Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (7): 1036-1041.doi: 10.3969/j.issn.2095-4344.2015.07.010
Previous Articles Next Articles
Atrial fibrosis and tissue remodeling: the role of transforming growth factor-beta1 and other neurohormonal media
Yuan Xiao-fei, Xu Quan-fu, Li Yi-gang
- Department of Cardiology, Xinhua Hospital of Shanghai Jiao Tong University, Shanghai 200092, China
-
Online:2015-02-12Published:2015-02-12 -
Contact:Li Yi-gang, Professor, Doctoral supervisor, Department of Cardiology, Xinhua Hospital of Shanghai Jiao Tong University, Shanghai 200092, China -
About author:Yuan Xiao-fei, Master, Department of Cardiology, Xinhua Hospital of Shanghai Jiao Tong University, Shanghai 200092, China
CLC Number:
Cite this article
Yuan Xiao-fei, Xu Quan-fu, Li Yi-gang. Atrial fibrosis and tissue remodeling: the role of transforming growth factor-beta1 and other neurohormonal media[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(7): 1036-1041.
share this article
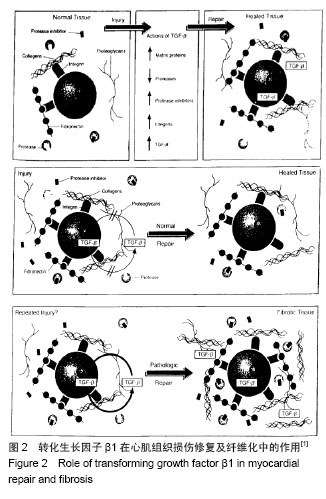
2.1 转化生长因子β1在心房纤维化和结构重构中的作用 研究证明转化生长因子β1在心脏纤维化的发展中起着核心作用,是重要的促纤维化细胞因子之一 [1-2]。转化生长因子β1隶属转化生长因子超家族,由多种细胞分泌的一类具有广泛生物学效应的生长因子,对心血管系统有多种病理生理作用(图2)。 转化生长因子β1发挥其生物学功能需要多种细胞内信号转导和调节分子的协同作用,Smads蛋白是其中的一员。转化生长因子β1具有广泛的生物学作用。依据靶细胞的不同,转化生长因子β1对细胞增殖分化表现出促进或抑制的双重作用[3]。现已明确,转化生长因子β1与细胞外基质沉积的关系最为密切,被公认为器官纤维化的治疗靶点[4]。转化生长因子β1及其受体在心脏的心肌细胞和非心肌细胞中均有表达,心脏中的转化生长因子β1主要由心脏成纤维细胞和肌成纤维细胞产生[3]。 转化生长因子β1在心房纤维化的发生中发挥着重要作用,心房纤维化发生的显著标志是细胞外基质的形成,而这种特定的结构改变是转化生长因子β1的下游介质结缔组织生长因子(connective tissue growth factor,CTGF)介导产生的。转化生长因子β1直接作用于内皮细胞和平滑肌细胞,刺激结缔组织生长因子的表达和分泌,促进细胞外基质的形成[5]。 研究表明,血管紧张素可激活转化生长因子β1的信使核糖核酸转录,诱导蛋白表达[6]。