Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (33): 5372-5376.doi: 10.3969/j.issn.2095-4344.2014.33.022
Previous Articles Next Articles
Mechanism of estrogen and estrogen-related compounds in osteoarthritis: joint protection and repair of bone cartilage cells
Wang Jian, Tao Hai-rong
- Department of Orthopedics, Shanghai Third People’s Hospital, School of Medicine of Shanghai Jiao Tong University, Shanghai 201900, China
-
Online:2014-08-13Published:2014-08-13 -
Contact:Tao Hai-rong, M.D., Associate chief physician, Master’s supervisor, Department of Orthopedics, Shanghai Third People’s Hospital, School of Medicine of Shanghai Jiao Tong University, Shanghai 201900, China -
About author:Wang Jian, Studying for master’s degree, Department of Orthopedics, Shanghai Third People’s Hospital, School of Medicine of Shanghai Jiao Tong University, Shanghai 201900, China
CLC Number:
Cite this article
Wang Jian, Tao Hai-rong. Mechanism of estrogen and estrogen-related compounds in osteoarthritis: joint protection and repair of bone cartilage cells[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(33): 5372-5376.
share this article
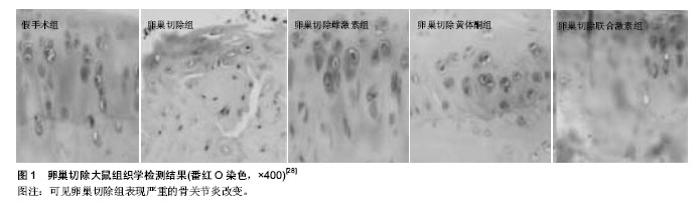
2.1 在骨关节炎发病机制中雌激素对骨代谢的影响 雌激素受体存在于成骨细胞、破骨细胞和骨细胞中。雌激素,尤其是雌二醇可以作用于成骨细胞和破骨细胞表面的受体直接影响骨的形成与吸收,也可以刺激这些细胞分泌局部调节因子来影响骨代谢,雌激素通过刺激成骨细胞表达骨保护素来对抗骨吸收[7]。在分子水平,雌激素通过影响多条骨代谢调控途径参与了骨关节炎骨代谢的调控。在调控骨吸收方面,骨保护素/核因子κB受体活化因子/及其配体系统是近年来发现的在破骨细胞分化过程中的一个重要信号传导通路。成骨细胞及骨髓基质细胞表达核因子κB受体活化因子配体,与破骨细胞前体细胞或破骨细胞表面上的核因子κB受体活化因子结合后,促进破骨细胞的分化及骨吸收活性。成骨细胞及骨髓基质细胞分泌表达骨保护素与核因子κB受体活化因子配体竞争性结合,阻止核因子κB受体活化因子配体与核因子κB受体活化因子之间的结合。因此骨保护素干扰核因子κB受体活化因子配体与核因子κB受体活化因子结合的程度决定了骨吸收的速度[8]。