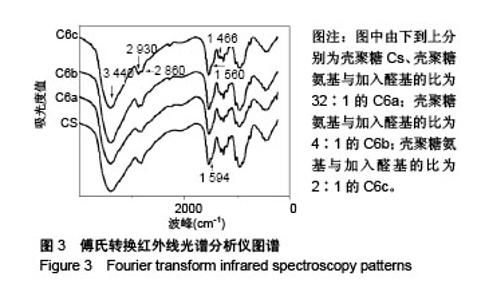
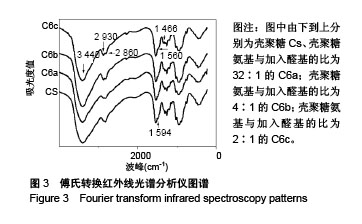
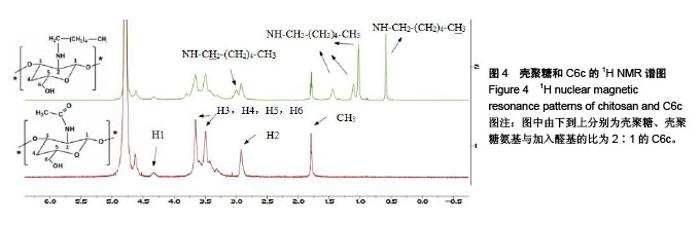
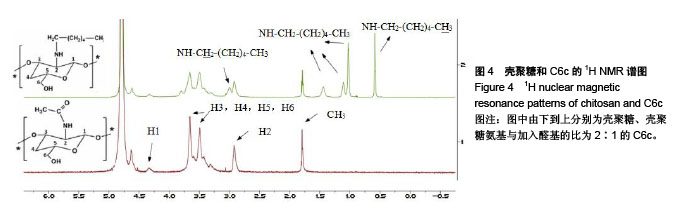
Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (22): 3520-3526.doi: 10.3969/j.issn.2095-4344.0775
Previous Articles Next Articles
Preparation of N-hexane chitosan and its cytotoxicity and coagulation property
Liu Meng-yuan1, Jing Miao-lei1, Guan Jing2, Huang Shu-jie2, Yang Jian2, Li Zhi-hong2
- 1Department of Textile, Tianjin University of Technology, Tianjin 300387, China; 2Institute of Medical Equipment, Academy of Military Medical Sciences, Tianjin 300161, China
-
Received:2018-01-20Online:2018-08-08Published:2018-08-08 -
Contact:Jing Miao-lei, Master, Associate professor, Department of Textile, Tianjin University of Technology, Tianjin 300387, China -
About author:Liu Meng-yuan, Master, Department of Textile, Tianjin University of Technology, Tianjin 300387, China -
Supported by:the National Natural Science Foundation of China, No. 31570956
CLC Number:
Cite this article
Liu Meng-yuan, Jing Miao-lei, Guan Jing, Huang Shu-jie, Yang Jian, Li Zhi-hong. Preparation of N-hexane chitosan and its cytotoxicity and coagulation property[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(22): 3520-3526.
share this article
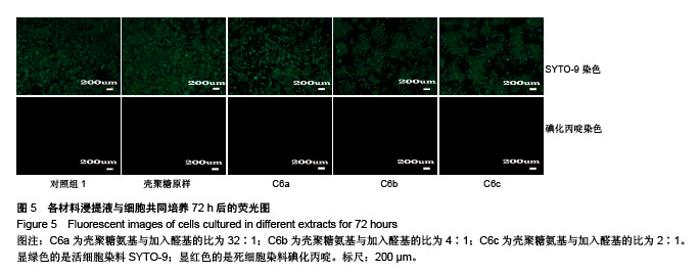
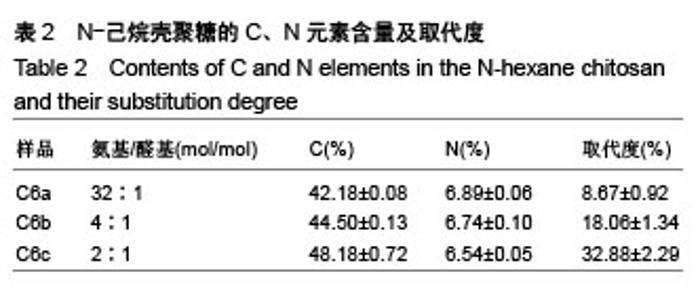
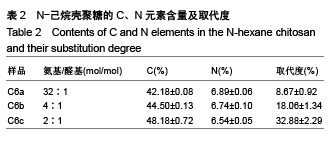
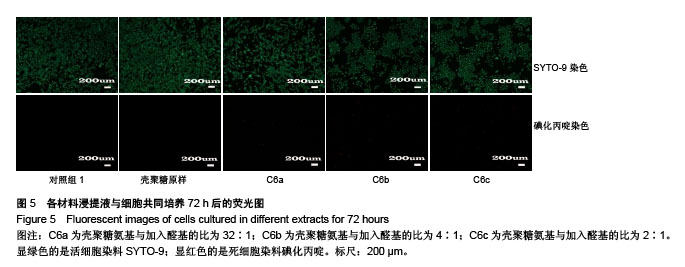
2.4 细胞毒性 2.4.1 细胞增殖率 对照组细胞增殖率为100%,壳聚糖原样细胞增殖率为102.30%,N-己烷壳聚糖(C6a,C6b,C6c)的细胞增殖率分别为99.31%、96.45%、98.18%,说明细胞生长状态良好。研究结果表明,实验中N-己烷壳聚糖的合成方法不会造成有害有毒物质的残留。根据国标细胞毒性分级标准(GB/T 16886.5-2003),细胞增殖率大于100%以及在80%-99%之间的材料,其毒性分级分别为0级和1级,因此可知N-己烷壳聚糖的毒性分级是0级和1级,证实合成的N-己烷壳聚糖是一种安全的生物材料[29]。 2.4.2 荧光染色观察 在荧光显微镜下显绿色的是活细胞染料SYTO-9;显红色的是死细胞染料碘化丙啶。各材料浸提液与细胞共同培养72 h后的荧光照片显示各组绿色荧光丰富,表明细胞生长良好;各组基本没有红色荧光,表明死细胞数量极少。MTT和荧光染色结果均表明各材料几乎无毒性作用,未引起大量活细胞死亡(图5)。"
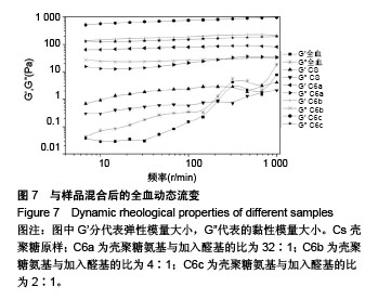
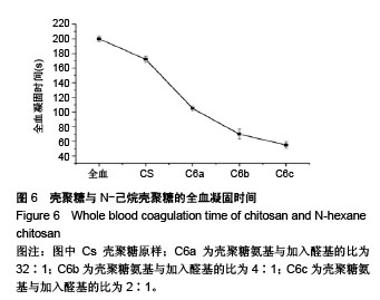
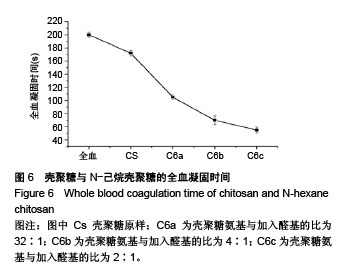
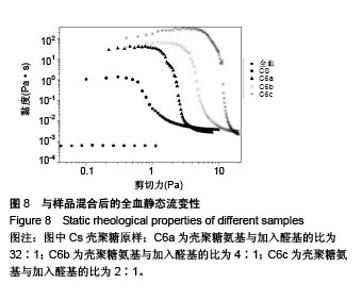
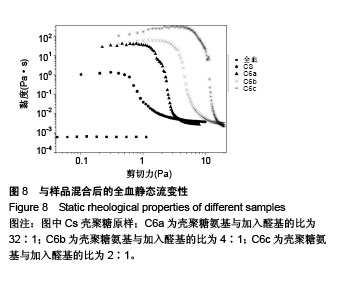
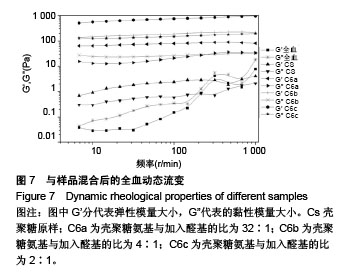
