Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6880-6891.doi: 10.12307/2026.401
Previous Articles Next Articles
Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane
Song Muze1, Liu Chuyi2, 3, Tang Qingjuan1, Dai Yuankun2, 3, Song Wenshan2, Li Bafang2, Wang Yuanyuan2
- 1School of Food Science and Engineering, 3School of Medicine and Pharmacy, Ocean University of China, Qingdao 266003, Shandong Province, China; 2Marine Biomedical Research Institute of Qingdao, Qingdao 266000, Shandong Province, China
-
Accepted:2025-12-17Online:2026-09-18Published:2026-03-12 -
Contact:Wang Yuanyuan, Senior engineer, Marine Biomedical Research Institute of Qingdao, Qingdao 266000, Shandong Province, China -
About author:Song Muze, MS, School of Food Science and Engineering, Ocean University of China, Qingdao 266003, Shandong Province, China
CLC Number:
Cite this article
Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
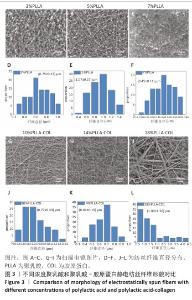
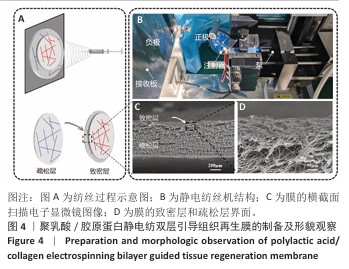
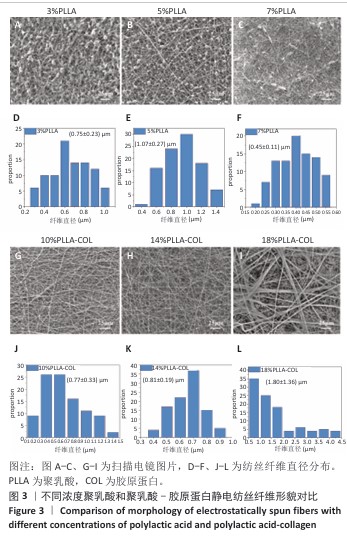
2.1 静电纺丝液浓度对纤维的影响 不同浓度电纺液制备的纤维形貌如图3所示。使用单纯聚乳酸溶液进行纺丝时,3%聚乳酸组溶剂过多,未完全挥发形成了大量液滴,液滴与液滴之间仅有少量的纤维相连,纤维直径为(0.75±0.23) μm;随聚乳酸浓度升高,液滴逐渐减少,5%聚乳酸组仍有较多液滴,但纤维明显增多且纤维直径变粗且粗细不均一;7%聚乳酸组仅有少量液滴,此时纤维极细,平均直径仅有(0.45±0.11) μm,纤维粗细均一。此外,随聚乳酸浓度升高,纤维之间的孔隙也逐渐变小。 使用聚乳酸-胶原蛋白溶液进行纺丝时均无液滴出现,10%聚乳酸-胶原蛋白组纤维存在弯曲牵伸不足的现象,并且纤维直径不均一,平均直径为(0.77±0.33) μm;14%聚乳酸-胶原蛋白组纤维不存在弯曲,纤维直径分布呈现正态分布,说明纤维尺寸均一性较好,平均直径为(0.81±0.19) μm;18%聚乳酸-胶原蛋白组静电纺丝时会出现泰勒锥不稳定、纺丝液堵塞针头的情况,调整电压和推注速度均无法解决,纺丝纤维可以看到出现纤维黏附情况,纤维直径分布不规律且不均一。 综合以上结果,7%聚乳酸溶液和14%聚乳酸-胶原蛋白溶液制备的纺丝纤维直径粗细有别,分别为(0.45±0.11) μm和(0.81±0.19) μm,大体可见两者孔隙大小不一,可以达到相对致密和相对疏松的要求,并且纤维直径符合正态分布,因此选择7%聚乳酸溶液和14%聚乳酸-胶原蛋白溶液分别作为致密层纺丝液和疏松层纺丝液,用作后续双层膜的制备。 "
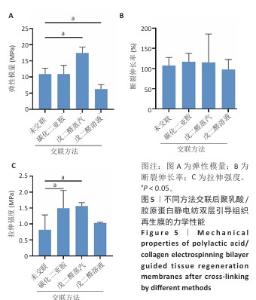
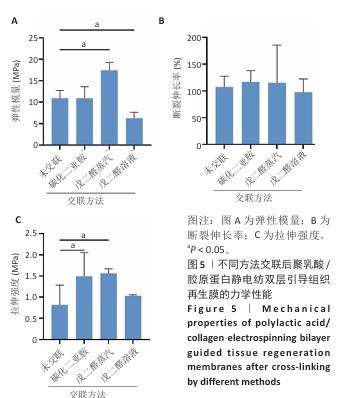
2.3 静电纺丝双层口腔修复膜的力学性能检测结果 弹性模量是衡量材料产生弹性变形难易程度的指标,拉伸强度是评价材料力学强度性能的特征值,断裂伸长率是纤维受外力作用至拉断时拉伸后伸长长度与拉伸前长度的比值。交联前后聚乳酸/胶原蛋白静电纺双层引导组织再生膜的力学性能参数结果,如图5所示。戊二醛蒸汽交联后膜的弹性模量为(17.38±1.88) MPa,拉伸强度为(1.57±0.11) MPa,相比未交联膜分别提高了59.60%和92.60%(P < 0.05)。戊二醛溶液交联后膜的弹性模量为(6.21±1.45) MPa,相比未交联膜降低了43.04%(P < 0.05),拉伸强度为(1.03±0.03) MPa,相比未交联膜提高了26.64%。碳化二亚胺/羟基琥珀酰亚胺交联后膜的弹性模量为(10.86±2.73) MPa,与未交联膜相比无显著差异(P > 0.05),拉伸强度为(1.49±0.55) MPa, 相比未交联膜提高了83.61%(P < 0.05)。3种交联方法后膜的断裂伸长率与未交联膜相比无显著差异(P > 0.05)。引导组织再生膜需要具备良好的机械性能,不易破碎且抗拉伸,才能更好地适应组织再生复杂的内环境。戊二醛蒸汽交联后膜的弹性模量和拉伸强度显著提高,综合以上结果表明,戊二醛蒸汽交联方法对膜的力学性能提高效果最佳,因此后续实验采用戊二醛蒸汽交联法进行交联,该方法交联度为(17.42±1.67)%。 "
