Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6710-6718.doi: 10.12307/2026.804
Previous Articles Next Articles
In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds
Zhao Zhanghong1, Jin Dongsheng1, Ruan Shiqiang1, Huang Wenliang2, Wan Yu1, Tian Renyuan1, Deng Jiang1
- 1Department of Orthopedics, Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China; 2Department of Orthopedics, Moutai Hospital of Guizhou Province, Renhuai 564500, Guizhou Province, China
-
Accepted:2025-12-11Online:2026-09-18Published:2026-03-10 -
Contact:Deng Jiang, Professor, Chief physician, Department of Orthopedics, Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China -
About author:Zhao Zhanghong, Master candidate, Physician, Department of Orthopedics, Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81660367 (to DJ); Guizhou Provincial Science and Technology Project, No. ZK(2021)387 (to HWL); Guizhou Provincial Science and Technology Project, No. LC[2024]019 (to HWL)
CLC Number:
Cite this article
Zhao Zhanghong, Jin Dongsheng, Ruan Shiqiang, Huang Wenliang, Wan Yu, Tian Renyuan, Deng Jiang. In vitro osteogenic and anti-inflammatory properties of icariin sustained-release microsphere three-dimensional scaffolds[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6710-6718.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

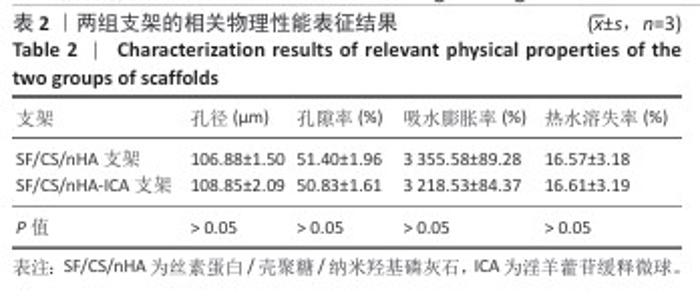
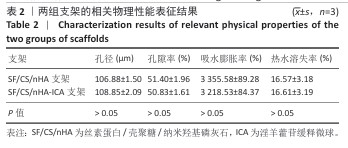
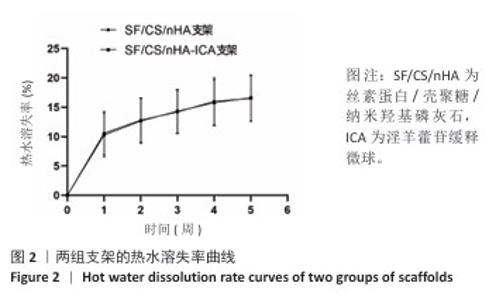
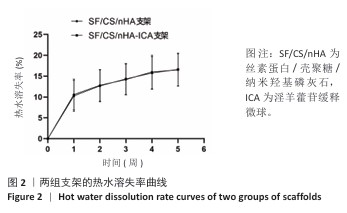
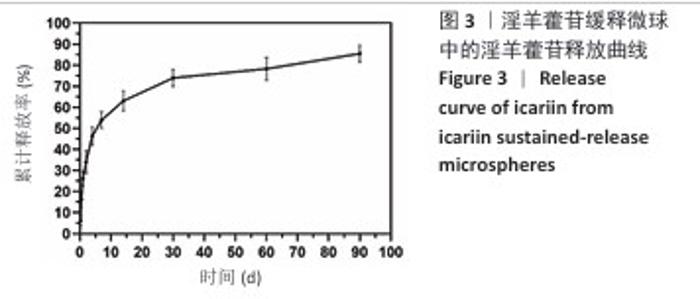
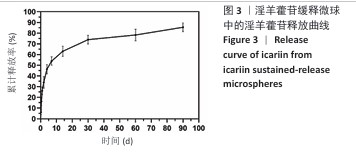
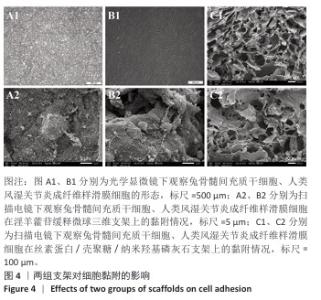
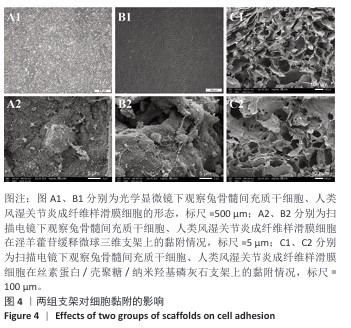
2.1 支架与淫羊藿苷缓释微球相关表征结果 SF/CS/nHA支架和SF/CS/nHA-ICA支架在外观上无显著差异,都是白色圆柱体,大小可根据需要塑形,支架的切割面光滑平坦(图1 A1、B1),可以为细胞黏附和生长提供足够的空间。扫描电镜下可见两组支架内部呈蜂窝状多孔结构,孔隙互连,利于营养物质交换和细胞渗入(图1 A2、B2)。干燥后淫羊藿苷缓释微球为白色粉末,扫描电镜下见微球呈类球形,表面略显粗糙,部分区域可见轻微塌陷(图1 C1、C2)。扫描电镜见淫羊藿苷缓释微球均匀分布于SF/CS/nHA支架表面,微球与支架基体结合紧密,未见明显剥离现象,局部放大见微球表面存在细微孔洞,有利于药物分子释放(图1 D1、D2)。 两组支架的孔径及孔隙率比较差异无显著性意义(P > 0.05),见表2。SF/CS/nHA支架和SF/CS/nHA-ICA支架的吸水膨胀率分别为(3 355.58±89.28)%和(3 218.53±84.37)%,表明两组支架均具有较强的吸水性,在骨修复过程中可为细胞生长提供所需的水分及营养物质。 两组支架的热水溶失率曲线见图2。SF/CS/nHA支架和SF/CS/nHA-ICA支架的总热水溶失率分别为(16.57±3.18)%和(16.61±3.19)%,组间比较差异无显著性意义(P > 0.05),表明两组支架可为细胞生长提供相对稳定的空间,同时具有一定的生物降解性。上述数据表明,添加淫羊藿苷缓释微球后不影响SF/CS/nHA支架的相关物理性能。 淫羊藿苷缓释微球的载药率为(29.38±0.04)%、包封率为(52.01±0.09)%。淫羊藿苷缓释微球存在一定的突释效应,7 d内总体药物释放速率快,此后药物释放速率减慢,90 d药物累积缓释率达85.47%(图3)。 "

