Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6814-6825.doi: 10.12307/2026.760
Previous Articles Next Articles
Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy
Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng
- Department of Arthroplasty and Sports Medicine, The Third People's Hospital of Chengdu, Chengdu 610031, Sichuan Province, China
-
Accepted:2025-09-25Online:2026-09-18Published:2026-03-11 -
Contact:Tao Qifeng, MS, Department of Arthroplasty and Sports Medicine, The Third People's Hospital of Chengdu, Chengdu 610031, Sichuan Province, China -
About author:Zhao Wenbo, PhD, Department of Arthroplasty and Sports Medicine, The Third People's Hospital of Chengdu, Chengdu 610031, Sichuan Province, China -
Supported by:Chengdu Science and Technology Project Support, No. 2024-YF05-00119-SN (to ZWB); Chengdu Medical Research Project Support, No. 2024169 (to ZWB)
CLC Number:
Cite this article
Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng. Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6814-6825.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
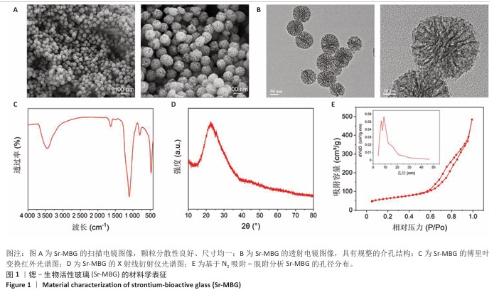
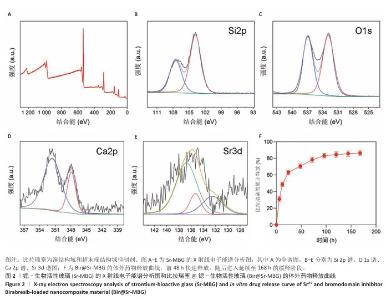
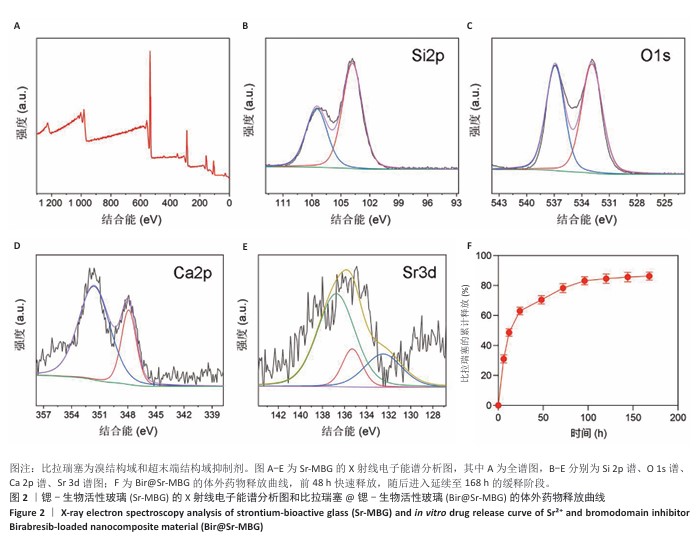
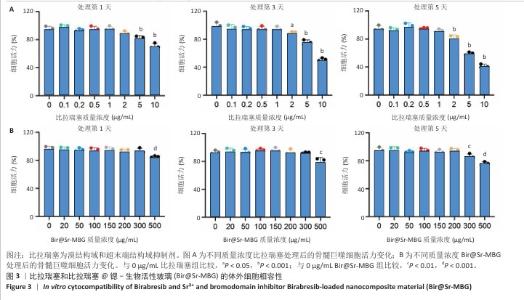
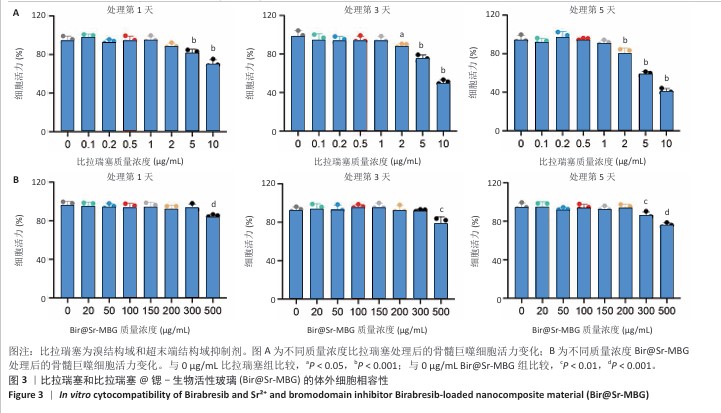
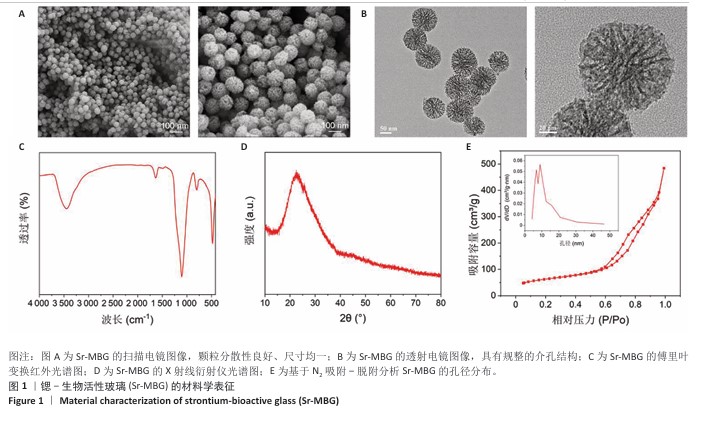
2.1 材料学表征结果 扫描电镜观察显示Sr-MBG颗粒分散性良好、尺寸均一,粒径约为100?nm(图1A),为药物递送等生物医用应用提供了理想基础。透射电镜观察显示Sr-MBG具有规整的介孔结构(图1B),有利于药物的负载与控释。 傅里叶变换红外光谱分析显示,Sr-MBG具备典型的硅氧骨架结构,468?cm-1(Si-O-Si摇摆)、798?cm-1(Si-O-Si弯曲)与1 089?cm-1(Si-O-Si伸缩)为它的主要特征峰(图1C)[29-30]。X射线衍射结果显示,Sr-MBG在2θ=20°-30°出现宽峰(图1D),证实该材料为无定形状态。 氮气吸附-脱附等温线呈现典型Ⅳ型曲线,表明Sr-MBG具备介孔特征(图1E)。Sr-MBG孔径集中于7.62?nm,分布窄,结构均一;Sr-MBG比表面积高达223.82?m2/g,提供了良好的药物负载能力,凸显它在递药系统中的应用潜力。 通过X射线光电子能谱分析确认Sr-MBG的元素组成,在全谱图中可清晰观察到C 1s、O 1s、Si 2p、Ca 2p与Sr 3d特征峰(图2A)。在Si 2p谱图中,103.6 eV主峰对应Si-O-Si桥氧键,107.3 eV次峰归因于表面Si-OH基团,反映典型的硅氧骨架结构(图2B)。O 1s谱图显示主峰位于532.5 eV,归属于材料骨架中的Si-O-Si和O-Si键(图2C)。