Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6736-6743.doi: 10.12307/2026.409
Previous Articles Next Articles
3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities
Pi Zhilong1, Li Jiayuan1, Tan Zhichao2, Lu Xiaomei3, Zhang Zhiqiang3, Ye Xiangling2
- 1Graduate School of Guangzhou University of Chinese Medicine, Guangzhou 510006, Guangdong Province, China; 2Dongguan Hospital, Guangzhou University of Chinese Medicine, Dongguan 523000, Guangdong Province, China; 3Dongguan Eighth People's Hospital, Dongguan 523320, Guangdong Province, China
-
Accepted:2025-11-14Online:2026-09-18Published:2026-03-10 -
Contact:Ye Xiangling, MD, Attending physician, Dongguan Hospital, Guangzhou University of Chinese Medicine, Dongguan 523000, Guangdong Province, China -
About author:Pi Zhilong, MS candidate, Graduate School of Guangzhou University of Chinese Medicine, Guangzhou 510006, Guangdong Province, China -
Supported by:Guangdong Basic and Applied Basic Research Foundation, No. 2023A1515110833 (to YXL); China Postdoctoral Science Foundation, No. 2023M740862 and No. 2024T170198 (to YXL); Guangdong Province Basic and Applied Funds Basic Research Project-Regional Joint Fund-Regional Cultivation Project, No. 2021B1515140002 (to LXM); Dongguan Social Development Science and Technology Project (Key Project), No. 20231800935402 (to TZC)
CLC Number:
Cite this article
Pi Zhilong, Li Jiayuan, Tan Zhichao, Lu Xiaomei, Zhang Zhiqiang, Ye Xiangling. 3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6736-6743.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
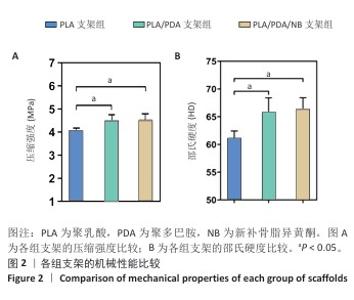
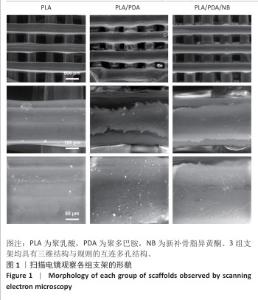
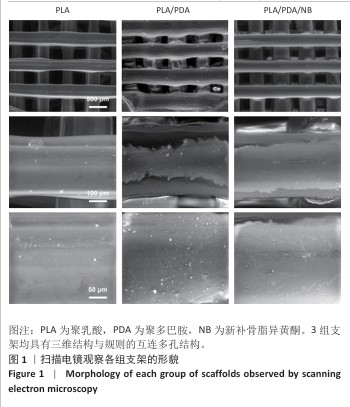
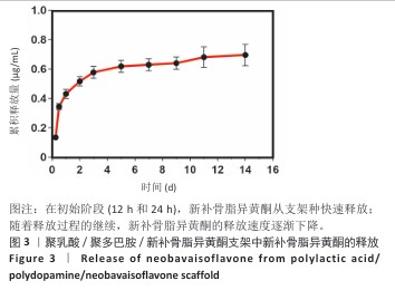
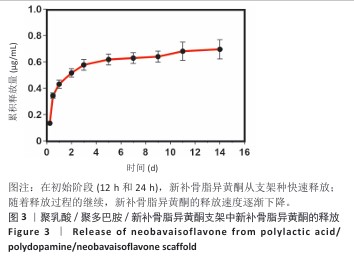
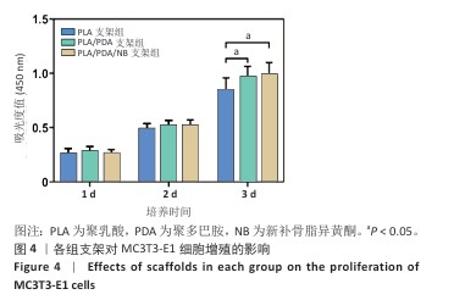
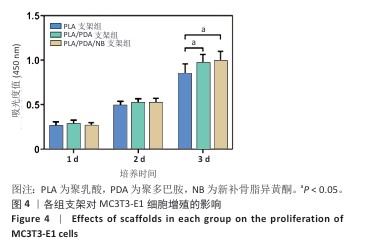
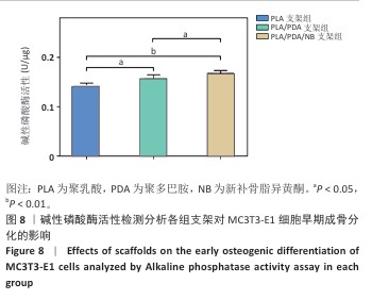
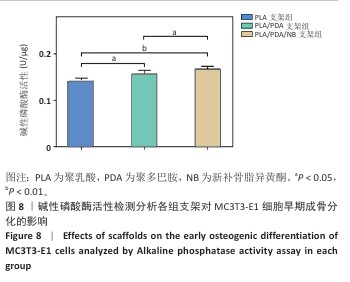
2.2 支架力学性能测试结果 图2显示了3组支架的力学性能。聚乳酸支架、聚乳酸/聚多巴胺支架与PLA/PDA/NB支架的压缩强度分别为(4.06±0.13),(4.52±0.23),(4.55±0.24) MPa,聚乳酸/聚多巴胺支架和PLA/PDA/NB支架的压缩强度大于聚乳酸支架 (P < 0.05)。聚乳酸支架、聚乳酸/聚多巴胺支架与PLA/PDA/NB支架的邵氏硬度分别为(61.17±1.26),(65.83±2.57),(66.33±2.08) HD,聚乳酸/聚多巴胺支架和PLA/PDA/NB支架的邵氏硬度大于聚乳酸支架(P < 0.05)。力学测试结果表明,聚乳酸/聚多巴胺支架和PLA/PDA/NB支架的力学性能较聚乳酸支架有一定提升。"
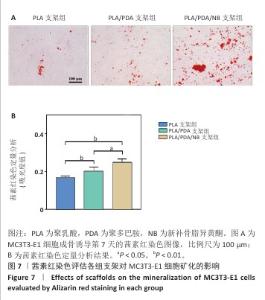
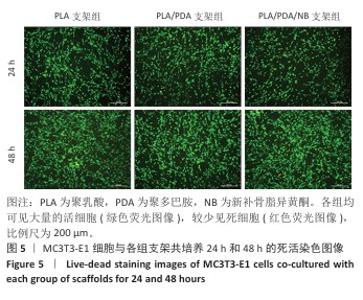
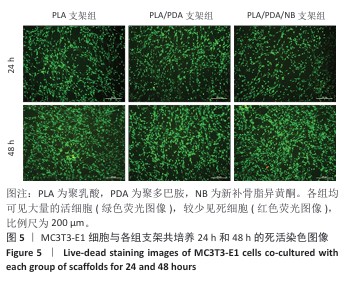
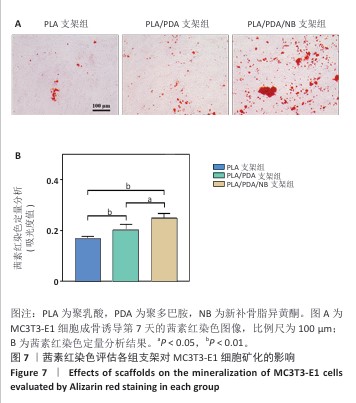