转化生长因子β1激活通过Smad信号通路,刺激胶原蛋白的生成[7]。在快速心室起搏诱导的充血性心力衰竭模型中血管紧张素可以迅速提升激活转化生长因子β1在心房的表达。而血管紧张素Ⅱ1型受体拮抗剂可以抑制转化生长因子β1的表达[8]。组成型活性转化生长因子β1可在心脏心肌组织中优先表达,导致选择性心房纤维化,引起传导异质性,从而诱导房颤发生。有研究表明,在房颤患者中心房纤维化可能是一个双相反应过程,心房纤维化早期转化生长因子β1表达增加,纤维化达到一定程度后转化生长因子β1表达相应减少,这表明纤维化的进展可导致转化生长因子β1信号通路代偿性改变[9]。 已有报道转化生长因子β1血清水平可能与房颤消融术后和电复律后复发相关。研究认为,血浆中高浓度的转化生长因子β1可能是预测非瓣膜性房颤发生的有效指标[10]。纤维化传导阻滞可能与转化生长因子β1基因多态性有关,而这种传导阻滞使房颤易感性增加[11]。也有报道称转化生长因子β1可作为血管紧张素的调节剂[12],但其确切机制尚需进一步研究。 血管紧张素调控转化生长因子β1 mRNA的转录,蛋白的表达,而血管紧张素受体1(AT-1)的阻断剂抑制转化生长因子β1的表达。Kapur等[12]报道,心脏成纤维细胞内转化生长因子β1信号传导需要内分泌激素,通过降低内分泌腺体的活性选择性地抑制转化生长因子β1信号传导,减弱心房纤维化,并且可以提高心脏衰竭小鼠模型的生存率。 2.2 RAAS促纤维化机制 目前普遍认为RAAS在致心律失常和心脏结构重塑中的作用是显而易见的。房颤、高血压性心脏病等心脏疾病可激活RAAS,而RAAS的激活可引起心肌纤维化,导致可促进多种心脏疾病发生的心脏结构重构和电重构的发生。血管紧张素转化酶(ACE)促进血管紧张素Ⅰ(Ang-Ⅰ)转化成血管紧张素Ⅱ(Ang-Ⅱ),激活血管紧张素受体1(AT-1),血管紧张素受体1通过G蛋白介导途径引发磷酸化瀑布链式反应并作用于胞外信号激酶1,2(ERK-1、ERK-2),活化转录因子Elk-1和c-fos作用于细胞丝裂原蛋白活化激酶(MAPKs),刺激成纤维细胞的增殖、肥大与凋亡。 有临床研究表明,RAAS抑制剂可显著降低房颤的发病率和房颤复律后的复发风险[13]。因此,房颤的发生和复发与RAAS密切相关,其中心房肌成纤维细胞,通过激活RAAS起着核心的作用。 在心脏组织,各种纤维化途径通过血管紧张素1型受体被激活[14]。血管紧张素1型受体的刺激促进心房成纤维细胞转化为肌成纤维细胞并合成转化生长因子β1。肌成纤维细胞通过激活转化生长因子β1/Smad信号转导途径促进心房纤维化的发生[15],并引起细胞外基质通过粘着相关蛋白促进心房重塑。在心脏中,转化生长因子β1已被证明优先诱导心房纤维化。此外,RAAS的分子机制亦可通过特定的信号转导途径,促进阵发性心房颤动发展至持续性心房颤动[16]。 2.3 Galectin-3在心房纤维化和结构重构中的作 用 Galectin-3是一种半乳糖苷结合凝集素,在纤维化组织中高度表达。研究表明静脉输注Galectin-3引起心肌纤维化[16],这可能是通过中和抗纤维化肽N-乙酰-丝氨酰-天冬-赖氨酰-脯氨酸(AC-SDKP)复合体引起的。因此,Galectin-3在诱导心肌纤维化中发挥着重要作用。 Galectin-3分为C末端和N末端,C末端是由具有凝集素活性的糖类识别结构域组成,N末端是串联排列的氨基酸重复序列。Galectin-3通过C末端与糖类配体(如N-乙酰氨基乳糖)结合,通过N末端与非糖基化分子配体(如细胞表面受体、细胞外受体)结合来发挥生物学作用,并由非经典分泌途径分泌到细胞外,与细胞表面受体和糖蛋白相互作用,参与跨膜信号转导。 正常心肌低度表达Galectin-3,肥大心肌表达Galectin-3的程度明显升高。