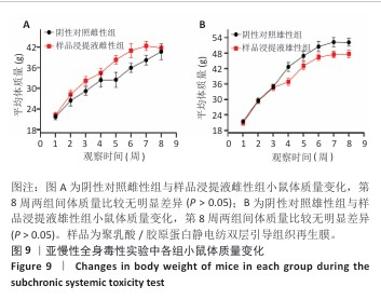
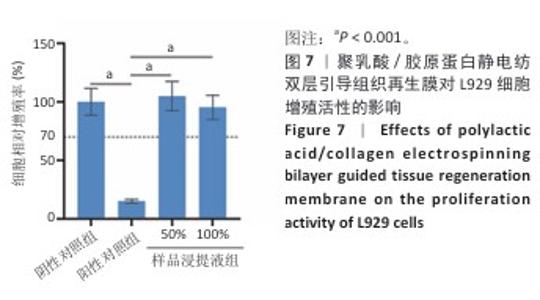
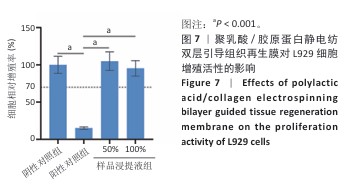
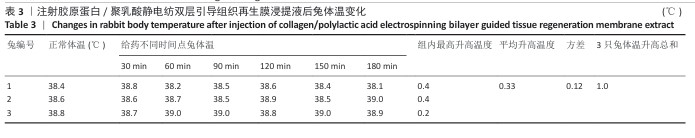
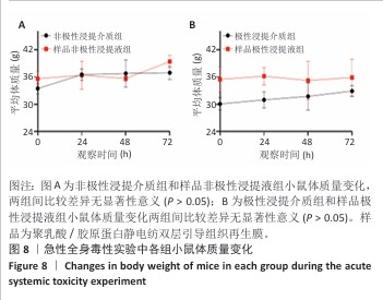
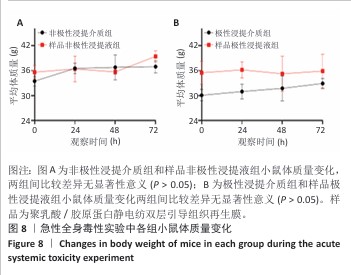
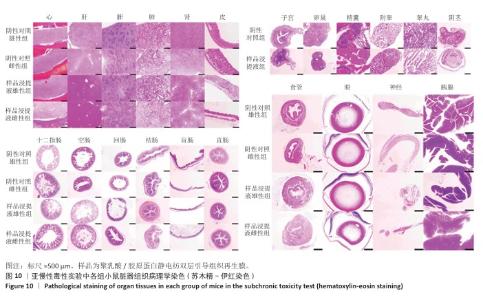
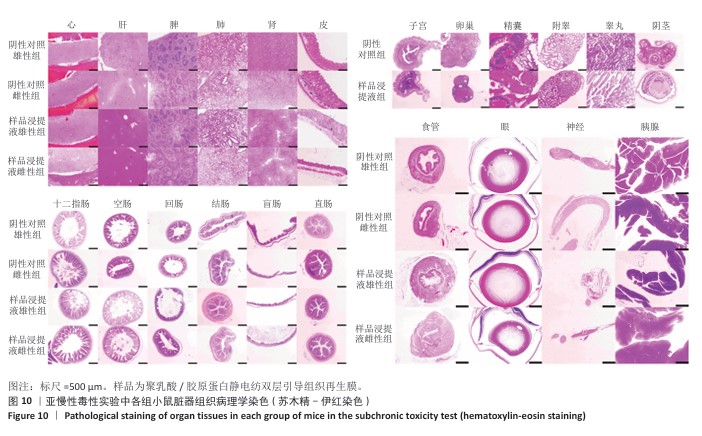
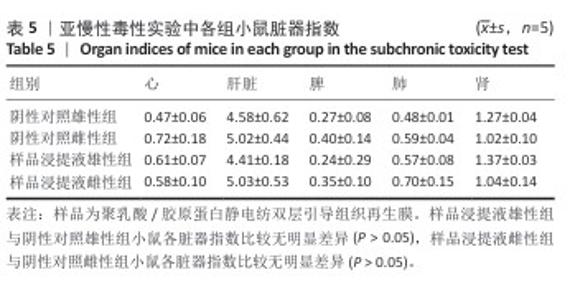
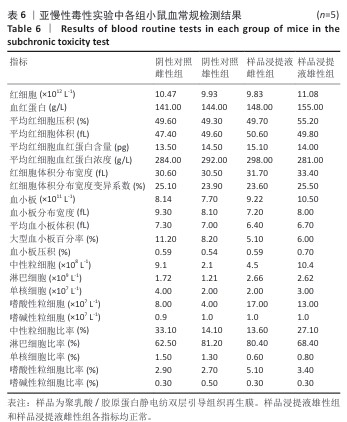
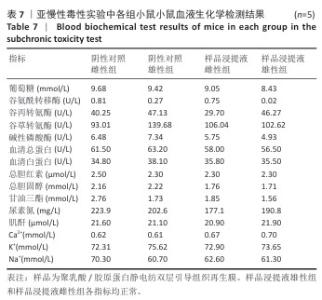
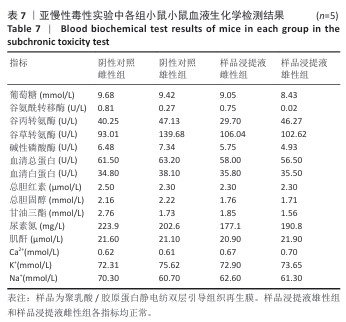
2.9 亚慢性全身毒性实验结果 实验周期内4组小鼠均未出现死亡,小鼠运动正常,未出现虚脱震颤、呼吸困难、反应降低、腹泻等症状,临床观察未见毒性反应。各组小鼠体质量变化如图9所示,样品浸提液雄性组与阴性对照雄性组小鼠体质量变化趋势一致,第8周实验结束后两组小鼠体质量相比无显著差异(P > 0.05);样品浸提液雌性组与阴性对照雌性组小鼠体质量变化趋势一致,第8周实验结束后两组小鼠体质量相比无显著差异(P > 0.05)。 各组小鼠脏器组织病理学染色结果如图10所示,样品浸提液雄性组和样品浸提液雌性组小鼠脏器均未见异常。各组小鼠脏器指数结果如表5所示,样品浸提液雄性组与阴性对照雄性组小鼠各脏器指数比较无明显差异(P > 0.05),样品浸提液雌性组与阴性对照雌性组小鼠各脏器指数比较无明显差异(P > 0.05)。 "
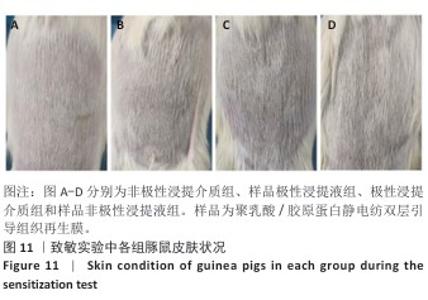
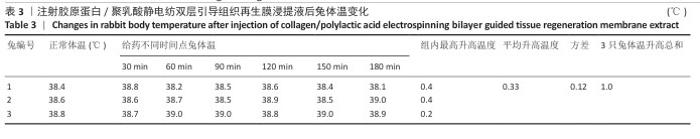
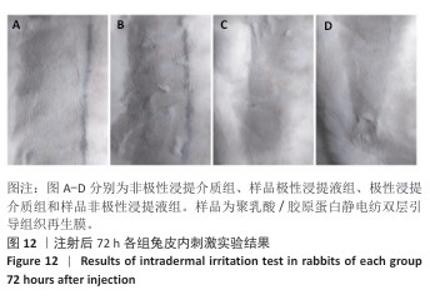
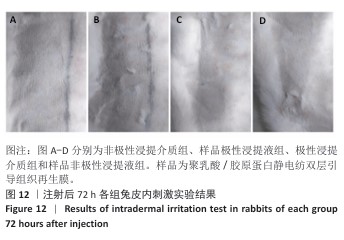
2.10 致敏实验结果 实验周期内所有豚鼠敷贴牢固、未脱落,实验结束拆除敷贴后豚鼠腹部皮肤状况如图11所示,皮肤均未出现红斑、焦痂、水肿反应,对照分级标准,分级平均为0级。聚乳酸/胶原蛋白静电纺双层引导组织再生膜极性和非极性浸提液诱导豚鼠皮肤后并未出现任何过敏反应,与对照组(极性浸提介质组、非极性浸提介质组)豚鼠皮肤状况相同。 2.11 皮内刺激实验结果 聚乳酸/胶原蛋白静电纺双层引导组织再生膜样品皮内刺激实验结果,如图12所示,注射24,48,72 h后,样品极性浸提液组和样品非极性浸提液组均未出现水肿和红斑,未见任何刺激反应,皮肤状况与极性浸提介质组、非极性浸提介质组一致,并且皮内反应计分均为0分。"
| [1] XIA D, YANG F, ZHENG Y. Research status of biodegradable metals designed for oral and maxillofacial applications: A review. Bioact Mater. 2021;276(11):4186-4208. [2] ALQAHTANI AM. Guided Tissue and Bone Regeneration Membranes: A Review of Biomaterials and Techniques for Periodontal Treatments. Polymers (Basel). 2023;15(16):3355. [3] GALLI M, YAO Y, GIANNOBILE WV, et al. Current and future trends in periodontal tissue engineering and bone regeneration. Plast Aesthet Res. 2021;8:3. [4] CHEN H, XU J, DUN Z, et al. Emulsion electrospun epigallocatechin gallate-loaded silk fibroin/polycaprolactone nanofibrous membranes for enhancing guided bone regeneration. Biomed Mater. 