Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (32): 5132-5139.doi: 10.3969/j.issn.2095-4344.2014.32.009
Previous Articles Next Articles
Effects and mechanisms of human umbilical cord-derived mesenchymal stem cells on the thymus development in nude mice
Wang Li-ming1, Wang Li-hua2, Li Ming1, Wang Qian-yun1, Yang Jie1, Liu Guang-yang2, Cong Xiu-li2,Liu Yong-jun2
- 1Cell Therapy Center, the 323 Hospital of Chinese PLA, Xi’an 710054, Shaanxi Province, China; 2Alliancells Institute of Stem Cells and Translational Regenerative Medicine, Tianjin 300381, China
-
Received:2014-07-08Online:2014-08-06Published:2014-09-18 -
Contact:Liu Yong-jun, M.D., Professor, Alliancells Institute of Stem Cells and Translational Regenerative Medicine, Tianjin 300381, China -
About author:Wang Li-ming, Master, Chief physician, Cell Therapy Center, the 323 Hospital of Chinese PLA, Xi’an 710054, Shaanxi Province, China -
Supported by:the Military Medicine and Health Project, No. CLZ12GA23
CLC Number:
Cite this article
Wang Li-ming, Wang Li-hua, Li Ming, Wang Qian-yun, Yang Jie, Liu Guang-yang, Cong Xiu-li,Liu Yong-jun. Effects and mechanisms of human umbilical cord-derived mesenchymal stem cells on the thymus development in nude mice[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(32): 5132-5139.
share this article
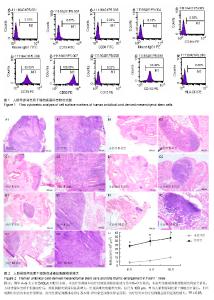
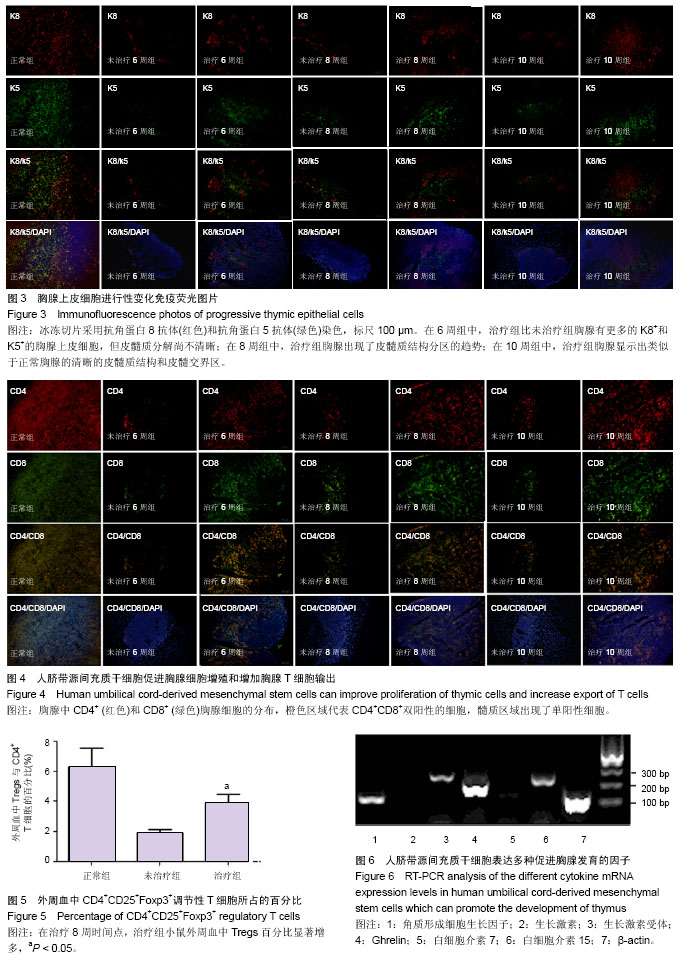
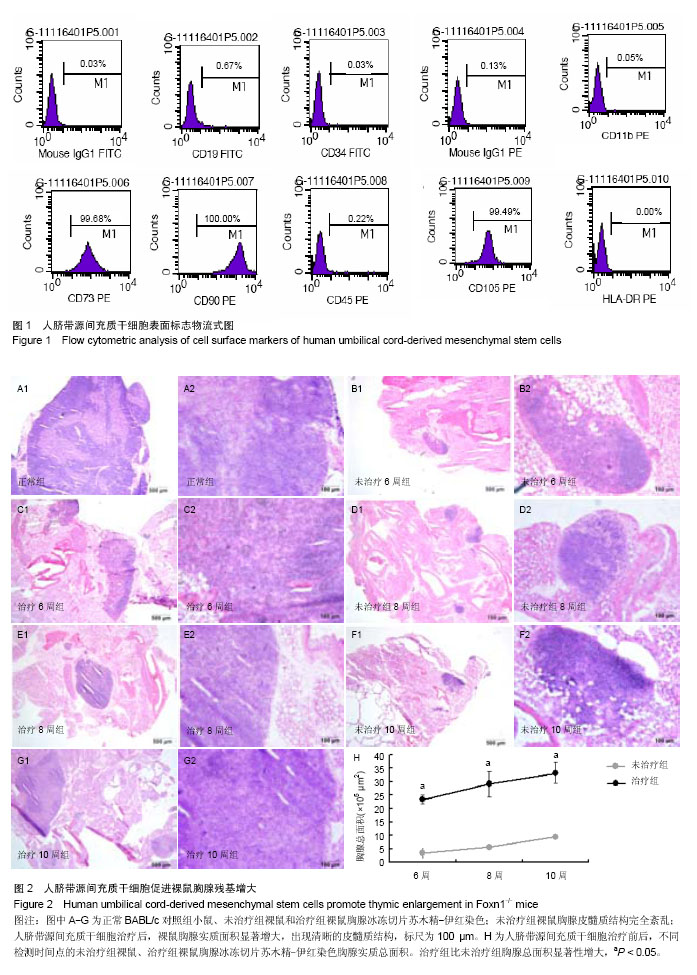
2.1 人脐带源间充质干细胞表型鉴定 流式细胞仪检测结果显示,分离培养的间充质干细胞达到国际细胞治疗协会(ISCT)的鉴定标准[23](图1)。 2.2 人脐带源间充质干细胞能够促进裸鼠胸腺残基体积增大 Foxn1基因变异导致胸腺发育异常,但在裸鼠体内的胸腺部位,仍然存在着胸腺样器官,它由紧密的淋巴样组织组成,周围是一层薄薄的纤维囊和囊下界限不清的成熟的脂肪组织。 为了确定人脐带源间充质干细胞是否能够促进胸腺发育,实验从大小和组织学两个方面来分析不同治疗时间点的治疗组和未治疗组小鼠胸腺的差别。从外观和质量来看,治疗组和未治疗组无显著性差异。但苏木精-伊红染色显示,治疗组小鼠比未治疗组小鼠的正常胸腺组织学结构区域显著性增大。而且,治疗组较未治疗组小鼠的胸腺组织出现了明显的皮髓质交界结构(图2)。 这些数据表明,用人脐带源间充质干细胞治疗6周开始就可见胸腺结构的重塑,并且随着持续反复的治疗,胸腺结构不断向正常胸腺改善。 2.3 人脐带源间充质干细胞治疗促进裸鼠胸腺皮质和髓质结构重建以及胸腺上皮细胞的分化 裸鼠的胸腺体积较小,且结构异常,没有明显的皮髓质区域之分,其中的大多数胸腺上皮细胞分化只停留在祖细胞阶段[24-29]。K8K5分别是成熟的皮质上皮细胞和髓质上皮细胞的标志,可用来评价胸腺上皮细胞的定位和组织特征。在正常胸腺中,髓质上皮细胞(medullary TECs,mTECs)组成K5+的髓质区域,周围被K8+的皮质所包绕。 本实验中,人脐带源间充质干细胞治疗后6,8,10周的胸腺组织抗角蛋白免疫荧光染色显示,未治疗组裸鼠胸腺上皮细胞散在分布;相反,治疗组胸腺出现清晰的皮质(K5-K8+ TECs)和髓质(K5+K8- TECs)区域。此外,在治疗组胸腺的皮质和髓质区域均出现了紧密的上皮细胞网状结构,皮髓质交界区的结构接近正常胸腺(图3)。这表明人脐带源间充质干细胞能够促进胸腺上皮细胞的分化和增殖,重建裸鼠的胸腺结构。 2.4 人脐带源间充质干细胞增加胸腺生成能力,促进胸腺T细胞的输出 研究发现人脐带源间充质干细胞能够明显改善裸鼠胸腺结构的基础上,实验进一步探讨胸腺T细胞的能力是否提高。在正常的胸腺组织,大多数的胸腺细胞是CD4+CD8+双阳的,而在裸鼠,由于胸腺上皮细胞和胸腺祖细胞缺乏相互作用,CD4+CD8+T细胞数量极少。