Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (50): 8072-8079.doi: 10.3969/j.issn.2095-4344.2014.50.007
Previous Articles Next Articles
Different concentrations of angiotensin II induce directional differentiation of bone marrow mesenchymal stem cells into cardiomyocytes
Shi Jing-bao, Xing Wan-hong
- Department of Cardiothoracic Surgery, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2014-11-08Online:2014-12-03Published:2014-12-03 -
Contact:Xing Wan-hong, M.D., Associate chief physician, Master’s supervisor, Department of Cardiothoracic Surgery, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Shi Jing-bao, Studying for master’s degree, Department of Cardiothoracic Surgery, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:the Science and Technology Plan of Shanxi Province, No. 20120313021-3; Shanxi Scientific Research Fund for Returned Scholars, No. 2012091
CLC Number:
Cite this article
Shi Jing-bao, Xing Wan-hong. Different concentrations of angiotensin II induce directional differentiation of bone marrow mesenchymal stem cells into cardiomyocytes[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(50): 8072-8079.
share this article
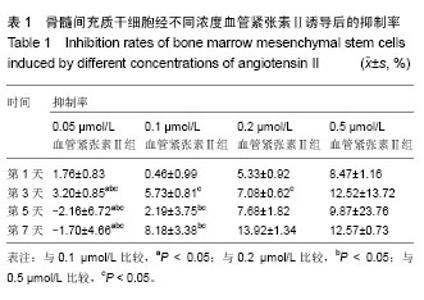
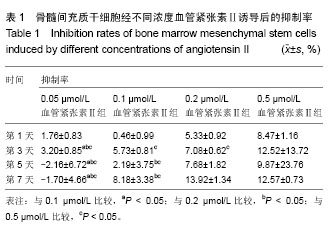
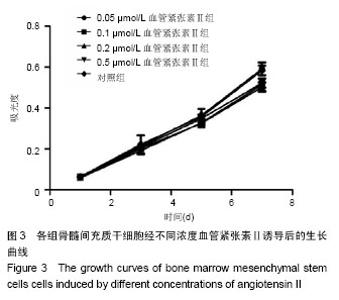
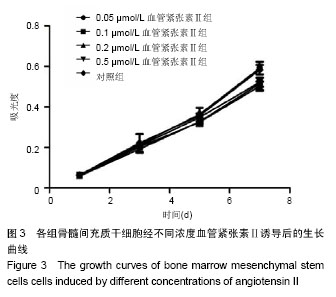
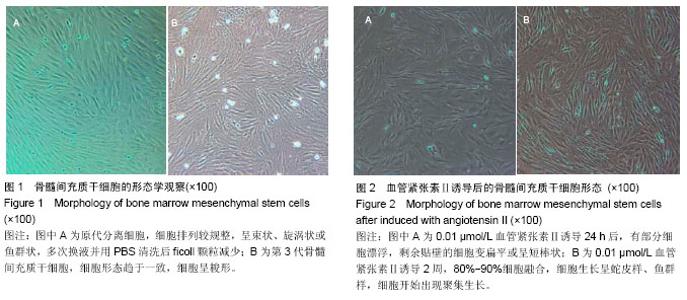
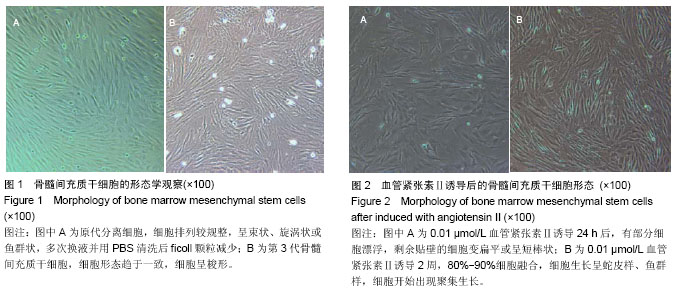
2.1 骨髓间充质干细胞的分离培养和形态学观察 通过密度梯度离心+贴壁法获得骨髓间充质干细胞,培养48 h可见有纺锤形、三角形、短小梭形细胞,细胞折光性好,但可见细胞表面黏附有ficoll颗粒,并且培养基中有大量的ficoll分离液干扰,72 h成为可见细长纺锤、细长梭形细胞。细胞生长较零散,成簇的细胞生长较快,可形成折光双联体。第4天,细胞开始增殖,细胞密度较低,细胞排列不规则,可见三角形、梭形细胞及少量圆形细胞,第7天有70%-80%的细胞融合,细胞排列较规整,呈束状、旋涡状或鱼群状。多次换液并用PBS清洗后ficoll颗粒减少(图1A)。细胞传至第2,3代,细胞形态趋于一致,细胞呈梭形细胞样(图1B)。 2.2 诱导后的细胞形态观察 第3代骨髓间充质干细胞经不同浓度血管紧张素Ⅱ诱导液分别诱导24 h,0.5 μmol/L组可见贴壁的部分细胞脱落,其余各组细胞均有死亡,且随药物浓度的升高细胞死亡率升高。剩余贴壁的细胞变扁平或呈短棒状(图2A)。2 d后存活细胞可见细胞核的分裂,开始增殖,细胞呈细长梭形,少数细胞呈三角形或多边形。培养2周后,80%-90%细胞融合,细胞生长呈蛇皮样、鱼群样,且细胞出现聚集生长(图2B),3周时可见有克隆样聚集生长现象,细胞呈细长梭形,部分细胞有分支。不同浓度血管紧张素Ⅱ诱导4周后均未见自发性搏动。 2.3 大鼠骨髓间充质干细胞诱导后增殖能力及抑制率 诱导后的前5 d诱导组与空白对照组细胞的增殖曲线无显著差异(图3)。第7天,0.05 μmol/L血管紧张素Ⅱ组的吸光度值与0.1,0.2,0.5 μmol/L血管紧张素Ⅱ组比较,差异有显著性意义(P < 0.05),0.2,0.5 μmol/L血管紧张素Ⅱ组的吸光度值较空白对照组差异有显著性意义(P < 0.05)。通过对抑制率的计算,显示第3代骨髓间充质干细胞在诱导培养1 d内,各组间的增殖抑制能力大致相同,于第2天已开始增殖,随着诱导时间的延长,各诱导组相对空白对照组来说药物抑制率逐渐增强;第3天0.1 μmol/L组与0.2 μmol/L组的抑制率差异无显著性意义(P > 0.05),0.05 μmol/L组于第5天开始出现负值,与其他组比较差异均有显著性意义(P < 0.05)。第5天0.2 μmol/L与0.5 μmol/L组的抑制率差异显著性无意义(P > 0.05),第7天0.2 μmol/L与0.5 μmol/L组的抑制率差异显著性无意义(P > 0.05),均与0.1 μmol/L组的抑制率差异有显著性意义(P < 0.05),见表1。"

