Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (7): 991-996.doi: 10.3969/j.issn.2095-4344.2014.07.002
Previous Articles Next Articles
Large vascularized tissue-engineered bone for repair of bone defects
Li Yao-hua
- Department of Orthopedics, the Second Affiliated Hospital of Hebei North University, Zhangjiakou 075100, Hebei Province, China
-
Revised:2013-12-01Online:2014-02-12Published:2014-02-12 -
About author:Li Yao-hua, Attending physician, Department of Orthopedics, the Second Affiliated Hospital of Hebei North University, Zhangjiakou 075100, Hebei Province, China -
Supported by:the Supporting Program of the Second Affiliated Hospital of Hebei North University, No. HBBFXY-20120340
CLC Number:
Cite this article
Li Yao-hua. Large vascularized tissue-engineered bone for repair of bone defects[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(7): 991-996.
share this article
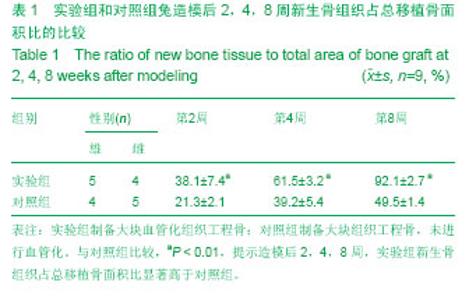
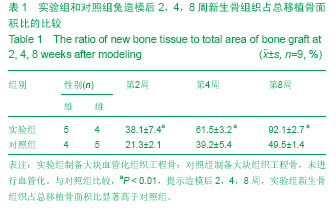
2.1 实验动物数量分析 造模未造成动物死亡,进入结果分析最终为18只兔,无脱失,实验组和对照组各9只。 2.2 大体观察 造模后2周时,实验组新生组织软骨层内充满不成熟软骨样组织,胶原纤维杂乱排列,可以观察到明显的纤维结缔组织连接;对照组仅有少量骨痂形成。 造模后4周,实验组有大量连续性骨痂形成,并有血管长入支架内,且血管周围形成编织骨;对照组仅有骨痂形成较前有所增多。 造模后8周,实验组大块血管化组织工程骨直径和厚度基本和宿主骨相同,其中有2只比宿主骨还要厚;对照组有大骨痂形成,但是仍然骨不连。 2.3 影像学检查 造模后2周时,实验组X射线摄片大块血管化组织工程骨处可见大片云雾状密度增高影;对照组仅显支架上下端有少许云雾状密度增高影。 造模后4周,实验组有X射线摄片可见大块血管化组织工程骨周围有片状阻射影,骨连接处界线模糊;对照组云雾状密度增高影有所增多,但是骨连接处界线清晰。 造模后8周,实验组X射线摄片可见大块血管化组织工程骨显骨性连续性连接,为连续性高密度影;对照组有大片状密度增高影,骨连接处界线仍然模糊可见。 2.4 组织学检查 造模后2周时,实验组可见大量毛细血管增生周边可见新生骨小梁,大块血管化组织工程骨周围有丰富的纤维组织;对照毛细血管少,且较短小未能长入支架中,新生骨小梁也明显少于实验组,支架周围纤维组织也少。 造模后4周,实验组工程骨周围血管丰富,基本布满整个工程骨,纤维组织下有大量新生骨组织,并可见大量活跃的成骨细胞;对照毛细血管较前增多,周围有少量新骨形成。 造模后8周,实验组的大块血管化组织工程骨已经和宿主骨被骨组织桥接,并逐渐形成板成骨内有髓腔样结构;对照组也有较多成骨细胞核新生骨,但是和实验组比较差别明显。 2.5 新生骨组织占总移植骨的面积比 通过对比造模后2,4,8周新生骨组织占总移植骨面积比,2组之间差异有显著性意义(P < 0.01,表1)。"
| [1] 杨强,彭江,卢世璧,等.组织工程骨软骨复合体的研究进展及展望[J]. 中国矫形外科杂志,2008,16(8): 606-609.[2] Naito Y, Shinoka T, Duncan D, et al. Vascular tissue engineering: towards the next generation vascular grafts. Adv Drug Deliv Rev. 2011;163(4-5):312-323.[3] Kanczler JM, Oreffo RO. Osteogenesis and angiogenesis: the potential for engineering bone. Eur Cell Mater. 2008;15:100-114.[4] Zhou M, Peng X, Mao C, et al. Primate mandibular reconstruction with prefabricated, vascularized tissue-engineered bone flaps and recombinant human bone morphogenetic protein-2 implanted in situ. Biomaterials. 2010; 31(18):4935-4943. [5] Johnson EO, Troupis T, Soucacos PN. Tissue-engineered vascularized bone grafts: basic science and clinical relevance to trauma and reconstructive microsurgery. Microsurgery. 2011; 31(3):176-182.[6] Kneser U, Schaefer DJ, Polykandriotis E, et al. Tissue engineering of bone: the reconstructive surgeon's point of view. J Cell Mol Med. 2006;10(1):7-19.[7] Zhou J, Lin H, Fang T, et al. The repair of large segmental bone defects in the rabbit with vascularized tissue engineered bone. Biomaterials. 