Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7639-7647.doi: 10.12307/2026.276
Previous Articles Next Articles
Signaling pathways related to active ingredients of ginseng in the treatment of musculoskeletal degenerative diseases
Xia Tiange1, 2, Zhou Yi1, 2, Li Shaoshuo2, Wang Jianwei2, Shao Yang2
- 1Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; 2Department of Orthopedics, Wuxi TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Wuxi 214071, Jiangsu Province, China
-
Received:2025-08-09Revised:2025-12-08Online:2026-10-18Published:2026-03-06 -
Contact:Wang Jianwei, MD, Professor, Doctoral supervisor, Department of Orthopedics, Wuxi TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Wuxi 214071, Jiangsu Province, China Co-corresponding author: Shao Yang, MD, Master’s supervisor, Department of Orthopedics, Wuxi TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Wuxi 214071, Jiangsu Province, China -
About author:Xia Tiange, MS candidate, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; Department of Orthopedics, Wuxi TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Wuxi 214071, Jiangsu Province, China -
Supported by:National Natural Science Foundation of China (General Program), No. 82274546 (to WJW); National Natural Science Foundation of China (General Program), No. 82405520 (to LSS); Wuxi “Double Hundred” Top-notch Young and Middle-aged Medical and Health Talents Project, No. HB2023072 (to SY); Wuxi Health Commission Scientific Research Project, No. Q202232 (to LSS); Wuxi "Double Hundred" Top-notch Young and Middle-aged Medical and Health Talents Project, No. HB2023074 (to LSS); Jiangsu Province Traditional Chinese Medicine Science and Technology Development Plan (Young Talent Project), No. QN202322 (to LSS); Jiangsu Province Graduate Research and Practice Innovation Program Project, No. SJCX25_1080 (to ZY)
CLC Number:
Cite this article
Xia Tiange, Zhou Yi, Li Shaoshuo, Wang Jianwei, Shao Yang. Signaling pathways related to active ingredients of ginseng in the treatment of musculoskeletal degenerative diseases[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7639-7647.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
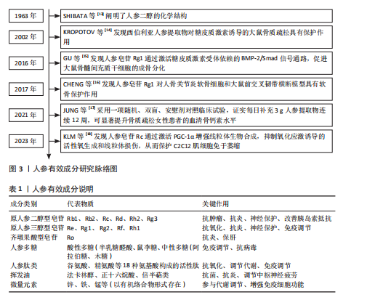
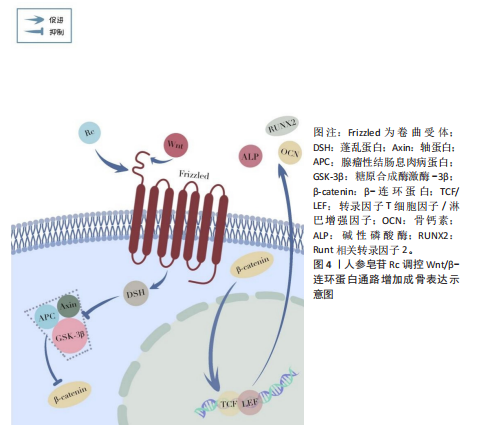
2.1 人参有效活性成分研究时间图 1963年SHIBATA等[13]基于化学转化、红外光谱及立体模型分析,阐明人参二醇及其12-表差向异构体的构型与构象,确立其顺式C/D环连接的立体化学结构,并首次确认天然人参二醇属顺式C/D 13-H四环三萜,该成果为人参皂苷分类研究奠定基础。2002年KROPOTOV等[14]发现西伯利亚人参提取物可缓解糖皮质激素诱导的大鼠骨质疏松,其在降低尿钙/羟脯氨酸排泄及维持骨强度方面的效果,与阳性对照药依普黄酮相当。2016年GU等[15]证实人参皂苷Rg1通过激活糖皮质激素受体依赖的骨形态发生蛋白2/Smad通路,促进大鼠骨髓间充质干细胞向成骨细胞分化,显著缩短胫骨骨折愈合周期。2017年CHENG等[16]的研究进一步表明,Rg1对人骨关节炎软骨细胞及大鼠前交叉韧带断裂模型具有明确软骨保护作用。2021年JUNG等[17]通过随机双盲安慰剂对照临床试验发现,骨质疏松女性每日口服3 g人参提取物12周后,血清骨钙素水平显著升高,骨吸收/形成比值降低,膝关节骨关节炎症状改善,且未出现安全性问题。2023年KIM等[18]发现人参皂苷Rc通过激活过氧化物酶体增殖物激活受体γ共激活因子1α(peroxisome proliferator-activated receptor gamma co-activator 1 alpha,PGC-1α)增强线粒体生物合成,抑制氧化应激诱导的活性氧生成和线粒体损伤,从而有效阻止C2C12肌细胞萎缩并恢复ATP合成功能。见图3及表1。 2.2 人参防治骨骼肌肉退行性疾病的有效成分 人参的药用价值与其复杂的化学成分密切相关。其根茎组织中不仅含有以人参皂苷Rb1、Rg1为代表的三萜皂苷类化合物,还包含多糖、肽类及倍半萜类挥发油成分。值得注意的是,该药材亦富含铁、锰等微量元素,这些成分的协同作用共同构成了其多元化的生物活性基础[19]。基于国内外最新研究进展,此文对人参中具有显著改善骨骼肌肉疾病作用的活性成分展开系统性综述。 2.2.1 人参皂苷 人参皂苷作为该药材的核心药用成分,隶属于四环三萜类化合物家族,主要来源于根部组织。现有研究表明,其化学结构主要分为三种类型:原人参二醇型、原人参三醇型及齐墩果酸型。其中,原人参二醇型包含Ra1-3、Rb1-3、Rc、Rd等皂苷成分,原人参三醇型则含有Re、Rf、Rg1-2等衍生物,这两类成分共同构成总皂苷的主要部分。