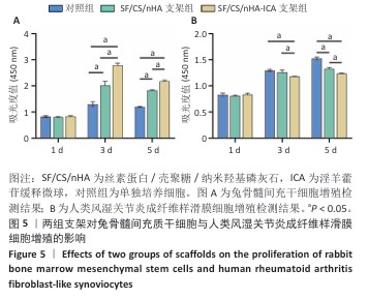
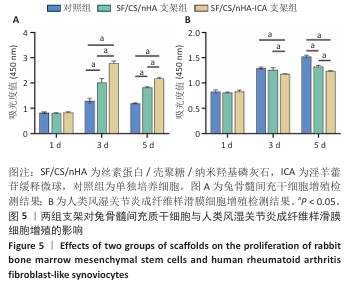
2.3 支架对兔骨髓间充质干细胞与人类风湿关节炎成纤维样滑膜细胞增殖的影响 兔骨髓间充质干细胞:CCK-8检测结果显示,培养第1天,各组细胞增殖吸光度值比较差异无显著性意义(P > 0.05);培养第3,5天,SF/CS/nHA支架组、SF/CS/nHA-ICA支架组细胞增殖吸光度值高于对照组(P < 0.05),SF/CS/nHA-ICA支架组细胞增殖吸光度值高于SF/CS/nHA支架组(P < 0.05),见图5A。表明SF/CS/nHA-ICA支架可促进兔骨髓间充质干细胞的增殖。 人类风湿关节炎成纤维样滑膜细胞:CCK-8检测结果显示,培养第1天,各组细胞增殖吸光度值比较差异无显著性意义(P > 0.05);培养第3天,SF/CS/nHA-ICA支架组细胞增殖吸光度值低于对照组、SF/CS/nHA支架组(P < 0.05);培养第5天,SF/CS/nHA支架组、SF/CS/nHA-ICA支架组细胞增殖吸光度值低于对照组(P < 0.05),SF/CS/nHA-ICA支架组细胞增殖吸光度值低于SF/CS/nHA支架组(P < 0.05),见图5B。表明SF/CS/nHA-ICA支架可抑制人类风湿关节炎成纤维样滑膜细胞的增殖。 "
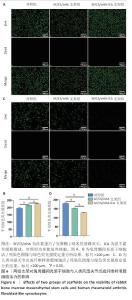
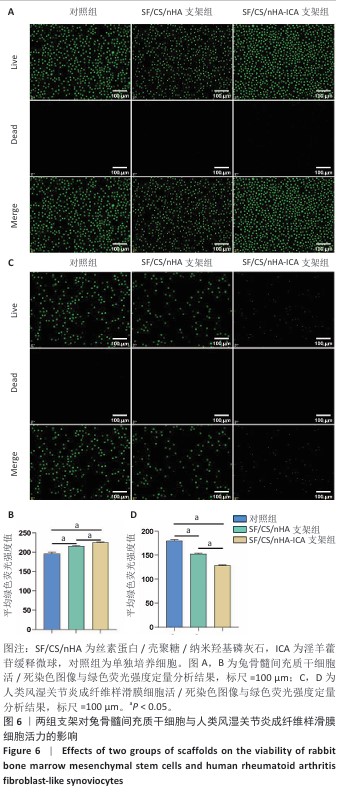
2.4 支架对兔骨髓间充质干细胞与人类风湿关节炎成纤维样滑膜细胞活力的影响 兔骨髓间充质干细胞:活/死染色显示,各组活细胞(绿色)占主导地位,死细胞(红色)数量较少,并且与对照组、SF/CS/nHA支架组相比,SF/CS/nHA-ICA支架组绿色荧光强度更高(P < 0.05),见图6A,B。表明SF/CS/nHA-ICA支架提升兔骨髓间充质干细胞活力。 人类风湿关节炎成纤维样滑膜细胞:活/死染色显示,各组可见大量绿色荧光信号,红色信号稀少,但SF/CS/nHA-ICA支架组绿色荧光强度低于对照组、SF/CS/nHA支架组(P < 0.05),见图6C,D。表明SF/CS/nHA-ICA支架可抑制人类风湿关节炎成纤维样滑膜细胞活力。 "
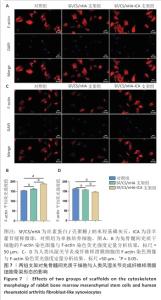
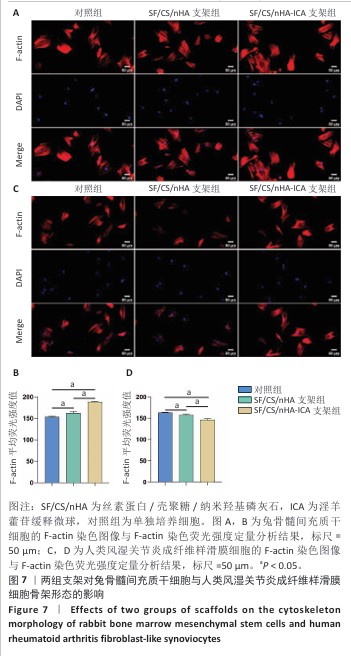
2.5 支架对兔骨髓间充质干细胞与人类风湿关节炎成纤维样滑膜细胞骨架形态的影响 兔骨髓间充质干细胞:F-actin染色显示各组细胞分布均匀,细胞核形态及大小一致,细胞核未见明显异染及碎裂表现,SF/CS/nHA-ICA支架组F-actin染色荧光强度高于对照组、SF/CS/nHA支架组(P < 0.05),见图7A,B。表明SF/CS/nHA-ICA支架可促进兔骨髓间充质干细胞的增殖。 人类风湿关节炎成纤维样滑膜细胞:与对照组比较,SCN-FLS-ICA组、SCN-FLS组细胞骨架形态稍有变形,并且细胞数量减少,SF/CS/nHA-ICA支架组F-actin染色荧光强度低于对照组、SF/CS/nHA支架组(P < 0.05),见图7C,D。表明SF/CS/nHA-ICA支架可抑制人类风湿关节炎成纤维样滑膜细胞的增殖。"
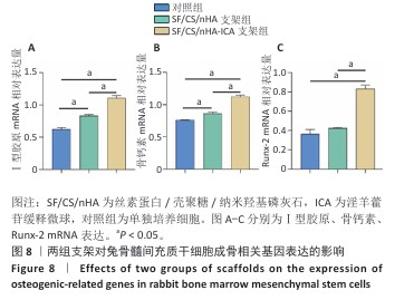
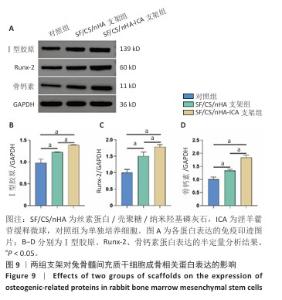
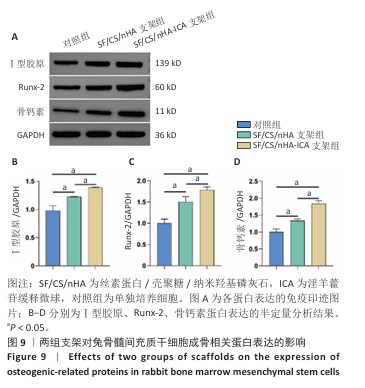
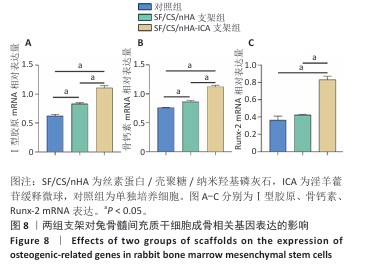
2.6 两组支架对兔骨髓间充质干细胞成骨相关基因、蛋白表达的影响 RT-qPCR 检测结果显示,SF/CS/nHA支架组Ⅰ型胶原、骨钙素mRNA表达高于对照组(P < 0.05),SF/CS/nHA-ICA支架组Runx-2、骨钙素、Ⅰ型胶原mRNA表达高于对照组、SF/CS/nHA支架组(P < 0.05),见图8。Western blot检测结果显示,SF/CS/nHA支架组Runx-2、Ⅰ型胶原、骨钙素蛋白表达高于对照组(P < 0.05),SF/CS/nHA-ICA支架组Runx-2、骨钙素、Ⅰ型胶原蛋白表达高于SF/CS/nHA支架组(P < 0.05),见图9。结果表明,SF/CS/nHA-ICA支架具有一定诱导成骨潜在作用。"