2024;19(5). doi: 10.1088/1748-605X/ad6dc8. [5] KARAHALILOGLU Z, ERCAN B, TAYLOR EN, et al. Antibacterial Nanostructured Polyhydroxybutyrate Membranes for Guided Bone Regeneration. J Biomed Nanotechnol. 2015;11(12):2253-2263. [6] KARFELDSULZER LS, GHAYOR C, SIEGENTHALER B, et al. Comparative study of NMP-preloaded and dip-loaded membranes for guided bone regeneration of rabbit cranial defects. J Tissue Eng Regen Med. 2017;11(2):425-433. [7] LINDNER C, ALKILDANI S, STOJANOVIC S, et al. In Vivo Biocompatibility Analysis of a Novel Barrier Membrane Based on Bovine Dermis-Derived Collagen for Guided Bone Regeneration (GBR). Membranes. 2022;12(4):378. [8] 刘蓉,潘涛华,朱丽雷.侧向转位瓣联合GTR治疗牙龈瘤伴根分叉病变患牙1例[J].临床口腔医学杂志,2023,12(39):753-754. [9] 杨浩然,刘青,徐力群,等.应用引导组织再生术及植骨术治疗种植体周围炎1例[J].山东大学学报,2024,62(4):101-107. [10] SHAOQIONG L, CHAUSANG L, KUN L, et al. Swee Hin Teoh,Marine collagen scaffolds in tissue engineering. Curr Opin Biotechnol. 2022;74: 92-103. [11] TIAN Z, SIWEI C, XINXIN D, et al. Fabrication and Characterization of Collagen/PVA Dual-Layer Membranes for Periodontal Bone Regeneration. Front Bioeng Biotechnol. 2021;9:630977. [12] ANDONEGI M, IRASTORZA A, IZETA A, et al. A Green Approach towards Native Collagen Scaffolds: Environmental and Physicochemical Assessment. Polymers. 2020;12(7):1597. [13] AKHTER M, AHMAD I, ALSHAHRAN M, et al. Drug Delivery Challenges and Current Progress in Nanocarrier-Based Ocular Therapeutic System. Gels. 2022;8(2):82. [14] SHAKOOR S, KIBBLE E, El-JAWHARI JJ. Bioengineering Approaches for Delivering Growth Factors: A Focus on Bone and Cartilage Regeneration.Bioengineering. 2022;9(5):223. [15] ZHANG KR, GAO HL, PAN XF, et al. Multifunctional Bilayer Nanocomposite Guided Bone Regeneration Membrane. Matter. 2019;1(3):770-781. [16] YOSHIMOTO I, SASAKI JI, TSUBOI R, et al. Development of layered PLGA membranes for periodontal tissue regeneration. Dent Mater. 2018;34(3):538-550. [17] ABE GL, SASAKI JI, KATATA C, et al. Fabrication of novel poly(lactic acid/caprolactone) bilayer membrane for GBR application. Dent Mater. 2020;36(5):626-634. [18] FENELON M, GALVEZ P, KALBERMATTEN D, et al. Emerging Strategies for the Biofabrication of Multilayer Composite Amniotic Membranes for Biomedical Applications. Int J Mol Sci. 2023;24(19):14424. [19] LIU C, ZHANG X, ZHAO L, et al. Multilayer amnion-PCL nanofibrous membrane loaded with celecoxib exerts a therapeutic effect against tendon adhesion by improving the inflammatory microenvironment. Heliyon. 