Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (24): 3863-3869.doi: 10.3969/j.issn.2095-4344.2014.24.016
Previous Articles Next Articles
Nerve growth factor promotes the expression of vascular endothelial growth factor in the fracture healing process
Liu Yu-peng1, 2, Zhao De-wei2, Wang Wei-ming2, Zhang Yao2, Li Fang2, Liu Zhen-gang2
- 1 Department of Biomedical Engineering, Dalian University of Technology, Dalian 116024, Liaoning Province, China
2 Department of Orthopaedic Surgery, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China
-
Revised:2014-05-19Online:2014-06-11Published:2014-06-11 -
Contact:Liu Yu-peng, Master, Chief physician, Department of Biomedical Engineering, Dalian University of Technology, Dalian 116024, Liaoning Province, China; Department of Orthopaedic Surgery, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
About author:Liu Yu-peng, Master, Chief physician, Department of Biomedical Engineering, Dalian University of Technology, Dalian 116024, Liaoning Province, China; Department of Orthopaedic Surgery, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China
CLC Number:
Cite this article
Liu Yu-peng, Zhao De-wei, Wang Wei-ming, Zhang Yao, Li Fang, Liu Zhen-gang. Nerve growth factor promotes the expression of vascular endothelial growth factor in the fracture healing process[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(24): 3863-3869.
share this article
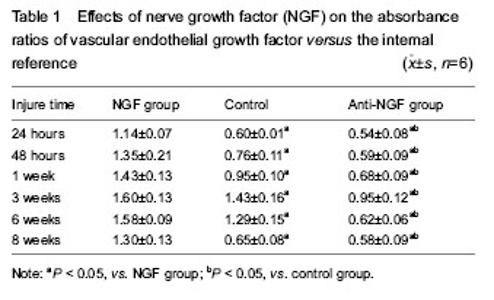
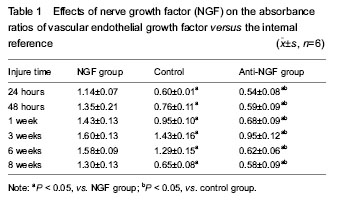
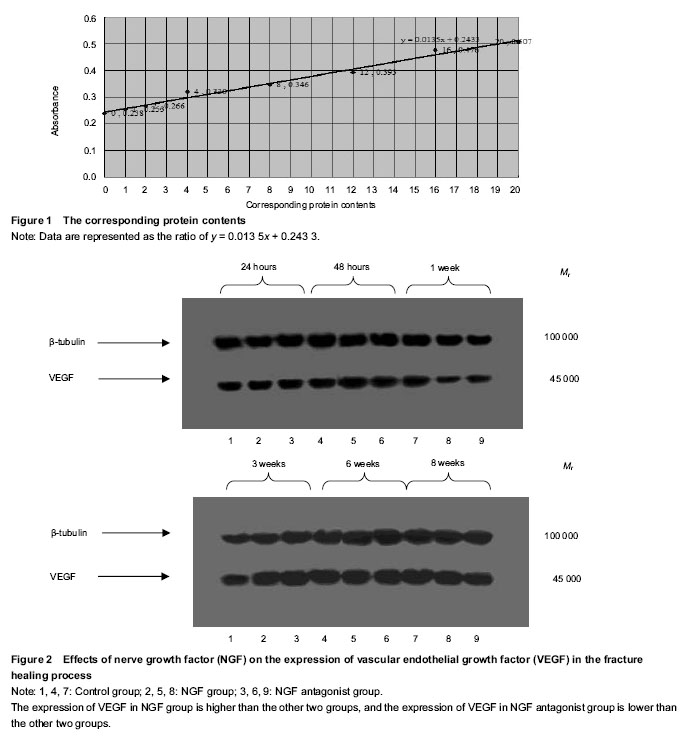
Quantitative analysis of experimental animals All experimental rabbits were involved in the results analysis, without any loss. General observation All rabbits in the three groups survived with no deaths and ate properly. The surgical incisions exhibited no signs of infection or purulence and healed at approximately 1 postoperative week. At 24 hours postoperatively, the samples in the three groups all exhibited subcutaneous swelling at the incision sites, with no hematomas formed at the fracture sites. At 48 hours postoperatively, initial hematomas at the fracture sites were formed in all three groups. At 1 week postoperatively, hematoma organization was formed at the fracture sites (NGF group > control group > NGF antagonist group), with discontinuous fractures. At 3 weeks postoperatively, continuous fractures were observed, with primary callus formation, and strong bony connections were formed at the fracture sites (NGF group > control group > NGF antagonist group). At 6 weeks postoperatively, the callus sizes had shrunk compared with the primary calluses (NGF group < control group < NGF antagonist group). At 8 weeks postoperatively, the NGF group and the control group showed no obvious fracture lines, while the NGF antagonist group still showed a small amount of callus. Western blot analysis and statistical analysis results Images of immunoblots showing VEGF expression with an internal reference in the bone tissues at different time points in the three groups are shown in Figure 2. The absorbance ratios and the means with standard deviations of VEGF target protein expression versus the internal reference in the bone tissues at different time points in the three groups are shown in Table 1, and their changing trend over time is displayed in Figure 2. Finally, a statistical analysis of variance was conducted for the absorbance ratios of VEGF expression