2010;31(6):1171-1179. [8] Brey EM, Uriel S, Greisler HP, et al. Therapeutic neovascularization: contributions from bioengineering. Tissue Eng. 2005;11(3-4):567-584.[9] Frhlich M, Grayson WL, Wan LQ, et al. Tissue engineered bone grafts: biological requirements, tissue culture and clinical relevance. Curr Stem Cell Res Ther. 2008;3(4):254-264.[10] Hibino N, Villalona G, Pietris N, et al. Tissue-engineered vascular grafts form neovessels that arise from regeneration of the adjacent blood vessel. FASEB J. 2011;25(8):2731- 2739. [11] Colton CK. Implantable biohybrid artificial organs. Cell Transplant. 1995;4(4):415-436. [12] Brandi ML, Collin-Osdoby P. Vascular biology and the skeleton. J Bone Miner Res. 2006;21(2):183-192.[13] Correia C, Grayson WL, Park M, et al. In vitro model of vascularized bone: synergizing vascular development and osteogenesis. PLoS One. 2011;6(12):e28352. [14] Koike N, Fukumura D, Gralla O, et al. Tissue engineering: creation of long-lasting blood vessels. Nature. 2004;428 (6979):138-139.[15] Tsigkou O, Pomerantseva I, Spencer JA, et al. Engineered vascularized bone grafts. Proc Natl Acad Sci U S A. 2010; 107(8):3311-3316. [16] Buschmann J, Welti M, Hemmi S, et al. Three-dimensional co-cultures of osteoblasts and endothelial cells in DegraPol foam: histological and high-field magnetic resonance imaging analyses of pre-engineered capillary networks in bone grafts. Tissue Eng Part A. 2011;17(3-4):291-299.[17] Arkudas A, Beier JP, Pryymachuk G, et al. Automatic quantitative micro-computed tomography evaluation of angiogenesis in an axially vascularized tissue-engineered bone construct. Tissue Eng Part C Methods. 2010;16(6): 1503-1514.[18] Beier JP, Horch RE, Hess A, et al. Axial vascularization of a large volume calcium phosphate ceramic bone substitute in the sheep AV loop model. J Tissue Eng Regen Med. 2010; 4(3):216-223.[19] Marquass B,Somerson JS,Hepp P,et al.A novel MSC-seededtriphasic construct for the repair ofosteochondral defects.J Orthop Res. 2010;28(12):1586-1599.[20] 荆立忠,郭秦炜,胡跃林.不同年龄阶段家兔髂软骨组织学特点[J].中国运动医学杂志,2012,31(2):124-128.[21] 王海彬,何伟,袁浩.骨细胞和I型胶原[J].中国中医骨伤科杂志, 2003,11(3):56-59.[22] Deng L Lv J,Pang J,et al.Construction oftissue-engineered OS-teochondral composites and repair of large joint defects in rabbit. J Tissue Eng Regen Med. 2012. [Epub ahead of print].[23] Castro NJ,Hacking SA,Zhang LG.Recent progress in interfacialtissue engineering approaches for osteochondral defects. Ann Biomed Eng. 2012;40(8):1628-1640.[24] Khanarian NL,Haney NM, Burga RA,et al.Afunctional agarose-hydroxyapatite scafold for osteochondral interface regeneration.Biomaterials. 2012;33(21):5247-5258.[25] Rodrigues MT,Lee SJ,Gomes ME,et al.Bilayered constructsaimed at osteochondral strategies: the influence of mediumsupplements in the osteogenic and chondrogenic diferentiation ofamniotic fluid-derived stem cells.Acta Biomater. 2012;8(7):2795-2806.