Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (2): 283-288.doi: 10.3969/j.issn.2095-4344.2014.02.020
Previous Articles Next Articles
Adipose-derived stem cells for repair of urinary system injury: current situation and problems in application
Guo Ting-ting, Li Li-xi, Tian Hong
- Department of Medical Genetics, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China
-
Received:2013-11-26Online:2014-01-08Published:2014-01-08 -
Contact:Tian Hong, M.D., Associate professor, Department of Medical Genetics, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China -
About author:Guo Ting-ting, Studying for master’s degree, Department of Medical Genetics, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China -
Supported by:the National Natural Science Foundation of China, No. 31271043
CLC Number:
Cite this article
Guo Ting-ting, Li Li-xi, Tian Hong. Adipose-derived stem cells for repair of urinary system injury: current situation and problems in application[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(2): 283-288.
share this article
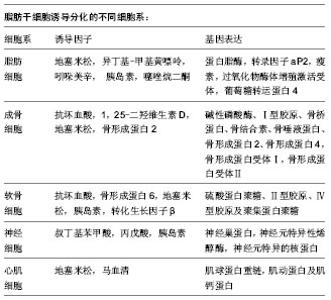
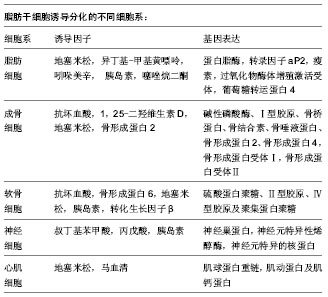
2.1 脂肪干细胞的获取 2.1.1 脂肪干细胞的来源 干细胞最初是在骨髓中分离出来的,随后在脂肪组织、肌肉组织、牙根、胎盘、羊水及脐血中均分离出了干细胞[5-6]。自从2001年Zuk从人脂肪组织中成功分离培养出脂肪源性干细胞,脂肪干细胞以其取材方便的独特优势,逐渐成为人们研究的热点。在人体中脂肪组织可占到正常体质量的10%-29%[7],哺乳动物主要有两种脂肪组织:棕色脂肪组织和白色脂肪组织,虽然二者同属脂肪组织,但却在形态、分布和功能上相差甚远。有研究称棕色脂肪组织仅在婴儿期发挥作用[8],然而最新研究表明在低温环境下成人体内也可产生棕色脂肪细胞,该类细胞可通过高比率燃烧脂肪,保持体温[9]。白色脂肪组织主要分布在腹腔内的网膜、肠、肾周及臀部、大腿、腹部的皮下区域,在人体,白色脂肪组织起储存及释放能量的调节器作用。两类脂肪组织均存在脂肪来源干细胞,然而棕色脂肪组织中干细胞的数量和分化潜能都不如白色脂肪组织中的干细胞[10-11],因此研究重点集中在白色脂肪组织。 脂肪细胞是一种终末分化期细胞,当机体出现细胞坏死或发生细胞凋亡时,脂肪来源干细胞会分化成脂肪细胞补充损失的脂肪细胞[12],研究表明1 g脂肪组织可以提取约5×103个干细胞,是1 g骨髓中提取干细胞数量的100-500倍[13],脂肪干细胞以其易大量获取、提取方法简单且不影响供区功能的优势,成为再生医学理想的种子细胞。 2.1.2 脂肪干细胞的分离培养 脂肪来源干细胞的分离培养在文献中已有很详细描述[14-22],综合多篇文献,以下操作为多见:手术分离脂肪组织,PBS清洗3-5遍,剪碎成2 mm3/小块。用0.1% Ⅰ型胶原酶37 ℃水浴消化1 h,400×g室温离心10 min,弃上清。用PBS重悬洗涤,100 μm滤网滤除未消化的组织,400×g室温离心10 min,弃去上清,培养基重悬并用移液管反复吹打,使之成为单细胞悬液,接种到含体积分数为10%胎牛血清的DMEM培养液中,置 37 ℃、体积分数为5%CO2的饱和湿热培养箱培养。脂肪干细胞是贴壁细胞,原代接种24 h后可见大量细胞贴壁,初呈短梭形或多角形[23],接下来2 d,每天换液1次,并用PBS轻轻洗涤,弃去组织块和未贴壁的细胞,以后每3 d换液1次。48 h后分裂增殖速度加快,细胞体积增大,呈长梭形,形成集落呈涡轮状紧密排列[24]。当细胞达80%- 90%融合时,0.25%胰酶常规消化,进行传代,传代密度为6 700/cm2,传代培养后的细胞增殖速度明显加快。其中值得注意的几个关键因素,如细胞种植密度、血清浓度、氧浓度,它们都能影响到细胞的生长和分化。通常细胞种植密度为(1.0-2.0)×109 L-1,血清体积分数为10%,氧体积分数为21%。 2.2 脂肪干细胞的鉴定 2.2.1 脂肪干细胞的标志物 目前干细胞研究领域认为:脂肪干细胞尚未发现特异性的细胞表面标志物,其既表达其他成体干细胞中广泛表达的标志物如CD44、CD90、CD105,但又不完全和其他成体干细胞相同。研究者们对此做了大量研究,提取的原代脂肪来源干细胞多还混杂有内皮细胞、红细胞和成纤维细胞等细胞群,随着传代次数增加会逐步得到纯化,通常选取传代至第3代的脂肪干细胞进行细胞免疫表型的检测。通过对多篇文献的查阅发现[8, 25-29],脂肪干细胞表达CD9、CD10、CD13、CD29、CD44、CD49d、CD49e、CD54、CD55、CD58、CD59、CD90、CD105、CD146、CD166、SH-3等,其中CD29、CD44、CD90和CD105均高表达[30];不表达CD31、CD34、CD45、HLA-DR,但对CD34和STRO-1的表达尚存在争议[25,31-33]。此外脂肪干细胞所取位置、细胞所处时期、供体的年龄、手术类型、培养环境、细胞密度不同以及供体种的不同都有可能引起细胞表面标志物的差异[32-34],如CD90在人干细胞中高度表达,而在小鼠中不表达,除此之外随着细胞的传代CD90表达也会下降;林立新等[35]研究也发现CD31、CD49d虽然在原代脂肪干细胞呈阳性表达,但在第2代转为阴性表达,CD29、CD105、CD166、Stro-1、Flk-1在第3代脂肪干细胞中的表达量也明显多于原代脂肪干细胞。因此免疫组化和流式细胞仪检测等免疫表型的结果对确认脂肪干细胞也只能起到辅助作用,而鉴定其具有干细胞特性的最好方法是进行诱导分化,并进行相应检测以证明诱导分化成功[36]。 2.2.2 脂肪干细胞的分化潜能 干细胞之所以受到人们的关注,除了其快速增长的特点外,还因为它的增殖分化潜能,在不同细胞因子的诱导下,脂肪干细胞可以分化成脂肪细胞、成骨细胞、软骨细胞、肌细胞等,除此之外有研究发现脂肪干细胞经相应诱导因子的诱导后,检测到神经细胞、心肌细胞标志物的表达[37]。Gentile等[19]对脂肪来源干细胞诱导分化的常见不同细胞系进行了总结。"
