Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6768-6778.doi: 10.12307/2026.374
Previous Articles Next Articles
Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions
Zhou Yunqi1, 2, Liu Xu1, Xiao Dongqin1, Li Xingping2, Shi Feng3, Zhang Bo1, Pu Chao2, Luo Xuwei1, Zhang Chengdong1
- Research Institute of Tissue Engineering and Stem Cells, Department of Orthopedics of Nanchong Central Hospital, The Second Clinical College of North Sichuan Medical College, Nanchong 637000, Sichuan Province, China; 2Department of Orthopedics, Chengfei Hospital, Chengdu 610091, Sichuan Province, China; 3Collaboration Innovation Center for Tissue Repair Material Engineering Technology, China West Normal University, Nanchong 637000, Sichuan Province, China
-
Accepted:2025-09-12Online:2026-09-18Published:2026-03-11 -
Contact:Luo Xuwei, PhD, Associate chief physician, Research Institute of Tissue Engineering and Stem Cells, Department of Orthopedics of Nanchong Central Hospital, The Second Clinical College of North Sichuan Medical College, Nanchong 637000, Sichuan Province, China Zhang Chengdong, PhD, Assistant research fellow, Research Institute of Tissue Engineering and Stem Cells, Department of Orthopedics of Nanchong Central Hospital, The Second Clinical College of North Sichuan Medical College, Nanchong 637000, Sichuan Province, China -
About author:Zhou Yunqi, Master candidate, Research Institute of Tissue Engineering and Stem Cells, Department of Orthopedics of Nanchong Central Hospital, The Second Clinical College of North Sichuan Medical College, Nanchong 637000, Sichuan Province, China; Department of Orthopedics, Chengfei Hospital, Chengdu 610091, Sichuan Province, China -
Supported by:Natural Science Foundation of Sichuan Province, No. 2023NSFSC1740 (to ZCD); Nanchong City-University Collaborative Research Project, No. 22SXJCQN0002 (to XDQ); Sichuan Medical Association Youth Innovation Project, No. Q2024026 (to LXW); Sichuan Medical Association Youth Innovation Project, No. Q22034 (to LXP)
CLC Number:
Cite this article