Ca 2p谱图在347.1 eV和350.6 eV分别出现2p?/?与2p?/?双峰(图2D),表明Ca2?成功掺入硅酸盐网络。Sr 3d谱图显示出2个双峰结构,其中主峰(3d?/?:134.2 eV,3d?/?:135.3 eV)代表玻璃网络中的Sr2?,次峰(3d?/?:138.3 eV)归因于Sr-O-Ca配位形成(图2E),提示材料中存在复杂的离子相互作用。 Bir@Sr-MBG的药物包封率为44.82%、载药率为7.47%。在PBS(pH=7.4)条件下,Bir@Sr-MBG的释放曲线呈现典型双阶段特征:前48 h快速释放,随后进入延续至168 h的缓释阶段,最终累计释放量超过80%(图2F)。该控制释放行为得益于Sr-MBG的分级介孔结构:表面连通孔隙介导初期释放,内部介孔通道延缓药物扩散,从而实现持续释放并提升治疗效果。 "
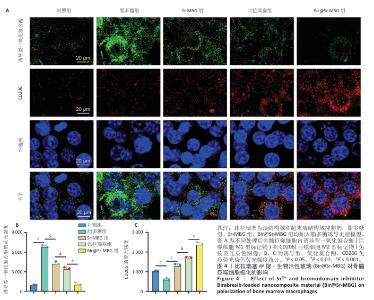
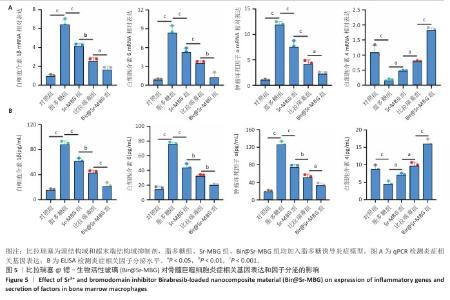
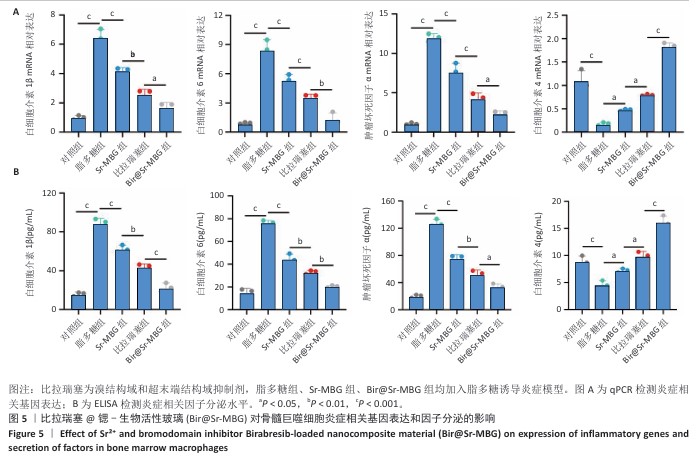
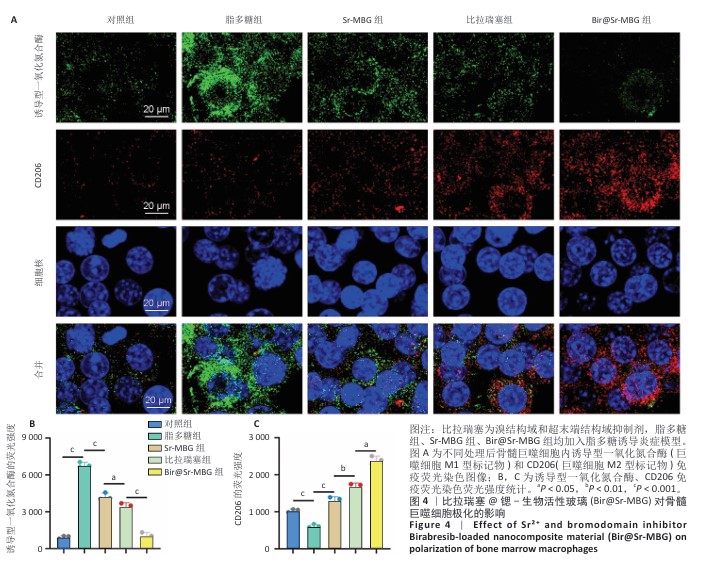
2.3 Bir@Sr-MBG的体外调控巨噬细胞极化作用分析结果 免疫荧光结果显示,脂多糖刺激显著诱导巨噬细胞M1型标志物诱导型一氧化氮合酶表达,而比拉瑞塞、Sr-MBG、Bir@Sr-MBG处理可有效下调诱导型一氧化氮合酶荧光信号,并且Bir@Sr-MBG处理下调诱导型一氧化氮合酶荧光信号的作用强于比拉瑞塞、Sr-MBG(图4A,B),提示Bir@Sr-MBG具有抗炎特性。脂多糖刺激显著抑制巨噬细胞M2型标志物CD206的表达,而比拉瑞塞、Sr-MBG、Bir@Sr-MBG处理可有效上调CD206的表达,并且Bir@Sr-MBG处理上调CD206表达的作用强于比拉瑞塞、Sr-MBG(图4A,C)。上述结果表明,Bir@Sr-MBG可通过调节巨噬细胞极化状态改善炎症微环境,从而为缓解炎性破骨活性提供可能的分子机制支持。 qPCR检测结果显示,与对照组比较,脂多糖组骨髓巨噬细胞M1型相关基因白细胞介素1β、白细胞介素6、肿瘤坏死因子α mRNA表达升高,M2型相关基因白细胞介素4 mRNA表达降低,差异均有显著性意义;与脂多糖组比较,比拉瑞塞组、Sr-MBG组细胞介素1β、白细胞介素6、肿瘤坏死因子α mRNA表达均降低,白细胞介素4 mRNA表达升高,差异均有显著性意义;与Sr-MBG组、比拉瑞塞 组比较,Bir@Sr-MBG组细胞介素1β、白细胞介素6、肿瘤坏死因子α mRNA表达均降低,白细胞介素4 mRNA表达升高,差异均有显著性意义,见图5A,提示Bir@Sr-MBG可诱导抗炎性转录重编程。 ELISA检测结果显示,与对照组比较,脂多糖组细胞上清中促炎因子白细胞介素1β、白细胞介素6、肿瘤坏死因子α水平均升高,抗炎因子白细胞介素4水平降低,差异均有显著性意义;与脂多糖组比较,比拉瑞塞组、Sr-MBG组细胞上清中白细胞介素1β、白细胞介素6、肿瘤坏死因子α水平均降低,抗炎因子白细胞介素4水平升高,差异均有显著性意义;与Sr-MBG组、比拉瑞塞组比较,Bir@Sr-MBG组细胞上清中白细胞介素1β、白细胞介素6、肿瘤坏死因子α水平均降低,抗炎因子白细胞介素4水平升高,差异均有显著性意义(图5B)。这一调控过程可能依赖于对核因子κB信号通路的抑制,从而有效减弱脂多糖诱导的炎症反应。机制研究结果进一步表明,Bir@Sr-MBG在转录及分泌层面对巨噬细胞具有精确的免疫调节作用。因此,Bir@Sr-MBG通过诱导巨噬细胞向M2表型转化重塑抗炎微环境,进而抑制破骨细胞形成并促进成骨,为基于免疫调控的骨质疏松治疗提供了理论支持与实验依据。"
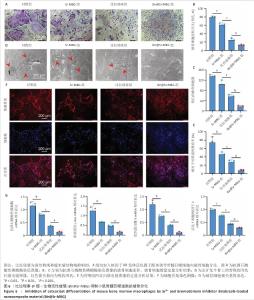
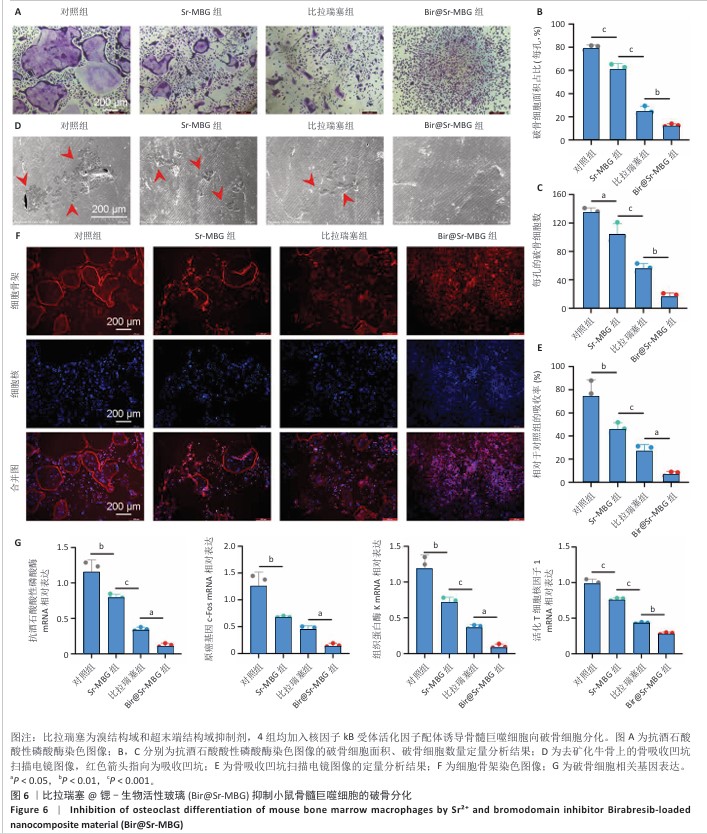