齐墩果酸型属于五环三萜皂苷,结构与前两类显著不同,以人参皂苷Ro为典型代表,虽含量较少,但具有独特的生物活性特征[20]。 (1)人参皂苷对骨质疏松症的影响:骨质疏松症是以骨密度下降及骨微结构受损为特征的代谢性骨病,其病理改变直接导致骨骼脆性增高与骨折风险显著提升[21]。骨稳态失衡是其核心病理机制:正常生理状态下,破骨细胞介导的骨吸收与成骨细胞主导的骨形成维持动态平衡,而这一调节机制的紊乱将引发持续性骨量丢失,最终形成病理性骨转换状态[22]。 成骨细胞:成骨细胞作为骨组织构建与修复的核心功能单元,通过合成Ⅰ型胶原基质及调控羟基磷灰石沉积,在骨再生与重建过程中发挥双重调控作用。其分泌的骨钙素、骨桥蛋白等非胶原蛋白,与矿化基质的空间排列共同构成骨骼力学稳定性的基础。近年研究表明,人参皂苷可通过多维度调控成骨细胞生物学行为干预骨质疏松进程,多种人参皂苷已被证实能增加其关键标志物的表达。 YE等[23]的研究显示,人参皂苷Rb1可提升类固醇诱导的股骨头缺血性坏死大鼠模型中碱性磷酸酶和骨钙素活性,上调血管内皮生长因子及其受体、Runt相关转录因子2、骨形态发生蛋白2的蛋白表达。LIU等[24]发现Rb2能有效改善生酮饮食诱导的骨质疏松小鼠的骨代谢状况:生酮饮食显著降低骨特异性碱性磷酸酶浓度,而Rb2干预可逆转这一变化;同时,生酮饮食导致抗酒石酸酸性磷酸酶浓度显著升高,Rb2则使其明显下降。在骨组织中,生酮饮食会减少骨钙素表达、增加抗酒石酸酸性磷酸酶表达;而Rb2可促进骨钙素表达、抑制抗酒石酸酸性磷酸酶表达,并通过抑制过氧化物酶体增殖物激活受体γ和组织蛋白酶K的表达,促进骨髓间充质干细胞向成骨细胞分化、抑制其向脂肪细胞分化,进而维持骨代谢平衡。YANG等[25]证实人参皂苷Rc可刺激Ⅰ型胶原蛋白、Runt相关转录因子2、碱性磷酸酶等骨形成相关基因表达,进而促进MC3T3-E1前体细胞的体外成骨分化与矿化。 破骨细胞:破骨细胞起源于造血谱系,为负责骨吸收的大型多核细胞,分泌的多种生长因子和细胞因子参与骨骼形成过程。破骨细胞与成骨细胞的功能耦合是维持骨骼结构稳定性的关键。研究表明,包括人参皂苷Rb1和Rg3在内的多种人参皂苷可抑制破骨细胞生成与活性。 CHENG等[26]利用核因子κB受体活化因子配体诱导的Raw264.7细胞破骨分化模型发现,人参皂苷Rb1通过三重协同机制抑制破骨分化:选择性阻断c-Jun氨基末端激酶(c-Jun N-terminal Kinase,JNK)/ p38磷酸化、抑制核因子κB核转位,下调c-Fos、活化T细胞核因子胞质1基因及肿瘤坏死因子α mRNA水平,在无细胞毒性浓度范围内有效干预破骨细胞生成。ZHANG等[27]研究揭示,人参皂苷Rg3通"

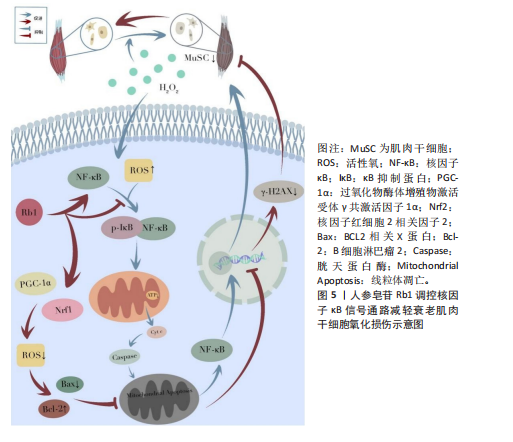
过靶向核转运蛋白α2抑制破骨分化:Rg3以剂量和时间依赖性方式下调核转运蛋白α2表达;显著减少抗酒石酸酸性磷酸酶、活化T细胞核因子胞质1等破骨标志物表达,并阻断p65核转位;进一步实验显示,核转运蛋白α2过表达可部分逆转Rg3对破骨分化的抑制作用,证实核转运蛋白α2是Rg3调控破骨细胞生成的关键靶标。 间充质干细胞:间充质干细胞在骨形成过程中具有关键作用,不仅是成骨细胞的来源,还参与骨形成微环境的调控。研究发现,人参皂苷可通过促进转录因子激活,诱导骨髓间充质干细胞的增殖与成骨分化。 DING等[28]系统阐释了人参皂苷CK通过Wnt/β-连环蛋白(beta-catenin,β-catenin)通路促进骨折修复的机制:在10-40 μmol/L安全浓度范围内,人参皂苷CK显著增强大鼠骨髓间充质干细胞活性,通过激活Wnt/β-连环蛋白信号通路促进β-连环蛋白核转位,并上调Runt相关转录因子2、骨桥蛋白等成骨关键基因表达。WANG等[29]通过实验证实,人参皂苷Rg1可显著抑制人骨髓间充质干细胞的衰老特征,表现为降低SA-β-gal染色阳性率及P53、P16表达;同时调控其分化,促进成骨与软骨分化、抑制脂肪分化。机制上,Rg1能抑制糖原合成酶激酶3β的过度磷酸化,减少β-连环蛋白的核转位;而糖原合成酶激酶3β抑制剂LiCl可逆转Rg1对细胞分化的调控效应,这为间充质干细胞再生治疗提供了药理依据。 (2)人参皂苷对骨关节炎的影响:骨关节炎是一种以软骨和软骨下骨破坏、结构变形为特征的退行性疾病,主要病理表现为关节疼痛与功能丧失[30]。作为全球致残率最高的肌肉骨骼疾病,骨关节炎与低度慢性炎症密切相关[31]。促炎细胞因子如白细胞介素1β、肿瘤坏死因子α通过激活核因子κB、丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)等信号通路,诱导软骨细胞外基质降解和软骨细胞凋亡,驱动疾病进展[32]。 LUAN等[33]研究以单碘乙酸诱导的大鼠骨关节炎模型及软骨细胞为研究对象,证实人参皂苷Rb1通过抑制miR-21-5p表达、上调成纤维细胞生长因子18活性,减轻单碘乙酸诱导的骨关节炎病理进程。动物实验表明,Rb1剂量依赖性改善大鼠关节功能、减少关节组织炎症细胞浸润,并抑制白细胞介素6、白细胞介素1β、肿瘤坏死因子α等炎症因子释放;细胞实验显示,Rb1可增强单碘乙酸处理后软骨细胞活力,下调miR-21-5p,上调成纤维细胞生长因子18及Ⅱ型胶原蛋白表达,抑制血小板结合蛋白基序的解聚蛋白样金属蛋白酶5活性。机制研究发现,miR-21-5p直接靶向成纤维细胞生长因子18的3’UTR,恢复miR-21-5p水平可逆转Rb1的保护效应,证实其通过miR-21-5p成纤维细胞生长因子18信号轴发挥抗骨关节炎作用。ARAVINTHAN等[34]研究人员对卵巢切除术后单碘乙酸诱导的骨关节炎大鼠进行每周3次关节内Rb1注射(持续4周),结果显示Rb1显著缓解炎症症状,降低血清炎性细胞因子水平,增强骨形态发生蛋白2和胶原蛋白表达。PAN等[35]的研究表明,人参皂苷Rh2具有显著抗炎活性,在脂多糖诱导的RAW 264.7巨噬细胞炎症模型中,人参皂苷Rh2在0–80 μg/mL浓度范围内无明显细胞毒性,可有效抑制脂多糖诱导的肿瘤坏死因子α、白细胞介素6和一氧化氮释放。并且Rh2通过直接结合Toll样受体4和髓样分化蛋白2,阻断脂多糖与细胞表面受体结合,抑制Toll样受体4/髓样分化蛋白2复合物二聚化,进而抑制Toll样受体4/核因子κB信号通路激活。鉴于炎症反应在骨关节炎发病机制中的核心作用,抑制该通路可能有效缓解骨关节炎病理进程,提示人参皂苷Rh2有望成为骨关节炎治疗的潜在候选药物。 (3)人参皂苷对肌肉减少症的影响:肌肉减少症是指由衰老引起的肌肉功能丧失,主要表现为肌肉力量和质量进行性下降[36]。肌肉减少症的核心病理机制是年龄相关的免疫衰老、慢性低度炎症与氧化应激的协同作用[37-38]。 PARK等[39]在2020年开展的随机、双盲、安慰剂对照试验中,对59例45-75岁的2型糖尿病患者进行了24周的干预,结果显示服用韩国红参(含Rg1、Re、Rh1等)可显著改善肌肉减少症相关标志物——血清卵泡抑素和性激素结合球蛋白的水平。ZHU等[12]通过骨骼肌衰老模型发现,人参皂苷Rh4通过靶向激活沉默信息调节因子1(silent information regulator 1,SIRT1)通路,显著改善肌肉减少症相关的病理改变:其通过抑制核因子红细胞2相关因子2(nuclear factor erythroid 2-related factor 2,Nrf2)乙酰化以增强抗氧化能力,使骨骼肌中丙二醛和活性氧水平降低,超氧化物歧化酶、谷胱甘肽过氧化物酶活性显著提升;同时通过调控过氧化物酶体增殖物激活受体γ共激活因子1α-线粒体转录因子A信号轴增强线粒体生物合成,并抑制缺氧诱导因子 1α-c-Myc 轴改善线粒体功能障碍,最终通过维持线粒体稳态延缓骨骼肌衰老,为肌肉减少症的干预提供了实验依据。 (4)人参皂苷对椎间盘退变的影响:椎间盘退变是腰痛的主要病理基础之一,约40%的腰痛直接源于椎间盘退变,即椎间盘源性腰痛[40]。正常椎间盘由上下终板、中央的髓核(含髓核细胞)和外周的纤维环构成。髓核细胞分泌的Ⅱ型胶原蛋白和蛋白聚糖形成细胞外基质,维持椎间盘的结构与功能[41]。然而,多种因素会打破平衡:年龄增长使椎间盘失去弹性、纤维环变脆;遗传因素增加椎间盘退变易感性;机械创伤直接损伤椎间盘[42];并且炎症反应在椎间盘退变进程中起核心作用,白细胞介素1β、肿瘤坏死因子α等炎症因子大量表达,通过激活相关信号通路,引发细胞凋亡、氧化应激和细胞外基质代谢失衡,加速椎间盘退变发展[43]。 YANG等[44]构建了椎间盘退变大鼠模型并分组处理,同时体外处理髓核细胞,研究发现,人参皂苷Rg1可抑制Yes相关蛋白1/转录共激活因子(Yes-associated protein 1/transcriptional coactivator with PDZ-binding motif,YAP1/TAZ)信号通路,减少炎症因子分泌、降低氧化应激水平、抑制细胞外基质降解和髓核细胞凋亡,对椎间盘退变有调控作用。CHEN等[45]以体外培养的髓核细胞为对象,探究人参皂苷Rg3对椎间盘退变的调控机制。研究发现,肿瘤坏死因子α诱导的髓核细胞出现凋亡增加、增殖受抑、细胞外基质代谢失衡及氧化应激损伤加剧,人参皂苷Rg3处理后这些变化呈浓度依赖性改善;进一步发现人参皂苷Rg3可能通过阻断核因子κB信号通路发挥作用,该通路激活后影响Bcl-2、Bax等凋亡蛋白表达,调节氧化应激水平,调控细胞外基质代谢基因,这为椎间盘退变治疗提供潜在靶点。 2.2.2 人参多糖 人参多糖是由多种单糖通过糖苷键连接形成的天然高分子聚合物,主要含中性糖及少量酸性糖[46],该成分不仅存在于人参药用主根和根茎,在传统非药用部位(茎、叶、浆果)中含量也较高[47]。人参多糖具有抗氧化、抗疲劳、免疫调节及维持肠道菌群稳态等多重生物活性,在抗炎与代谢性疾病干预中具潜在价值[48]。 免疫稳态的精准调控是缓解骨骼肌肉疾病炎症反应、促进组织修复的关键。KIM等[49]利用RAW 264.7巨噬细胞模型证实:白参多糖、红参多糖及热加工人参多糖的免疫激活效应随加工温度升高而降低,3种多糖均能促进一氧化氮、肿瘤坏死因子α和白细胞介素6分泌,其中白参多糖(500 μg/mL)通过激活丝裂原活化蛋白激酶-核因子κB p65信号轴,促使细胞外信号调节激酶(extracellular signal-regulated kinase,ERK)、JNK、p38、核因子κB p65发生磷酸化,同时上调诱导型一氧化氮合酶蛋白表达,其免疫激活活性显著高于红参多糖与热加工人参多糖。FANG等[50]实验显示,非熏硫多糖能有效改善免疫低下小鼠的体质量、免疫器官指数及淋巴细胞亚群比例,而熏硫多糖对炎症表型的调控作用显著降低。机制研究表明非熏硫多糖通过恢复肠道菌群多样性及增加短链脂肪酸生成增强免疫,硫熏处理则可能改变多糖-微生物互作模式降低疗效。这些发现揭示了加工工艺对人参多糖功能的影响,为优化制剂工艺适配骨骼肌肉疾病治疗提供了理论支撑。 2.2.3 人参肽类 近年来,人参肽类因其独特的生物活性受到广泛关注,在骨骼肌肉疾病的防治中表现出多维度潜力。 LIU等[51]研究表明,人参多肽可通过抑制炎症反应和氧化应激,激活磷脂酰肌醇3-激酶/蛋白激酶B(phosphatidylinositol 3-kinase/protein kinase B,PI3K/Akt)通路,并抑制丝裂原活化蛋白激酶通路的过度激活,从而改善糖尿病小鼠的胰岛素抵抗及肝、肾等多器官损伤。肝肾功能的改善及糖脂代谢的调节,可能通过维持代谢稳态与减少炎症,间接促进骨骼肌健康。此外,ZHU等[52]研究发现人参寡肽通过NAD?/沉默信息调节因子1/过氧化物酶体增殖物激活受体γ共激活因子1α通路促进线粒体生物合成,显著延缓成纤维细胞衰老进程,并通过抑制核因子κB激活减少白细胞介素6、白细胞介素1β等炎症因子释放,为肌少症的干预提供了分子机制依据。ZHU等[53]进一步在氧化应激诱导的细胞模型中证实,人参寡肽通过增强线粒体膜电位、促进ATP生成及抑制Beclin1、LC3B等自噬相关蛋白表达,保护内皮细胞和神经元免受氧化损伤,这一机制可能对维持肌肉细胞能量代谢稳态及抗萎缩具有重要意义。综上,人参来源的肽类通过抗炎、抗氧化、线粒体功能调控及关键再生信号通路激活,为骨骼肌疾病的治疗提供了创新策略。 2.3 人参有效成分调控相关信号通路 2.3.1 Wnt/β-连环蛋白信号通路 Wnt/β-连环蛋白信号通路在骨代谢中发挥核心调控作用,尤其在成骨细胞分化、骨形成与骨吸收平衡中至关重要。当Wnt信号未激活时,细胞质中的β-连环蛋白会与轴蛋白、腺瘤性息肉病coli蛋白、糖原合成酶激酶3β和酪蛋白激酶1α形成复合物。在这个复合物中,糖原合成酶激酶3β和酪蛋白激酶1α作为激酶,促使β-连环蛋白磷酸化,磷酸化后的β-连环蛋白被E3泛素连接酶识别并降解,使得β-连环蛋白在细胞内维持较低水平。此时转录因子T细胞因子/淋巴增强因子,抑制下游靶基因的转录。当Wnt信号激活时,Wnt配体与细胞膜上的Frizzled受体和LRP5/6共受体结合,招募蓬乱蛋白。蓬乱蛋白的激活抑制糖原合成酶激酶3β的活性,使得β-连环蛋白无法被磷酸化,从而在细胞质中稳定积累。积累的β-连环蛋白进入细胞核,与转录因子T细胞因子/淋巴增强因子转录因子结合,形成β-连环蛋白/转录因子T细胞因子-淋巴增强因子复合物,该复合物与下游靶基因的启动子区域结合,启动转录过程,促进成骨细胞的增殖、分化以及骨基质的合成,如上调骨钙素、碱性磷酸酶等成骨相关基因的表达,增强骨形成能力[54-55]。 YANG等[25]通过体内外实验揭示了人参皂苷Rc通过激活Wnt/β-连环蛋白信号通路促进骨形成的机制。在卵巢切除骨质疏松小鼠模型中,50 mg/kg人参皂苷Rc显著提升骨密度并改善骨小梁结构,同时上调β-连环蛋白、磷酸化糖原合成酶激酶3β及成骨分化标志物(Runt相关转录因子2、Ⅰ型胶原蛋白、碱性磷酸酶等)的表达;细胞实验中,50 μmol/L人参皂苷Rc通过增强Wnt通路活性促进MC3T3-E1细胞的成骨分化与矿化,而Wnt抑制剂XAV-939可完全逆转此效应,证实其机制依赖β-连环蛋白信号传导。研究结果表明,人参皂苷Rc作为天然活性成分,可通过靶向Wnt/β-连环蛋白通路调控成骨功能,为治疗骨质疏松提供潜在策略。见图4。 2.3.2 核因子κB信号通路 核因子κB转录因子家族包含RelA(p65)、RelB、c-Rel、p50及p52亚基。静息时,经典通路二聚体受κB抑制蛋白(inhibitor of κB,IκB)抑制滞留胞质;非经典通路二聚体受前体p100抑制。经典途径由肿瘤坏死因子α、白细胞介素1β、细菌毒素等激活,依赖IκB激酶介导IκB降解,促使二聚体入核调控炎症基因;非经典途径由核因子κB受体活化因子配体等激活,依赖核因子κB诱导激酶、IκB激酶α加工二聚体,调控淋巴发育及骨代谢基因。在骨骼疾病中,核因子κB受体活化因子配体通过激活核因子κB双通路驱动破骨细胞分化与骨吸收,导致类风湿关节"
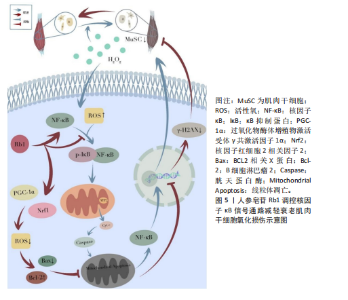
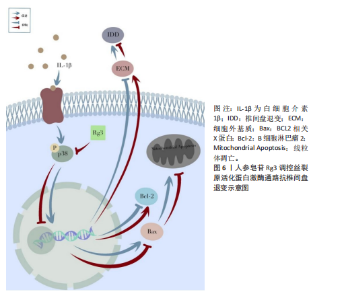