Galectin-3促进巨嗜细胞游走、成纤维细胞增生和肥大,在心肌重塑和心力衰竭的病理发展过程中具有重要作用[17-18]。Galectin-3可以调节转化生长因子β1等细胞因子的活性,通过复杂的分子生物学途径,经由磷酸化的刺激促进促纤维化分子的转录和Smad2/3复合物核易位,诱导心房纤维化的形成。心房纤维化导致心房内不均匀性传导,为折返形成创造了条件,促进了房颤的发生[19-20]。 Yu等[21]研究表明通过药物抑制Galectin-3的表达可减轻心肌纤维化及左心室功能障碍,并可抑制随后的心脏衰竭的发展。 Galectin-3可通过细胞因子介导的信号通路促进基因的转录,代表心房结构重构和电重构共同的上游环节,导致心房纤维化并进行性发展至房颤。几种类型的Galectin-3抑制剂已被文献报道可作为分子机制水平上的疾病修饰治疗剂,用以抑制心房重构和延缓房颤的进展[21]。 2.4 炎症在心房纤维化和结构重构中的作用 心房纤维化是心房颤动发生的主要机制之一。炎症促进心房纤维化的主要机制为细胞外基质(如Ⅰ型胶原、Ⅲ型胶原)和纤维连接蛋白的沉积导致心房传导异质性,促进心房结构重构和电重构。此外,基质-暴露细胞因子,黏附分子和生长因子是炎症发生和扩散的主要因子,同时它们在纤维化的发病机制中的作用亦同样重要[22]。 事实上,炎症在心房结构重塑中是一个重要的混杂因素,为房颤的发生提供了物质基础[23-24]。白细胞已被确定在房颤患者心房组织重塑中起着重要作用[23],同时射频消融术后心房组织白细胞浸润的程度可独立预测术后房颤的复发[25-26]。值得注意的是,存储在白细胞中的酶系统如髓过氧化物酶与基质金属蛋白酶2等,是已知的促进组织重塑的酶类,也可预测射频消融术后房颤的复发[27]。 白细胞,特别是中性粒细胞(白细胞中最丰富的子集和先天免疫系统的重要成分)的激活和浸润,取决于其内皮释放的局部细胞因子的激活状态和黏附分子的活性。中性粒细胞的黏附主要依赖于整合素在白细胞膜外的表达:中性粒细胞表达整合素CD11b/CD18(MAC1)可结合黏附分子如细胞间黏附分子1(ICAM-1),结合后可激活内皮细胞表面,来发挥中性粒细胞的黏附作用[28]。值得注意的是,CD11b/CD18不仅与细胞蛋白相互作用,还可与细胞外基质的纤维蛋白和胶原成分如高亲和性多糖的硫酸乙酰肝素等相互作用[29-30],促进纤维组织的形成。 CD11b/CD18下游的效应途径包括NADPH氧化酶的活化、超氧化物和颗粒蛋白的形成和释放[31]。中性粒细胞已被证明可通过释放髓过氧化物酶与内皮细胞相连接,这种连接是通过直接激活CD11b/CD18的整合素来介导的,而这种连接正是心房纤维化结构重构的起始步骤[32]。髓过氧化物酶是由存储在中性粒细胞的初级颗粒的细胞活化后释放,可促进心房纤维化和降低心房颤动发生的阈值,提高房颤易感性[32]。 目前的研究已经扩展了炎症的作用机制,包括参与心房重构的途径,以及在心房颤动的发生发展中的作用。目前的数据表明,白细胞,特别是中性粒细胞,作为循环效应中的最佳蛋白载体,可以诱导胶原蛋白沉积,促进细胞外基质形成,导致心房重构,促进心房纤维化形成。此外,中性粒细胞与心房组织细胞密切接触,释放髓过氧化物酶激活CD11b/CD18的整合素介导细胞间连接,亦可导致心房纤维化形成。当然,这可能不是唯一的途径,在心房纤维化中,中性粒细胞中的超氧化物歧化酶,通过解耦的NADPH氧化酶,一氧化氮合酶等亦与心肌纤维化和房颤的发生密切相关。而纤维化程度与炎症细胞浸润程度是否相关目前不得而知。明确纤维化程度在房颤中的作用非常重要,心房纤维化的程度可预测房颤消融术后复发的风险[33-34]。 2.5 松弛素在心房纤维化和结构重构中的作用 松弛素是一种自然发生的人类肽激素,它最初被确定为生殖激素。松弛素具有多效性[35]。