2.7 茜素红染色评估支架对MC3T3-E1细胞矿化的影响 图7A显示了各组MC3T3-E1细胞成骨诱导7d后茜素红染色的代表性图像,与聚乳酸支架组相比,聚乳酸/聚多巴胺支架组、PLA/PDA/NB支架组MC3T3-E1细胞具有更多的钙结节红色染色面积和最多的矿化结节;此外,镜下可见PLA/PDA/NB支架组MC3T3-E1细胞产生大量钙晶体,部分逐渐融合成簇。图7B显示了茜素红染色矿化结节定量分析结果,聚乳酸/聚多巴胺支架组、PLA/PDA/NB支架组矿化结节形成量多于聚乳酸支架组(P < 0.05,P < 0.01),PLA/PDA/NB支架组矿化结节形成量多于聚乳酸/聚多巴胺支架组(P < 0.05)。以上结果表明,PLA/PDA/NB支架有助于促进成骨细胞矿化结节的产生。 "
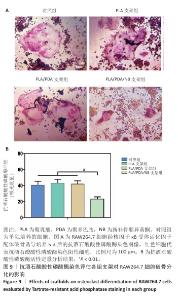
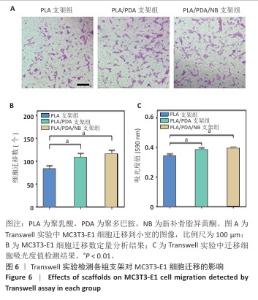
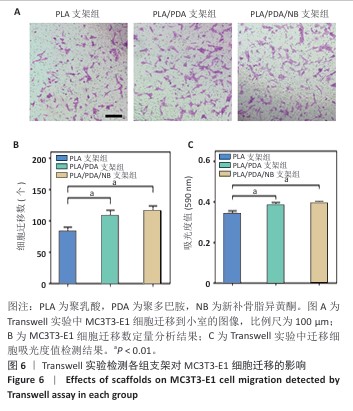
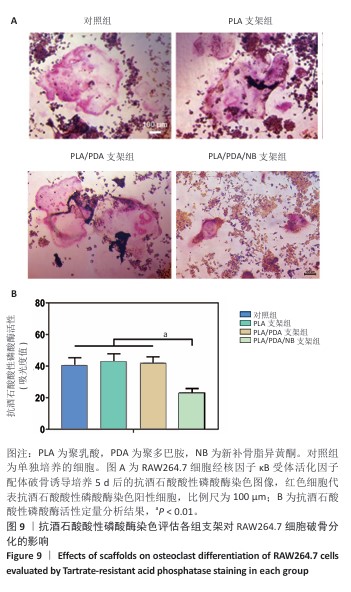
2.9 抗酒石酸酸性磷酸酶染色评估支架对RAW264.7细胞破骨分化的影响 破骨诱导培养5 d后的抗酒石酸酸性磷酸酶染色显示,对照组多个RAW264.7细胞融合形成了具有多核的巨大破骨细胞,这些巨噬细胞在经过抗酒石酸酸性磷酸酶染色后呈红色,说明诱导成功;同时聚乳酸支架组和聚乳酸/聚多巴胺支架组RAW264.7细胞同样融合形成了具有多核的巨大破骨细胞,这说明聚乳酸支架和聚乳酸/聚多巴胺支架并没有抑制破骨细胞骨吸收的能力;PLA/PDA/NB支架组破骨细胞细胞融合显著少于其他3组,多核抗酒石酸酸性磷酸酶染色阳性细胞数量明显减少,见图9A。图9B显示了抗酒石酸酸性磷酸酶活性定量分析结果,PLA/PDA/NB支架组抗酒石酸酸性磷酸酶活性低于其他3组(P < 0.01)。结果表明,PLA/PDA/NB支架具有较强抑制破骨细胞骨吸收的效果。"
| [1] ZHANG LY, BI Q, ZHAO C, et al. Recent Advances in Biomaterials for the Treatment of Bone Defects. Organogenesis. 2020;16(4):113-125. [2] WANG J, LIU M, YANG C, et al. Biomaterials for bone defect repair: Types, mechanisms and effects. Int J Artif Organs. 2024;47(2):75-84. [3] BELLO SAMIR A, CRUZ-LEBRÓN J, RODRÍGUEZ-RIVERA OA, et al. Bioactive Scaffolds as a Promising Alternative for Enhancing Critical-Size Bone Defect Regeneration in the Craniomaxillofacial Region. ACS Appl Bio Mater. 2023;6:4465-4503. [4] REN Y, ZHANG CR, LIU YH, et al. Advances in 3D Printing of Highly Bioadaptive Bone Tissue Engineering Scaffolds. ACS Biomater Sci Eng. 2024;10:255-270. [5] ZHU TY, ZHOU HB, CHEN XJ, et al. Recent advances of responsive scaffolds in bone tissue engineering. Front Bioeng Biotechnol. 2023;11:1296881. [6] MAMIDI N, IJADI F, NORAHAN MH. Leveraging the Recent Advancements in GelMA Scaffolds for Bone Tissue Engineering: An Assessment of Challenges and Opportunities. Biomacromolecules. 2023;25(4):2075-2113. [7] KUMAWAT VS, BANDYOPADHYAY-GHOSH S, GHOSH SB. An overview of translational research in bone graft biomaterials. J Biomater Sci Polym Ed. 2023; 34(4):497-540. [8] MAYFIELD CK, AYAD M, LECHTHOLZ-ZEY E, et al. 3D-Printing for Critical Sized Bone Defects: Current Concepts and Future Directions. Bioengineering (Basel). 2022;9(11):680. [9] ANSARI MAA, GOLEBIOWSKA AA, DASH M, et al. Engineering biomaterials to 3D-print scaffolds for bone regeneration: practical and theoretical consideration. Biomater Sci. 2022;10:2789-2816. [10] KIM HD, AMIRTHALINGAM S, KIM SL, et al. Biomimetic Materials and Fabrication Approaches for Bone Tissue Engineering. Adv Healthc Mater. 