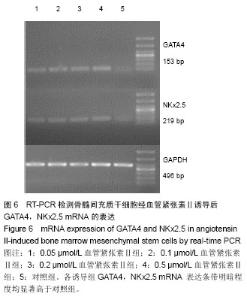
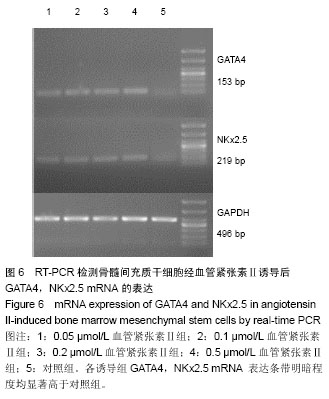
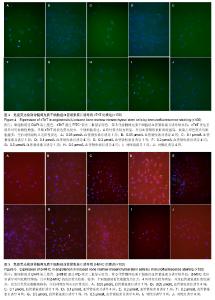
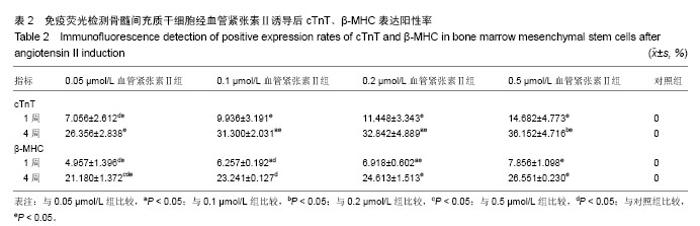
2.4 免疫荧光检测骨髓间充质干细胞诱导后的特异蛋白表达 第3代骨髓间充质干细胞经诱导培养1周后,各浓度诱导组个别细胞表达cTnT,弱表达β-MHC;随着诱导时间的延长,各诱导组均阳性表达cTnT和β-MHC,且呈诱导时间依赖性增强;而空白对照组均未见阳性表达。 0.05 μmol/L组诱导培养1周后,cTnT表达阳性率为7.056%,0.5 μmol/L组cTnT表达阳性率为14.682%,明显高于0.05 μmol/L组,差异有显著性意义(P < 0.05)。诱导4周后,0.2 μmol/L组cTnT表达阳性率与0.1,0.5 μmol/L组比 较差异无显著性意义(P > 0.05)。0.1 μmol/L组cTnT表达阳性率与0.5 μmol/L组比较差异有显著性意义(P < 0.05)。 β-MHC在诱导初期表现为个别细胞被染色,在诱导1周时0.2 μmol/L组β-MHC表达阳性率与0.1,0.5 μmol/L组比较差异无显著性意义(P > 0.05)。在诱导培养4周后,各组诱导率均较1周时增加,0.05 μmol/L组β-MHC表达阳性率与0.2,0.5 μmol/L组比较差异有显著性意义(P < 0.05)。0.1 μmol/L组β-MHC表达阳性率与0.5 μmol/L组比较差异有显著性意义(P < 0.05)。0.2 μmol/L组β-MHC表达阳性率与0.1,0.5 μmol/L组比较差异无显著性意义 (P > 0.05),见图4,5和表2。 2.5 RT-PCR检测各诱导组GATA4、Nkx2.5的表达 各诱导组可见明显条带,对照组无条带,说明诱导组表达心肌早期形成转录因子GATA-4、Nkx2.5基因,对照组不表达心肌早期形成转录因子基因。第3代骨髓间充质干细胞经诱导培养4周后各诱导组可见阳性表达,GATA4、NKx2.5基因在对照组不表达(图6)。"
| [1] Chen XQ, Chen LL, Fan L,et al.Stem cells with FGF4-bFGF fused gene enhances the expression of bFGF and improves myocardial repair in rats.Biochem Biophys Res Commun. 2014;447(1):145-151. [2] Igura K, Okada M, Kim HW,et al.Identification of small juvenile stem cells in aged bone marrow and their therapeutic potential for repair of the ischemic heart.Am J Physiol Heart Circ Physiol. 2013;305(9):H1354-1362. [3] Valarmathi MT, Goodwin RL, Fuseler JW,et al.A 3-D cardiac muscle construct for exploring adult marrow stem cell based myocardial regeneration.Biomaterials. 2010; 31(12): 3185-200. [4] 徐信群,王泉兰,应国秋,等.骨髓间充质干细胞移植对心肌梗死大鼠损伤心肌的修复效果[J].南昌大学学报:医学版,2013,53(6): 1-4,21. [5] 王海萍,张雷,赵静,等.大鼠骨髓间充质干细胞移植治疗心肌梗死的实验研究[J].中国临床解剖学杂志,2012,30(2):209-213. [6] 田茂,朴海南,陈宇,等. 骨髓间充质干细胞体外构建工程化心肌组织的生存[J].中国组织工程研究,2014,18(20):3133-3138. [7] 马红芬,张晓刚,史若飞,等.人工脑膜复合大鼠骨髓间充质干细胞修复心肌梗死[J].中国组织工程研究,2013,17(14): 2552-2557. [8] 苑媛,吕安林,陈丹,等.血管紧张素诱导人骨髓间充质干细胞分化为心肌样细胞[J].心脏杂志,2006,18(3):258-261 . [9] Xing Y, Lv A, Wang L,et al.The combination of angiotensin II and 5-azacytidine promotes cardiomyocyte differentiation of rat bone marrow mesenchymal stem cells.Mol Cell Biochem. 