2023;9(12):e23214. [20] MA M, LIU Q, YE C, et al. Preparation of P3HB4HB/(Gelatin + PVA) Composite Scaffolds by Coaxial Electrospinning and Its Biocompatibility Evaluation. Biomed Res Int. 2017;2017:9251806. [21] YE K, KUANG H, YOU Z, et al. Electrospun Nanofibers for Tissue Engineering with Drug Loading and Release. Pharmaceutics. 2019;11(4):182. [22] DZOBO K, TURNLEY T, WISHART A, et al. Fibroblast-Derived Extracellular Matrix Induces Chondrogenic Differentiation in Human Adipose-Derived Mesenchymal Stromal/Stem Cells in Vitro. Int J Mol Sci. 2016;17(8):1259. [23] WANG Y, LIU Y, ZHANG X, et al. Engineering Electrospun Nanofibers for the Treatment of Oral Diseases. Front Chem. 2021;9:797523. [24] 李可,李未扬,唐毓婧,等.引导组织再生膜的研究进展[J].石油化工,2024,53(8):1163-1169. [25] ALI IH, KHALIL IA, EL-SHERBINY IM. Design, development, in-vitro and in-vivo evaluation of polylactic acid-based multifunctional nanofibrous patches for efficient healing of diabetic wounds. Sci Rep. 2023;13(1):3215. [26] SROUJI S, BENDAVID D, LOTAN R, et al. Slow-release human recombinant bone morphogenetic protein-2 embedded within electrospun scaffolds for regeneration of bone defect: in vitro and in vivo evaluation. Tissue Eng Part A. 2011;17(3-4):269-277. [27] HUNTER K, MA T. In vitro evaluation of hydroxyapatite-chitosan-gelatin composite membrane in guided tissue regeneration. J Biomed Mater Res A. 2013;101(4):1016-1025. [28] HIGUCHI J, FORTUNATO G, WOZNIAK B, et al. Polymer Membranes Sonocoated and Electrosprayed with Nano-Hydroxyapatite for Periodontal Tissues Regeneration. Nanomaterials. 2019;9(11):1625. [29] THOMAS NG, SANIL GP, GOPIMOHAN R, et al. Biocompatibility and cytotoxic evaluation of drug-loaded biodegradable guided tissue regeneration membranes. J Indian Soc Periodontol. 2012;16(4):498-503. [30] XUE J, HE M, LIU H, et al. Drug loaded homogeneous electrospun PCL/gelatin hybrid nanofiber structures for anti-infective tissue regeneration membranes. Biomaterials. 2014;35(34):9395-9405. [31] ZHOU T, CHEN S, DING X, et al. Fabrication and Characterization of Collagen/PVA Dual-Layer Membranes for Periodontal Bone Regeneration. Front Bioeng Biotechnol. 