Zhou Yunqi, Liu Xu, Xiao Dongqin, Li Xingping, Shi Feng, Zhang Bo, Pu Chao, Luo Xuwei, Zhang Chengdong. Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6768-6778.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
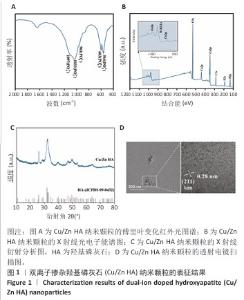
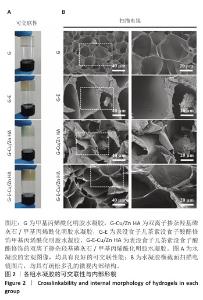
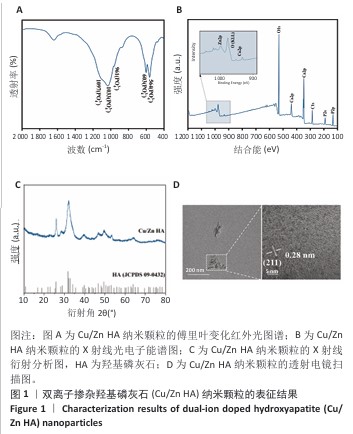
2.1 双离子掺杂羟基磷灰石纳米颗粒的表征 傅里叶变化红外光谱分析显示,双离子掺杂羟基磷灰石纳米颗粒在562,603 cm-1处的吸收峰归属于PO4-3的弯曲振动峰,在961,1 033和1 097 cm-1处的3个峰归属于PO4-3的对称伸缩振动峰,见图1A。 X射线光电子能谱表明,双离子掺杂羟基磷灰石纳米颗粒的主峰包含O1s(530.6 eV)、Ca2p (346.8 eV、438.8 eV)、C1s(284.6 eV)、 P2s(190.5 eV)、P2p(132.9eV);此外还观察到Cu2p(933.9 eV)和Zn2p(1022.3 eV)的峰,证实了Cu2+、Zn2+已成功掺杂在羟基磷灰石纳米颗粒中,见图1B。 X射线衍射分析显示,双离子掺杂羟基磷灰石纳米颗粒符合羟基磷灰石标准衍射图谱(JCPDS 09-0432),见图1C。透射电镜观察微观结构显示,双离子掺杂羟基磷灰石纳米颗粒呈现细针状晶体形态,直径为50-100 nm。使用DM软件(Digital Micrograph,美国)分析双离子掺杂羟基磷灰石纳米颗粒晶格条纹的物相晶格间距,计算得出该晶格条纹间距为0.28 nm,归属于羟基磷灰石(211)晶面,见图1D。综合以上结果表明成功获得了双离子掺杂羟基磷灰石纳米颗粒。 "
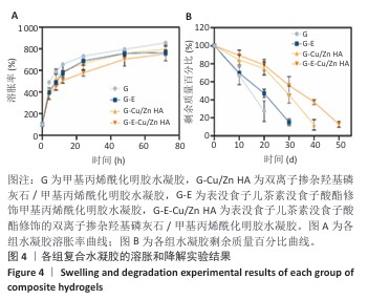
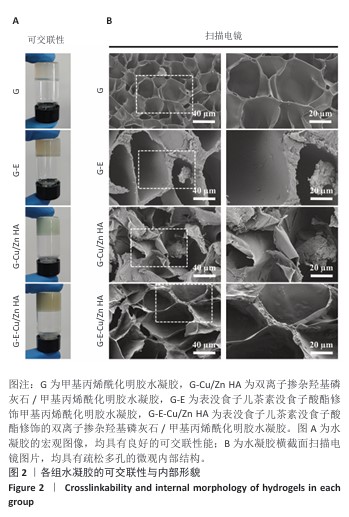
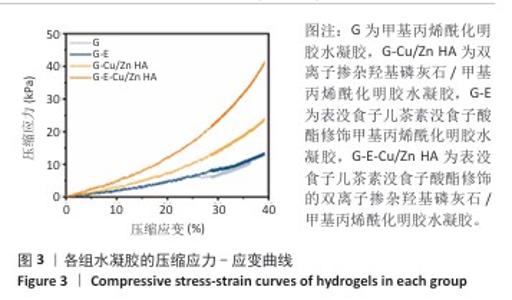
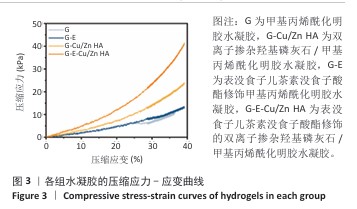
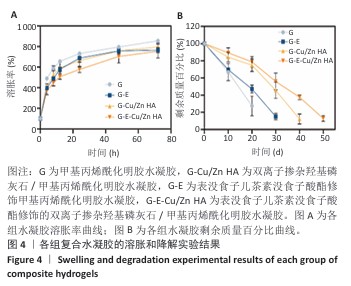
2.4 水凝胶的溶胀及降解检测结果 溶胀实验结果显示,4组水凝胶在最初的4 h内都表现出快速的溶胀行为(> 350%),G水凝胶约在72 h达到溶胀平衡(约为800%),而G-E、G-Cu/Zn HA和G-E-Cu/Zn HA水凝胶约在48 h时已达到溶胀平衡(约为700%),见图4A。 降解实验结果显示,4组水凝胶均表现出其良好的体外生物降解性,G、G-E、G-Cu/Zn HA、G-E-Cu/Zn HA水凝胶在PBS中浸泡第30 天的剩余质量百分比分别约为0%,15%,45%,55%;浸泡第50天,G、G-E、G-Cu/Zn HA水凝胶均已完全降解,仅G-E-Cu/Zn HA水凝胶仍然拥有约13%的剩余质量百分比,见图4B。 "
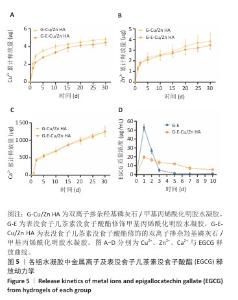
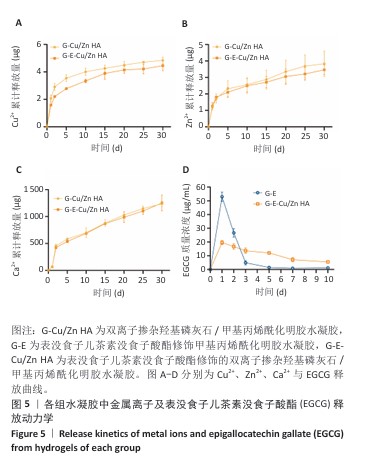
2.5 水凝胶中金属离子及EGCG释放动力学检测结果 金属离子释放动力学检测显示,在30 d的时间内,随着时间的推移,G-Cu/Zn HA和G-E-Cu/Zn HA水凝胶中的Cu2+、Zn2+、Ca2+表现出持续释放趋势,前2 d,水凝胶中Cu2+、Zn2+、Ca2+突释,这是由于水凝胶表面吸附的离子快速解离形成的;5 d后释放速率减缓并趋于稳定;释放30 d后,G-Cu/Zn HA水凝胶中Cu2+、Zn2+、Ca2+释放总量分别为4.85,3.85,1 236.71 μg,G-E-Cu/Zn HA水凝胶中Cu2+、Zn2+、Ca2+释放总量分别为4.45,3.47 ,1 251.8 μg,见图5A-C。 EGCG释放动力学曲线显示,G-E水凝胶呈现典型的两阶段释放特征:初始阶段EGCG释放速率显著升高,第1,2天时释放的EGCG质量浓度分别达到53.1,26.7 μg/mL,约为总释放量的60.2%和30.2%;随后进入缓释平台期,释放速率显著降低,第3,5,7,10天释放的EGCG质量浓度分别为4.9,1.5,0.9及1.2 μg/mL,第3-10天内释放量仅占EGCG总释放量的9.6%;不同于G-E水凝胶,G-E-Cu/Zn HA水凝胶第1,2,3,5,7,10天释放的EGCG质量浓度分别为19.6,16.7,13.7,12.1,7.1,5.5 μg/mL,呈梯度递减趋势,见图5D,这是由于EGCG分子中的邻苯二酚基团与Cu2+、Zn2+通过配位键形成螯合显著抑制了EGCG的突释现象。 "
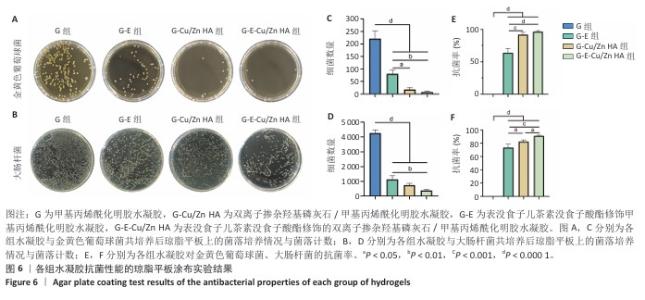
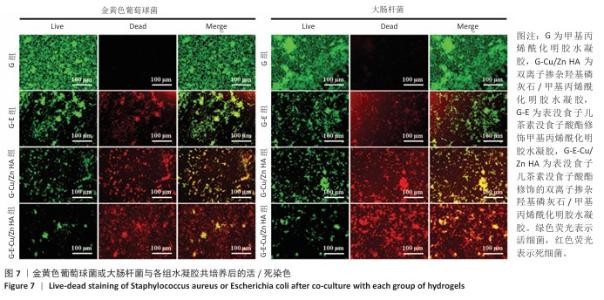