应用人脐带源间充质干细胞后,CD4+CD8+胸腺细胞显著增加,并在髓质区域可见少量成熟的单阳性细胞(图4)。 2.5 人脐带源间充质干细胞增加裸鼠外周血中调节性T细胞数量 Tregs主要产生于胸腺,在成年健康小鼠外周血中,CD4+CD25+Foxp3+Tregs占CD4+T细胞的5%-10%[30],通过抑制CD4+效应性T细胞的功能,在自身免疫耐受和免疫平衡中发挥着核心作用[31]。已有大量数据表明,缺乏CD4+CD25+Foxp3+Tregs与自身免疫性疾病的发生密切相关[32]。本研究发现,治疗后外周血中Tregs占CD4+T细胞的百分比较未治疗组显著增加(从1.95%平均增加到3.89%,图5)。 2.6 人脐带源间充质干细胞表达细胞因子和生长因子 研究表明,脐带源间充质干细胞能够在表面黏附分子的介导下[1],从血管中迁移到组织中[33]。 作者用CM-Dil标记人脐带源间充质干细胞来观察腹腔注射后细胞在胸腺组织中的分布,结果表明,人脐带源间充质干细胞能够迁移并且定植到胸腺,主要存在于胸腺组织的边缘,在胸腺组织中心区域也有散在分布。 此外,用real-time PCR检测到人脐带源间充质干细胞能够高表达多种与胸腺发育相关的细胞因子和生长因子,其中包括角质形成细胞生长因子、Ghrelin、白细胞介素15(图6)。"
| [1] Uccelli A, Moretta L, Pistoia V.Mesenchymal stem cells in health and disease.Nat Rev Immunol. 2008;8(9):726-736. [2] Le Blanc K, Rasmusson I, Sundberg B,et al.Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells.Lancet. 2004;363 (9419):1439-1441. [3] Sun L, Akiyama K, Zhang H,et al.Mesenchymal stem cell transplantation reverses multiorgan dysfunction in systemic lupus erythematosus mice and humans.Stem Cells. 2009; 27(6):1421-1432. [4] González MA, Gonzalez-Rey E, Rico L,et al.Treatment of experimental arthritis by inducing immune tolerance with human adipose-derived mesenchymal stem cells.Arthritis Rheum. 2009;60(4):1006-1019. [5] Koetz K, Bryl E, Spickschen K,et al.T cell homeostasis in patients with rheumatoid arthritis.Proc Natl Acad Sci U S A. 2000;97(16):9203-9208. [6] Kayser C, Alberto FL, da Silva NP,et al.Decreased number of T cells bearing TCR rearrangement excision circles (TREC) in active recent onset systemic lupus erythematosus.Lupus. 2004;13(12):906-911. [7] Augello A, Tasso R, Negrini SM,et al.Cell therapy using allogeneic bone marrow mesenchymal stem cells prevents tissue damage in collagen-induced arthritis.Arthritis Rheum. 2007;56(4):1175-1186. [8] Miller JF.Immunological function of the thymus.Lancet. 1961; 2(7205):748-749. [9] Erickson M, Morkowski S, Lehar S, et al. Regulation of thymic epithelium by keratinocyte growth factor.Blood. 2002;100(9): 3269-3278. [10] Gill J, Malin M, Holländer GA,et al.Generation of a complete thymic microenvironment by MTS24(+) thymic epithelial cells. Nat Immunol. 