Shevde等[9]研究发现,雌激素可通过雌激素受体抑制核因子κB受体活化因子配体和破骨细胞形成。Xu 等[10]对卵巢切除大鼠(雌性大鼠)骨保护素表达的研究发现,去卵巢大鼠给予雌激素后,腰椎骨保护素mRNA表达升高。雌激素上调成骨细胞骨保护素的表达,对核因子κB受体活化因子配体表达则有下调作用。 当雌激素缺乏时,破骨细胞前细胞数量和破骨细胞数量增加。由于成骨细胞上的骨保护素可以竞争性阻断核因子κB受体活化因子与核因子κB受体活化因子结合,从而抑制了破骨细胞的成熟,所以当补充雌激素后骨保护素-核因子κB受体活化因子配体结合表达明显上升[11]。说明雌激素可通过增加成骨细胞中骨保护素和核因子κB受体活化因子配体的表达,抑制破骨性骨吸收,防止骨关节炎的发生和进展。West等[12]的实验证明,绝经前妇女组的骨保护素水平明显高于绝经后妇女组,表明雌激素分泌减少,影响骨保护素表达。体内调控破骨细胞的多种因子和激素几乎都是通过影响骨保护素和核因子κB受体活化因子配体的分泌实现的[13]。 雌激素已被证明在破骨细胞不同的生长的阶段(即破骨细胞形成,骨吸收和细胞凋亡阶段)都能产生直接的抗破骨细胞的作用。直接抑制多核破骨细胞的形成被认为是通过抑制破骨细胞前体细胞NfKappa配体的受体激活剂诱导的c-Junand上的c-JunN末端激酶活性引起的[14]。雌激素也被证明对成熟破骨细胞有直接影响。一些研究表明,雌激素下调溶酶体酶的活性和生成,下调的组织蛋白酶K和抗酒石酸酸性磷酸酶可以减少骨吸收[15]。为进一步验证这方面的发现,一项最新研究发现,循环池中细胞表达核因子κB受体活化因子配体(骨髓基质细胞,T细胞和B细胞)在早期绝经后妇女是增加的。此外,在骨质疏松症的发病机制中,这些细胞核因子κB受体活化因子配体的表达与骨吸收率密切相关[16]。作者还发现,雌激素治疗能显著降低核因子κB受体活化因子配体表达的细胞数量。 临床试验中,雌激素会增加骨保护素的水平,显示出核因子κB受体活化因子配体-骨保护素轴在雌激素调节骨代谢中的重要作用。此外,研究表明雌激素能显著提高骨保护素的表达水平,因此雌激素去势降低骨保护素在体内的表达,从而增加破骨细胞的活性。这并不奇怪,选择性雌激素受体调节剂影响雌激素的抗破骨细胞作用,研究表明,他莫昔芬、雷洛昔芬和奥培米芬通过上调成骨细胞骨保护素的表达来抑制破骨细胞的形成。除了通过对破骨细胞和成骨细胞的骨保护素/核因子κB受体活化因子配体比率的调节直接作用控制骨吸收,雌激素也调节了一系列其他的细胞因子的表达来影响骨细胞活性,如转化生长因子β,白细胞介素1,白细胞介素6,肿瘤坏死因子α。总之,雌激素上调抗破骨细胞的细胞因子,而下调亲破骨细胞的细胞因子[17]。 已经确认Wnt/catenin途径(经典Wnt途径)、骨形态发生蛋白途径参与调控骨形成过程[18]。雌激素通过抑制Wnt信号通路可使成骨细胞分化进程受阻而诱导Wnt家族高表达则可以使成骨细胞特异性基因表达增加,促进骨形成。骨形态发生蛋白是促进骨形成和诱导成骨细胞分化最重要的细胞外信号分子之一,雌激素激活Smads信号传导和调节成骨基因转录而发挥其成骨作用。应用基因芯片与定量PCR技术,Hopwood等[18]研究者观察骨关节炎患者成骨细胞的相关基因表达,发现Wnt信号系统与骨形态发生蛋白信号系统相关基因(Wnt5b,Pten,Runx2,骨形态发生蛋白5,Smad3,Smad4)与正常对照组比较呈现差异表达,提示雌激素通过Wnt与骨形态发生蛋白信号系统也参与了骨关节炎患者骨代谢的调控。 2.2 在骨关节炎发病机制中雌激素对软骨代谢的影响 雌激素作用于软骨细胞、破骨细胞和成骨细胞3种靶细胞,在细胞水平上的已被证实具有多种效果。相关研究表明雌激素或选择性雌激素受体调节剂作用于软骨细胞上的雌激素受体调节软骨细胞功能[19]。另外,雌激素对软骨细胞的直接影响高度依赖于雌激素的剂量,低剂量可以防止软骨退变,促进软骨的形成。