它具有抗炎作用,影响细胞外基质的沉积的作用,拮抗血管紧张素Ⅱ的作用,有效的抗纤维化活性。 RAAS在心房纤维化中起主要作用的是血管紧张素Ⅱ。血管紧张素Ⅱ通过激活血管紧张素Ⅱ1型受体,促进成纤维细胞的基因表达(包括胶原蛋白),增加成纤维细胞密度,促进心肌纤维化的形成和心房重构的发生。在众多的非临床研究中,松弛素已被证明能预防或逆转神经激素血管紧张素Ⅱ和内皮素对组织器官的影响。在体外,松弛素可以拮抗血管紧张素Ⅱ增加胶原沉积的作用,可能是通过抑制Smad转录因子的细胞外基质的合成来发挥作用的[36]。 在心房和心室纤维化的大鼠模型中,松弛素明显抑制转化生长因子β1和血管紧张素Ⅱ介导的胶原蛋白的产生和沉积[37]。在小鼠的心肌梗死模型中,松弛素延缓了心肌梗死引起的心肌纤维化进展,这与减少转化生长因子β1表达,抑制肌成纤维细胞的分化相关[34]。注射松弛素作为一种抗纤维化策略在高血压和糖尿病性心肌病模型已经显示出一致的抑制和逆转心肌纤维化的作用[38]。松弛素能最终改善组织纤维化导致的结构重构,减少胶原沉积,有效逆转心肌纤维化。 在影响细胞外基质积聚中最重要的介质是金属蛋白酶。永久性房颤患者中,松弛素和金属蛋白酶的激活相关,在心脏重塑中起着重要作用。人THP-1细胞中,松弛素引起的组织重构,是通过增加基质金属蛋白酶9的表达完成的[39]。值得注意的是,虽然松弛素降低了异常的胶原蛋白的积累,但它不影响健康的基底组织胶原含量,突出其治疗的安全性[40]。 此外,松弛素还具有调节心脏离子电流的能力。有研究证明松弛素可以抑制心肌细胞传导速度,降低动作电位时程[41-42]。因此,松弛素可通过抑制分子信号转导通路,抑制关键酶促反应和离子通道来控制心房纤维化的发生,从而为在分子水平上的房颤的治疗提供新的研究方向。 "
| [1]Lijnen PJ, Petrov VV, Fagard RH.Induction of cardiac fibrosis by transforming growth factor-beta(1).Mol Genet Metab. 2000; 71(1-2):418-435.
[2]Rosenkranz S.TGF-beta1 and angiotensin networking in cardiac remodeling.Cardiovasc Res. 2004;63(3):423-432.
[3]Rahimi RA, Leof EB.TGF-beta signaling: a tale of two responses.J Cell Biochem. 2007;102(3):593-608.
[4]Verrecchia F, Mauviel A.Transforming growth factor-beta and fibrosis.World J Gastroenterol. 2007;13(22):3056-3062.
[5]Dean RG, Balding LC, Candido R,et al.Connective tissue growth factor and cardiac fibrosis after myocardial infarction.J Histochem Cytochem. 2005;53(10):1245-1256.
[6]Lee AA, Dillmann WH, McCulloch AD,et al.Angiotensin II stimulates the autocrine production of transforming growth factor-beta 1 in adult rat cardiac fibroblasts.J Mol Cell Cardiol. 1995;27(10):2347-2357.
