.
[14] HUANG GJ, ZHONG S, SUSARLA SM, et al. Craniofacial reconstruction with poly(methyl methacrylate) customized cranial implants. J Craniofac Surg. 2015;26(1):64-70.
[15] ZANOTTI B, ZINGARETTI N, VERLICCHI A, et al. Cranioplasty: Review of Materials. J Craniofac Surg. 2016;27(8):2061-2072.
[16] GARCIA-GONZALEZ D, JAYAMOHAN J, SOTIROPOULOS SN, et al. On the mechanical behaviour of PEEK and HA cranial implants under impact loading. J Mech Behav Biomed Mater. 2017;69:342-354.
[17] YANG X, LIU P, XING H, et al. Skull modulated strategies to intensify tumor treating fields on brain tumor: a finite element study. Biomech Model Mechanobiol. 2022;21(4):1133-1144.
[18] YAO S, ZHANG Q, MAI Y, et al. Outcome and risk factors of complications after cranioplasty with polyetheretherketone and titanium mesh: A single-center retrospective study. Front Neurol. 2022;13:926436.
[19] MIAN SH, MOIDUDDIN K, ELSEUFY SM, et al. Adaptive Mechanism for Designing a Personalized Cranial Implant and Its 3D Printing Using PEEK. Polymers (Basel). 2022;14(6):1266.
[20] MARCIÁN P, NARRA N, BORÁK L, et al. Biomechanical performance of cranial implants with different thicknesses and material properties: A finite element study. Comput Biol Med. 2019; 109:43-52.
[21] VAN DE VIJFEIJKEN SECM, MÜNKER TJAG, SPIJKER R, et al. Autologous Bone Is Inferior to Alloplastic Cranioplasties: Safety of Autograft and Allograft Materials for Cranioplasties, a Systematic Review. World Neurosurg. 2018;117:443-452.e8.
[22] 何征晖,惠纪元,冯军峰,等.创伤性脑损伤去骨瓣术后聚醚醚酮颅骨修补的疗效[J].中华创伤杂志,2022,38(4):340-345.
[23] WANG J, CHI Y, YANG B, et al. The application of biomaterials in osteogenesis: A bibliometric and visualized analysis. Front Bioeng Biotechnol. 2022;10:998257.
[24] SEDLÁKOVÁ V, MOURCOS S, PUPKAITĖ J, et al. Biomaterials for direct cardiac repair-A rapid scoping review 2012-2022. Acta Biomater. 2024; 180:61-81.
[25] ZHOU H, CHEN Y, Yan W, et al. Advances and challenges in biomaterials for tendon and enthesis repair. Bioact Mater. 2025;47:531-545.
[26] MALOO LM, TOSHNIWAL SH, RECHE A, et al. A Sneak Peek Toward Polyaryletherketone (PAEK) Polymer: A Review. Cureus. 2022;14(11):e31042.
[27] SIGNORELLI F, DELLA PEPA GM, MARZIALI G, et al. Bedside Ultrasound for Ventricular Size Monitoring in Patients with PEEK Cranioplasty: A Preliminary Experience of Technical Feasibility in Neurotrauma Setting. Neurocrit Care. 2022;37(3):705-713.
[28] MOZAFFARI K, RANA S, CHOW A, et al. Customized polyetheretherketone (PEEK) implants are associated with similar hospital length of stay compared to autologous bone used in cranioplasty procedures. J Neurol Sci. 2022;434:120169.
[29] SIRACUSA V, MAIMONE G, ANTONELLI V. State-of-Art of Standard and Innovative Materials Used in Cranioplasty. Polymers (Basel). 2021;13(9):1452.
[30] SAUVIGNY T, GIESE H, HÖHNE J, et al. A multicenter cohort study of early complications after cranioplasty: results of the German Cranial Reconstruction Registry. J Neurosurg. 2021; 137(2):591-598.