高剂量则具有相反的效果,即分解代谢。在分子水平上的雌激素调节的一系列细胞因子和生长因子的表达水平,如转化生长因子β,胰岛素样生长因子1,肿瘤坏死因子α,白细胞介素1β和白细胞介素6,在使用特定剂量时能防止软骨退变和刺激新的细胞合成。这些效果也表现出对软骨细胞蛋白水解活性水平的调节,其中雌激素诱导基质金属蛋白酶/基质金属蛋白酶抑制剂比例的下调,从而减少软骨的降解[19]。雌激素通过雌激素受体复合物与胰岛素样生长因子1基因启动子相互作用对胰岛素样生长因子1水平进行调控,同时,雌激素还可促进关节软骨细胞合成和胰岛素样生长因子结合蛋白3,胰岛素样生长因子结合蛋白3通过调节胰岛素样生长因子1的活性,进而调节关节软骨内胰岛素样生长因子结合蛋白2 水平,最终使关节软骨细胞合成的软骨基质增加,保护关节软骨。 细胞因子肿瘤坏死因子α、白细胞介素1β和白细胞介素6等作用于软骨细胞诱导基质金属蛋白酶、促分解代谢因子和其他炎症因子基因的表达,引起软骨基质的降解。雌激素对某一种或多种炎症因子具有直接或间接抑制效应,从而对关节软骨起到保护作用。体外培养软骨细胞在雌激素缺乏的情况下,细胞中介导的活性细胞因子肿瘤坏死因子α、白细胞介素1β和白细胞介素6表达和分泌增 加[20]。尽管雌激素调节的分子机制尚未完全清楚,但已有发现雌激素作用于软骨细胞雌激素受体,降低细胞中白细胞介素1β mRNA的稳态水平。雌激素下调白细胞介素1β、肿瘤坏死因子α启动子的活性,这种影响可能是间接的,因为白细胞介素1β、肿瘤坏死因子α的启动子中无雌激素作用元件[21]。白细胞介素1β、肿瘤坏死因子α诱导基质细胞和成骨细胞产生白细胞介素6,这种诱导作用随雌激素增加而抑制,随雌激素减少而被促进。实验证实去卵巢小鼠的骨髓培养中白细胞介素6产生增加,这种效应至少在小鼠中被认为是雌激素间接影响了白细胞介素6启动子近端225 bp的转录活性[22]。另一方面雌激素也能通过糖皮质激素的介导,对炎性细胞因子产生间接抑制作用。因为雌激素可以通过下丘脑-垂体-肾上腺轴的作用促进肾上腺皮质分泌糖皮质激素,从而间接抑制基质金属蛋白酶类的产生,对关节软骨起到保护作用。 实验证明糖皮质激素可以抑制经白细胞介素1β诱导变性的软骨细胞表达基质金属蛋白酶1、基质金属蛋白酶3,同时能刺激被白细胞介素1β抑制的软骨细胞重新表达基质金属蛋白酶抑制剂1。体外实验证明药理浓度 (10-6 mol/L)和低浓度雌二醇(10-12 mol/L)既能诱导正常软骨细胞表达基质金属蛋白酶1,又能抑制经白细胞介素1β诱导变性的软骨细胞表达基质金属蛋白酶1[23]。体现了雌激素对关节软骨代谢并非简单的单向调节,而是随环境、对象的变化而改变,动态地维持其代谢平衡。同时,雌激素能减少软骨细胞基质金属蛋白酶1、基质金属蛋白酶3、基质金属蛋白酶13的mRNA表达[24],刺激被白细胞介素1β抑制的软骨细胞重新表达基质金属蛋白酶抑制剂1,改善软骨细胞分泌基质金属蛋白酶和基质金属蛋白酶抑制剂的不平衡状态[25],表明对已发生变性的软骨而言,雌激素也许能延缓其退变过程,从而达到保护软骨的作用。 此外,雌激素刺激软骨基质合成,即蛋白多糖的和2型胶原蛋白,因此雌激素在整体水平上对软骨非常有益。 越来越多的研究致力于雌激素在骨关节炎的发病机制中的作用。骨关节炎的病理改变是以软骨下骨硬化,骨小梁结构改变,骨髓水肿病变和骨赘形成为重要特征[15]。 雌激素是一种能直接和间接抑制破骨细胞生成和减少骨吸收的骨转换抑制剂,能直接和间接的作用于软骨[14]。在180只接受为期3年雌激素替代疗法治疗的雌性猕猴实验中证实雌激素对预防骨关节炎有长期疗效[26]。与对照组相比,雌激素替代疗法雌激素替代疗法组显著减少骨关节炎的软骨病变。此外,一些实验模型已证实雌激素对关节软骨的保护作用[27],同时也表现出抑制骨吸收的作用。