Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (41): 7241-7247.doi: 10.3969/j.issn.2095-4344.2013.41.009
Previous Articles Next Articles
Selective regeneration of rat femoral nerve after crush injury and freeze injury
Aikeremujiang•Muheremu1, 2, Cao Peng1, Wu Zhong-yan1, Wang Xin-ling1, Jia Min1
- 1Department of Orthopedics, the Fifth Hospital Affiliated to Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China; 2Beijing Jishuitan Hospital of Tsinghua University, Beijing 100035, China
-
Received:2013-07-24Revised:2013-08-05Online:2013-10-08Published:2013-11-01 -
Contact:Wu Zhong-yan, Chief physician, Department of Orthopedics, the Fifth Hospital Affiliated to Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China wzyfah@163.com -
About author:Aikeremujiang?Muheremu☆, Studying for doctorate, Physician, Department of Orthopedics, the Fifth Hospital Affiliated to Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China; Beijing Jishuitan Hospital of Tsinghua University, Beijing 100035, China taklimakan321@hotmail.com -
Supported by:Scientific Research and Innovation Fund of Xinjiang Medical University, No. XJC2012122*, XJC2012117*, XJC2012123*
CLC Number:
Cite this article
Aikeremujiang•Muheremu, Cao Peng, Wu Zhong-yan, Wang Xin-ling, Jia Min. Selective regeneration of rat femoral nerve after crush injury and freeze injury[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(41): 7241-7247.
share this article

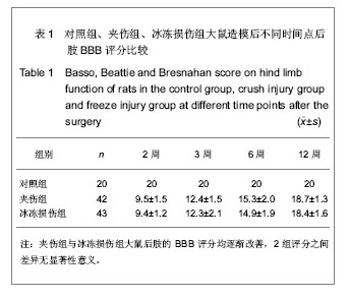
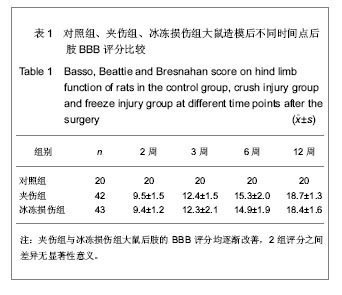
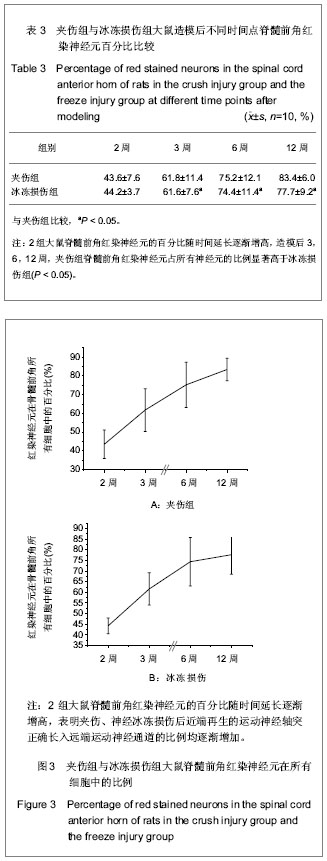
各组大鼠分别于造模后2,3,6,12周根据BBB评分量表进行行为学评分。 夹伤组与冰冻损伤组大鼠术侧后腿活动范围均缩小,伸腿功能均受限。 夹伤组与冰冻损伤组大鼠后肢BBB评分造模后随时间延长而增加,夹伤组与冰冻损伤组的BBB评分显著低于对照组(P < 0.05),夹伤组与冰冻损伤组之间各时间点BBB评分差异无显著性意义(P > 0.05)。 2.3 电生理检测 术侧电极刺激吻合口近端神经,对侧刺激相对应位置。于股四头肌处可记录到运动诱发电位。 夹伤组潜伏期为(1.78±0.33) ms,波幅(4.21± 0.87) mV,对照组潜伏期(1.66±0.25) ms,波幅(5.83±1.20) mV;冰冻损伤组潜伏期(1.80±0.41) ms,波幅(4.81±1.52) mV。 与对照组大鼠正常神经比较,夹伤组与冰冻损伤组的潜伏期延长(P < 0.05),波幅降低(P < 0.05),但夹伤组与冰冻损伤组大鼠之间差异无显著性意义(P > 0.05)。 2.4 逆行示踪 脊髓冰冻切片后,置于荧光显微镜下,在不同颜色的激发光下,观察到对照组中脊髓前角仅有红色标记的运动神经元,见图1;而夹伤组与冰冻损伤组可同时观察到红色、蓝色以及紫色染的神经元,见图2及表2。"

| [1] Kehoe S, Zhang XF, Boyd D. FDA approved guidance conduits and wraps for peripheral nerve injury: a review of materials and efficacy. Injury. 2012;43(5):553-572.