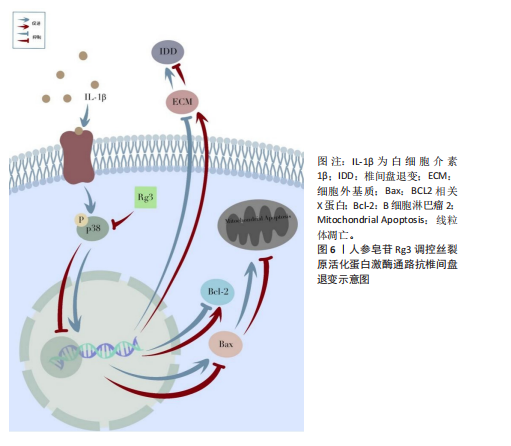
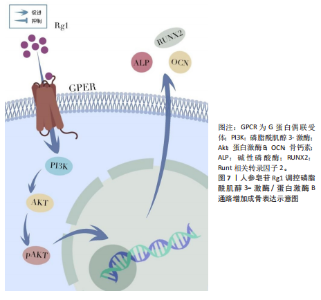
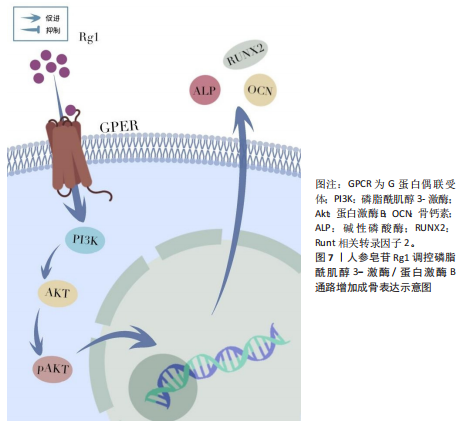
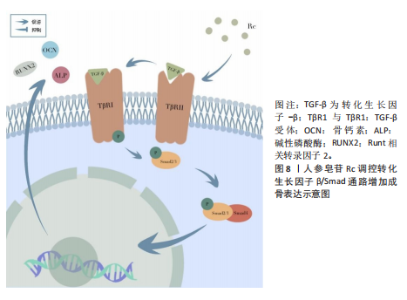
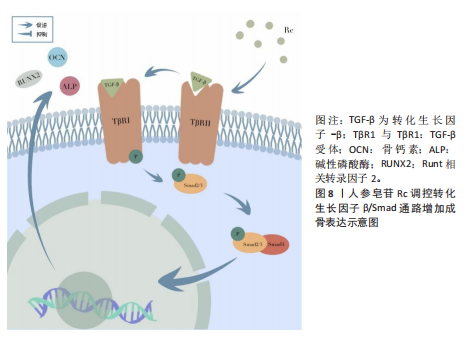
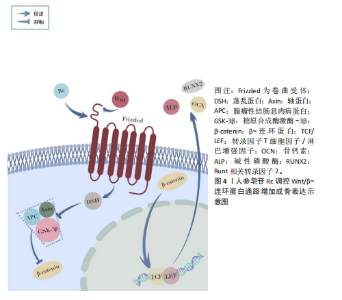
炎及骨质疏松。在肌肉疾病中,核因子κB过度激活:诱导肿瘤坏死因子α、白细胞介素6等促炎因子分泌,加重炎症;直接上调萎缩基因促进蛋白降解,共同导致肌炎及肌肉萎缩。靶向核因子κB通路是治疗骨骼肌肉退行性病变的重要策略[56-57]。 DONG等[58]通过整合C2C12成肌细胞与老年小鼠模型,阐明人参皂苷Rb1通过靶向核因子κB通路改善骨骼肌退行性变的分子机制。研究证实,Rb1通过多途径协同作用:一方面直接结合核因子κB蛋白并抑制其磷酸化激活,阻断下游信号传导;另一方面有效清除活性氧以维持线粒体稳态,表现为促进线粒体生物合成关键因子(过氧化物酶体增殖物激活受体γ共激活因子1α、核因子红细胞2相关因子1)表达并抑制细胞色素C异常释放。在凋亡调控层面,Rb1显著抑制胱天蛋白酶级联反应并上调Bcl-2表达。动物实验进一步验证其能维持老年骨骼肌PAX7+干细胞库,减少细胞凋亡并改善肌肉功能,该研究为基于核因子κB通路调控的肌少症干预策略提供了理论依据。见图5。 ZHANG等[27]研究发现,人参皂苷Rg3通过靶向核转运蛋白核转运蛋白α2抑制核因子κB信号通路,从而拮抗破骨细胞分化。在核因子κB受体活化因子配体诱导的RAW264.7细胞模型中,Rg3显著下调核转运蛋白α2表达并阻断核因子κB关键蛋白p65的核易位。核转运蛋白α2敲降实验证实其通过激活核因子κB通路调控破骨分化标志物表达;而核因子κB抑制剂JSH-23可拮抗核转运蛋白α2过表达的促破骨效应。并且Rg3对破骨分化的抑制作用可被核转运蛋白α2过表达部分逆转,提示核转运蛋白α2-核因子κB轴是Rg3调控骨稳态的核心靶点。 2.3.3 丝裂原活化蛋白激酶信号通路 丝裂原活化蛋白激酶信号通路通过3级激酶磷酸化级联反应调控细胞增殖、分化和凋亡,在骨骼肌稳态和疾病进展中发挥核心作用。其分支细胞外信号调节激酶通路的异常激活可抑制肌源性分化因子,导致肌细胞再生障碍;而JNK/p38通路的过度活化则通过促炎因子加剧肌肉炎症和蛋白降解,与肌萎缩侧索硬化症等疾病密切相关[59-60]。 LEE等[61]的研究发现,40 μmol/L人参皂苷Rg2可抑制核因子κB受体活化因子配体诱导的骨髓巨噬细胞向破骨细胞分化,无细胞毒性,且呈剂量依赖性。其分子机制表现为,通过降低细胞外信号调节激酶、JNK和p38的磷酸化水平抑制丝裂原活化蛋白激酶信号通路,下调转录因子c-Fos和活化T细胞核因子胞质1的mRNA及蛋白表达,进而抑制破骨细胞特异性基因Acp5、Oscar的转录,这为其抗骨质疏松作用提供了实验依据。 CHEN等[62]的研究证实,人参皂苷Rg3可通过抑制p38 丝裂原活化蛋白激酶通路减轻椎间盘退变。在白细胞介素1β诱导的髓核细胞模型中,Rg3能下调促凋亡蛋白Bax及分解代谢酶(基质金属蛋白酶2、基质金属蛋白酶3、血小板结合蛋白基序的解聚蛋白样金属蛋白酶),同时上调抗凋亡蛋白Bcl-2,从而减少细胞凋亡和细胞外基质降解,其核心机制在于抑制p38 丝裂原活化蛋白激酶的磷酸化激活。在椎间盘退变大鼠模型中,Rg3能有效改善椎间盘高度、修复纤维环排列紊乱、保留蛋白聚糖基质,且同样通过抑制p38 丝裂原活化蛋白激酶通路减轻退变进程。见图6。 2.3.4 磷脂酰肌醇3-激酶/蛋白激酶B信号通路 磷脂酰肌醇3-激酶/蛋白激酶B信号通路在细胞生命活动中至关重要。当细胞表面受体,如G蛋白偶联受体与配体结合后,磷脂酰肌醇3-激酶被激活。磷脂酰肌醇3-激酶的Class I类为异二聚体,激活后催化磷脂酰肌醇-4,5-二磷酸生成磷脂酰肌醇-3,4,5-三磷酸。磷脂酰肌醇-3,4,5-三磷酸招募3-磷酸肌醇依赖性蛋白激酶1和蛋白激酶B,3-磷酸肌醇依赖性蛋白激酶1使蛋白激酶B的Thr308位点磷酸化,哺乳动物雷帕霉素靶蛋白复合体2使蛋白激酶B的Ser473位点磷酸化,从而激活蛋白激酶B。激活的蛋白激酶B可磷酸化下游靶蛋白,调控细胞增殖、分化、存活和代谢等过程,抑制叉头框O转录因子来抗凋亡[63]。并且磷脂酰肌醇3-激酶/蛋白激酶B信号通路对骨质疏松的发生发展具有关键作用,它能促进成骨细胞的增殖与分化,抑制其凋亡,进而增加骨形成[64]。 JIANG等[65]揭示人参皂苷Rg1通过G蛋白偶联雌激素受体-磷脂酰肌醇3-激酶/蛋白激酶B信号轴促进成骨的新机制。在斑马鱼骨质疏松模型中,Rg1通过激活G蛋白偶联雌激素受体显著增强蛋白激酶B磷酸化,进而改善骨形成;而G蛋白偶联雌激素受体抑制剂G15或磷脂酰肌醇3-激酶抑制剂LY294002均可阻断该效应。细胞实验证实,Rg1通过上调G蛋白偶联雌激素受体表达激活磷脂酰肌醇3-激酶/蛋白激酶B通路促进MC3T3-E1细胞成骨分化,该作用可被G15抑制且被G蛋白偶联雌激素受体激动剂G1部分恢复。值得注意的是,磷脂酰肌醇3-激酶/蛋白激酶B通路抑制剂完全能消除Rg1的促成骨效应,证实该通路是G蛋白偶联雌激素受体下游的关键执行者。研究首次阐明Rg1通过G蛋白偶联雌激素受体介导的磷脂酰肌醇3-激酶/蛋白激酶B信号级联调控骨代谢,为靶向该通路治疗骨质疏松提供了理论依据。见图7。 2.3.5 转化生长因子β/Smad信号通路 转化生长因子β通常与潜伏期相关肽、潜伏期转化生长因子结合蛋白形成复合物,经含KRFK基序蛋白、基质金属蛋白酶、RGD基序识别的整合素及活性氧等激活后,与转化生长因子β受体Ⅰ、转化生长因子β受体Ⅱ结合,使Smad2/3磷酸化并与Smad4形成复合物,调节靶基因表达。该通路与骨质疏松紧密相关,正常情况下,转化生长因子β可维持成骨细胞和破骨细胞功能平衡,其通路异常会增加骨质疏松风险;而转化生长因子β通路激活机制异常,也可能影响骨重建,进而导致骨质疏松[66-68]。 WANG等[69]通过建立卵巢切除大鼠骨质疏松模型及培养原代人颅骨成骨细胞,证实人参皂苷Rc通过激活转化生长因子β/Smad信号通路,即上调转化生长因子β及磷酸化Smad2/3,显著促进成骨细胞分化和Ⅰ型胶原合成,从而改善卵巢切除大鼠的骨质疏松。并且抑制该通路可完全逆转Rc的促骨形成效应,验证了转化生长因子β/Smad的核心调控作用。"
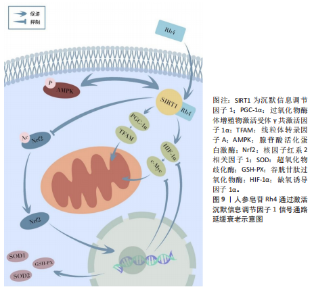
现,沉默信息调节因子1抑制会加重含有α-突触核蛋白的衰老相关内含物的形成,增多多巴胺能神经元的年龄依赖性退化数量,进而加速相关疾病的发展[72]。此外,过量产生的自由基会损伤细胞,引发氧化和亚硝化应激,破坏免疫功能,诱发一系列衰老相关疾病,而沉默信息调节因子1能够在体内外调控活性氧的生成与积累[73]。 ZHU等[12]研究利用D-半乳糖诱导的骨骼肌衰老模型,揭示人参皂苷Rh4通过靶向激活沉默信息调节因子1信号通路延缓衰老的分子机制。Rh4特异性结合沉默信息调节因子1的Ser267和Pro323残基激活其去乙酰化功能,触发多重保护效应:在抗氧化层面,沉默信息调节因子1介导核因子红细胞2相关因子2去乙酰化促进核转位,上调超氧化物歧化酶1、超氧化物歧化酶2和谷胱甘肽过氧化物酶等抗氧化酶表达,抑制白细胞介素6、肿瘤坏死因子α等炎症因子释放;在线粒体稳态层面,沉默信息调节因子1通过去乙酰化修饰激活过氧化物酶体增殖物激活受体γ共激活因子1α,激活后的过氧化物酶体增殖物激活受体γ共激活因子1α与核呼吸因子1、核呼吸因子2协同促进线粒体转录因子A的表达,进而增强线粒体生物合成,并抑制缺氧诱导因子1α与c-Myc的相互作用以改善线粒体功能。此外,Rh4通过腺苷酸活化蛋白激酶,形成腺苷酸活化蛋白激酶-沉默信息调节因子1双向调控环路,进一步增强能量代谢效率。此研究系统阐释了Rh4通过沉默信息调节因子1调控线粒体稳态与氧化应激网络对抗骨骼肌衰老的机制。见图9。"