| [1] GVARAMIA D, KERN J, JAKOB Y, et al. Regenerative Potential of Perichondrium: A Tissue Engineering Perspective. Tissue Eng Part B Rev. 2022;28(3):531-541. [2] LIU B, XIAN Y, CHEN X, et al. Inflammatory Fibroblast-Like Synoviocyte-Derived Exosomes Aggravate Osteoarthritis via Enhancing Macrophage Glycolysis. Adv Sci (Weinh). 2024;11(14):e2307338. [3] YANG J, LI S, LI Z, et al. Targeting YAP1-regulated Glycolysis in Fibroblast-Like Synoviocytes Impairs Macrophage Infiltration to Ameliorate Diabetic Osteoarthritis Progression. Adv Sci (Weinh). 2024;11(5):e2304617. [4] XIE Y, ZINKLE A, CHEN L, et al. Fibroblast growth factor signalling in osteoarthritis and cartilage repair. Nat Rev Rheumatol. 2020;16(10):547-564. [5] RUIZ-ALONSO S, LAFUENTE-MERCHAN M, CIRIZA J, et al. Tendon tissue engineering: Cells, growth factors, scaffolds and production techniques. J Control Release. 2021;333:448-486. [6] LIANG W, ZHOU C, LIU X, et al. Current status of nano-embedded growth factors and stem cells delivery to bone for targeted repair and regeneration. J Orthop Translat. 2025;50:257-273. [7] REN Y, XU Z, XU Y, et al. Advanced Strategies in Bone Tissue Engineering: “Membrane-Jelly” Hydrogel System to Improve Bone Marrow Stem Cell Osteogenic Differentiation and Bone Regeneration. ACS Appl Mater Interfaces. 2025;17(24):34982-34996. [8] BISCONTI F, VILARDO B, CORALLO G, et al. An Assist for Arthritis Studies: A 3D Cell Culture of Human Fibroblast‐Like Synoviocytes by Encapsulation in a Chitosan‐Based Hydrogel. Advanced Therapeutics. 2024;7(12). doi:10.1002/adtp.202470027. [9] 冯美杰,李蕾,王颖航,等.黄芪甲苷对人类风湿关节炎成纤维样滑膜细胞增殖及白细胞介素6分泌的影响[J].中国中医基础医学杂志,2020, 26(10):1484-1487. [10] WANG X, TANG P, YANG K, et al. Regulation of bone homeostasis by traditional Chinese medicine active scaffolds and enhancement for the osteoporosis bone regeneration. J Ethnopharmacol. 2024;329:118141. [11] GAO ZR, FENG YZ, ZHAO YQ, et al. Traditional Chinese medicine promotes bone regeneration in bone tissue engineering. Chin Med. 2022;17(1):86. [12] 薛春阳,王秀会.淫羊藿苷调节酸性微环境减轻绝经后老年骨质疏松性疼痛[J].中国组织工程研究,2024,28(28):4461-4468. [13] 何文凤,薛成,郑健康,等.淫羊藿苷联合可注射壳聚糖/胶原复合水凝胶对心肌梗死大鼠血管新生的作用[J].中国组织工程研究,2023,27(25): 3992-3998. [14] CUI J, LIN L, HAO F, et al. Comprehensive review of the traditional uses and the potential benefits of epimedium folium. Front Pharmacol. 2024;15:1415265. [15] MIAO M, LI M, SHENG Y, et al. Epimedium-Curculigo herb pair enhances bone repair with infected bone defects and regulates osteoblasts through LncRNA MALAT1/miR-34a-5p/SMAD2 axis. J Cell Mol Med. 2024;28(13):e18527. [16] CAI Z, SUN F, WANG Q, et al. Icariin alleviates cardiomyocyte pyroptosis through AMPK-NLRP3 pathway to ameliorates diabetic cardiomyopathy. Int Immunopharmacol. 