作者提出高水平的骨代谢等同于高风险的软骨退化,外源性雌激素通过抑制骨的吸收可能会有助于延缓骨关节炎的发展。Yang等[28]研究雌激素和黄体酮激素对切除卵巢的老鼠骨及软骨的影响,结果发现联合激素治疗组与切除卵巢组相比,明显减少软骨退化(图1)。 通过前交叉韧带和部分内侧半月板切除术诱导骨关节炎实验证实雌激素去势和骨吸收增加导致骨质疏松症和骨关节炎恶化[29]。与没有骨质疏松症动物相比,骨质疏松症增加了骨关节炎的严重程度。在内侧半月板不稳定的动物中研究骨关节炎的发展,这些动物有一半是睾丸摘除(雄性小鼠)或卵巢切除(雌性小鼠)。雄性小鼠骨关节炎严重程度明显高于雌性小鼠。然而,与对照组相比,卵巢切除的雌性小鼠发展为更严重的骨性关节炎,而睾丸切除的雄性小鼠骨性关节炎的严重程度并不明显[30]。 Christgau等[31]已证实雌激素在软骨退变中的积极作用。选择性雌激素受体调节剂有防止骨吸收和软骨退变的作用,发现使用激素替代疗法的女性软骨退化显著低于对照组,虽然与年龄匹配的绝经前妇女组相比,绝经后的妇女软骨退化是显著增高的[31]。与安慰剂相比,妇女服用雌激素可以减少45%的关节手术[32]。 相对于绝经前的人群,患有骨关节炎的绝经后妇女有更显著的软骨退变。一项流行病学研究支持这一概念,除了以年龄作为一个危险因素,女性比男性具有更高的风 险[33]。这间接支持这一假设:雌激素与骨关节炎的发病率相关。此外,在猴子和大鼠的实验研究表明,雌激素去势增加骨代谢[26-27],并加速软骨破裂。这些观察的事实有些复杂了,软骨细胞已被证明表达雌激素受体和雌激素的干预效应,但是,这并不说明雌激素缺乏是软骨退变的惟一不利影响因素。 2.3 在骨关节炎发病机制中雌激素对滑膜炎症的影响 骨关节炎除了涉及骨、软骨改变尤其是在后期阶段还受到滑膜的影响。雌激素对炎症的作用,主要来源于雌激素对类风湿性关节炎作用的研究。类风湿性关节炎是机体产生的促炎症介质作用于关节骨骼的炎症,导致骨质流失。类风湿性关节炎是一种自身免疫性疾病,在女性中更为常见。发病的高峰是在女性更年期,这期间卵巢产生的性激素急剧减少。动物研究已经证明雌激素对类风湿关节炎有显著的疗效,并且有报告称激素替代疗法对绝经期妇女类风湿性关节炎有积极的作用[34]。 雌激素和雌激素相关的化合物对自身免疫疾病导致的炎症有良好的作用,在骨关节炎进展的后期阶段可能会抑制滑膜炎症和炎症递质导致的软骨流失。 "
| [1]Lugo L, Villalvilla A,Largo R.Selective estrogen receptor modulators (SERMs): New alternatives for osteoarthritis? Maturitas. 2014;77(4):380-384.
[2]Felson DT, Neogi T.Osteoarthritis: is it a disease of cartilage or of bone?Arthritis Rheum 2004;50:341-344.
[3]Wang P, Liu C, Yang XT, et al.Effect of extracorporeal shock wave therapy on cartilage and subchondral bone remodeling in rabbits with ACLT-induced osteoarthritis. 2014;45(1):120-125.
[4]Henrotin Y, Lambert C, Richette P.Importance of synovitis in osteoarthritis: Evidence for the use of glycosaminoglycans against synovial inflammation.Semin Arthritis Rheum. 2013.