2.4 Bir@Sr-MBG的体外抗破骨分化效果 抗酒石酸酸性磷酸酶染色结果显示,比拉瑞塞组、Sr-MBG组、抗酒石酸酸性磷酸酶阳性多核破骨细胞数量少于对照组,Bir@Sr-MBG组抗酒石酸酸性磷酸酶阳性多核破骨细胞数量少于比拉瑞塞组、Sr-MBG组,差异均有显著性意义(图6A-C),提示Bir@Sr-MBG可有效抑制核因子κB受体活化因子配体诱导的破骨细胞生成。 去矿化牛骨切片上的骨吸收实验结果显示,对照组出现大量吸收坑,比拉瑞塞组、Sr-MBG组吸收坑明显减少,而Bir@Sr-MBG组几乎未见明显吸收区(图6D,E),表明Bir@Sr-MBG可显著削弱破骨细胞的骨吸收能力。 FITC标记鬼笔环肽染色结果显示,对照组中成熟破骨细胞具有典型的F-肌动蛋白环结构,比拉瑞塞组、Sr-MBG组、Bir@Sr-MBG处理可抑制该环的形成,其中Bir@Sr-MBG处理的抑制作用最强(图6F),进一步支持Bir@Sr-MBG对成熟破骨细胞骨架功能的抑制作用。 qPCR检测结果显示,与对照组比较,比拉瑞塞组、Sr-MBG组抗酒石酸酸性磷酸酶、原癌基因c-Fos、组织蛋白酶K、活化T细胞核因子1 mRNA表达均降低,差异均有显著性意义;与比拉瑞塞组、Sr-MBG组比较,Bir@Sr-MBG组抗酒石酸酸性磷酸酶、原癌基因c-Fos、组织蛋白酶K、活化T细胞核因子1 mRNA表达降低,差异均有显著性意义,见图6G。 综上所述,Bir@Sr-MBG可显著抑制核因子κB受体活化因子配体诱导的破骨细胞分化与骨吸收功能,显示出该材料在调控骨重塑过程中的潜在治疗价值。 "
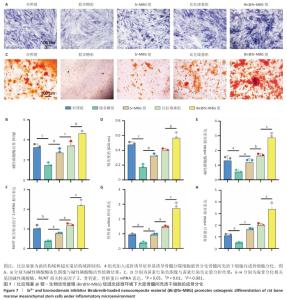
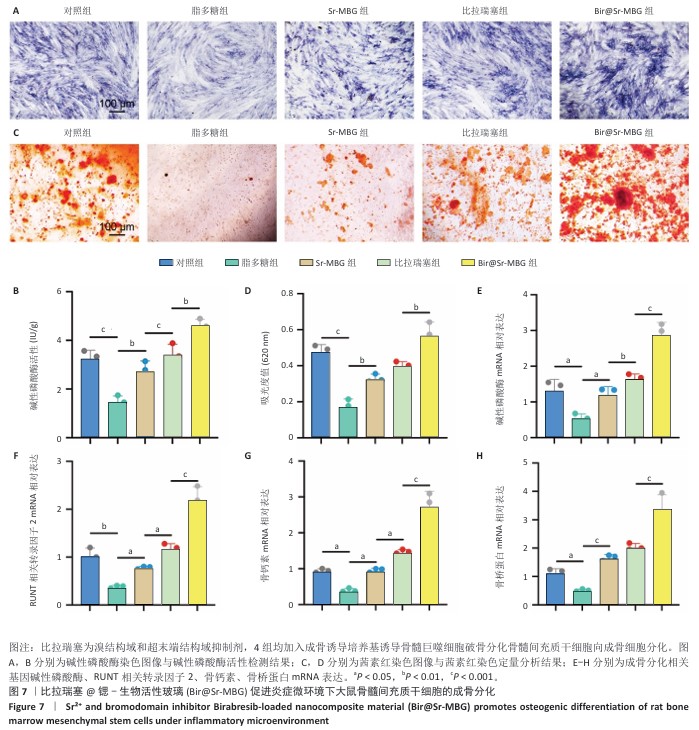
2.5 Bir@Sr-MBG的体外促成骨分化效果 碱性磷酸酶染色与活性测定结果显示,脂多糖组碱性磷酸酶活性低于对照组,比拉瑞塞组、Sr-MBG组碱性磷酸酶活性高于脂多糖组,Bir@Sr-MBG组碱性磷酸酶活性高于比拉瑞塞组、Sr-MBG组,差异均有显著性意义(图7A,B),提示Bir@Sr-MBG可有效促进早期成骨分化。 茜素红染色结果显示,脂多糖组钙沉积少于对照组,比拉瑞塞组、Sr-MBG组钙沉积多于脂多糖组,Bir@Sr-MBG组钙沉积多于比拉瑞塞组、Sr-MBG组,差异均有显著性意义(图7C,D),表明Bir@Sr-MBG可增强晚期矿化能力。 qPCR检测结果显示,与对照组比较,脂多糖组碱性磷酸酶、RUNT相关转录因子2、骨钙素、骨桥蛋白 mRNA表达均降低,差异均有显著性意义;与脂多糖组比较,比拉瑞塞组、Sr-MBG组碱性磷酸酶、RUNT相关转录因子2、骨钙素、骨桥蛋白 mRNA表达均升高,差异均有显著性意义;与比拉瑞塞组、Sr-MBG组比较,Bir@Sr-MBG组碱性磷酸酶、RUNT相关转录因子2、骨钙素、骨桥蛋白 mRNA表达均升高,差异均有显著性意义,见图7E-H。 总的来说,Bir@Sr-MBG在模拟炎症条件下展现出“促成骨-抗炎”的双重调控能力,显著促进骨髓间充质干细胞的成骨分化全过程,具备良好的骨再生应用潜力。 "
| [1] HUANG Y, CHEN T, REN C, et al. High-Strength Gelatin Hydrogel Scaffold with Drug Loading Remodels the Inflammatory Microenvironment to Enhance Osteoporotic Bone Repair. Adv Mater. 2025;37(13):2501051. [2] HUO S, LIU X, ZHANG S, et al. p300/CBP inhibitor A-485 inhibits the differentiation of osteoclasts and protects against osteoporotic bone loss. Int Immunopharmacol. 2021;94:107458. [3] XIE Q, DU X, LIANG J, et al. FABP4 inhibition suppresses bone resorption and protects against postmenopausal osteoporosis in ovariectomized mice. Nat Commun. 2025;16(1):4437. [4] KAHWATI LC, KISTLER CE, BOOTH G, et al. Screening for Osteoporosis to Prevent Fractures: A Systematic Evidence Review for the US Preventive Services Task Force. JAMA. 2025;333(6):509-531. [5] BOLLAND MJ, NISA Z, MELLAR A, et al. Fracture Prevention with Infrequent Zoledronate in Women 50 to 60 Years of Age. N Engl J Med. 2025;392(3):239-248. [6] ZHAO W, QIAN J, LI J, et al. From death to birth: how osteocyte death promotes osteoclast formation. Front Immunol. 