2017;6(23).doi: 10.1002/adhm.201700612. [11] YAHARA Y, NGUYEN T, ISHIKAWA K, et al. The origins and roles of osteoclasts in bone development, homeostasis and repair. Development. 2022;149(8): dev199908. [12] ELSON A, ANUJ A, BARNEA-ZOHAR M, et al. The origins and formation of bone-resorbing osteoclasts. Bone. 2022;164:116538. [13] DURDAN MM, AZARIA RD, WEIVODA MM. Novel insights into the coupling of osteoclasts and resorption to bone formation. Semin Cell Dev Biol. 2022;123: 4-13. [14] JIANG T, XIA T, QIAO F, et al. Role and Regulation of Transcription Factors in Osteoclastogenesis. Int J Mol Sci. 2023;24(22):16175. [15] RUAN B, KONG LY, TAKAYA Y, et al. Studies on the chemical constituents of Psoralea corylifolia L. J Asian Nat Prod Res. 2007;9(1):41-44. [16] 李雅茹,杨霞,赵仁双,等.新补骨脂异黄酮通过caspase-3/GSDME通路诱导肝细胞癌Huh-7细胞焦亡[J].中国肿瘤生物治疗杂志,2023,30(4):318-323. [17] 陈辉文.新补骨脂异黄酮抑制破骨细胞分化、减轻小鼠去卵巢后骨质丢失的作用及机制研究[D].上海:中国人民解放军海军军医大学,2021. [18] 徐小昆,陈志永,李卓清,等.新补骨脂异黄酮在Caco-2细胞模型中的吸收机制研究[J].中国中药杂志,2016,41(15):2922-2926. [19] DON MJ, LIN LC, CHIOU WF. Neobavaisoflavone stimulates osteogenesis via p38-mediated up-regulation of transcription factors and osteoid genes expression in MC3T3-E1 cells. Phytomedicine. 2012;19(6):551-561. [20] CHEN H, FANG C, ZHI X, et al. Neobavaisoflavone inhibits osteoclastogenesis through blocking RANKL signalling-mediated TRAF6 and c-Src recruitment and NF-κB, MAPK and Akt pathways. J Cell Mol Med. 2020;24(16):9067-9084. [21] MURRAY DJ, VESELY MJJ, NOVAK CB, et al. Bisphosphonates and avascular necrosis of the mandible: Case report and review of the literature. J Plast Reconstr Aesthet Surg. 2008;61(1):94-98. [22] TAKEMOTO RC, MCLAURIN TM, TEJWANI N, et al. Evolution of atypical femur fractures and the association with bisphosphonates. Bull Hosp Jt Dis (2013). 2014;72(1):104-109. [23] KIM WJ, RYU JH, KIM JW, et al. Bone-targeted lipoplex-loaded three-dimensional bioprinting bilayer scaffold enhanced bone regeneration. Regen Biomater. 2024;11:rbae055. [24] WANG H, LIU D, YANG Z, et al. AssoIciation of bone morphogenetic protein-2 gene polymorphisms with susceptibility to ossification of the posterior longitudinal ligament of the spine and its severity in Chinese patients. Eur Spine J. 2008;17(7):956-964. [25] ZHAO Y, GUO J, MU Q, et al. Exploring quality evaluation markers of Fructus Psoraleae based on chemometric analysis integrated with network pharmacology. Phytochem Anal. 2024;35(2):321-335. [26] SZLISZKA