versus the internal reference in the bone tissues at different time points in the three groups (Table 1). In the fracture-healing process, VEGF expression in the callus continuously increased, reaching a peak at postoperative 3 weeks, and then gradually decreased (Figure 2). VEGF expression in the callus was higher in the NGF group than in the other two groups, while VEGF expression in the callus was lower in the NGF antagonist group than in the other two groups (Table 1). The one-way analysis of variance for the absorbance ratios of the target protein VEGF versus the internal reference protein in each group revealed that, compared with the NGF antagonist group and the control group, the results of the NGF group were higher at all time points, with statistically significant differences (P < 0.05). These results indicate that NGF had a positive role in promoting VEGF expression at the fracture site. The differences between the NGF antagonist group and the control group at the 24- and 48-hour and the 8-week time points were not statistically significant (P > 0.05), which may indicate that the NGF antibody concentrations had not yet reached effective levels at the early stages of fracture healing and that the anti-NGF antibody concentration was completely attenuated at the late stage of fracture healing (Table 1)."
| [1] Street J, Bao M, Leo de Guzman, et al. Vascular endothelial growth factor stimulates bone repair by promoting angiogenesis and bone turnover. Proc Natl Acad Sci U S A. 2002;99(15):9656-9661. [2] Tong G, Lei J, Zhong C, Cai W, Lin X. The curative effect and safety of collagen sponge with different pH and content of protein implanted into orthopedic bone defect. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2012;29(6): 1125-1130. [3] Lyons FG, Gleeson JP, Partap S, et al. Novel microhydroxyapatite particles in a collagen scaffold: a bioactive bone void filler? Clin Orthop Relat Res. 2014; 472(4):1318-1328. [4] Hou J, Wang J, Cao L, et al. Segmental bone regeneration using rhBMP-2-loaded collagen/chitosan microspheres composite scaffold in a rabbit model. Biomed Mater. 2012; 7(3):035002. [5] Fan BS, Lou JY. Enhancement of angiogenic effect of co-transfection human NGF and VEGF genes in rat bone marrow mesenchymal stem cells. Gene. 2011;485(2): 167-171. [6] Zhang YG, Lu SB, Wang JF, et al. Experimental study of expression of BMP, TGF-β and bFGF in fracture healing. Zhonghua Chuangshang Zazhi. 2000;3(16):170-172. [7] Einhorn TA. The science of fracture healing. J Orthop Trauma. 2005;19(10S):S4-S6. [8] Liu YP, Zhao DW, Lu DC, et al. Expression of nerve growth factors in the fractured bone of rat. Zhongguo Linchuang Kangfu. 2006;0(17):107-109. [9] Sisask G, Bjurholm A, Ahmed M, et al. Ontogeny of sensory nerves in the developing skeleton. Anat Rec. 1995;243: 234-240. [10] Sisask G, Silfverswärd CJ, Bjurholm A, et al. Ontogeny of sensory and autonomic nerves in the developing mouse skeleton. Auton Neurosci. 2013;177(2):237-243. [11] Yasui M, Shiraishi Y, Ozaki N, et al. Nerve growth factor and associated nerve sprouting contribute to local mechanical hyperalgesia in a rat model of bone injury. Eur J Pain. 2012; 16(7):953-965. [12] Mo Y, Yang Z, Zhao J, et al. Preliminary study on appropriate concentration gradient of nerve growth factor in promoting fracture healing. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2011;25(5):581. [13] Eppley BL, Snyders RV, Windelmann T, et al. Effects of nerve growth factor on craniofacial onlay bone graft survival: preliminary findings. J Craniofac Surg. 1992;2(4):174-180. [14] Dobnig H, Sipos A, Jiang Y, et al. Early changes in biochemical markers of bone formation correlate with improvements in bone structure during teriparatide therapy. J Clin Endocrinol Metab. 2005;90(7):3970-3977. [15] Obrant KJ, Ivaska KK, Gerdhem P, et al. Biochemical markers of bone turnover are influenced by recently sustained fracture. J Bone. 2005;36(5):786-792. [16] Nakanishi T, Takahashi K, Aoki C, et al. Expression of nerve growth factor family neurotrophins in a mouse osteoblastic cell line. Biochem Biophys Res Commun. 1994; 198(3): 891-897. [17] Chim SM, Tickner J, Chow ST, et al. Angiogenic factors in bone local environment. Cytokine Growth Factor Rev. 2013;24(3):297-310. [18] Yasko AW, Lane JM, Fellinger EJ, et al. The healing of segmental bone defects, induced by recombinant human bone morphogenetic protein (rh-BMP). J Bone Joint Surg (Am). 1992;74:659. [19] Hayami T, Funaki H, Yaoeda K, et al. Expression of the cartilage derived anti-angiogenic factor chondromodulin-I decreases in the early stage of experimental osteoarthritis. J Rheumatol. 2003;30(10):2207-2217. [20] Brown AP, Courtney CL, King LM, et al. Cartilage dysplasia and tissue mineralization in the rat following administration of a FGF receptor tyrosine kinase inhibitor. Toxicol Pathol. 2005;33(4):449-455. [21] Petersen W, Pufe T, Zantop T, et al. Expression of VEGFR-1 and VEGFR-2 in degenerative Achilles tendons. J Orthop Res. 2004;420(3):286-291. [22] Street JT, Wang JH, Wu QD, et al. The angiogenic response to skeletal injury is preserved in the elderly. J Orthop Res. 2001;19(6):1057-1066. [23] Jimenez-Andrade JM, Ghilardi JR, Castañeda-Corral G, et al. Preventive or late administration of anti-NGF therapy attenuates tumor-induced nerve sprouting, neuroma formation, and cancer pain.Pain. 2011;152(11):2564-2574. [24] Mogi M, Kondo A, Kinparak, et al. Anti-apoptotic action of nerve growth factor in mouse osteoblastic cell line. Life Sci. 2000;67(10):1197-1206. [25] Zhuang YF, Li J. Serum EGF and NGF levels of patients with brain injury and limb fracture. Asian Pac J Trop Med. 2013;6(5):383-386. [26] Iannone F, De Bari C, Dell'Accio F, et al. Increased expression of nerve growth factor (NGF) and high affinity NGF receptor (p140 TrkA) in human osteoarthritic chondrocytes. Rheumatology (Oxford). 2002;41(12): 1413-1418. [27] Grills BL, Schuijers JA, Ward AR. Topical application of nerve growth factor improves fracture healing in rats. J Orthop Res. 1997;15(2):235-242. [28] Jehan F, Naveilhan P, Neveu I, et al. Regulation of NGF, BDNF and LNGFR gene expression in ROS 17/2.8 cells. Molcell Endocrinol. 1996;116(2):149-156. [29] Lazarovici P, Gazit A, Staniszewska I, et al. Nerve growth factor (NGF) promotes angiogenesis in the quail chorioallantoic membrane. Endothelium. 2006;13(1):51-59. [30] Furfaro F1, Ang ES, Lareu RR, et al. A histological and micro-CT investigation in to the effect of NGF and EGF on the periodontal, alveolar bone, root and pulpal healing of replanted molars in a rat model-a pilot study. Prog Orthod. 2014;15:2 [31] Garcia R, Aguiar J, Alberti E, et al. Bone marrow stromal cells produce nerve growth factor and glial cell line-derived neurotrophic factors. Biocjem Biophys Res Commun. 2004; 316(3):753-754. [32] Cao J, Wang L, Lei DL, et al. Local injection of nerve growth factor via a hydrogel enhances bone formation during mandibular distraction osteogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(1):48-53. [33] Cai WX, Zheng LW, Li CL, et al. Effect of different rhbmp-2 and tg-vegf ratios on the formation of heterotopic bone and neovessels. Biomed Res Int. 2014;2014:571510. |