2025;16:1551542. [7] MELIS S, TROMPET D, CHAGIN AS, et al. Skeletal stem and progenitor cells in bone physiology, ageing and disease. Nat Rev Endocrinol. 2025;21(3):135-153. [8] FU W, CHEN M, WANG K, et al. Tau is a receptor with low affinity for glucocorticoids and is required for glucocorticoid-induced bone loss. Cell Res. 2025;35(1):23-44. [9] SHARMA G, LEE YH, KIM JC, et al. Bone Regeneration Enhanced by Quercetin-Capped Selenium Nanoparticles via miR206/Connexin43, WNT, and BMP signaling pathways. Aging Dis. 2025. doi: 10.14336/AD.2025.0025. [10] SHAN C, WANG W, LU H, et al. A spatially organized Cd24a+/Pax9+ stem cell core governs postnatal tooth establishment. Sci Adv. 2025; 11(23):eadu5653. [11] MENG S, LIU Q, DAI R, et al. Development of a novel macroscopic regulation and microscopic intervention mode nanosystem for osteoporosis treatment. Mater Today Bio. 2025;32:101829. [12] SAPRA L, SAINI C, MISHRA PK, et al. Bacillus coagulans ameliorates inflammatory bone loss in post-menopausal osteoporosis via modulating the “Gut-Immune-Bone” axis. Gut Microbes. 2025;17(1): 2492378. [13] CUI Y, LV B, LI Z, et al. Bone-Targeted Biomimetic Nanogels Re-Establish Osteoblast/Osteoclast Balance to Treat Postmenopausal Osteoporosis. Small. 2024;20(6):e2303494. [14] XUE P, WANG J, FU Y, et al. Material-Mediated Immunotherapy to Regulate Bone Aging and Promote Bone Repair. Small. 2025;21(11): e2409886. [15] KUBI JA, BRAH AS, CHEUNG KMC, et al. Low-molecular-weight estrogenic phytoprotein suppresses osteoporosis development through positive modulation of skeletal estrogen receptors. Bioact Mater. 