[2] Lin MY, Manzano G, Gupta R. Nerve allografts and conduits in peripheral nerve repair. Hand Clin. 2013;29(3): 331-348.
[3] Ruiter G, Malessy M, Alaid A. Misdirection of regenerating motor axons after nerve injury and repair in the rat sciatic nerve model. Exp Neurol . 2011:(28): 339-350.
[4] Kou Y, Peng J, Wu Z, et al. Small gap sleeve bridging can improve the accuracy of peripheral nerve selective regeneration. Artif Cells Nanomed Biotechnol. 2013. [Epub ahead of print]
[5] Bongso A, Fong CY. The Therapeutic Potential, Challenges and Future Clinical Directions of Stem Cells from the Wharton's Jelly of the Human Umbilical Cord. Stem Cell Rev. 2013;9(2):226-240.
[6] Popovich PG, Tovar CA, Wei P, et al. A reassessment of a classic neuroprotective combination therapy for spinal cord injured rats. Exp Neurol. 2012;233(2):677-685.
[7] Behera D, Behera S, Jacobs KE, et al. Bilateral peripheral neural activity observed in vivo following unilateral nerve injury. Am J Nucl Med Mol Imaging. 2013;3(3): 282-290.
[8] Kim JR, Oh SH, Kwon GB, et al. Acceleration of Peripheral Nerve Regeneration through Asymmetrically Porous Nerve Guide Conduit Applied with Biological/Physical Stimulation. Tissue Eng Part A. 2013.[Epub ahead of print]
[9] Ring D. Symptoms and disability after major peripheral nerve injury. Hand Clin. 2013;29(3):421-425.
[10] Zaverucha-do-Valle C, Gubert F, Bargas-Rega M, et al. Bone marrow mononuclear cells increase retinal ganglion cell survival and axon regeneration in the adult rat. Cell Transplant. 2010;20:391-406.
[11] Allodi I, Udina E, Navarro X. Specificity of peripheral nerve regeneration: interactions at the axon level. Prog Neurobiol. 2012;98(1):16-37. [12] Ryan JN, Scott L, Zin ZK, et al. Functional characterization of optimized acellular peripheral nerve graft in a rat sciatic nerve injury model. Neurol Res. 2011;33(6):600-608.
[13] Ruiter G, Malessy M, Alaid A. Misdirection of regenerating motor axons after nerve injury and repair in the rat sciatic nerve model. Exp Neurol. 2008;211:339-350.
[14] Eng L, Tsang S, Yasuhiko T, et al. Genetically-manipulated adult stem cells as therapeutic agents and gene delivery vehicle for wound repair and regeneration. J Control Release. 2012; 157(3):321-330.
[15] Ratzka A, Baron O, Stachowiak MK, et al. Fibroblast growth factor 2 regulates dopaminergic neuron development in vivo. J Neurochem. 2012;122(1):94-105.
[16] Ebert AD, Laussmann M, Wegehingel S, et al. Tec-kinase- mediated phosphorylation of fibroblast growth factor 2 is essential for unconventional secretion. Traffic. 2010;11: 813-826.
[17] Ilary A, Laura CD, Eva SN, et al. FGF-2 Low Molecular Weight Selectively Promotes Neuritogenesis of Motor Neurons In Vitro. Mol Neurobiol. 2013;47(2): 770-781.