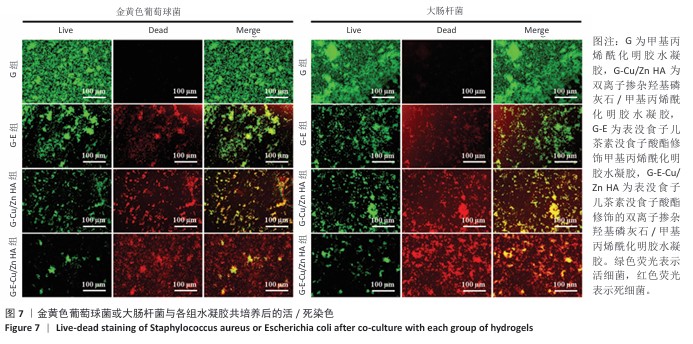
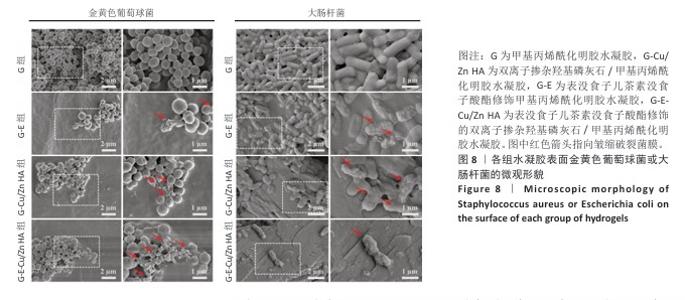
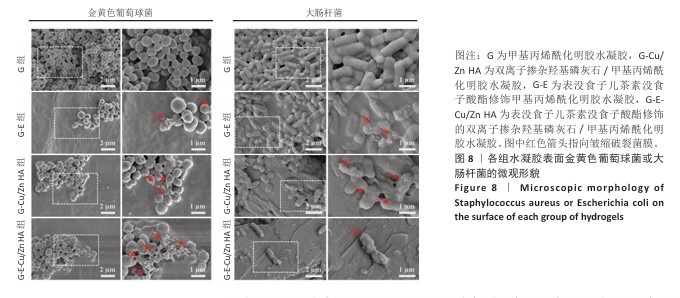
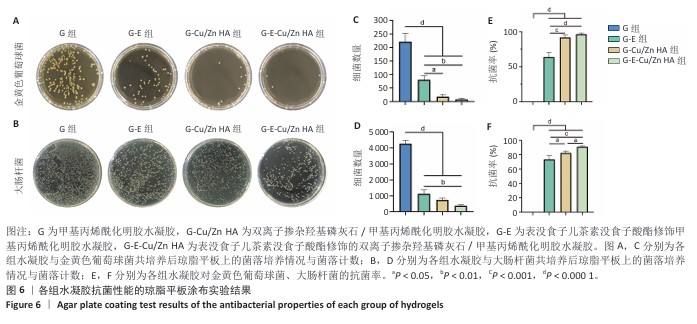
2.6 水凝胶的体外抗菌活性检测结果 对琼脂平板上的金黄色葡萄球菌及大肠杆菌菌落进行观察并计数,结果显示,与G组比较,G-E组、G-Cu/Zn组金黄色葡萄球菌及大肠杆菌细菌数量均有所下降,G-E-Cu/Zn HA组金黄色葡萄球菌及大肠杆菌细菌数量降低更为明显(图6A-D)。G-E、G-Cu/Zn HA、G-E-Cu/Zn HA水凝胶对于金黄色葡萄球菌的抑菌率分别为(63.8±5.5)%,(91.9±3.0)%,(96.4±1.7)%,对于大肠杆菌的抑菌率分别为(73.5±4.4)%,(82.6±2.3)%,(91.1±1.2)%,见图6E,F。 活/死染色结果显示,G组金黄色葡萄球菌或大肠杆菌表现为大量绿色荧光且无红色荧光,证明大部分金黄色葡萄球菌或大肠杆菌处于存活状态,细菌无明显死亡;相较于G组,G-E组、G-Cu/Zn HA组、G-E-Cu/Zn HA组金黄色葡萄球菌或大肠杆菌绿色荧光减少、红色荧光增多,并且以G-E-Cu/Zn HA组表现最明显,证明金黄色葡萄球菌或大肠杆菌出现明显死亡(图7)。 利用扫描电镜对各组水凝胶表面生长的细菌进行观察,结果显示:G组中金黄色葡萄球菌或大肠杆菌大量生长,呈典型的球形或长杆状,菌膜光滑完整;G-E组、G-Cu/Zn HA组、G-E-Cu/Zn HA组金黄色葡萄球菌或大肠杆菌形态发生改变,密度明显减小,部分细菌菌膜已发生皱缩破裂(红色箭头指向为已皱缩破裂菌膜),尤其以G-E-Cu/Zn HA组表现最明显,见图8。 "
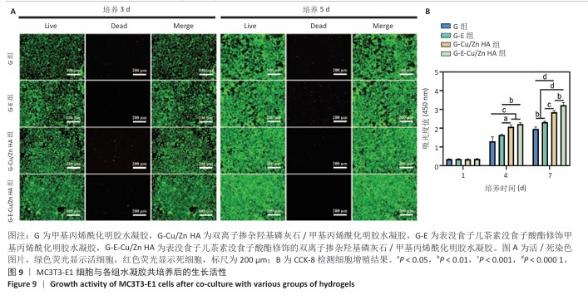
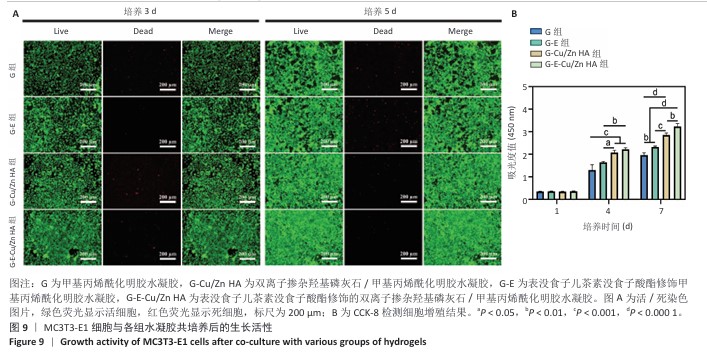
2.7 水凝胶的细胞相容性评估结果 活/死染色结果显示,与4组水凝胶共培养3,5 d后的MC3T3-E1细胞均生长良好、形态正常,仅见少量死细胞,并且各组培养5 d的活细胞数量明显多于培养3 d,见图9A。CCK-8检测结果显示,随着培养时间的延长,与4组水凝胶共培养的MC3T3-E1细胞均出现增殖,培养1 d后,各组间细胞数量无明显差异(P > 0.05);培养4 d后,G-Cu/Zn HA组及G-E-Cu/Zn HA组细胞数量多于G组、G-E组(P < 0.05,P < 0.01,P < 0.001);培养7 d后,G-E-Cu/Zn HA组细胞数量多于其他3组 (P < 0.01,P < 0.000 1),见图9B。活/死细胞染色和CCK-8检测表明4组水凝胶均具有良好的细胞相容性,并且G-E-Cu/Zn HA水凝胶可促进MC3T3-E1细胞增殖。"
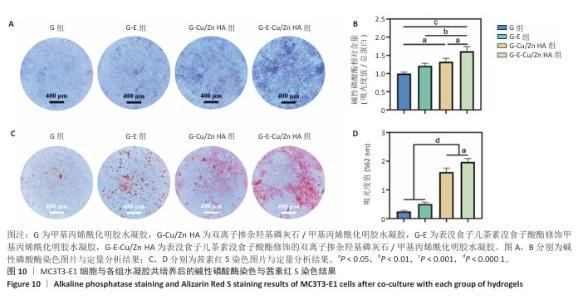
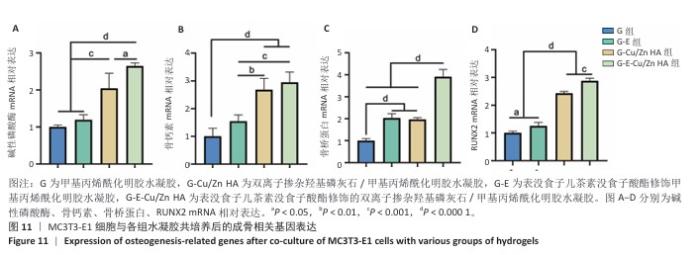
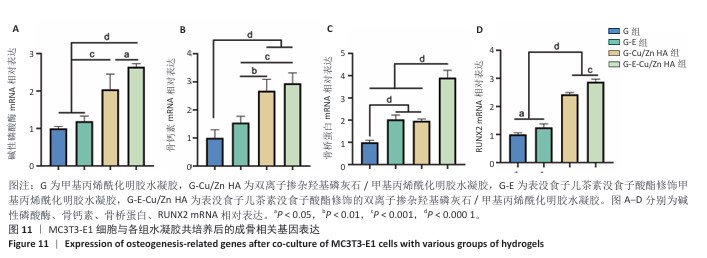
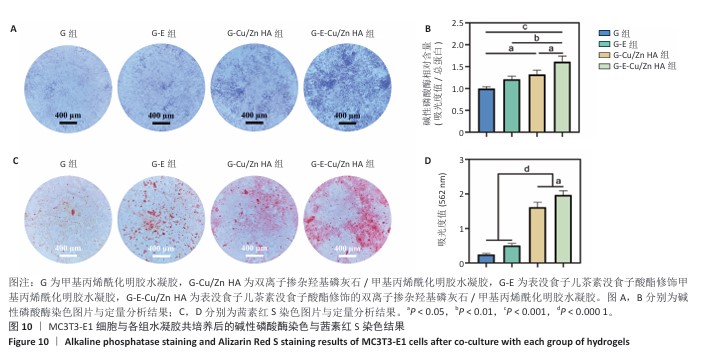
2.8 水凝胶的促成骨活性检测结果 碱性磷酸酶染色:碱性磷酸酶是评估成骨分化的早期标志物,此次研究通过检测碱性磷酸酶评估各组水凝胶对MC3T3-E1细胞成骨分化的影响,结果显示,与G组、G-E组和G-Cu/Zn HA组相比,G-E-Cu/Zn HA组碱性磷酸酶阳性染色面积更大,蓝紫色更深,见图10A。碱性磷酸酶活性定量分析结果显示,G-E-Cu/Zn HA组碱性磷酸酶含量分别较G组、G-E组和G-Cu/Zn HA组增高了61.1%,32.9%,22.0%,见图10B。 茜素红S染色:茜素红S染色图像显示,G组桔红色钙结节沉积最少,G-E-Cu/Zn HA组钙结节沉积最多,见图10C。茜素红S染色定量分析结果显示,G-E-Cu/Zn HA组骨矿化水平相较于G组、G-E组和G-Cu/Zn HA组分别升高了700.8%,290.1%,21.7%,见图10D。 成骨相关基因表达:RT-qPCR测定结果显示,G-E-Cu/Zn HA组碱性磷酸酶、骨桥蛋白、RUNX2 mRNA表达高于其他3组,G-Cu/Zn HA组和G-E-Cu/Zn HA组骨钙素mRNA表达高于其他2组,差异均有显著性意义,见图11,表明G-E-Cu/Zn HA水凝胶具有显著的促成骨相关基因表达能力。"