2024; 42:299-315. [16] KALYANARAMAN H, PAL CHINA S, CASTEEL DE, et al. Crosstalk between androgen receptor and protein kinase G signaling in bone: implications for osteoporosis therapy. Trends Pharmacol Sci. 2025;46(3):279-294. [17] LIU X, LI X, HUO S, et al. Magnesium bioactive glass hybrid functionalized polyetheretherketone with immunomodulatory function to guide cell fate and bone regeneration. Colloids Surf B Biointerfaces. 2023;230:113523. [18] HUO S, LYU Z, SU X, et al. Formation of a novel Cu-containing bioactive glass nano-topography coating with strong bactericidal capability and bone regeneration. Compos B Eng. 2023;253:110521. [19] HUO S, LIU S, LIU Q, et al. Copper-Zinc-Doped Bilayer Bioactive Glasses Loaded Hydrogel with Spatiotemporal Immunomodulation Supports MRSA-Infected Wound Healing. Adv Sci (Weinh). 2024;11(5):e2302674. [20] WANG L, JIANG S, ZHOU J, et al. From hard tissues to beyond: Progress and challenges of strontium-containing biomaterials in regenerative medicine applications. Bioact Mater. 2025;49:85-120. [21] WU D, WU H, YU P, et al. Unraveling the molecular mechanism of aqueous extract of Sargentodoxa cuneata against ulcerative colitis from serum metabolomics and bioinformatics perspectives. J Chromatogr B Analyt Technol Biomed Life Sci. 2024;1249:124372. [22] SU X, LYU Z, WU Y, et al. Strontium-doped bioactive glass/PDA functionalized polyetheretherketone with immunomodulatory property for enhancing photothermal clearance of Staphylococcus aureus. Mater Design. 2023;225:111552. [23] BAO X, HUO S, WANG Z, et al. Multifunctional biomimetic hydrogel dressing provides anti-infection treatment and improves immunotherapy by reprogramming the infection-related wound microenvironment. J Nanobiotechnology. 2024;22(1):80. [24] HUO S, WANG F, LYU Z, et al. Dual-functional polyetheretherketone surface modification for regulating immunity and bone metabolism. Chem Eng J. 2021;426:130806. [25] MIAO L, ZIHAO X, JUNHAO S, et al. A New Nanoplatform Under NIR Released ROS Enhanced Photodynamic Therapy and Low Temperature Photothermal Therapy for Antibacterial and Wound Repair. Int J Nanomedicine. 