| [1] ZHENG YL, SONG G, GUO JB, et al. Interactions Among lncRNA/circRNA, miRNA, and mRNA in Musculoskeletal Degenerative Diseases. Front Cell Dev Biol. 2021;9:753931. [2] WEN ZQ, LIN J, XIE WQ, et al. Insights into the underlying pathogenesis and therapeutic potential of endoplasmic reticulum stress in degenerative musculoskeletal diseases. Mil Med Res. 2023;10:54. [3] GBD 2021 Osteoarthritis Collaborators. Global, regional, and national burden of osteoarthritis, 1990-2020 and projections to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023; 5(9):e508-e522. [4 ] SI L, WINZENBERG TM, JIANG Q, et al. Projection of osteoporosis-related fractures and costs in China: 2010-2050. Osteoporos Int. 2015;26(7): 1929-1937. [5] YIN P, JIANG Y, FANG X, et al. Cell-Based Therapies for Degenerative Musculoskeletal Diseases. Adv Sci (Weinheim, Baden-Wurttemberg, Germany). 2023; 10(21): e2207050. [6] COMPSTON J. Practical guidance for the use of bisphosphonates in osteoporosis. Bone. 2020; 136:115330. [7] TAI TW, CHEN HY, SHIH CA, et al. Asia-Pacific consensus on long-term and sequential therapy for osteoporosis. Osteoporos Sarcopenia. 2024; 10(1):3-10. [8] WU PH, KIM HS, JANG IT. Intervertebral Disc Diseases PART 2: A Review of the Current Diagnostic and Treatment Strategies for Intervertebral Disc Disease. Int J Mol Sci. 2020; 21(6):2135. [9] SUN Q, LIU F, FANG J, et al. Strontium ranelate retards disc degradation and improves endplate and bone micro-architecture in ovariectomized rats with lumbar fusion induced – Adjacent segment disc degeneration. Bone Rep. 2024;20: 101744. [10] FAN S, ZHANG Z, SU H, et al. Panax ginseng clinical trials: Current status and future perspectives. Biomed Pharmacother. 2020;132:110832. [11] KO SY. Therapeutic Potential of Ginsenosides on Bone Metabolism: A Review of Osteoporosis, Periodontal Disease and Osteoarthritis. Int J Mol Sci. 2024;25(11):5828. [12] ZHU A, DUAN Z, CHEN Y, et al. Ginsenoside Rh4 delays skeletal muscle aging through SIRT1 pathway. Phytomedicine. 2023;118:154906. [13] SHIBATA S, TANAKA O, NAGAI M, et al. Studies on the Constituents of Japanese and Chinese Crude Drugs. XII. Panaxadiol, A Sapogenin of Ginseng Roots. (2). Chem Pharm Bull (Tokyo). 1963;11(6):762-765. [14] KROPOTOV AV, KOLODNYAK OL, KOLDAEV VM. Effects of Siberian ginseng extract and ipriflavone on the development of glucocorticoid-induced osteoporosis. Bull Exp Biol Med. 2002; 133(3):252-254. [15] GU Y, ZHOU J, WANG Q, et al. Ginsenoside Rg1 promotes osteogenic differentiation of rBMSCs and healing of rat tibial fractures through regulation of GR-dependent BMP-2/SMAD signaling. Sci Rep. 2016;6:25282. [16] CHENG W, JING J, WANG Z, et al. Chondroprotective Effects of Ginsenoside Rg1 in Human Osteoarthritis Chondrocytes and a Rat Model of Anterior Cruciate Ligament Transection. Nutrients. 2017;9(3):263. [17] JUNG SJ, OH MR, LEE DY, et al. Effect of Ginseng Extracts on the Improvement of Osteopathic and Arthritis Symptoms in Women with Osteopenia: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients. 