2021;9:630977. [32] CARDENAS-AGUAZACO W, CAMACHO B, GOMEZ-PACHON EY, et al. Electrospun Scaffolds of Polylactic Acid, Collagen, and Amorphous Calcium Phosphate for Bone Repair. Pharmaceutics. 2023;15(11):2529. [33] HALL BARRIENTOS IJ, PALADINO E, SZABO P, et al. Electrospun collagen-based nanofibres: A sustainable material for improved antibiotic utilisation in tissue engineering applications. Int J Pharm. 2017;531(1): 67-79. [34] ZHANG S, CHEN L, JIANG Y, et al. Bi-layer collagen/microporous electrospun nanofiber scaffold improves the osteochondral regeneration. Acta Biomater. 2013;9(7):7236-7247. [35] HUANG WY, HIBINO T, SUYE SI, et al. Electrospun collagen core/poly-l-lactic acid shell nanofibers for prolonged release of hydrophilic drug. RSC Adv. 2021;11(10):5703-5711. [36] XU Y, CUI W, ZHANG Y, et al. Hierarchical Micro/Nanofibrous Bioscaffolds for Structural Tissue Regeneration. Adv Healthc Mater. 2017;6(13):10. [37] CHEN H, ZHANG H, SHEN Y, et al. Instant in-situ Tissue Repair by Biodegradable PLA/Gelatin Nanofibrous Membrane Using a 3D Printed Handheld Electrospinning Device. Front Bioeng Biotechnol. 2021;9:684105. [38] NING Y, SHEN W, AO F. Application of blocking and immobilization of electrospun fiber in the biomedical field. RSC Adv. 2020;10(61): 37246-37265. [39] KONG B, LIU R, GUO J, et al. Tailoring micro/nano-fibers for biomedical applications. Bioact Mater. 2023;19:328-347. [40] GREINER A, WENDORFF JH. Electrospinning: a fascinating method for the preparation of ultrathin fibers. Angew. Chem. 2007;46(30):5670-5703. [41] APABLAZA JA, LEZCANO MF, LOPEZ MARQUEZ A, et al. Main Morphological Characteristics of Tubular Polymeric Scaffolds to Promote Peripheral Nerve Regeneration-A Scoping Review. Polymers. 2021;13(15):2563. [42] SUN D, CAO R, WU H, et al. Harsh Environmental-Tolerant and High-Performance Triboelectric Nanogenerator Based on Nanofiber/Microsphere Hybrid Membranes. Materials (Basel, Switzerland). 2023; 16(2):562. [43] VANHEUSDEN C, VANMINSEL J, REDDY N, et al. Fabrication of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) Fibers Using Centrifugal Fiber Spinning: Structure, Properties and Application Potential. Polymers. 