2012;360(1-2):279-287. [10] 张卫泽,樊艳,陈永清,等.血管紧张素Ⅱ对成人脂肪间充质干细胞向心肌细胞分化的影响[J].第四军医大学学报, 2008,29(8): 692-695. [11] 顾健,王爱玲,郝玉瑜,等.大鼠骨髓间充质干细胞体外诱导分化为心肌样细胞的研究[J].医学综述,2014,20(6):1109-1111,1116. [12] 陈玲玲,尹立雪.超声辐照微泡介导5-氮杂胞苷诱导人骨髓间充质干细胞心肌样分化的实验研究[J].中华超声影像学杂志,2013, 22(11):991-996. [13] 孙庆国,赵文静,王日中,等.体外定向诱导大鼠骨髓间充质干细胞分化为心肌样细胞的实验研究[J].中国实验诊断学,2012,16(12): 2202-2204. [14] 邸军,李梓菲,陈艳,等.大鼠骨髓间充质干细胞体外向心肌细胞诱导分化的机制[J].中国老年学杂志,2013,33(12):2835-2836. [15] 王宁,李新华,邢万红,等.细胞代数与5-氮胞苷浓度对体外诱导骨髓间充质干细胞向心肌样细胞定向分化的影响[J].中西医结合心脑血管病杂志,2010, 8(10):1220-1222. [16] 王宁,邢万红,李新华,等.大鼠骨髓间充质干细胞体外心肌定向诱导及心肌组织构建研究[J].国际生物医学工程杂志,2010,33(3): 163-166. [17] Makino S, Fukuda K, Miyoshi S,et al.Cardiomyocytes can be generated from marrow stromal cells in vitro.J Clin Invest. 1999;103(5):697-705. [18] Fukuda K.Development of regenerative cardiomyocytes from mesenchymal stem cells for cardiovascular tissue engineering. Artif Organs. 2001;25(3):187-193. [19] Liu Y, Song J, Liu W,et al.Growth and differentiation of rat bone marrow stromal cells: does 5-azacytidine trigger their cardiomyogenic differentiation?Cardiovasc Res. 2003;58(2): 460-468. [20] Xu J, Lin SC, Chen J,et al.CCR2 mediates the uptake of bone marrow-derived fibroblast precursors in angiotensin II-induced cardiac fibrosis.Am J Physiol Heart Circ Physiol. 2011;301(2):H538-547. [21] Sopel MJ, Rosin NL, Lee TD,et al.Myocardial fibrosis in response to Angiotensin II is preceded by the recruitment of mesenchymal progenitor cells.Lab Invest. 2011;91(4): 565-578. [22] Qian C, Schoemaker RG, van Gilst WH,et al.The role of the renin-angiotensin-aldosterone system in cardiovascular progenitor cell function.Clin Sci (Lond). 2009;116(4):301-314. [23] Hakuno D, Fukuda K, Makino S,et al.Bone marrow-derived regenerated cardiomyocytes (CMG Cells) express functional adrenergic and muscarinic receptors.Circulation. 2002;105(3): 380-386. [24] Wang X, Phillips MI, Mehta JL.LOX-1 and angiotensin receptors, and their interplay.Cardiovasc Drugs Ther. 2011; 25(5):401-417. [25] Wang X, Khaidakov M, Ding Z,et al.Cross-talk between inflammation and angiotensin II: studies based on direct transfection of cardiomyocytes with AT1R and AT2R cDNA.Exp Biol Med (Maywood). 