2025;156:114690. [17] HAN L, TIAN X, YANG X, et al. The pathogenesis of hepatocellular carcinoma: ERK/ULK1/NCOA4-mediated inhibition of iron autophagy, and Epimedium extract targeted modulation of this pathway to treat hepatocellular carcinoma. Phytomedicine. 2025;141:156666. [18] 熊伟,袁灵梅,钱国文,等. “补肾壮骨”中药应用于骨组织工程支架修复节段性骨缺损[J].中国组织工程研究,2023,27(21):3438-3444. [19] XIE Y, SUN W, YAN F, et al. Icariin-loaded porous scaffolds for bone regeneration through the regulation of the coupling process of osteogenesis and osteoclastic activity. Int J Nanomedicine. 2019;14:6019-6033. [20] XU Z, SUN Y, DAI H, et al. Engineered 3D-Printed Polyvinyl Alcohol Scaffolds Incorporating β-Tricalcium Phosphate and Icariin Induce Bone Regeneration in Rat Skull Defect Model. Molecules. 2022;27(14):4535. [21] LIU T, ZHANG X, LUO Y, et al. Slowly Delivered Icariin/Allogeneic Bone Marrow-Derived Mesenchymal Stem Cells to Promote the Healing of Calvarial Critical-Size Bone Defects. Stem Cells Int. 2016;2016:1416047. [22] XIE L, LIU N, XIAO Y, et al. In Vitro and In Vivo Osteogenesis Induced by Icariin and Bone Morphogenetic Protein-2: A Dynamic Observation. Front Pharmacol. 2020;11:1058. [23] WU Y, CAO L, XIA L, et al. Evaluation of Osteogenesis and Angiogenesis of Icariin in Local Controlled Release and Systemic Delivery for Calvarial Defect in Ovariectomized Rats. Sci Rep. 2017;7(1):5077. [24] YANG S, ZHANG X, LIAO X, et al. Icariin regulates RANKL-induced osteoclast differentiation via the ERα/c-Src/RANK signaling. Biomed Mater. 2024;19(2). doi: 10.1088/1748-605X/ad2554. [25] PAN L, ZHANG Y, CHEN N, et al. Icariin Regulates Cellular Functions and Gene Expression of Osteoarthritis Patient-Derived Human Fibroblast-Like Synoviocytes. Int J Mol Sci. 2017;18(12):2565. [26] HSIEH TP, SHEU SY, SUN JS, et al. Icariin inhibits osteoclast differentiation and bone resorption by suppression of MAPKs/NF-κB regulated HIF-1α and PGE(2) synthesis. Phytomedicine. 2011;18(2-3):176-185. [27] RUAN SQ, DENG J, YAN L, et al. Composite scaffolds loaded with bone mesenchymal stem cells promote the repair of radial bone defects in rabbit model. Biomed Pharmacother. 2018;97:600-606. [28] XIAO H, HUANG W, XIONG K, et al. Osteochondral repair using scaffolds with gradient pore sizes constructed with silk fibroin, chitosan, and nano-hydroxyapatite. Int J Nanomedicine. 