2.6 流变学测试 如图7所示,在动态流变实验中,模量是频率的函数。未加样品的全血显示出一种黏稠状态,在整个检测区域内,它的黏性模量G”均大于弹性模量G’,说明全血未形成凝胶。但相反地,样品与全血混合后呈现一个基本的物理凝胶弹性响应的典型特征:即在整个检测区域内,它的弹性模量G’均大于黏性模量G”,且两者都弱依赖于频率。壳聚糖原样与C6a、C6b、C6c的G’/G”分别为3.07、4.10、4.96、7.34,可知所有N-己烷壳聚糖的G’和G’/G”比值均大于壳聚糖原样,且随着取代度的增大而增大。说明N-己烷壳聚糖止血效果比壳聚糖原样好,且取代度越大,促凝作用越好。 如图8所示,在静态流变实验中,黏度是剪切应力的函数。可以看出全血的黏度恒定,大约是0.001 Pa.s。在低剪切力作用下,壳聚糖/全血的黏度大约为1 Pa.s,N-己烷壳聚糖/全血的黏度在60-100 Pa.s之间,而在剪切力增大到一个数值时黏度急速下降,表明这个应力是凝胶的屈服应力,这代表凝胶很难在低于这个值的压力下流动。壳聚糖原样与C6a、C6b、C6c的屈服应力分别约为0.6 Pa、 1.21 Pa、2.58 Pa、4.83 Pa,说明N-己烷壳聚糖使血液形成的凝胶更为牢固,且取代度越大,屈服应力越大,形成的血栓凝胶越牢固。"
| [1] Schmid BC, Rezniczek GA, Rolf N, et al. Postpartum hemorrhage: use of hemostatic combat gauze. Am J Obstet Gynecol. 2012; 206(1): e12-e13.[2] Eldibany RM. Platelet rich fibrin versus Hemcon dental dressing following dental extraction in patients under anticoagulant therapy. Tant Dent J. 2014;11(2):75-84.[3] Wu Y, He J, Cheng W, et al. Oxidized regenerated cellulose-based hemostat with microscopically gradient structure. Carbohydr Polym.2012;88(3):1023-1032.[4] Gerlach T, Grayson J K, Pichakron K O, et al. Preliminary study of the effects of smectite granules (WoundStat) on vascular repair and wound healing in a swine survival model. J Trauma Acute Care Surg. 2010; 69(5):1203-1209.[5] 张晓菲,刘丽宏,丁春雷,等.印迹壳低聚糖在小鼠体内的代谢与组织分布[J].中国药理学与毒理学杂志,2011,25(3): 314-319.[6] Du T, Chen Z, Li H, et al. Modification of collagen–chitosan matrix by the natural crosslinker alginate dialdehyde. International journal of biological macromolecules. 2016; 82: 580-588.[7] Chen Z, Du T, Tang X, et al. Comparison of the properties of collagen–chitosan scaffolds after γ-ray irradiation and carbodiimide cross-linking. J Biomater Sci Polym Ed.2016; 27(10): 937-953.[8] Fan L, Yang H, Yang J, et al. Preparation and characterization of chitosan/gelatin/PVA hydrogel for wound dressing. Carbohydr Polym. 2016; 146: 427-434.[9] Tranquilan-Aranilla C, Barba BJD, Vista JRM, et al. Hemostatic efficacy evaluation of radiation crosslinked carboxymethyl kappa-carrageenan and chitosan with varying degrees of substitution. Radiat Phys Chem Oxf Engl 1993. 2016;124:124-129.[10] Chan LW, Kim CH, Wang X, et al. PolySTAT-modified chitosan gauzes for improved hemostasis in external hemorrhage. Acta Biomater.2016; 31:178-185.[11] Devlin JJ, Kircher S, Kozen BG, et al. Comparison of ChitoFlex®, CELOXTM, and QuikClot® in control of hemorrhage. J Emerg Med. 2011; 41(3): 237-245. [12] 田浩,吴良平,张玉新.战创伤快速止血材料及其临床应用[J].临床军医杂志,2008,36(6): 989-991.[13] Martins AF, Facchi SP, Follmann HDM, et al. Antimicrobial activity of chitosan derivatives containing N-quaternized moieties in its backbone: a review. Int J Mol Sci. 2014; 15(11): 20800-20832.