| [1] NAUTH A, SCHEMITSCH E, NORRIS B, et al. Critical-Size bone defects: Is there a consensus for diagnosis and treatment? J Orthop Trauma. 2018;32(3):S7-S11. [2] BALDWIN P, LI DJ, AUSTON DA, et al. Autograft, allograft, and bone graft substitutes: Clinical evidence and indications for use in the setting of orthopaedic trauma surgery. J Orthop Trauma. 2019;33(4):203-213. [3] TAKAMOTO M, TAKECHI M, OHTA K, et al. Risk of bacterial contamination of bone harvesting devices used for autogenous bone graft in implant surgery. Head Face Med. 2013;9(1):1-5. [4] 任波,唐永亮,李妮,等.温敏抗菌水凝胶治疗感染性骨缺损[J].中国组织工程研究,2025,29(34):7269-7277. [5] SAINT-PASTOU TERRIER C, GASQUE P. Bone responses in health and infectious diseases: A focus on osteoblasts. J Infect. 2017;75(4):281-292. [6] INZANA JA, SCHWARZ EM, KATES SL, et al. Biomaterials approaches to treating implant-associated osteomyelitis. Biomaterials. 2016;81(10):58-71. [7] ZHANG LY, BI Q, ZHAO C, et al. Recent advances in biomaterials for the treatment of bone defects. Organogenesis. 2020;16(4):113-125. [8] YUE K, TRUJILLO-DE SANTIAGO G, ALVAREZ MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73(12):254-271. [9] SUN M, SUN X, WANG Z, et al. Synthesis and properties of gelatin methacryloyl (GelMA) hydrogels and their recent applications in load-bearing tissue. Polymers - Basel. 2018;10(11):1290-1310. [10] DONG Z, YUAN Q, HUANG K, et al. Gelatin methacryloyl (GelMA)-based biomaterials for bone regeneration. RSC Adv. 2019;9(31):17737-17744. [11] WANG J, WANG X, LIANG Z, et al. Injectable antibacterial Ag-HA/ GelMA hydrogel for bone tissue engineering. Front Bioeng Biotech. 2023;11(7): 1219460-1219473. [12] GEORGE SM, NAYAK C, SINGH I, et al. Multifunctional hydroxyapatite composites for orthopedic applications: A review. ACS Biomater Sci Eng. 2022;8(8): 3162-3186. [13] LÓPEZ EO, BERNARDO PL, CHECCA NR, et al. Hydroxyapatite and lead-substituted hydroxyapatite near-surface structures: Novel modelling of photoemission lines from X-ray photoelectron spectra. Appl Surf Sci. 2022;571(10):151310-151322. [14] LV XF, ZHOU DM, SUN XH, et al. Nano sized hydroxyapatite-polylactic acid-vancomycin in alleviation of chronic osteomyelitis. Drug Des Dev Ther. 2022; 16(7):1983-1993. [15] LIM PN, WANG Z, TONG SY, et al. Silver,silicon co-substituted hydroxyapatite modulates bacteria-cell competition for enhanced osteogenic function. Biomed Mater. 