[26] Harichandan A, Buhring HJ. Prospective isolation of human MSC. Best Pract Res Clin Haematol. 2011;24(1):25-36.[27] Nery AA, Nascimento IC, Glaser T, et al. Humanmesenchymal stem cells:from immunophenotyping by flowcytometry to clinical applications. Cytometry A. 2013; 831(1):48-61.[28] Park JS, Shim MS, Shim SH, et al. Chondrogenic potential ofstem cells derived from amniotic fluid,adipose tissue,or bonemarrow encapsulated in fibrin gels containing TGF-β3. Biomaterials. 2011;32(32):8139-8149.[29] Perrier E, Ronziere MC, Bareille R, et al. Analysis of collagenexpression during chondrogenic induction of human bonemarrow mesenchymal stem cells. Biotechnol Lett. 2011; 33(10):2091-2101.[30] Mahnoudifar N, Doran PM. Chondrogenesis and cartilage tissue engineering the longer road to technology development. Trends Biotechnol. 2012;30(3):166-176.[31] 张清林,吕惠成,吴一民.转化生长因子β1联合骨形态发生蛋白2诱导骨髓间充质干细胞体外向软骨细胞的分化[J].中国组织工程研究与临床康复,2010,14(24):4371-4375.[32] Park JS, Yang HN, Woo DG, et al. The promotion ofchondrogenesis, osteogenesis, and adipogenesis of human mesenchymal stem cells by multiple growth factors incorporated into nanosphere-coated microspheres. Biomaterials. 2011;32(1):28-38.[33] Bosetti M, Boccafoschi F, Leigheb M, et al. Chondrogenic induction of human mesenchymal stem cells using combined growth factors for cartilage tissue engineering. J Tissue Eng Regen Med. 2012;6(3):205-213.[34] Hildner F, Concaro S, Peterbauer A, et al. Human adipose-derived stem cells contribute to chondrogenesis in coculture with human articular chondrocytes. Tissue Eng Pt A. 2009;15(12):3961-3969.[35] Mo XT, Guo SC, Xie HQ, et al. Variations in the ratios of co-cultured mesenchymal stem cells and chondrocytes regulate the expression of cartilaginous and osseous phenotype in alginate constructs. Bone. 2009;45(1):42-51.[36] Tew SR, Murdoch AD, Rauchenberg RP, et al. Cellular methods in cartilage research:Primary human chondrocytes in culture and chondrogenesis in human bone marrow stem cells. Methods. 2008;45(1):2-9.[37] Fischer J, Dickhut A, Rickert M, et al. Human articul archondrocytes secrete parathyroid hormone- related protein and inhibit hypertrophy of mesenchymal stem cells in coculture during chondro- genesis. Arthritis Rheum. 2010; 62(9):2696-2706.[38] Chen WH, Lai MT, Wu AT, et al. In vitro stage-specific chondrogenesis of mesenchymal stem cells committed to chondrocytes. Arthritis Rheum. 2009;60(2):450-459.[39] Niyibizi C, Wang S, Mi Z, et al. The fate of mesenchymal stem cells transplanted into immunecompetent neonatal mice:implications for skeletal gene therapy via stem cells. Mol Ther. 2004;9(9):955-963.[40] Quintana L, zur Nieden NI, Semino CE. Morphogenetic and regulatory mechanisms during developmental chondrogenesis: new paradigms for cartilage tissue engineering. Tissue Eng Part B Rev. 2009;15(1):29-41. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||