2024;19:7509-7527. [26] ZHANG S, HUO S, LI H, et al. Flufenamic acid inhibits osteoclast formation and bone resorption and act against estrogen-dependent bone loss in mice. Int Immunopharmacol. 2020;78:106014. [27] HUO S, LIU Y, XU Z, et al. Specific Activation of the STING Pathway by Engineering Piezoelectric Hydrogel Microspheres for Boosting Implant Infection Immunotherapy. ACS Nano. 2025;19(17):16383-16404. [28] ZHANG X, WANG S, YANG Q, et al. 3D Printed Microsphere-Hydrogel Scaffold Facilitates Restoration of Reinnervation in Bone Regeneration through Programmable Release of NGF/BMP-2 Mimetic Peptides. Adv Healthc Mater. 2025;14(20):e2501594. [29] SU XJ, LIU S, HUO SC, et al. Polyetheretherketone surface modification by lithium-doped bioglass nanospheres to regulate bone immunity and promote osseointegration. Mater Design. 2024;238:112646. [30] CHEN Y, WANG T, YAN Z, et al. Bioactive Glass-Induced B Cell Depletion Remodels the Osteoimmunological Microenvironment to Enhance Osteogenesis. Acta Biomater. 2025;201:648-664. [31] BAI L, LI J, LI G, et al. Skeletal interoception and prospective application in biomaterials for bone regeneration. Bone Res. 2025;13(1):1. [32] ARTAMONOV MY, SOKOV EL, KORNILOVA LE, et al. The Intraosseous Environment: Physiological Parameters, Regulatory Mechanisms, and Emerging Insights in Bone Biology. Int J Mol Sci. 2025;26(8):3876. [33] YAO Y, CAI X, CHEN Y, et al. Estrogen deficiency-mediated osteoimmunity in postmenopausal osteoporosis. Med Res Rev. 2025; 45(2):561-575. [34] MA S, LU Y, SUI S, et al. Unraveling the triad of immunotherapy, tumor microenvironment, and skeletal muscle biomechanics in oncology. Front Immunol. 