[7]Evans RA, Tian YC, Steadman R,et al.TGF-beta1-mediated fibroblast-myofibroblast terminal differentiation-the role of Smad proteins.Exp Cell Res. 2003;282(2):90-100.
[8]Hao J, Wang B, Jones SC, et al.Interaction between angiotensin II and Smad proteins in fibroblasts in failing heart and in vitro.Am J Physiol Heart Circ Physiol. 2000;279(6):H3020-H 3030.
[9]Gramley F, Lorenzen J, Koellensperger E, et al. Atrial fibrosis and atrial fibrillation: the role of the TGF-b1 signaling pathway. Int J Cardiol.2010;143(3):405-413.
[10]Kim SK, Pak HN, Park JH, et al. Clinical and serological predictors for the recurrence of atrial fibrillation after electrical cardioversion. Europace. 2009;11(12):1632-1638.
[11]Clancy RM, Backer CB, Yin X, et al. Cytokine polymorphisms and histologic expression in autopsy studies: contribution of TNF-alpha and TGF-β1 to the pathogenesis of autoimmune-associated congenital heart block.J Immunol. 2003;171(6):3253-3261.
[12]Kapur NK, Wilson S, Yunis AA, et al. Reduced endoglin activity limits cardiac fibrosis and improves survival in heart failure. Circulation.2012;125(22): 2728-2738.
[13]Ehrlich JR, Hohnloser SH, Nattel S. Role of angiotensin system and effects of its inhibition in atrial fibrillation: clinical and experimental evidence. Eur Heart J. 2006;27(5): 512-518.
[14]He X, Gao X, Peng L, et al. Atrial fibrillation induces myocardial fibrosis through angiotensin II type 1 receptor-specific Arkadia-mediated downregulation of Smad7. Circ Res. 2011;108(2):164-175.
[15]Dixon IM.The soluble interleukin 6 receptor takes its place in the pantheon of interleukin 6 signaling proteins: phenoconversion of cardiac fibroblasts to myofibroblasts. Hypertension. 2010;56(2):193-195.
[16]Liu YH, D'Ambrosio M, Liao T, et al. N-acetyl-seryl-aspartyl- lysyl-proline prevents cardiac remodeling and dysfunction induced by galectin-3, a mammalian adhesion/growth- regulatory lectin.Am J Physiol Heart Circ Physiol. 2009; 296(2):H404-H412.
[17]de Boer RA, Voors AA, Muntendam P, et al. Galectin-3: a novel mediator of heart failure development and progression.Eur J Heart Fail. 2009;11(9):811-817.
[18]Grandin EW, Jarolim P, Murphy SA, et al. Galectin-3 and the development of heart failure after acute coronary syndrome: pilot experience from PROVE IT-TIMI 22. Clin Chem. 2012; 58(1):267-273.
[19]Shinagawa K, Shi YF, Tardif JC, et al. Dynamic nature of atrial fibrillation substrate during development and reversal of heart failure in dogs. Circulation. 2002;105(22): 2672-2678.
[20]Okuyama Y, Miyauchi Y, Park AM, et al.High resolution mapping of the pulmonary vein and the vein of Marshall during induced atrial fibrillation and atrial tachycardia in a canine model of pacing-induced congestive heart failure. J Am Coll Cardiol. 2003; 42(2):348-360.
[21]Yu L, Ruifrok WP, Meissner M, et al. Genetic and pharmacological inhibition of galectin-3 prevents cardiac remodeling by interfering with myocardial fibrogenesis. Circ Heart Fail. 2013;6(1):107-117.
[22]Goudis CA, Kallergis EM, Vardas PE. Extracellular matrix alterations in the atria: insights into the mechanisms and perpetuation of atrial fibrillation. Europace. 2012;14(5): 623-630.
[23]Narducci ML, Pelargonio G, Dello Russo A, et al. Role of tissue C-reactive protein in atrial cardiomyocytes of patients undergoing catheter ablation of atrial fibrillation: pathogenetic implications. Europace. 2011; 13(8):1133-1140.