[18] Madison RD, Robinson GA,Chadaram SR. The speci?city of motor neurone regeneration (preferential reinnervation). Acta Physiol. 2007;189: 201-206.
[19] Lundborg G. A 25-year perspective of peripheral nerve surgery: evolving neuroscientific concepts and clinical significance. J Hand Surg. 2000;25(3):391-414.
[20] Cajal RS. Experiments dealing with the transplantation of nerves and their products designed to prove especially an attractive or neurotrophic action on nerve sprouts. Degeneration and Regeneration of the Nervous System. New York, NY: Hafner Press, 1928: 329-361.
[21] Forssman J. Ueber de Ursachen, Welche die wachstumsachtung der peripheren nervenfasern bei der regeneration bestimmen. Beitp Pathol Anat. 1898; 24: 55-100.
[22] Weiss P, Taylor AC. Histiomechanical analysis of nerve reunion in the rat after tubular splicing. Arch Surg. 1943; 47: 419-447.
[23] Weiss P, Taylor AC. Further experimental evidence against “neurotrophism” in nerve regeneration. J Exp Zool. 1944; 95: 233-257.
[24] Lundborg G, Dablin L, Danielsen N, et al. Tissue specificity in nerve regeneration. Scan J Plast Reconstr Surg. 1986; 20: 279-283.
[25] Politis MJ. Specificity in mammalian peripheral nerve regeneration at the level of the nerve trunk. Brain Res. 1985; 328: 271-276.
[26] Politis MJ, Ederle K, Spencer PS. Tropism in nerve regeneration in vivo. Attraction of regenerating axons by diffusible factors derived from cells in distal nerve stumps of transected peripheral nerves. Brain Res. 1982; 253: 1-12.
[27] Mackinnon S, Dellon L, Lundborg G, et al. A study of neurotrophism in a primate model. J Hand Surg. 1986;11A: 888-896.
[28] Seckel BR, Chiu TH, Nyilas E, et al. Nerve regeneration through synthetic biodegradable nerve guides: regulation by the target organ. Plast Reconstr Surg. 1984; 74: 173-180.
[29] Brunelli G, Fontana G, Jager C, et al. Chemotactic arrangement of axons inside and distal to a venous graft. J Reconstr Microsurg. 1987; 3(2): 87-93.
[30] Brushart TME. Motor axons preferentially reinnervate motor pathways. J Neurosci. 1993; 13: 2730-2738.
[31] Chiu DT, Janecka I, Krizek TJ, et al. Autogenous vein graft as a conduit for nerve regeneration. Surgery. 1982; 91: 226-232.
[32] Chiu DT, Lovelace RE, Yu LT, et al. Comparative electrophysiologic evaluation of nerve grafts and autogenous vein grafts as nerve conduits: an experimental study. J Reconstr Microsurg. 1988; 4(4): 303-310.
[33] Chiu DT, Strauch B. A prospective clinical evaluation of autogenous vain grafts used as a nerve conduit for distal sensory nerve defects of 3 cm or less. Plast Reconstr Surg. 1990; 86: 928-934.
[34] Coughlin MD. Target organ stimulation of parasympathetic nerve growth in the developing mouse submandibular gland. Dev Biol. 1975; 43(1): 140-158.
[35] Politis MJ, Spencer PS. An in vivo assay of neurotropic activity. Brain Res. 1983; 278(1-2): 229-231.
[36] Madison RD, Robinson GA,Chadaram SR. The speci?city of motor neurone regeneration (preferential reinnervation). Acta Physiol. 2007;189:201-206.
[37] Hoke A, Redett R, Hameed H, et al. Schwann cells express motor and sensory phenotypes that regulate axon regeneration. J Neurosci. 2006;26(38):9646-9655.
[38] Anjan KD, Renjitha G, Ishwar P. Current Status and Prospective Application of Stem Cell-Based Therapies for Spinal Cord Injury. Curr Stem Cell Res Ther. 2011;6:93-104.