2021;13(10):3352. [18] KIM A, PARK SM, KIM NS, et al. Ginsenoside Rc, an Active Component of Panax ginseng, Alleviates Oxidative Stress-Induced Muscle Atrophy via Improvement of Mitochondrial Biogenesis. Antioxidants (Basel). 2023;12(8):1576. [19] SINGH S, AGRAWAL R, SHARMA H. Comprehensive PRISMA Based Systematic Review: Exploring the Phytochemistry, Pharmacological Profile and Clinical aspects of Panax ginseng. Curr Top Med Chem. 2025; 25(2):172-195. [20] PENG Y, PAN W, CAO X, et al. Potential Oral Health Benefits of Ginseng and Its Extracts. Int Dent J. 2023;73(4):473-480. [21] JOHNSTON CB, DAGAR M. Osteoporosis in Older Adults. Med Clin North Am. 2020;104(5): 873-884. [22] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Ther. 2022;237:108168. [23] YE J, WEI D, PENG L. Ginsenoside Rb1 prevents steroid‑induced avascular necrosis of the femoral head through the bone morphogenetic protein‑2 and vascular endothelial growth factor pathway. Mol Med Rep. 2019;20(4):3175-3181. [24] LIU Q, ZHOU J, YANG Z, et al. The Ginsenoside Exhibits Antiosteoporosis Effects in Ketogenic-Diet-Induced Osteoporosis via Rebalancing Bone Turnover. Front Pharmacol. 2020;11:593820. [25] YANG N, ZHANG X, LI L, et al. Ginsenoside Rc Promotes Bone Formation in Ovariectomy-Induced Osteoporosis In Vivo and Osteogenic Differentiation In Vitro. Int J Mol Sci. 2022;23(11):6187. [26] CHENG B, LI J, DU J, et al. Ginsenoside Rb1 inhibits osteoclastogenesis by modulating NF-κB and MAPKs pathways. Food and Chemical Toxicology. 2012;50(5):1610-1615. [27] ZHANG X, HUANG F, LIU J, et al. Molecular Mechanism of Ginsenoside Rg3 Alleviation in Osteoporosis via Modulation of KPNA2 and the NF-κB Signalling Pathway. Clin Exp Pharmacol Physiol. 2025;52(3):e70019. [28] DING L, GU S, ZHOU B, et al. Ginsenoside Compound K Enhances Fracture Healing via Promoting Osteogenesis and Angiogenesis. Front Pharmacol. 2022;13:855393. [29] WANG Z, JIANG R, WANG L, et al. Ginsenoside Rg1 Improves Differentiation by Inhibiting Senescence of Human Bone Marrow Mesenchymal Stem Cell via GSK-3β and β-Catenin. Stem Cells Int. 2020; 2020:2365814. [30] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. The Lancet. 2019;393(10182):1745-1759. [31] DRIBAN JB, HARKEY MS, LIU SH, et al. Osteoarthritis and Aging: Young Adults with Osteoarthritis. Curr Epidemiol Rep. 2020;7(1): 9-15. [32] ZHU L, VINCENT TL. Genome-Wide Association Studies to Drug: Identifying Retinoic Acid Metabolism Blocking Agents to Suppress Mechanoflammation in Osteoarthritis. DNA Cell Biol. 2023;42(9):527-531. [33] LUAN J, CHE G, MAN G, et al. Ginsenoside Rb1 from Panax ginseng attenuates monoiodoacetate-induced osteoarthritis by inhibiting miR-21-5p/FGF18-mediated inflammation. J Food Biochem. 2022;46(10):e14340. [34] ARAVINTHAN A, HOSSAIN MA, KIM B, et al. Ginsenoside Rb1 inhibits monoiodoacetate-induced osteoarthritis in postmenopausal rats through prevention of cartilage degradation. J Ginseng Res. 2021;45(2):287-294. [35] PAN S, PENG L, YI Q, et al. Ginsenoside Rh2 Alleviates LPS-Induced Inflammatory Responses by Binding to TLR4/MD-2 and Blocking TLR4 Dimerization. Int J Mol Sci. 2024;25(17):9546. [36] LARSSON L, DEGENS H, LI M, et al. Sarcopenia: Aging-Related Loss of Muscle Mass and Function. Physiological Reviews. 