| [1] Yoo JJ, Olson J, Atala A,et al. Regenerative medicine strategies for treating neurogenic bladder.Int Neurourol J. 2011;15(3):109-119.[2] Atala A.Tissue engineering of human bladder.Br Med Bull. 2011;97:81-104. [3] Petrovic V, Stankovic J, Stefanovic V.Tissue engineering of the urinary bladder: current concepts and future perspectives.ScientificWorldJournal. 2011;11:1479-1488.[4] 张钦,徐月敏.脂肪干细胞在组织工程技术治疗泌尿系统疾病中的研究现状[J].中国修复重建外科杂志,2011,25(4): 494-497.[5] Bianco P, Robey PG, Simmons PJ. Mesenchymal stem cells: revisiting history, concepts, and assays.Cell Stem Cell. 2008 ; 2(4):313-319. [6] Shin S, Kim Y, Jeong S,et al.The therapeutic effect of human adult stem cells derived from adipose tissue in endotoxemic rat model.Int J Med Sci. 2013;10(1):8-18.[7] Kahn CR. Medicine. Can we nip obesity in its vascular bud. Science. 2008;322(5901):542-543. [8] 徐巧瑜.人脂肪干细胞(ADSCs)体外培养的研究及应用进展[J].中国美容医学, 2012, 21(7): 1265-1267.[9] Wang QA, Tao C, Gupta RK,et al.Tracking adipogenesis during white adipose tissue development, expansion and regeneration.Nat Med. 2013;19(10):1338-1344.[10] Prunet-Marcassus B, Cousin B, Caton D,et al. From heterogeneity to plasticity in adipose tissues: site-specific differences.Exp Cell Res. 2006;312(6):727-736. [11] Gimble J, Guilak F.Adipose-derived adult stem cells: isolation, characterization, and differentiation potential.Cytotherapy. 2003;5(5):362-369.[12] Szöke K, Brinchmann JE.Concise review: therapeutic potential of adipose tissue-derived angiogenic cells.Stem Cells Transl Med. 2012;1(9):658-667. [13] Kitagawa Y, Korobi M, Toriyama K, et al. History of discovery of human adipose-derived stem cells and their clinical application. Jpn J Plast Reconstr Surg. 2006;49: 1097-1104.[14] Zachar V, Rasmussen JG, Fink T.Isolation and growth of adipose tissue-derived stem cells.Methods Mol Biol. 2011; 698:37-49.[15] Williams KJ, Godke RA, Bondioli KR. Isolation and culture of porcine adipose tissue-derived somatic stem cells.Methods Mol Biol. 2011;702:77-86.[16] Levi B, James AW, Nelson ER,et al. Human adipose derived stromal cells heal critical size mouse calvarial defects.PLoS One. 2010;5(6):e11177.[17] Yu G, Floyd ZE, Wu X,et al. Isolation of human adipose-derived stem cells from lipoaspirates.Methods Mol Biol. 2011;702:17-27. [18] De Ugarte DA, Alfonso Z, Zuk PA,et al. Differential expression of stem cell mobilization-associated molecules on multi-lineage cells from adipose tissue and bone marrow. Immunol Lett. 2003;89(2-3):267-270.