2021;16(5):055018-055057. [16] MENDES SOARES IP, ANSELMI C, KITAGAWA FA, et al. Nano-hydroxyapatite-incorporated polycaprolactone nanofibrous scaffold as a dentin tissue engineering-based strategy for vital pulp therapy. Dent Mater. 2022;38(6): 960-977. [17] NOORI A, HOSEINPOUR M, KOLIVAND S, et al. Exploring the various effects of Cu doping in hydroxyapatite nanoparticle. Sci Rep. 2024;14(1):3421-3434. [18] GUPTA S, QAYOOM I, MAIRAL A, et al. Local delivery of exosomes and antibiotics in hydroxyapatite-based nanocement for osteomyelitis management. ACS Infect Dis. 2024;10(11):3994-4008. [19] WU Q, XU S, WANG X, et al. Complementary and synergistic effects on osteogenic and angiogenic properties of copper-incorporated silicocarnotite bioceramic: In vitro and in vivo studies. Biomaterials. 2021;268(5):515-525. [20] HU X, HE J, QIAO L, et al. Multifunctional dual network hydrogel loaded with novel tea polyphenol magnesium nanoparticles accelerates wound repair of MRSA infected diabetes. Adv Funct Mater. 2024;34(22):2312140-2312158. [21] ZHA K, TAN M, HU Y, et al. Regulation of metabolic microenvironment with a nanocomposite hydrogel for improved bone fracture healing. Biomed Mater. 2024;37(9):424-438. [22] ZHANG X, TANG Y, WANG P, et al. A review of recent advances in metal ion hydrogels: mechanism,properties and their biological applications. New J Chem. 2022;46(29):13838-13855. [23] 程新奇,邵龙辉,沈华侨,等.铜锶二元掺杂硅酸钙涂层改性钛合金的促成骨和抗菌效应[J].中国组织工程研究,2025,29(22):4639-4646. [24] PENG X, MCCLEMENTS DJ, LIU X, et al. EGCG-based nanoparticles: synthesis, properties, and applications. Crit Rev Food Sci Nutr. 2024;43(4):1-22. [25] WU T, CUI C, FAN C, et al. Tea eggs-inspired high-strength natural polymer hydrogels. Biomed Mater. 2021;6(9):2820-2828. [26] CAPASSO L, DE MASI L, SIRIGNANO C, et al. Epigallocatechin gallate (EGCG): Pharmacological properties, biological activities and therapeutic potential. Molecules. 