E, CZUBA ZP, SEDEK L, et al. Enhanced TRAL-mediated apoptosis in prostate cancer cells by the bioactive compounds neobavaisoflavone and psoralidin isolated from Psoralea corylifolia. Pharmacol Rep. 2011;63(1):139-148. [27] CHEN Y, QIU Z, HU X, et al. Biofunctional supramolecular injectable hydrogel with spongy-like metal-organic coordination for effective repair of critical-sized calvarial defects. Asian J Pharm Sci. 2025;20(1):100988. [28] LEE H, DELLATORE SM, MILLER WM, et al. Mussel-inspired surface chemistry for multifunctional coatings. Science. 2007;318(5849):426-430. [29] CHIEN HW, CHIU TH. Stable N-halamine on polydopamine coating for high antimicrobial efficiency. Eur Polym J. 2020;130:109654. [30] DING Y, YANG Z, BI CWC, et al. Modulation of protein adsorption, vascular cell selectivity and platelet adhesion by mussel-inspired surface functionalization. J Mater Chem B. 2014;2(24):3819-3829. [31] LEUNG JM, BERRY LR, ATKINSON HM, et al. Surface modification of poly(dimethylsiloxane) with a covalent antithrombin-heparin complex for the prevention of thrombosis: use of polydopamine as bonding agent. J Mater Chem B. 2015;3(29):6032-6036. [32] QIN T, LIU L, CAO H, et al. Polydopamine modified cellulose nanocrystals (CNC) for efficient cellulase immobilization towards advanced bamboo fiber flexibility and tissue softness. Int J Biol Macromol. 2023;253(Pt 6):126734. [33] DENG ZJ, WANG WW, XU X, et al. Biofunction of Polydopamine Coating in Stem Cell Culture. ACS Appl Mater Interfaces. 2021;13:10748-10759. [34] YANG J, BI X, LI M. Osteoclast Differentiation Assay. Pancreatic Cancer. Humana Press, New York, NY, 2019:143-148. [35] ZHU G, ZHANG T, CHEN M, et al. Bone physiological microenvironment and healing mechanism: Basis for future bone-tissue engineering scaffolds . Bioact Mater. 2021;6(11):4110-4140. [36] KENKRE JS, BASSETT JHD. The bone remodelling cycle. Ann Clin Biochem. 2018; 55(3):308-327. [37] XU J, YU L, LIU F, et al. The effect of cytokines on osteoblasts and osteoclasts in bone remodeling in osteoporosis: a review. Front Immunol. 2023;14:1222129. [38] SIMS NA, MARTIN TJ. Osteoclasts Provide Coupling Signals to Osteoblast Lineage Cells Through Multiple Mechanisms. Ann Rev Physiol. 2020;82:507-529. [39] SIDDIQUI JA, PARTRIDGE NC. Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement. Physiology (Bethesda). 2016;31(3):233-245. [40] AL-BARI AA, AL MAMUN A. Current advances in regulation of bone homeostasis. FASEB Bioadv. 