2025;16:1572821. [35] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Ther. 2022;237:108168. [36] LU J, WU H, JIN H, et al. The influence of modified Qing E Formula on the differential expression of serum exosomal miRNAs in postmenopausal osteoporosis patients. Front Pharmacol. 2024;15: 1467298. [37] ZENG Q, DU S, YUAN R, et al. Self-Healing Hydrogel Dressing with Solubilized Flavonoids for Whole Layer Regeneration of Diabetic Wound. Adv Healthc Mater. 2025;14(11):e2500734. [38] WU Y, HUO S, LIU S, et al. Cu-Sr Bilayer Bioactive Glass Nanoparticles/Polydopamine Functionalized Polyetheretherketone Enhances Osteogenic Activity and Prevents Implant-Associated Infections through Spatiotemporal Immunomodulation. Adv Healthc Mater. 2023;12(32):e2301772. [39] LIU XL, ZHANG CJ, SHI JJ, et al. Nacre-mimetic cerium-doped nano-hydroxyapatite/chitosan layered composite scaffolds regulate bone regeneration via OPG/RANKL signaling pathway. J Nanobiotechnology. 2023;21(1):259. [40] LIANG QL, XU HG, YU L, et al. Binding-induced fibrillogenesis peptide inhibits RANKL-mediated osteoclast activation against osteoporosis. Biomaterials. 2023;302:122331. [41] CASEY AM, RYAN DG, PRAG HA, et al. Pro-inflammatory macrophages produce mitochondria-derived superoxide by reverse electron transport at complex I that regulates IL-1β release during NLRP3 inflammasome activation. Nat Metab. 2025;7(3):493-507. [42] JIN Z, XU H, ZHAO W, et al. Macrophage ATF6 accelerates corticotomy-assisted orthodontic tooth movement through promoting Tnfα transcription. Int J Oral Sci. 2025;17(1):28. |
| [1] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [2] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [3] | Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718. |
| [4] | Pi Zhilong, Li Jiayuan, Tan Zhichao, Lu Xiaomei, Zhang Zhiqiang, Ye Xiangling. 3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6736-6743. |
| [5] | Zhou Yunqi, Liu Xu, Xiao Dongqin, Li Xingping, Shi Feng, Zhang Bo, Pu Chao, Luo Xuwei, Zhang Chengdong. Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6768-6778. |