2025;30(3):654. [27] YAO Y, ZHANG H, WANG Z, et al. Reactive oxygen species (ROS)-responsive biomaterials mediate tissue microenvironments and tissue regeneration. J Mater Chem B. 2019;7(33):5019-5037. [28] LEE JY, LIM H, AHN JW, et al. Design of a 3D BMP-2-delivering tannylated PCL scaffold and its anti-oxidant, anti-inflammatory, and osteogenic effects in vitro. Int J Mol Sci. 2018;19(11):3602. [29] ISMIYATIN K, YUANITA T, SARASWATI W, et al. Mixing ratio of nano hydroxyapatite and Epigallocatechin-3-gallate (EGCG) towards viscosity and antibacterial effect as a potential pulp capping Material: An experimental study. Saudi Dent J. 2024;36(7):1006-1009. [30] WEI H, QIN J, HUANG Q, et al. Epigallocatechin-3-gallate (EGCG) based metal-polyphenol nanoformulations alleviates chondrocytes inflammation by modulating synovial macrophages polarization. Biomed Pharmacother. 2023;161(11):114366-114380. [31] ZHANG X, HASANI-SADRABADI MM, ZARUBOVA J, et al. Immunomodulatory microneedle patch for periodontal tissue regeneration. Matter. 2022;5(2): 666-682. [32] SUN Z, CHEN X, MIAO F, et al. Engineering Ag-decorated graphene oxide nano-photothermal platforms with enhanced antibacterial properties for promoting infectious wound healing. Int J Nanomed. 2024;19(8):8901-8927. [33] XIAO D, YANG F, ZHAO Q, et al. Fabrication of a CuZn co-incorporated calcium phosphate scaffold-derived GDF-5 sustained release system with enhanced angiogenesis and osteogenesis properties. RSC Adv. 2018;8(52):29526-29534. [34] ZHANG C, YANG F, XIAO D, et al. Repair of segmental rabbit radial defects with CuZn co-doped calcium phosphate scaffolds incorporating GDF-5 carrier. RSC Adv. 2020;10(4):1901-1909. [35] SALAM N, GIBSON IR. Lithium ion doped carbonated hydroxyapatite compositions: Synthesis, physicochemical characterisation and effect on osteogenic response in vitro. Biomater Adv. 2022;140(9):213068-213082. [36] QIAN J, ZHANG W, CHEN Y, et al. Osteogenic and angiogenic bioactive collagen entrapped calcium/zinc phosphates coating on biodegradable Zn for orthopedic implant applications. Biomater Adv. 2022;136(10):212792-212813. [37] HUANG Q, LI X, ELKHOOLY TA, et al. The Cu-containing TiO2 coatings with modulatory effects on macrophage polarization and bactericidal capacity prepared by micro-arc oxidation on titanium substrates. Colloid Surface B. 2018;170(5):242-250. [38] MING P, LI B, LI Q, et al. Multifunctional sericin-based biomineralized nanoplatforms with immunomodulatory and angio/osteo-genic activity for accelerated bone regeneration in periodontitis. Biomaterials. 