[24]Galea R, Cardillo MT, Caroli A, et al. Inflammation and C-reactive protein in atrial fibrillation: cause or effect? Tex Heart Inst J. 2014; 41(5):461-468.
[25]Ramlawi B, Otu H, Mieno S, et al. Oxidative stress and atrial fibrillation after cardiac surgery: a case-control study. Ann Thorac Surg. 2007;84(4): 1166-1173.
[26]Kim YM, Kattach H, Ratnatunga C, et al.Association of atrial nicotinamide adenine dinucleotide phosphate oxidase activity with the development of atrial fibrillation after cardiac surgery. J Am Coll Cardiol. 2008; 51(1):68-74.
[27]Rudolph V, Andrié RP, Rudolph TK, et al. Myeloperoxidase acts as a profibrotic mediator of atrial fibrillation. Nature Med.2010;16(4): 470-474.
[28]Phillipson M, Heit B, Colarusso P, et al. Intraluminal crawling of neutrophils to emigration sites: a molecularly distinct process from adhesion in the recruitment cascade. J Exp Med. 2006; 203(12):2569-2575.
[29]Ross GD.Role of the lectin domain of Mac-1/CR3 (CD11b/CD18) in regulating intercellular adhesion.Immunol Res. 2002;25(3):219-227.
[30]Xia Y, Borland G, Huang J, et al.Function of the lectin domain of Mac-1/complement receptor type 3 (CD11b/CD18) in regulating neutrophil adhesion.J Immunol. 2002; 169(11):6417-6426.
[31]Jakus Z, Berton G, Ligeti E, et al. Responses of neutrophils to anti-integrin antibodies depends on costimulation through low affinity Fc gamma Rs: full activation requires both integrin and nonintegrin signals. J Immunol. 2004; 173(3):2068-2077.
[32]Jerke U, Rolle S, Purfürst B, et al. β2 integrin-mediated cell-cell contact transfers active myeloperoxidase from neutrophils to endothelial cells.J Biol Chem.2013;288(18): 12910-12919.
[33]Reil JC, Hohl M, Oberhofer M, et al. Cardiac Rac1 overexpression in mice creates a substrate for atrial arrhythmias characterized by structural remodelling. Cardiovasc Res. 2010;87(3): 485-493.
[34]Adam O, Lavall D, Theobald K, et al.Rac1-induced connective tissue growth factor regulates connexin 43 and N-cadherin expression in atrial fibrillation.J Am Coll Cardiol. 2010; 55(5):469-440.
[35]Zhao Z, Liu T, Li G. Relaxin-2 may offer therapeutic advantages in end-stage heart failure. Int J Cardiol. 2012; 57(3): 432-433.
[36]Samuel CS, Unemori EN, Mookerjee I, et al. Relaxin modulates cardiac fibroblast proliferation, differentiation, and collagen production and reverses cardiac fibrosis in vivo. Endocrinology, 2004;145(9): 4125-4133.
[37]Mookerjee I, Unemori EN, DU XJ, et al.Relaxin modulates fibroblast function, collagen production, and matrix metalloproteinase-2 expression by cardiac fibroblasts.Ann N Y Acad Sci. 2005;1041:190-193.
[38]Samuel CS, Cendrawan S, Gao XM, et al. Relaxin remodels fibrotic healing following myocardial infarction. Lab Invest. 2011;91(5): 675-690.
[39]Lekgabe ED, Kiriazis H, Zhao C, et al. Relaxin reverses cardiac and renal fibrosis in spontaneously hypertensive rats. Hypertension.2005;46(2): 412-418.
[40]Jintang S, Alei F, Yun Z, et al. Fucoidan increases TNF-α-induced MMP-9 secretion in monocytic cell line U937. Inflamm Res. 2010;59(4): 271-276.