2020;2(11):668-679. [41] SUN Y, LI J, XIE X, et al. Recent Advances in Osteoclast Biological Behavior. Front Cell Dev Biol. 2021;9:788680. [42] HE Y, JIANG H, DONG S. Bioactives and Biomaterial Construction for Modulating Osteoclast Activities. Adv Healthc Mater. 2024;13(6):e2302807. [43] YOKOTA K. Osteoclast differentiation in rheumatoid arthritis. Immunol Med. 2024;47(1):6-11. [44] HASCOËT E, BLANCHARD F, BLIN-WAKKACH C, et al. New insights into inflammatory osteoclast precursors as therapeutic targets for rheumatoid arthritis and periodontitis. Bone Res. 2023;11(1):26. [45] KITAURA H, MARAHLEH A, OHORI F, et al. Osteocyte-Related Cytokines Regulate Osteoclast Formation and Bone Resorption. Int J Mol Sci. 2020;21(14):5169. [46] FOROUGHI AH, VALERI C, JIANG D, et al. Understanding compressive viscoelastic properties of additively manufactured PLA for bone-mimetic scaffold design. Med Eng Phys. 2023;114:103972. [47] SALAMANCA E, CHOY CS, AUNG LM, et al. 3D-Printed PLA Scaffold with Fibronectin Enhances In Vitro Osteogenesis. Polymers (Basel). 2023;15(12):2619. [48] SULTAN S, THOMAS N, VARGHESE M, et al. The Design of 3D-Printed Polylactic Acid-Bioglass Composite Scaffold: A Potential Implant Material for Bone Tissue Engineering. Molecules. 2022;27(21):7214. [49] SWANSON WB, OMI M, ZHANG Z, et al. Macropore design of tissue engineering scaffolds regulates mesenchymal stem cell differentiation fate. Biomaterials. 2021;272:120769. [50] BAHRAMINASAB M, DOOSTMOHAMMADI N, TALEBI A, et al. 3D printed polylactic acid/gelatin-nano-hydroxyapatite/platelet-rich plasma scaffold for critical-sized skull defect regeneration. Biomed Eng Online. 2022;21(1):86. [51] GUO W, YANG Y, LIU C, et al. 3D printed TPMS structural PLA/GO scaffold: Process parameter optimization, porous structure, mechanical and biological properties. J Mech Behav Biomed Mater. 2023;142:105848. [52] ZHANG Y, HUO M, WANG Y, et al. A tailored bioactive 3D porous poly(lactic-acid)-exosome scaffold with osteo-immunomodulatory and osteogenic differentiation properties. J Biol Eng. 2022;16(1):22. [53] WANG C, WANG H, CHEN Q, et al. Polylactic acid scaffold with directional porous structure for large-segment bone repair. Int J Biol Macromol. 2022;216:810-819. [54] YE X, ZHANG Y, LIU T, et al. Beta-tricalcium phosphate Enhanced Mechanical and Biological Properties of 3D Printed Polyhydroxyalkanoates Scaffold for Bone Tissue Engineering. Int J Biol Macromol. 2022;209(Pt A):1553-1561. [55] LEE BS. Myosins in Osteoclast Formation and Function. Biomolecules. 2018; 8(4):157. [56] 武瑞骐,章晓云,杨启培,等.补骨脂活性成分治疗骨质疏松症的相关信号通路的研究进展[J].解放军医学杂志,2024,49(5):578-585. |