[31] HENRY J, AMOO M, TAYLOR J, et al. Complications of Cranioplasty in Relation to Material: Systematic Review, Network Meta-Analysis and Meta-Regression. Neurosurgery. 2021;89(3):383-394.
[32] ZHANG R, ZHU M, LIU Y, et al. Hierarchical Micro-/Nanotopographical Cues on Polyether-Ether-Ketone Implants for Enhanced Osteogenesis and Angiogenesis. ACS Appl Mater Interfaces. 2025;17(29):41765-41780.
[33] MA H, HAN H, ZHAO X, et al. Engineering Multifunctional Polyether Ether Ketone Implant: Mechanics-Adaptability, Biominerialization, Immunoregulation, Anti-Infection, Osteointegration, and Osteogenesis. Adv Healthc Mater. 2023;12(12):e2202799.
[34] PÖPPE JP, SPENDEL M, GRIESSENAUER CJ, et al. Point-of-Care 3-Dimensional-Printed Polyetheretherketone Customized Implants for Cranioplastic Surgery of Large Skull Defects. Oper Neurosurg. 2024;27(4):449-454.
[35] QIN W, XING T, QIN S, et al. BMSCs-driven graphite oxide-grafted-carbon fibers reinforced polyetheretherketone composites as functional implants: in vivo biosafety and osteogenesis. J Biomater Sci Polym Ed. 2024;35(9):1343-1358.
[36] QI ML, LI M, YUAN K, et al. Fabrication and X-ray microtomography of sandwich-structured PEEK implants for skull defect repair. Sci Rep. 2024;14(1):28585.
[37] GAO Y, PANG Y, WEI S, et al. Amyloid-Mediated Nanoarchitectonics with Biomimetic Mineralization of Polyetheretherketone for Enhanced Osseointegration. ACS Appl Mater Interfaces. 2023;15(8):10426-10440.
[38] SUN C, KANG J, YANG C, et al. Additive manufactured polyether-ether-ketone implants for orthopaedic applications: a narrative review. Biomater Transl. 2022;3(2):116-133.
[39] SUN C, MENG Z, WANG L, et al. Development and Challenges of Additive Manufactured Customized Implant. Zhongguo Yi Liao Qi Xie Za Zhi. 2024;48(3):237-244.
[40] SHARAF MY, ESKANDER A, AFIFY MA. Novel PEEK Retentive Elements versus Conventional Retentive Elements in Mandibular Overdentures: A Randomized Controlled Trial. Int J Dent. 2022; 2022:6947756.
[41] ZHAO K, WU S, QIAN C, et al. Suitability and Trueness of the Removable Partial Denture Framework Fabricating by Polyether Ether Ketone with CAD-CAM Technology. Polymers (Basel). 2024;16(8):1119.
[42] MOHAMED OS, KARAWIA I, GABER AH, et al. Evaluation of the Shear Peel Bond Strength of the Computer-aided Design/Computer-aided Manufacturing Polyetheretherketone Band for Space Maintainer: An In Vitro Study. Int J Clin Pediatr Dent. 2025;18(1):75-79.
[43] TUSHAR, RANI P, ANANYA, et al. Evaluation of Impact Strength and Flexural Strength of Polyether Ether Ketone vs. Computer-Aided Design/Computer-Aided Manufacturing Polymethyl Methacrylate Denture Base Materials: An In-Vitro Study. Cureus. 2023;15(10):e47929.
[44] LI XX, LIU YS, SUN YC, et al. Evaluation of one-piece polyetheretherketone removable partial denture fabricated by computer-aided design and computer-aided manufacturing. Beijing Da Xue Xue Bao Yi Xue Ban. 2019;51(2):335-339.
[45] MOELLMANN HL, MEHR VN, KARNATZ N, et al. Evaluation of the Fitting Accuracy of CAD/CAM-Manufactured Patient-Specific Implants for the Reconstruction of Cranial Defects-A Retrospective Study. J Clin Med. 2022;11(7):2045.