2002;3(7):635-642. [11] Blackburn CC, Augustine CL, Li R,et al.The nu gene acts cell-autonomously and is required for differentiation of thymic epithelial progenitors.Proc Natl Acad Sci U S A. 1996;93(12): 5742-5746. [12] Guo J, Feng Y, Barnes P,et al.Deletion of FoxN1 in the thymic medullary epithelium reduces peripheral T cell responses to infection and mimics changes of aging.PLoS One. 2012;7(4): e34681. [13] Itoi M, Tsukamoto N, Yoshida H,et al.Mesenchymal cells are required for functional development of thymic epithelial cells. Int Immunol. 2007;19(8):953-964. [14] Anderson G, Jenkinson EJ, Moore NC,et al. MHC class II-positive epithelium and mesenchyme cells are both required for T-cell development in the thymus.Nature. 1993; 362(6415):70-73. [15] Ritter MA, Boyd RL.Development in the thymus: it takes two to tango.Immunol Today. 1993;14(9):462-469. [16] Holub M, Rossmann P, Tlaskalova H, et al.Thymus rudiment of the athymic nude mouse.Nature. 1975; 256 (5517):491-493. [17] Baksh D, Yao R, Tuan RS.Comparison of proliferative and multilineage differentiation potential of human mesenchymal stem cells derived from umbilical cord and bone marrow.Stem Cells. 2007;25(6):1384-1392. [18] Fry TJ, Sinha M, Milliron M,et al.Flt3 ligand enhances thymic-dependent and thymic-independent immune reconstitution. Blood. 2004;104(9):2794-2800. [19] Finch PW, Rubin JS.Keratinocyte growth factor/fibroblast growth factor 7, a homeostatic factor with therapeutic potential for epithelial protection and repair.Adv Cancer Res. 2004;91:69-136. [20] Voruganti VS, Tejero ME, Proffitt JM,et al.Characterization of ghrelin in pedigreed baboons: evidence for heritability and pleiotropy.Obesity (Silver Spring). 2008;16(4):804-810. [21] Holland AM, van den Brink MR. Rejuvenation of the aging T cell compartment.Curr Opin Immunol. 2009;21(4):454-459. [22] Kruyt MC, De Bruijn J, Veenhof M,et al.Application and limitations of chloromethyl-benzamidodialkylcarbocyanine for tracing cells used in bone Tissue engineering.Tissue Eng. 2003;9(1):105-115. [23] Dominici M, Le Blanc K, Mueller I,et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317. [24] Xiao S, Manley NR. Impaired thymic selection and abnormal antigen-specific T cell responses in Foxn1(Δ/Δ) mutant mice. PLoS One. 2010;5(11):e15396. [25] Su DM, Navarre S, Oh WJ,et al.A domain of Foxn1 required for crosstalk-dependent thymic epithelial cell differentiation. Nat Immunol. 2003;4(11):1128-1135. [26] Naquet P, Naspetti M, Boyd R.Development, organization and function of the thymic medulla in normal, immunodeficient or autoimmune mice.Semin Immunol. 