2023;15(5):1181. [44] ZHENG W, ZHANG W, JIANG X. Precise control of cell adhesion by combination of surface chemistry and soft lithography. Adv Healthcare Mater. 2013;2(1):95-108. [45] DALBY M J, GADEGAARD N, TARE R, et al. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat Mater. 2007;6(12):997-1003. [46] KARAGEORGIOU V, KAPLAN D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005;26(27):5474-5491. [47] DOS SANTOS VI, MERLINI C, ARAGONES Á, et al. In vitro evaluation of bilayer membranes of PLGA/hydroxyapatite/β-tricalcium phosphate for guided bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020; 112:110849. [48] CHANG, HI, WANG Y. Cell Responses to Surface and Architecture of Tissue Engineering Scaffolds. Regen Med Tissue Eng-Cells Biomater. 2010;8(29).doi: 10.5772/21983. [49] BAZGIR M, ZHANG W, ZHANG X, et al. Degradation and Characterisation of Electrospun Polycaprolactone (PCL) and Poly(lactic-co-glycolic acid) (PLGA) Scaffolds for Vascular Tissue Engineering. Materials. 2021; 14(17):4773. [50] RUHUNAGE C, DHAWAN V, NAWARATHEN CP, et al. Evaluation of Polymer-Coated Carbon Nanotube Flexible Microelectrodes for Biomedical Applications. Bioengineering. 2023;10(6):647. [51] LEE DY, SONG WH, LIM YS, et al. Fish Collagen Peptides Enhance Thymopoietic Gene Expression, Cell Proliferation, Thymocyte Adherence, and Cytoprotection in Thymic Epithelial Cells via Activation of the Nuclear Facto-κB Pathway, Leading to Thymus Regeneration after Cyclophosphamide-Induced Injury. Mar Drugs. 2023;21(10):531. [52] SREENA R, NATHANAEL AJ. Biodegradable Biopolymeric Nanoparticles for Biomedical Applications-Challenges and Future Outlook. Materials(Basel). 2023;16(6):2364. [53] CASTANEDA-RODRIGUES S, GONZALEZ-TORRES M, RIBAS-APARICIO RM, et al. Recent advances in modified poly (lactic acid) as tissue engineering materials. J Biol Eng. 2023;17(1):21. [54] QIAO Y, YU L, YANG P, et al. Spatiotemporal Immunomodulation and Biphasic Osteo-Vascular Aligned Electrospun Membrane for Diabetic Periosteum Regeneration. Adv Sci (Weinh). 