[46] HAMBÖCK M, HOSMANN A, SEEMANN R, et al. The impact of implant material and patient age on the long-term outcome of secondary cranioplasty following decompressive craniectomy for severe traumatic brain injury. Acta Neurochir (Wien). 2020;162(4):745-753.
[47] WANG YC, WU YC, CHANG CW, et al. An algorithmic approach of reconstruction for cranioplasty failure: A case series. Medicine (Baltimore). 2023;102(8):e33011.
[48] ZHANG S. Research Progress of 3D-Printed Polyetheretherketone in Implantable Medical Devices. Zhongguo Yi Liao Qi Xie Za Zhi. 2024; 48(3):251-256.
[49] PAN Y, LUO W, LIU X, et al. Enhanced antibacterial activity of copper sulfide/polyetheretherketone biocomposites. J Mater Sci Mater Med. 2025; 36(1):57.
[50] AL ALAWI K, AL FURQANI A, AL SHAQSI S, et al. Cranioplasty in Oman: Retrospective review of cases from the National Craniofacial Center 2012-2022. Sultan Qaboos Univ Med J. 2024;24(2): 250-258.
[51] CARBONARO R, GHIRINGHELLI G, AMENDOLA F, et al. Cranioplasty With Hydroxyapatite Implants: A Multidisciplinary Approach of Neurosurgeon and Plastic Surgeons to Improve Surgical Technique and Clinical Outcome. J Craniofac Surg. 2025;36(5):1470-1475.
[52] JACOB CC, PASQUINI L, MITCHELL KS, et al. Cranial Reconstruction for Infiltrative Meningioma Using 68Ga-DOTATATE Positron Emission Tomography/Computed Tomography and Individual Patient Solutions CaseDesigner®: A Case Series. Oper Neurosurg. 2026;30(1):42-49.
[53] JUNEJA M, PODDAR A, KHARBANDA M, et al. SCAI-Net: An AI-driven framework for optimized, fast, and resource-efficient skull implant generation for cranioplasty using CT images. Comput Biol Med. 2025;194:110504.
[54] CARBONARO R, GHIRINGHELLI G, NATALONI A, et al. Long-Term Series of Custom-Bone Hydroxyapatite Cranioplasty: Outcomes and Survival at 15 Years. J Craniofac Surg. 2025;36(4): 1263-1266.
[55] PARATE KP, NARANJE N, VISHNANI R, et al. Polyetheretherketone Material in Dentistry. Cureus. 2023;15(10):e46485.
[56] WEBBER MJ, APPEL EA, MEIJER EW, et al. Supramolecular biomaterials. Nat Mater. 2016; 15(1):13-26.
[57] LUONG JHT. Functional Biomaterials and Biomaterial Composites with Antimicrobial Properties. J Funct Biomater. 2024;15(9):267.
[58] KOH YG, PARK KM, LEE JA, et al. Total knee arthroplasty application of polyetheretherketone and carbon-fiber-reinforced polyetheretherketone: A review. Mater Sci Eng C Mater Biol Appl. 2019;100:70-81.
[59] JARMAN-SMITH M. Evolving uses for implantable PEEK and PEEK based compounds. Med Device Technol. 2008;19(6):12-15.
[60] SANCHEZ CV, KRAG AE, BARNETT S, et al. Polyetheretherketone Implant Cranioplasty for Large Cranial Defects: A Seven-Year Follow-Up. J Craniofac Surg. 2024. doi: 10.1097/SCS.0000000000010064.
[61] 中国医师协会神经外科医师分会,中国国际小儿神经外科医生同盟,儿童去骨瓣减压术后颅骨缺损修补专家共识协作组.儿童去骨瓣减压术后颅骨缺损修补专家共识(2025版)[J].中华医学杂志,2025,105(15):1157-1163.
[62] WANG J, WANG Y, SUN W, et al. Risk Factors and Prevention of Subcutaneous Fluid Collection After Polyetheretherketone Cranioplasty: A Retrospective Study. J Craniofac Surg. 2025. doi: 10.1097/SCS.0000000000011645.
|