[14] Kim TH, Jiang HL, Jere D, et al. Chemical modification of chitosan as a gene carrier in vitro and in vivo. Prog Polym Sci. 2007; 32(7): 726-753.[15] Prabaharan M, Mano JF. Chitosan-based particles as controlled drug delivery systems. Adv Drug Deliv Rev. 2005; 12(1): 41-57.[16] 张丽,张娜.壳聚糖及其衍生物在药物递送中的研究进展[J].中国新药与临床杂志.2014,33(1):9-14.[17] 黄玉芬,邹励宏,高洁,等.烷基化壳聚糖的制备及止血效果[J].中国组织工程研究,2016, 20(52):7878-7884.[18] Dowling MB, Kumar R, Keibler MA, et al. A self-assembling hydrophobically modified chitosan capable of reversible hemostatic action. Biomaterials. 2011; 32(13): 3351-3357.[19] Dowling MB, Smith W, Balogh P, et al. Hydrophobically-modified chitosan foam: description and hemostatic efficacy. J Surg Res. 2015; 193(1): 316-323. [20] 司泽兵,吴继功.临床止血材料的应用现状及研究进展[J].生物骨科材料与临床研究, 2015,12(6): 64-67.[21] 黄晓佳,王爱勤,袁光谱.N—烷基壳聚糖衍生物的合成及其对阳离子的吸附性能[D]. 2000.[22] 谢朝锋. N-长烷基壳聚糖季铵盐的合成研究[J].山东化工, 2016, 45(4):23-24.[23] 张高飞,黄国林,严松锦,等.壳聚糖的化学改性研究[J].广东化工, 2011,38(1): 80-81.[24] 郭立民,张谋真,刘勇.羧甲基壳聚糖的制备及应用[J].化学工程师, 2002, 2: 027.[25] Chen Z, Yao X, Liu L, et al. Blood coagulation evaluation of N-alkylated chitosan. Carbohydr Polym. 2017; 173:259.[26] 桑晶,朱社敏,匡荣.MTT比色法与相对增殖度法对输液用包装材料的细胞毒性检测结果的比较[J].中国现代应用药学, 2015, 32(5):571-574.[27] Chen Z, Li F, Liu C, et al. Blood clot initiation by mesoporous silica nanoparticles: dependence on pore size or particle size? J Mater Chem B. 2016; 4(44): 7146-7154.[28] 周秋菊,向俊锋,唐亚林,等.纯位移核磁共振氢谱及其应用[J].波谱学杂志,2016,33(3): 502-513.[29] 朱玲英,郭大伟,顾宁.纳米银细胞毒性体外检测方法研究进展[J].科学通报,2014,22:2145-2152.[30] 蓝广芊.壳聚糖/明胶复合止血材料的研制及其性能研究[D].西南大学, 2016.[31] 肖奇,孙树好,刘洋.OCP黏度指数改进剂的低温流变行为研究[J].润滑油,2013(1):51-59.[32] 尹刚,侯春林.浅析壳聚糖的止血作用及机理[J].生物骨科材料与临床研究, 2009, 6(4):18-19. |
| [1] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [2] | Liu Fang, Shan Zhengming, Tang Yulei, Wu Xiaomin, Tian Weiqun. Effects of hemostasis and promoting wound healing of ozone sustained-release hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3445-3449. |
| [3] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [4] | Chen Siyu, Li Yannan, Xie Liying, Liu Siqi, Fan Yurong, Fang Changxing, Zhang Xin, Quan Jiayu, Zuo Lin. Thermosensitive chitosan-collagen composite hydrogel loaded with basic fibroblast growth factor retards ventricular remodeling after myocardial infarction in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2472-2478. |