2019;99(1):427-511. [37] CHEI S, OH HJ, LEE K, et al. Dysfunction of B Cell Leading to Failure of Immunoglobulin Response Is Ameliorated by Dietary Silk Peptide in 14-Month-Old C57BL/6 Mice. Front Nutr. 2020;7:583186. [38] FOREMAN NA, HESSE AS, JI LL. Redox Signaling and Sarcopenia: Searching for the Primary Suspect. Int J Mol Sci. 2021;22(16):9045. [39] PARK K, AHN CW, KIM Y, et al. The effect of Korean Red Ginseng on sarcopenia biomarkers in type 2 diabetes patients. Arch Gerontol Geriatr. 2020;90:104108. [40] PENG B, LI Q, CHEN J, et al. Research on the role and mechanism of IL-17 in intervertebral disc degeneration. Int Immunopharmacol. 2024;132: 111992. [41] GUO HY, GUO MK, WAN ZY, et al. Emerging evidence on noncoding-RNA regulatory machinery in intervertebral disc degeneration: a narrative review. Arthritis Res Ther. 2020;22(1):27. [42] MOHD ISA IL, TEOH SL, MOHD NOR NH, et al. Discogenic Low Back Pain: Anatomy, Pathophysiology and Treatments of Intervertebral Disc Degeneration. Int J Mol Sci. 2022;24(1):208. [43] WANG Y, CHE M, XIN J, et al. The role of IL-1β and TNF-α in intervertebral disc degeneration. Biomed Pharmacother. 2020;131:110660. [44] YANG Y, GU X, HU H, et al. Ginsenoside Rg1 inhibits nucleus pulposus cell apoptosis, inflammation and extracellular matrix degradation via the YAP1/TAZ pathway in rats with intervertebral disc degeneration. J Orthop Surg Res. 2022;17:55. [45] CHEN J, LIU GZ, SUN Q, et al. Protective effects of ginsenoside Rg3 on TNF-α-induced human nucleus pulposus cells through inhibiting NF-κB signaling pathway. Life Sci. 2019; 216:1-9. [46] ZHANG S, DING C, LIU X, et al. Research Progress on Extraction, Isolation, Structural Analysis and Biological Activity of Polysaccharides from Panax Genus. Molecules. 2023;28(9):3733. [47] HWANG SH, SHIN MS, YOON TJ, et al. Immunoadjuvant activity in mice of polysaccharides isolated from the leaves of Panax ginseng C.A. Meyer. Int J Biol Macromol. 2018;107(Pt B):2695-2700. [48] HU Y, HE Y, NIU Z, et al. A review of the immunomodulatory activities of polysaccharides isolated from Panax species. J Ginseng Res. 2022; 46(1):23-32. [49] KIM SJ, BAEK SH, KANG KS, et al. Characterization of macrophage activation after treatment with polysaccharides from ginseng according to heat processing. Appl Biol Chem. 2023;66(1):15. [50] FANG J, LI YX, LUO HY, et al. Impacts of sulfur fumigation on the chemistry and immunomodulatory activity of polysaccharides in ginseng. Int J Biol Macromol. 2023;247:125843. [51] LIU T, ZHU C, DUAN Z, et al. Network Pharmacological Analysis Combined with Experimental Verification to Explore the Effect of Ginseng Polypeptide on the Improvement of Diabetes Symptoms in db/db Mice. J Agric Food Chem. 2024;72(33):18537-18551. [52] ZHU N, XU MH, LI Y. Bioactive Oligopeptides from Ginseng (Panax ginseng Meyer) Suppress Oxidative Stress-Induced Senescence in Fibroblasts via NAD+/SIRT1/PGC-1α Signaling Pathway. Nutrients. 