2025;314(38): 122885-122897. [39] LI J, KE H, LEI X, et al. Controlled-release hydrogel loaded with magnesium-based nanoflowers synergize immunomodulation and cartilage regeneration in tendon-bone healing. Biomed Mater. 2024;36(27):62-82. [40] FOWLER L, ENGQVIST H, ÖHMAN-MÄGI C. Effect of copper ion concentration on bacteria and cells. Materials (Basel). 2019;12(22):3798. [41] JENSEN LK, BJARNSHOLT T, KRAGH KN, et al. In vivo gentamicin susceptibility test for prevention of bacterial biofilms in bone tissue and on implants. Antimicrob. Agents Chemother. 2019;63(2):e01889-18. [42] ZHAO X, HU J, NIE J, et al. Immunomodulatory effect of Ti-Cu alloy by surface nanostructure synergistic with Cu2+ release. Colloid Surface B. 2023;231(17): 113586-113600 [43] HUANG Q, OUYANG Z, TAN Y, et al. Activating macrophages for enhanced osteogenic and bactericidal performance by Cu ion release from micro/nano-topographical coating on a titanium substrate. Acta Biomater. 2019;100(19): 415-426. [44] YOON JY, KIM TS, AHN JH, et al. Remifentanil promotes osteoblastogenesis by upregulating Runx2/osterix expression in preosteoblastic C2C12 cells. J Dent Anesth Pain Med. 2019;19(2): 91. [45] ZHANG C, ZHOU Z, LIU N, et al. Osteogenic differentiation of 3D-printed porous tantalum with nano-topographic modification for repairing craniofacial bone defects. Front Bioeng Biotechnol. 2023;11:1258030. |
| [1] | Liu Hongjie, Mu Qiuju, Shen Yuxue, Liang Fei, Zhu Lili. Metal organic framework/carboxymethyl chitosan-oxidized sodium alginate/platelet-rich plasma hydrogel promotes healing of diabetic infected wounds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1929-1939. |
| [2] | Min Changqin, Huang Ying. Construction of pH/near-infrared laser stimuli-responsive drug delivery system and its application in treatment of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1940-1951. |