2023;10(36):e2302874. [55] KAUR D, SHARMA RR, MARWAHA N. Defining an appropriate leucoreduction strategy by serial assessment of cytokine levels in platelet concentrates prepared by different methods. Asian J Transfus Sci. 2015; 9(1):31-35. [56] KRAMER M, PLUM PS, VELAZQUAZ O, et al. Cell type-specific transcriptomics of esophageal adenocarcinoma as a scalable alternative for single cell transcriptomics. Mol Oncol. 2020;14(6):1170-1184. [57] CHECKOURI E, REIGNIER F, ROBERTSilva C, et al. Evaluation of Polyphenol Content and Antioxidant Capacity of Aqueous Extracts from Eight Medicinal Plants from Reunion Island: Protection against Oxidative Stress in Red Blood Cells and Preadipocytes. Antioxidants. 2020;9(10):959. [58] FADILAH NIM, AHMAT N, HAO LQ, et al. Biological Safety Assessments of High-Purified Ovine Collagen Type I Biomatrix for Future Therapeutic Product: International Organisation for Standardisation (ISO) and Good Laboratory Practice (GLP) Settings. Polymers. 2023;15(11):2436. [59] WANI AL, ARA A, USMANI JA. Lead toxicity: a review. Interdiscip. Toxicol. 2015;8(2): 55-64. [60] XIONG W, WANG P, HU J, et al. A new sensitizer DVDMS combined with multiple focused ultrasound treatments: an effective antitumor strategy. Sci Rep. 2021;11(1):17288. [61] KIM TH, HEO SY, OH GW, et al. Biocompatibility and sub-chronic toxicity studies of phlorotannin/polycaprolactone coated trachea tube for advancing medical device applications. Sci Rep. 2024;14(1):3945. [62] MACHONA O, MUTANGA M, CHIDZWONDO F, et al. Sub-chronic toxicity determination of powdered Tenebrio molitor larvae as a novel food source. Toxicol Rep. 2024;12:111-116. [63] SINGH C, TIWRI KN, KUMAR P, et al. Toxicity profiling and antioxidant activity of ethyl acetate extract of leaves of Premna integrifolia L. for its application as protective agent against xenobiotics. Toxicol Rep. 2021;8:196-205. [64] HAN X, SUN Y, HUANGFU B, et al. Ultra-high-pressure passivation of soybean agglutinin and safety evaluations. Food Chem X. 2023;18: 100726. [65] HUZUM B, PUHA B, NECOARA RM, et al. Biocompatibility assessment of biomaterials used in orthopedic devices: An overview (Review). Exp Ther Med. 2021;22(5):1315. |
| [1] | Sun Lei, Zhang Qi, Zhang Yu. Pro-osteoblastic effect of chlorogenic acid protein microsphere/polycaprolactone electrospinning membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1877-1884. |