| [1] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [2] | Yang Weiqiang, Ding Tong, Yang Weike, Jiang Zhengang. Combined variable stress plate internal fixation affects changes of bone histiocyte function and bone mineral density at the fractured end of goat femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 890-894. |
| [3] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [4] | Cheng Shigao, , Wang Wanchun, Jiang Dong, Li Tengfei, Li Xun, Ren Lian. Comparison of the standard and long-stem bone cement prosthesis replacement in the treatment of intertrochanteric fractures in elderly patients [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 362-367. |
| [5] | Xie Yang, Lü Zhiyu, Zhang Shujiang, Long Ting, Li Zuoxiao. Effects of recombinant adeno-associated virus mediated nerve growth factor gene transfection on oligodendrocyte apoptosis and myelination in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3678-3683. |
| [6] | Mao Guoshu, Zhou Min, Li Xiaoming, Zhou Zihong, Yin Qudong. Effect of the third fragment on the healing of femoral shaft fractures after intramedullary nailing fixation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2405-2409. |
| [7] | Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279. |
| [8] | Yan Nan, Si Xiaofeng, Zeng Liang, Tian Wei, Shan Guangdong, Xiong Lishuo, Yang Weijie, Wang Zhengdong. Nerve growth factor interferes with proliferation and alpha-actin expression of skeletal muscle satellite cells in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2030-2035. |
| [9] | Wu Shengxiang, Liu Yuan, Lu Shuai. Mini-locking titanium plate system fixation in the treatment of carpal scaphoid fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1874-1878. |
| [10] | Xu Shaoce, Wang Shiyao, Zhou Jianwei, Pan Yixin, Wang Yuliang. Fibroblast growth factor receptor 3 regulation and mechanism in callus formation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1083-1088. |
| [11] | Liu Zhendong, Qin Sihe. Four-dimensional space events of fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(6): 903-910. |
| [12] | Cao Guolong, Tian Faming, Liu Jiayin. Lovastatin combined with insulin effects on fracture healing in rat models of bilateral ovariectomized type 2 diabetic mellitus [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 673-681. |
| [13] | Zhang Chao, Li Xingyong, Ma Guifu, Pu Xingyu, Luo Wenyuan. Hoxa9 silencing promotes tibial fracture healing by regulating osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5600-5606. |
| [14] | Li Yanqiang, Pan Deyue, Nan Feng, Han Xin. Treatment of senile osteoporotic patella fractures with double tension band [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(33): 5338-5342. |
| [15] | Yang Shun, Chen Keyi, Cheng Yabo, Xiang Wang, Zhang Jing, Gu Hongji, Chi Haotian. Wrist arthroscopy-assisted titanium internal fixator for the treatment of complex distal radius fractures [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 366-371. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||