| [3] | Wang Songpeng, Liu Yusan, Yu Huanying, Gao Xiaoli, Xu Yingjiang, Zhang Xiaoming, Liu Min. Bidirectional regulation of reactive oxygen species based on zeolitic imidazolate framework-8 nanomaterials: from tumor therapy and antibacterial activity to cytoprotection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2033-2013. |
| [4] | Sun Lei, Zhang Qi, Zhang Yu. Pro-osteoblastic effect of chlorogenic acid protein microsphere/polycaprolactone electrospinning membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1877-1884. |
| [5] | Lai Yu, Chen Yueping, Zhang Xiaoyun. Research hotspots and frontier trends of bioactive materials in treating bone infections [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2132-2144. |
| [6] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [7] | Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936. |
| [8] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [9] | Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718. |
| [10] | Pi Zhilong, Li Jiayuan, Tan Zhichao, Lu Xiaomei, Zhang Zhiqiang, Ye Xiangling. 3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6736-6743. |
| [11] | Chen Gang, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Wang Qianliang. Mechanism of ferrostatin-1 hydrogel in treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6807-6813. |
| [12] | Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng. Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6814-6825. |
| [13] | Zhou Lijing, Wang Shuang, Xiang Jinjiao, Wang Huichao, Chai Xuejiao. Apical sealing and resistance strength of C-Root BP material in in vitro environment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6868-6872. |
| [14] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [15] | Zhou Xiaohui, Wang Siyi, Zhou Qiyun, He Zhao, Jia Yujuan, Wang Yuanbin, Ma Jianwu, Chen Gang, Zheng Feng, Chu Genglei. Nanohydroxyapatite-polyether carbonate urethane electrospinning membrane promotes bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5134-5142. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||