| [1] | Min Changqin, Huang Ying. Construction of pH/near-infrared laser stimuli-responsive drug delivery system and its application in treatment of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1940-1951. |
| [2] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [3] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [4] | Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718. |
| [5] | Zhang Huifang, Li Hankai, Huang Haozhe, Liu Min, Wang Pin, Huang Haixia, Sun Libo, Lan Yuyan. Osteogenic and antibacterial properties of polyether ether ketone modified by multifunctional composite coating [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6719-6728. |
| [6] | Zhou Yunqi, Liu Xu, Xiao Dongqin, Li Xingping, Shi Feng, Zhang Bo, Pu Chao, Luo Xuwei, Zhang Chengdong. Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6768-6778. |
| [7] | Chen Gang, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Wang Qianliang. Mechanism of ferrostatin-1 hydrogel in treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6807-6813. |
| [8] | Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng. Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6814-6825. |
| [9] | Zhou Lijing, Wang Shuang, Xiang Jinjiao, Wang Huichao, Chai Xuejiao. Apical sealing and resistance strength of C-Root BP material in in vitro environment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6868-6872. |
| [10] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [11] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [12] | Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936. |
| [13] | Chen Weifei, Mei Yuandong, Ju Jihui. Repair of infected bone defect with dual-ion time-sequenced release multifunctional hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5188-5200. |
| [14] | Zhou Xiaohui, Wang Siyi, Zhou Qiyun, He Zhao, Jia Yujuan, Wang Yuanbin, Ma Jianwu, Chen Gang, Zheng Feng, Chu Genglei. Nanohydroxyapatite-polyether carbonate urethane electrospinning membrane promotes bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5134-5142. |
| [15] | Sun Danhe, Guo Xiaoling, Zhao Lingzhou. Construction and osteogenic activity of titanium dioxide nanotube and polydopamine composite coating on titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5167-5177. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||