| [6] | Chen Gang, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Wang Qianliang. Mechanism of ferrostatin-1 hydrogel in treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6807-6813. |
| [7] | Zhou Lijing, Wang Shuang, Xiang Jinjiao, Wang Huichao, Chai Xuejiao. Apical sealing and resistance strength of C-Root BP material in in vitro environment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6868-6872. |
| [8] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [9] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [10] | Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936. |
| [11] | Chen Weifei, Mei Yuandong, Ju Jihui. Repair of infected bone defect with dual-ion time-sequenced release multifunctional hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5188-5200. |
| [12] | Zhou Xiaohui, Wang Siyi, Zhou Qiyun, He Zhao, Jia Yujuan, Wang Yuanbin, Ma Jianwu, Chen Gang, Zheng Feng, Chu Genglei. Nanohydroxyapatite-polyether carbonate urethane electrospinning membrane promotes bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5134-5142. |
| [13] | Diao Youlu, Gao Jia, Pan Guoqing. Recruitable tissue repair biomaterials: advantages of regulating cell and factor migration and improving tissue integration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5270-5281. |
| [14] | Xu Yawei, Meng Shilong, Zhang Xu, Wang Chengjie, Yuan Yifeng, Shi Xiaolin, Wang Jiao, Liu Kang . Repairing bone defects with active ingredients of traditional Chinese medicine combined with hydrogels: successes and challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5295-5303. |
| [15] | Wang Zitong, Wu Zijian, Yang Aofei, Mao Tian, Fang Nan, Wang Zhigang. Biomaterials regulate microenvironment imbalance for treating spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5321-5330. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||