2019;14:2011-2027. [29] 叶鹏,田仁元,黄文良,等.丝素/壳聚糖/纳米羟基磷灰石构建的骨组织工程支架[J].中国组织工程研究,2013,17(29):5269-5274. [30] ZHANG Y, HUANG W, XIAO H, et al. NGF-BMSC-SF/CS composites for repairing knee joint osteochondral defects in rabbits: evaluation of the repair effect and potential underlying mechanisms. J Orthop Surg Res. 2024;19(1):443. [31] 金东升,赵张红,朱子银,等.淫羊藿苷缓释微球三维支架对兔骨髓间充质干细胞成骨分化的影响[J].中国组织工程研究,2026,30(7):1658-1668. [32] ARIAUDO D, CAVALIERI F, RINALDI A, et al. Alginate Microsponges as a Scaffold for Delivery of a Therapeutic Peptide against Rheumatoid Arthritis. Nanomaterials (Basel). 2023;13(19):2709. [33] PERCIVAL KM, PAUL V,HUSSEINI GA. Recent Advancements in Bone Tissue Engineering: Integrating Smart Scaffold Technologies and Bio-Responsive Systems for Enhanced Regeneration. Int J Mol Sci. 2024;25(11):6012. [34] CROCETTI L, VERGELLI C, GUERRINI G, et al. Novel formyl peptide receptor (FPR) agonists with pyridinone and pyrimidindione scaffolds that are potentially useful for the treatment of rheumatoid arthritis. Bioorg Chem. 2020;100:103880. [35] ZHAO Y, GAO C, LIU H, et al. Infliximab-based self-healing hydrogel composite scaffold enhances stem cell survival, engraftment, and function in rheumatoid arthritis treatment. Acta Biomater. 2021;121:653-664. [36] QUAN S, YANG J, HUANG S, et al. Silk fibroin as a potential candidate for bone tissue engineering applications. Biomater Sci. 2025;13(2):364-378. [37] NIKNAFS B, MESKARAF-ASADABADI M, HAMDI K, et al. Incorporating bioactive glass nanoparticles in silk fibroin/bacterial nanocellulose composite scaffolds improves their biological and osteogenic properties for bone tissue engineering applications. Int J Biol Macromol. 2024;266(Pt 1):131167. [38] RESSLER A. Chitosan-Based Biomaterials for Bone Tissue Engineering Applications: A Short Review. Polymers (Basel). 2022;14(16):3430. [39] RAY S, NANDI SK, DASGUPTA S. Enhanced bone regeneration usingAntheraea mylittasilk fibroin and chitosan based scaffold:in-vivoandin-vitrostudy. Biomed Mater. 