[39] Uccelli A, Benvenuto F, Laroni A, et al. Neuroprotective features of mesenchymal stem cells. Best Pract Res Clin Haematol. 2011;24(1):59-64.
[40] Yu W, Zhe Z, Zhiwu R, et al. Recellularized nerve allografts with differentiated mesenchymal stem cells promote peripheral nerve regeneration. Neurosci Lett. 2012;514(1): 96-101.
[41] Tsubokawa N, Maki Y, Yoshizu T, et al. Comparison of the neurotropic effects of motor and sensory Schwann cells during regeneration of peripheral nerves. Scand J Plast Reconstr Hand Surg.1999;33:379-385. |
| [1] | Liu Wei, Huang Jian. Applied research and progress of three-dimensional printing technology in joint replacement [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1123-1130. |
| [2] | Yang Bao-jia, Yang Kai-shun, Yao Ru-bin. Correlation of nicotine dose and lumbar posterolateral fusion rate: imaging and biomechanical testing [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(48): 7251-7260. |
| [3] | Wu Xiao-guang, Qiu Zhi-fu, Meng Jie, Zu Bing-xue, Li Meng-meng, Miao Hui. Effects of Buyanghuanwu decoction on the protein expression of PI3K, Akt, Bcl-2 and BAX in brain tissue of a rat model of cerebral hemorrhage [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5933-5938. |
| [4] | Zhang Jian-jun, Shi Huan-chang. Immunosuppressed rat model of cerebral hemorrhage: construction and assessment [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5939-5945. |
| [5] | Fu Feng, Zhao Ming-liang, Li Xiao-hong, Chen Chong, Wang Li-na, Sun Hong-tao, Tu Yue, Zhang Sai . Dynamic changes of brain cavity in rats after traumatic brain injury detected by MRI-based three-dimensional reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5946-5952. |
| [6] | Yu Yu-qin, Hu Nian-chun, Duan Ji-an, Li Da-peng, Liu Chang. Neuroprotective effects of sufentanil preconditioning on spinal cord injury in mouse models [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5966-5972. |
| [7] | Wang Zhan, Li Hao-peng, He Xi-jing, Hao Ding-jun, Zhang Kun, Chen Ming-xia, Lei Ting. Pathological changes in the spinal cord of a model of acute cauda equina compression [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5973-5978. |
| [8] | Zhao Yan-rui, Lv Wen-rui, Wang Dong, Zhou Jun-lin. Effects of carbonyl sulfide in a rat model of limb ischemia/reperfusion-induced acute lung injury [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5994-6000. |
| [9] | Fan Xu-hui, Yang Bo, Hu Xiang, Guan Fang-xia. Reactive hyperplasia of glial cells induced by spinal cord injury in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6001-6006. |
| [10] | Chen Wei-hua, Lv Guo-hua, Zhou Bin, Kang Yi-jun. Establishment of a dog model of pyogenic spinal infection [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6014-6020. |
| [11] | Ge Run, Yang Lan. Protective effects of piperine on alveolar bone and collagen in a periodontitis model [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6034-6040. |
| [12] | Song Qiao-qiao, Zhou Hui-liang, Zhen Hai-tao, Wang Na, Deng Jing, Wang Jin-xiang, Pan Xing-hua . Establishment and evaluation of a rhesus monkey model of experimental type 2 diabetes mellitus [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6048-6053. |
| [13] | Pan Lei, Zhang Jin-biao, Cui Rong-gang, Zhao Bao-hui, Li Hua, Zhang Zhong-yong, Wang Xu-chu. Establishment of nonalcoholic fatty liver C57BL/6 mouse models [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6054-6059. |
| [14] | Wang Shi-jun, Li Yu-ting, Li Chun-de. Establishment, application and progression of an animal model of cervical spondylosis [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 6067-6073. |
| [15] | Liu Ying-jie, Peng Jun, Liu Xiao-kang, Zhao Cheng, Yang Er-zhu, Xu Jian-guang. Experimental animal models of intervertebral fusion-induced adjacent segment degeneration [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(39): 5825-5833. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||