[19] Gentile P, Orlandi A, Scioli MG,et al. Concise review: adipose-derived stromal vascular fraction cells and platelet-rich plasma: basic and clinical implications for tissue engineering therapies in regenerative surgery.Stem Cells Transl Med. 2012;1(3):230-236. [20] Ji W, Zhang Y, Hu S,et al. Biocompatibility study of a silk fibroin-chitosan scaffold with adipose tissue-derived stem cells in vitro.Exp Ther Med. 2013;6(2):513-518.[21] 郭常敏,王达利,魏在荣,等. 人脂肪间充质干细胞体外培养鉴定与诱导分化的初步研究[J]. 遵义医学院学报,2013,36(1): 32-36.[22] 陆艳卉,元小冬,欧亚,等.成人脂肪基质细胞体外扩增生长和超微结构[J].中国组织工程研究,2013,17(10):1724-1729.[23] 马丽,黄鲁刚,林苹.体外诱导脂肪干细胞向内皮细胞及成血管的分化[J].中国组织工程研究,2013,17(10): 1730-1735.[24] 房艳,李德华,倪伟民,等.人脂肪源性干细胞多潜能分化特性[J].解剖科学进展,2013,19(3): 213-217.[25] Silva WA Jr, Covas DT, Panepucci RA,et al.The profile of gene expression of human marrow mesenchymal stem cells.Stem Cells. 2003;21(6):661-669.[26] Gronthos S, Franklin DM, Leddy HA,et al.Surface protein characterization of human adipose tissue-derived stromal cells.J Cell Physiol. 2001;189(1):54-63.[27] Zuk PA, Zhu M, Ashjian P,et al. Human adipose tissue is a source of multipotent stem cells.Mol Biol Cell. 2002;13(12): 4279-4295.[28] Aust L, Devlin B, Foster SJ,et al. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy. 2004;6(1):7-14.[29] Rodriguez AM, Pisani D, Dechesne CA,et al. Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse.J Exp Med. 2005;201(9):1397-1405.[30] 相新新,赵晶,李茵,等.脂肪干细胞定向分化的影响因素[J].生理科学进展,2010,41(5): 341-346.[31] Takemitsu H, Zhao D, Yamamoto I,et al. Comparison of bone marrow and adipose tissue-derived canine mesenchymal stem cells.BMC Vet Res. 2012;8:150. [32] Lin CS, Xin ZC, Deng CH,et al. Defining adipose tissue-derived stem cells in tissue and in culture.Histol Histopathol. 2010;25(6):807-815.[33] Rodriguez AM, Pisani D, Dechesne CA,et al.Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse.J Exp Med. 2005;201(9):1397-1405.[34] Cawthorn WP, Scheller EL, MacDougald OA. Adipose tissue stem cells meet preadipocyte commitment: going back to the future.J Lipid Res. 2012;53(2):227-246.[35] 林立新,黄勇,袁继龙,等.人脂肪来源干细胞部分生物学性状的实验研究[J].中国美容整形外科杂志, 2013, 24(4): 239-242.[36] 李战强,黄友章,彭勤建,等.成人脂肪来源间充质干细胞的分离培养与鉴定[J].第四军医大学学报,2006,27(1):52.[37] Mizuno H, Hyakusoku H.Fat grafting to the breast and adipose-derived stem cells: recent scientific consensus and controversy.Aesthet Surg J. 2010;30(3):381-387. [38] Park SH, Sim WY, Min BH,et al. Chip-based comparison of the osteogenesis of human bone marrow- and adipose tissue-derived mesenchymal stem cells under mechanical stimulation. PLoS One. 2012;7(9):e46689.[39] 王之发,翁雁鸣,刘彦普,等. 兔骨髓干细胞和脂肪干细胞增殖和分化能力的比较[J]. 现代生物医学进展,2013,13(11):2026- 2030.[40] Chhabra P, Brayman KL.The use of stem cells in kidney disease.Curr Opin Organ Transplant. 