2023;18(5). doi: 10.1088/1748-605X/acee3c. [40] CANCIANI E, STRATICÒ P, VARASANO V, et al. Polylevolysine and Fibronectin-Loaded Nano-Hydroxyapatite/PGLA/Dextran-Based Scaffolds for Improving Bone Regeneration: A Histomorphometric in Animal Study . Int J Mol Sci. 2023; 24(9):8137. [41] MO X, ZHANG D, LIU K, et al. Nano-Hydroxyapatite Composite Scaffolds Loaded with Bioactive Factors and Drugs for Bone Tissue Engineering. Int J Mol Sci. 2023;24(2):1291. [42] ZHANG XY, LI HN, CHEN F, et al. Icariin regulates miR-23a-3p-mediated osteogenic differentiation of BMSCs via BMP-2/Smad5/Runx2 and WNT/β-catenin pathways in osteonecrosis of the femoral head. Saudi Pharm J. 2021;29(12):1405-1415. [43] XU Y, JIANG Y, JIA B, et al. Icariin stimulates osteogenesis and suppresses adipogenesis of human bone mesenchymal stem cells via miR-23a-mediated activation of the Wnt/β-catenin signaling pathway. Phytomedicine. 2021;85: 153485. [44] SI Y, LI Y, GU K, et al. Icariin ameliorates osteoporosis in ovariectomized rats by targeting Cullin 3/Nrf2/OH pathway for osteoclast inhibition. Biomed Pharmacother. 2024;173:116422. [45] SINGH WR, DEVI HS, KUMAWAT S, et al. Angiogenic and MMPs modulatory effects of icariin improved cutaneous wound healing in rats. Eur J Pharmacol. 2019;858:172466. [46] WANG P, MENG Q, WANG W, et al. Icariin inhibits the inflammation through down-regulating NF-κB/HIF-2α signal pathways in chondrocytes. Biosci Rep. 2020;40(11): BSR20203107. [47] HUANG L, WANG J, YU J, et al. Picein alleviates oxidative stress and promotes bone regeneration in osteoporotic bone defect by inhibiting ferroptosis via Nrf2/HO-1/GPX4 pathway. Environ Toxicol. 2024;39(7):4066-4085. [48] PREETHI SOUNDARYA S, HARITHA MENON A, VIJI CHANDRAN S, et al. Bone tissue engineering: Scaffold preparation using chitosan and other biomaterials with different design and fabrication techniques. Int J Biol Macromol. 2018;119: 1228-1239. [49] WU B, WU L, HE Y, et al. Engineered PLGA microspheres for extended release of brexpiprazole: in vitro and in vivo studies. Drug Dev Ind Pharm. 2021;47(6): 1001-1010. [50] ZHAO H, TANG J, ZHOU D, et al. Electrospun Icariin-Loaded Core-Shell Collagen, Polycaprolactone, Hydroxyapatite Composite Scaffolds for the Repair of Rabbit Tibia Bone Defects. Int J Nanomedicine. 2020;15:3039-3056. [51] 魏丽,李德超,王静,等.聚乳酸-壳聚糖纤维/羟基磷灰石-硅酸钙复合支架材料的细胞相容性[J].中国组织工程研究与临床康复,2010,14(8): 1397-1401. |