[5]Bay-Jensen AC,Hoegh-Madsen S,Dam E,et al.Which elements are involved in reversible and irreversible cartilage degradation in osteoarthritis?Rheumatol Int 2010;30:435-442.
[6]Karsdal MA, Madsen SH, Christiansen C,et al.Cartilage degradation is fully reversible in the presence of aggrecanase but not matrix metalloproteinase activity. Arthritis Res Ther. 2008;10:R63. Epub 30 May 2008
[7]Wang QP, Yang L, Li XP,et al.Effects of 17β-estradiol on adiponectin regulation of the expression of osteoprotegerin and receptor activator of nuclear factor-κB ligand.Bone. 2012; 51(3):515-523.
[8]Rafeey M, Ghorbanihaghjo A, Masoumi F,et al.New perspective in osteoarthritis: the OPG and RANKL system as a potential therapeutic target?Iran Red Crescent Med J. 2013;15(10):e7591.
[9]Shevde LA,Samant RS,et al.Loss of N-Myc interactor promotes epithelial-mesenchymal transition by activation of TGF-β/SMAD signaling. Oncogene. 2013 Jun 17.doi: 10.1038/onc.2013.215.
[10]Xu Yong, Yang Hua, Qiao Jian-Ou, et al.High-bone-turnover Osteoporosis and Aortic Calcification in Opg Knockout Mice. Progress in Biochemistry and Biophysics.2007;34(3):260-266.
[11]Wang YD, Tao MF, Wang L,et al.Selective regulation of osteoblastic OPG and RANKL by dehydroepiandrosterone through activation of the estrogen receptor β-mediated MAPK signaling pathway.Horm Metab Res.2012;44(7):494-500.
[12]West SL, Lok CE, Jamal SA.Osteoprotegerin and fractures in men and women with chronic kidney disease.J Bone Miner Metab. 2013 Oct 11.
[13]Weitzmann MN.The Role of Inflammatory Cytokines, the RANKL/OPG Axis, and the Immunoskeletal Interface in Physiological Bone Turnover and Osteoporosis.Scientifica (Cairo).2013;2013:125705.
[14]Sorensen MG, Henriksen K, Dziegiel MH,et al. Estrogen directly attenuates human osteoclastogenesis, but has no effect on resorption by mature osteoclasts.DNA Cell Biol. 2006;25:475-483.
[15]Eghbali-Fatourechi G, Khosla S, Sanyal A,et al. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women.J Clin Invest.2003;111:1221-1230.
[16]Hofbauer LC, Schoppet M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases.JAMA 2004;292:490-495.
[17]Proff PR.The molecular mechanism behind bone remodelling a review. Clin Oral Investig.2009;13(4)355-362.
[18]Hopwood B, Tsykin AFindlay DM,et al.Microarray gene expression profiling of osteoarthritic bone suggests altered bone remodelling WNT and transforming growth factorbeta/ bone morphogenic protein signalling J.Arthritis Res Ther. 2007; 9(5):100-120.
[19]Tanko LB, Sondergaard BC, Oestergaard S,et al. An update review of cellular mechanisms conferring the indirect and direct effects of estrogen on articular cartilage. Climacteric. 2008;11:4-16.
[20]Jilka RL, Hangoc G, Girasole G, et al. Increased osteoclast development after estrogen loss: mediation by interleukin-6. Science.1992;257(5):88-91.
[21]Kimble RB,Srivastava S,Ross FP,et al.Estrogen deficiency increases the ability of stromal cells to support murine osteoclastogenesis via an interleukin-1and tumor necrosis factor-mediated stimulation of macrophage colony-stimulating factor production.J Biol Chem.1996;271(46):28890-28897.
[22]Ray A,Prefontaine KE,Ray P,et al.Down.modulation of interleukin-6 gene expression bv17 beta estradlol in the absence of high affinity DNA binding by the estrogen receptor. J Biol Chem.1994;269(17):12940-12946.
[23]万荣,杨庆铭.邓廉夫,等.不同浓度雌激素对软骨细胞金属蛋白酶 mRNA表达的影响[J].中华风湿病学杂志,2002,6(3):174.7.