[41]Zhao Z, Ng CY, Liu T, et al.Relaxin as novel strategy in the management of atrial fibrillation: potential roles and future perspectives. Int J Cardiol. 2014; 171(3):e72-73.
[42]Parikh A, Patel D, McTiernan CF, et al. Relaxin suppresses atrial fibrillation by reversing fibrosis and myocyte hypertrophy and increasing conduction velocity and sodium current in spontaneously hypertensive rat hearts. Circulation Res. 2013; 113(3): 313-321. |
| [1] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [2] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [3] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [4] | Zhang Lei, Yan Yu, Liu Yin, Xu Long, Yang Xinglei, Liu Yujia. An 8-week aerobic exercise improves obesity-induced myocardial fibrosis: role of nuclear factor-erythroid 2 p45-related factor 2 pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2650-2656. |
| [5] | Hu Sheng, Yuan Haiyan, Hu Meng, Jin Shanhu. Effects of moderate treadmill exercise on alpha-smooth muscle actin and type IV collagen in the liver of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1723-1727. |
| [6] | Li Xuan, Lu Min, Li Mingxing, Ao Meng, Tang Linmei, Zeng Zhen, Hu Jingwei, Huang Zhiqiang, Xuan Jiqing. In vitro multi-modal imaging of magnetic targeted nanoparticles and their targeting effect on hepatic stellate cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 566-571. |
| [7] | Zhu Shoulei, Yang Jiandong, Cai Jun, Zhang Yujie, Tian Yuan. Mechanism by which allicin inhibits proliferation and migration but promotes apoptosis of human fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5662-5667. |
| [8] | Cheng Xue, Fang Hong, Zhang Yunke, Wu Yingen. Interventional mechanism of Feibi prescription on extracellular matrix transformation in a mouse model of pulmonary fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5038-5043. |
| [9] | Li Qinwen, Liang Jie, Wang Dongmei, Shang Zhenghui. Fibrotic changes in rat dorsal root ganglion following chronic sciatic nerve compression [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4686-4691. |
| [10] | Yang Zhen, Li Hao, Gao Cangjian, Fu Liwei, Tian Guangzhao, Zha Kangkang, Sun Zhiqiang, Li Xu, Guo Weimin, Sui Xiang, Huang Jingxiang, Liu Shuyun, Lu Shibi, Guo Quanyi . Regulation of stem cells by transforming growth factor β3/polylactic acid-glycolic acid microspheres [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4540-4546. |
| [11] |
Ma Chao, Wang Huishan, Han Jinsong, Yin Zongtao, Zhang Xiling.
Cardiac valve prosthesis implantation and surgical maze ablation for the treatment of valvular disease with atrial fibrillation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4580-4587. |
| [12] |
Wu Zhen, Ma Wei, Zang Chenghao, Liu Kuangpin, Liu Wei, Liu Jie, Liang Yu, Li Chunyan, Chen Zhiming, Ru Jin, Fan Chuming, Yang Jinwei, Guo Jianhui, Li Liyan.
Protective effect of notoginsenoside R1 on carbon tetrachloride-induced liver fibrosis in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4213-4217. |
| [13] | Yin Lian, Zhao Jin, Lei Xuemei, Li Miaomiao, Wang Kun, Zhang Tingran, Luo Jiong. Effect of exercise-induced irisin on myocardial fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3730-3736. |
| [14] | Liu Yuetong, Wang Qin, Yang Ye, Zhu Jun, Abulikemu•Tuerdi. Effects of 1,25(OH)2D3 on microRNA-130b and transforming growth factor beta 1 in renal tissue of rat models of diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(2): 248-253. |
| [15] | Yang Ning, Huang Tao, Liu Xiaoran, Zhou Yue, Wang Ruiyuan, Li Junping. Changes of fibrotic factors in the recovery process of skeletal muscle strain [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(17): 2667-2674. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||