| [5] | Liu Feng, Zhang Yu, Wang Yanli, Luo Wei, Han Chaoshan, Li Yangxin. Application of temperature-sensitive chitosan hydrogel encapsulated exosomes in ischemic diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2479-2487. |
| [6] | Li Jie, Xu Jianzhen, Hu Ping, Lei Qiqi, Zhang Wenning, Ao Ningjian . Preparation and performance evaluation of carboxymethyl chitosan/oxidized glucomannan/Panax notoginseng compound sponge dressing for chronic wound [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2528-2534. |
| [7] | Ma Zhen, Liang Da, Wu Xiaolin, Zhong Wei . Safety evaluation of tranexamic acid in reducing perioperative blood loss in high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1847-1852. |
| [8] | Zhang Shuang, Xu Qinghua, Tong Lin, Cao Chen, Ye Hong . Current status and applicaton of absorbable hemostatic materials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1628-1634. |
| [9] | Feng Xiaoxia, Hou Weiwei, Jin Xiaoting, Wang Xinhua. Construction of periodontal biomimetic membrane with electrospun poly(lactic-co-glycolic acid) nanofibers and electrosprayed chitosan microspheres [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 511-516. |
| [10] | Li Li, Ma Li, Li He. Preparation and characterization of magnetic chitosan microspheres [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 577-582. |
| [11] | Liu Haiyan, Hu Yang, Wu Xiuping, Pan Haobo, Jing Xuan. Chitosan-based polysaccharide biomaterial for prevention and treatment of oral diseases [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 631-636. |
| [12] | Zhou Zhi, Xiong Yaqiong. Application of perioperative pulse irrigation combined with tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(36): 5806-5811. |
| [13] | Peng Ruhui, Zhou Dechun. Application of tranexamic acid combined with tourniquet optimization program in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(36): 5812-5817. |
| [14] | Liao Jian, Huang Xiaolin, Huo Hua, Zhou Qian, Cheng Yuting, Qi Yuhan, Wu Chao, Yang Tongjing, Liao Yunmao, Liang Xing. Effects of calcined bone/chitosan composite materials on proliferation and adhesion in osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5447-5453. |
| [15] | Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong. Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5493-5501. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||