2009;14(1):72-78. [41] Ebrahimi B, Eirin A, Li Z,et al. Mesenchymal stem cells improve medullary inflammation and fibrosis after revascularization of swine atherosclerotic renal artery stenosis.PLoS One. 2013;8(7):e67474.[42] 王巍巍,姜燕,王葳,等.低氧诱导因子1α基因修饰脂肪干细胞修复小鼠急性肾损伤[J]. 中国组织工程研究, 2012,16(41): 7651-7657.[43] Kolios G, Moodley Y.Introduction to stem cells and regenerative medicine.Respiration. 2013;85(1):3-10.[44] Matsunuma H, Kagami H, Narita Y,et al. Constructing a tissue-engineered ureter using a decellularized matrix with cultured uroepithelial cells and bone marrow-derived mononuclear cells.Tissue Eng. 2006;12(3):509-518.[45] Shen J, Fu X, Ou L,et al. Construction of ureteral grafts by seeding urothelial cells and bone marrow mesenchymal stem cells into polycaprolactone-lecithin electrospun fibers.Int J Artif Organs. 2010;33(3):161-170.[46] W?odarski KH, W?odarski P, Galus R,et al. Adipose mesenchymal stem cells. Their characteristics and potential application in tissue repair.Pol Orthop Traumatol. 2012;77: 97-99.[47] Rodríguez LV, Alfonso Z, Zhang R,et al. Clonogenic multipotent stem cells in human adipose tissue differentiate into functional smooth muscle cells.Proc Natl Acad Sci U S A. 2006;103(32):12167-12172.[48] Liu J, Huang J, Lin T,et al. Cell-to-cell contact induces human adipose tissue-derived stromal cells to differentiate into urothelium-like cells in vitro.Biochem Biophys Res Commun. 2009;390(3):931-936.[49] 张明,周哲,周娟,等.人脂肪干细胞与猪尿路上皮细胞隔离共培养后向尿路上皮样细胞分化[J]. 中华临床医师杂志:电子版, 2012, 6(8):2099-2102.[50] Zhang H, Qiu X, Shindel AW,et al. Adipose tissue-derived stem cells ameliorate diabetic bladder dysfunction in a type II diabetic rat model.Stem Cells Dev. 2012;21(9):1391-1400.[51] Salem SA, Hwie AN, Saim A,et al. Human adipose tissue derived stem cells as a source of smooth muscle cells in the regeneration of muscular layer of urinary bladder wall.Malays J Med Sci. 2013;20(4):80-87.[52] 朱卫东,徐月敏,冯超,等.脂肪干细胞复合膀胱脱细胞基质促进膀胱再生的实验研究[J].中华泌尿外科杂志, 2012, 33(2):111-116. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zeng Zhen, Hu Jingwei, Li Xuan, Tang Linmei, Huang Zhiqiang, Li Mingxing. Quantitative analysis of renal blood flow perfusion using contrast-enhanced ultrasound in rats with hemorrhagic shock during resuscitation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1201-1206. |
| [5] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [6] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [7] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [8] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [9] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [10] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [11] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [12] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [13] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||