| [2] | Yang Qiongqiong, Liu Wei. Comparison of performance and clinical effects of zirconia and titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2063-2071. |
| [3] | Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936. |
| [4] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [5] | Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718. |
| [6] | Pi Zhilong, Li Jiayuan, Tan Zhichao, Lu Xiaomei, Zhang Zhiqiang, Ye Xiangling. 3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6736-6743. |
| [7] | Zhou Yunqi, Liu Xu, Xiao Dongqin, Li Xingping, Shi Feng, Zhang Bo, Pu Chao, Luo Xuwei, Zhang Chengdong. Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6768-6778. |
| [8] | Chen Gang, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Wang Qianliang. Mechanism of ferrostatin-1 hydrogel in treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6807-6813. |
| [9] | Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng. Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6814-6825. |
| [10] | Lyu Tianyang, Li Ning, Huang Shuo, Liu Changkui, Guo Yayuan, Hu Kaijin. Preparation and biocompatibility of odanacatib microspheres-gel composite sustained-release carrier [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6840-6848. |
| [11] | Li Yujin, Ni Guansen, Mao Weiqing, Tang Jiayu, Li Xueqing. Biocompatibility and preclinical experiments of a Chinese-made 3D-printed minimally invasive tungsten alloy needle electrodes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6859-6867. |
| [12] | Zhou Lijing, Wang Shuang, Xiang Jinjiao, Wang Huichao, Chai Xuejiao. Apical sealing and resistance strength of C-Root BP material in in vitro environment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6868-6872. |
| [13] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [14] | Chen Weifei, Mei Yuandong, Ju Jihui. Repair of infected bone defect with dual-ion time-sequenced release multifunctional hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5188-5200. |
| [15] | Li Keyun, Yang Yuqi, Fei Yingying, Huang Shuai. Physicochemical properties and in vitro biological effects of resveratrol-eluting stents [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5243-5256. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||