| [1] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [2] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [3] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [4] | Pi Zhilong, Li Jiayuan, Tan Zhichao, Lu Xiaomei, Zhang Zhiqiang, Ye Xiangling. 3D printed neobavaisoflavone-coated scaffolds promote bone regeneration by regulating osteoblast/osteoclast activities [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6736-6743. |
| [5] | Zhou Yunqi, Liu Xu, Xiao Dongqin, Li Xingping, Shi Feng, Zhang Bo, Pu Chao, Luo Xuwei, Zhang Chengdong. Fabrication and characterization of hydrogels with both antibacterial and osteogenic functions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6768-6778. |
| [6] | Chen Gang, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Wang Qianliang. Mechanism of ferrostatin-1 hydrogel in treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6807-6813. |
| [7] | Zhao Wenbo, Miao Xin, Wang Yang, Liu Hao, Li Shengfa, Tao Qifeng. Sr/birabresib-loaded bioactive glass modulating bone microenvironment for osteoporosis therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6814-6825. |
| [8] | Zhou Lijing, Wang Shuang, Xiang Jinjiao, Wang Huichao, Chai Xuejiao. Apical sealing and resistance strength of C-Root BP material in in vitro environment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6868-6872. |
| [9] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [10] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [11] | Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936. |
| [12] | Zhou Xiaohui, Wang Siyi, Zhou Qiyun, He Zhao, Jia Yujuan, Wang Yuanbin, Ma Jianwu, Chen Gang, Zheng Feng, Chu Genglei. Nanohydroxyapatite-polyether carbonate urethane electrospinning membrane promotes bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5134-5142. |
| [13] | Sun Danhe, Guo Xiaoling, Zhao Lingzhou. Construction and osteogenic activity of titanium dioxide nanotube and polydopamine composite coating on titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5167-5177. |
| [14] | Zhan Lei, Wu Lina, Li Huan, Liu Min, Chen Tao, Pu Xiaobing, Zhou Changchun. Silk fibroin hydrogel loaded with icariin to promote tendon-bone healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5178-5787. |
| [15] | Chen Weifei, Mei Yuandong, Ju Jihui. Repair of infected bone defect with dual-ion time-sequenced release multifunctional hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5188-5200. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||