1999;11(1):47-55. [27] Gray D, Abramson J, Benoist C,et al.Proliferative arrest and rapid turnover of thymic epithelial cells expressing Aire.J Exp Med. 2007;204(11):2521-2528. [28] Bleul CC, Corbeaux T, Reuter A,et al. Formation of a functional thymus initiated by a postnatal epithelial progenitor cell.Nature. 2006;441(7096):992-996. [29] Loeffler J, Bauer R, Hebart H,et al.Quantification of T-cell receptor excision circle DNA using fluorescence resonance energy transfer and the LightCycler system.J Immunol Methods. 2002;271(1-2):167-175. [30] Itoh M, Takahashi T, Sakaguchi N,et al.Thymus and autoimmunity: production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance.J Immunol. 1999; 162(9):5317-5326. [31] Shevach EM, McHugh RS, Piccirillo CA,et al.Control of T-cell activation by CD4+ CD25+ suppressor T cells.Immunol Rev. 2001;182:58-67. [32] Yamazaki S, Inaba K, Tarbell KV,et al. Dendritic cells expand antigen-specific Foxp3+ CD25+ CD4+ regulatory T cells including suppressors of alloreactivity. Immunol Rev. 2006; 212:314-329. [33] Devine SM, Cobbs C, Jennings M,et al. Mesenchymal stem cells distribute to a wide range of tissues following systemic infusion into nonhuman primates.Blood. 2003;101(8): 2999-3001. [34] Nehls M, Pfeifer D, Schorpp M,et al.New member of the winged-helix protein family disrupted in mouse and rat nude mutations.Nature. 1994;372(6501):103-107. [35] Nehls M, Kyewski B, Messerle M,et al.Two genetically separable steps in the differentiation of thymic epithelium. Science. 1996;272(5263):886-889. [36] Chen L, Xiao S, Manley NR. Foxn1 is required to maintain the postnatal thymic microenvironment in a dosage-sensitive manner. Blood. 2009;113(3):567-574. [37] Wolf SS, Cohen A.Expression of cytokines and their receptors by human thymocytes and thymic stromal cells.Immunology. 1992;77(3):362-368. [38] Lu LL, Liu YJ, Yang SG,et al. Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica. 2006;91(8):1017-1026. [39] Goldberg GL, Zakrzewski JL, Perales MA,et al.Clinical strategies to enhance T cell reconstitution.Semin Immunol. 2007;19(5):289-296. [40] Dixit VD, Yang H, Sun Y,et al.Ghrelin promotes thymopoiesis during aging.J Clin Invest. 2007;117(10):2778-2790. [41] Rossi SW, Jeker LT, Ueno T,et al. Keratinocyte growth factor (KGF) enhances postnatal T-cell development via enhancements in proliferation and function of thymic epithelial cells. Blood. 2007;109(9):3803-3811. [42] Fontenot JD, Rudensky AY.A well adapted regulatory contrivance: regulatory T cell development and the forkhead family transcription factor Foxp3.Nat Immunol. 2005;6(4): 331-337. [43] Sakaguchi S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol. 2004;22:531-562. |