2012;237(12):1394-1401. [26] Coyne TM, Marcus AJ, Woodbury D,et al.Marrow stromal cells transplanted to the adult brain are rejected by an inflammatory response and transfer donor labels to host neurons and glia.Stem Cells. 2006;24(11):2483-2492. [27] Heikinheimo M, Scandrett JM, Wilson DB.Localization of transcription factor GATA-4 to regions of the mouse embryo involved in cardiac development.Dev Biol. 1994;164(2): 361-373. [28] Xing Y, Lv A, Wang L,et al.Engineered myocardial tissues constructed in vivo using cardiomyocyte-like cells derived from bone marrow mesenchymal stem cells in rats.J Biomed Sci. 2012;19:6. [29] Liu BW, Lü AL, Hou J,et al.Electrophysiological characteristics of cardiomyocyte-like cells from rat bone marrow derived mesenchymal stem cells by four inductors.Chin Med J (Engl). 2013;126(18):3528-3533. [30] Li L, Fan D, Wang C,et al.Angiotensin II increases periostin expression via Ras/p38 MAPK/CREB and ERK1/2/TGF-β1 pathways in cardiac fibroblasts.Cardiovasc Res. 2011;91(1): 80-89. [31] He JG, Chen SL, Huang YY,et al.The nonpeptide AVE0991 attenuates myocardial hypertrophy as induced by angiotensin II through downregulation of transforming growth factor-beta1/Smad2 expression.Heart Vessels. 2010;25(5): 438-443. [32] Wang N, Ren GD, Zhou Z,et al.Cooperation of myocardin and Smad2 in inducing differentiation of mesenchymal stem cells into smooth muscle cells.IUBMB Life. 2012;64(4):331-339. [33] Haddad GE, Coleman BR, Zhao A,et al.Regulation of atrial contraction by PKA and PKC during development and regression of eccentric cardiac hypertrophy.Am J Physiol Heart Circ Physiol. 2005;288(2):H695-704. [34] Li TF, O'Keefe RJ, Chen D.TGF-beta signaling in chondrocytes. Front Biosci. 2005;10:681-688. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [4] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [5] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [6] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [7] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [8] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [9] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [10] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [11] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [12] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [13] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||