[24]RicheRe P,Dumontier MF,Francois M,et al.Dual effects of 17β-oestradiol on interleukin-1β.induced proteoglycan degradation in chondrocytes.Ann Rheum Dis. 2004;63(2): 191-199.
[25]Lee YJ,Lee EB,Kwon YE,et al.Effect of estrogen on the expression of matrix metalloproteinase MMP-1,MMP-3,and MMP-13 and tissue inhibitor of metalloprotemase-1in osteoarthritis chondre cytes.Rheu-matol Intern. 2003:23(6): 282-288.
[26]Ham KD, Loeser RF, Lindgren BR,et al. Effects of long-term estrogen replacement therapy on osteoarthritis severity in cynomolgus monkeys.Arthritis Rheum, 2002;46:1956-1964.
[27]Oestergaard S, Sondergaard BC, Hoegh-Andersen P,et al.Effects of ovariectomy and estrogen therapy on type II collagen degradation and structural integrity of articular cartilage in rats: implications of the time of initiation.Arthritis Rheum 2006;54:2441-2451.
[28]Yang JH, Kim JH, Lim DS,et al.Effect of combined sex hormone replacement on bone/cartilage turnover in a murine model of osteoarthritis.Clin Orthop Surg. 2012;4(3):234-241.
[29]Calvo E, Castaneda S, Largo R, et al.Osteoporosis increases the severity of cartilage damage in an experimental model of osteoarthritis in rabbits.Osteoarthritis Cartilage. 2007;15:69-77.
[30]Ma HL, Blanchet TJ, Peluso D, et al.Osteoarthritis severity is sex dependent in a surgical mouse model.Osteoarthritis Cartilage. 2007;15:695-700.
[31]Christgau S, Tanko LB, Cloos PA,et al.Suppression of elevated cartilage turnover in postmenopausal women and in ovariectomized rats by estrogen and a selective estrogen- receptor modulator (SERM). Menopause. 2004;11:508-518.
[32]Cirillo DJ, Wallace RB, Wu L, et al.Effect of hormone therapy on risk of hip and knee joint replacement in the Women’s Health Initiative.Arthritis Rheum. 2006;54:3194-3204.
[33]Kerin A, Patwari P, Kuettner K,et al.Molecular basis of osteoarthritis: biomechanical aspects.Cell Mol Life Sci 2002; 59:27-35.
[34]Michael H, Harkonen PL, Kangas L,et al.Differential effects of selective oestrogen receptor modulators (SERMs) tamoxifen, ospemifene and raloxifene on human osteoclastsin vitro. Br J Pharmacol.2007;151:384-395. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [4] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [5] | Zheng Xiaolong, He Xiaoming, Gong Shuidi, Pang Fengxiang, Yang Fan, He Wei, Liu Shaojun, Wei Qiushi. Bone turnover characteristics in patients with alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 657-661. |
| [6] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [7] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [8] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [9] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [10] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [11] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [12] | Lü Jiaxing, Bai Leipeng, Yang Zhaoxin, Miao Yuesong, Jin Yu, Li Zhehong, Sun Guangpu, Xu Ying, Zhang Qingzhu. Evaluation of internal fixation with proximal femoral nail antirotation in elderly knee osteoarthritis patients with femoral intertrochanteric fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 391-396. |
| [13] | Zheng Li, Li Dadi, Hu Weifan, Tang Jinlong, Zhao Fengchao. Risk assessment of contralateral knee arthroplasty after unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 374-379. |
| [14] | Liu Jinfu, Zeng Ping, Nong Jiao, Fan Siqi, Feng Chengqin, Huang Jiaxing. Integrative analysis of biomarkers and therapeutic targets in synovium of patients with osteoarthritis by multiple microarrays [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3690-3696. |
| [15] | Chen Feng, Zhang Xiaoyun, Chen Yueping, Liao Jianzhao, Li Jiajun, Song Shilei, Lai Yu. Molecular mechanism of anhydroicaritin in the treatment of osteoarthritis: an analysis based on network pharmacology and bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3704-3710. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||