| [1] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [2] | Cui Lianxu, Jiang Wenkang, Lu Dahong, Xu Junrong, Liu Xiaocui, Wang Bingyun. Clinical-grade human umbilical cord mesenchymal stem cells affect the improvement of neurological function in rats with traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 835-839. |
| [3] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [4] | Su Meng, Wang Xin, Zhang Jin, Bei Ying, Huang Yu, Zhu Yanzhao, Li Jiali, Wu Yan. Nanocellular vesicles loaded with curcumin promote wound healing in diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1877-1883. |
| [5] | Yang Xu, Wang Feiqing, Zhao Jianing, Zhang Chike, Liang Huiling, Qi Shasha, Wu Dan, Liu Yanqing, Tang Dongxin, Liu Yang, Li Yanju. Effects of multiple myeloma cell conditioned medium on proliferation and differentiation of human umbilical cord mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1514-1520. |
| [6] | Ding Lili, Hu Mingzhi, Wu Zhihui, Sun Xiaolin, Wang Yongfu. Therapeutic effect of umbilical cord mesenchymal stem cells modified by miR-1-5p in MRL/lpr lupus mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1492-1500. |
| [7] | Ma Munan, Xie Jun, Sang Yuchao, Huang Lei, Zhang Guodong, Yang Xiaoli, Fu Songtao. Electroacupuncture combined with bone marrow mesenchymal stem cells in the treatment of chemotherapy-induced premature ovarian insufficiency in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 1-7. |
| [8] | Liu Siqi, Wu Mingrui, Qiao Lingran, Xie Liying, Chen Siyu, Han Zhibo, Zuo Lin. Effects of hydrogel loaded with human umbilical cord mesenchymal stem cells on diabetic wound repair in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 21-27. |
| [9] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [10] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [11] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [12] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [13] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [14] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [15] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||