2022;14(24):5289. [53] ZHU N, LI Y, XU M. Beneficial Effects of Small-Molecule Oligopeptides Isolated from Panax Ginseng C. A. Meyer on Cellular Fates in Oxidative Stress-Induced Damaged Human Umbilical Vein Endothelial Cells and PC-12. Int J Mol Sci. 2024;25(5):2906. [54] LIU J, XIAO Q, XIAO J, et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct Target Ther. 2022;7(1):3. [55] WANG Z, LI Z, JI H. Direct targeting of β-catenin in the Wnt signaling pathway: Current progress and perspectives. Med Res Rev. 2021;41(4): 2109-2129. [56] MATSUMORI A. Nuclear Factor-κB is a Prime Candidate for the Diagnosis and Control of Inflammatory Cardiovascular Disease. Eur Cardiol. 2023;18:e40. [57] GUO Q, JIN Y, CHEN X, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther. 2024; 9(1):53. [58] DONG W, CHEN W, ZOU H, et al. Ginsenoside Rb1 Prevents Oxidative Stress-Induced Apoptosis and Mitochondrial Dysfunction in Muscle Stem Cells via NF-κB Pathway. Oxid Med Cell Longev. 2022;2022:9159101. [59] CHAKRABORTY J, CHAKRABORTY S, CHAKRABORTY S, et al. Entanglement of MAPK pathways with gene expression and its omnipresence in the etiology for cancer and neurodegenerative disorders. Biochim Biophys Acta Gene Regul Mech. 2023;1866(4):194988. [60] MOUSTARDAS P, ABERDAM D, LAGALI N. MAPK Pathways in Ocular Pathophysiology: Potential Therapeutic Drugs and Challenges. Cells. 2023; 12(4):617 [61] LEE SH, PARK SY, KIM JH, et al. Ginsenoside Rg2 inhibits osteoclastogenesis by downregulating the NFATc1, c-Fos, and MAPK pathways. BMB Rep. 2023;56(10):551-556 [62] CHEN J, ZHANG B, WU L, et al. Ginsenoside Rg3 exhibits anti-catabolic and anti-apoptotic effects in IL-1β treated human disc nucleus pulposus cells and in a rat model of disc degeneration by inactivating the MAPK pathway. Cell Mol Biol (Noisy-le-grand). 2024;70(1):233-238. [63] GUPTA S, KUMAR M, CHAUDHURI S, et al. The non-canonical nuclear functions of key players of the PI3K-AKT-MTOR pathway. J Cell Physiol. 2022;237(8):3181-3204. [64] JING WB, JI H, JIANG R, et al. Astragaloside positively regulated osteogenic differentiation of pre-osteoblast MC3T3-E1 through PI3K/Akt signaling pathway. J Orthop Surg Res. 2021; 16(1):579. [65] JIANG Z, DENG L, LI M, et al. Ginsenoside Rg1 modulates PI3K/AKT pathway for enhanced osteogenesis via GPER. Phytomedicine. 2024;124: 155284. [66] CHIA ZJ, CAO YN, LITTLE PJ, et al. Transforming growth factor-β receptors: versatile mechanisms of ligand activation. Acta Pharmacol Sin. 2024; 45(7):1337-1348. [67] MASSAGUÉ J, SHEPPARD D. TGF-β signaling in health and disease. Cell. 2023;186(19):4007-4037. [68] EL-GAZZAR A, KANG H, FRATZL-ZELMAN N, et al. SMAD3 mutation in LDS3 causes bone fragility by impairing the TGF-β pathway and enhancing osteoclastogenesis. Bone Rep. 2022;17:101603. [69] WANG S, XU B, YIN H, et al. Ginsenoside Rc alleviates osteoporosis by the TGF-β/Smad signaling pathway. Cell Mol Biol (Noisy-le-grand). 2024;70(3):95-101. [70] SHAHGALDI S, KAHMINI FR. A comprehensive review of Sirtuins: With a major focus on redox homeostasis and metabolism. Life Sci. 2021;282: 119803. [71] ZIA A, POURBAGHER-SHAHRI AM, FARKHONDEH T, et al. Molecular and cellular pathways contributing to brain aging. Behav Brain Funct. 2021;17(1):6. [72] TRIPODI F, LOMBARDI L, GUZZETTI L, et al. Protective effect of Vigna unguiculata extract against aging and neurodegeneration. Aging (Albany NY). 2020;12(19):19785-19808. [73] SINGH V, UBAID S. Role of Silent Information Regulator 1 (SIRT1) in Regulating Oxidative Stress and Inflammation. Inflammation. 2020; 43(5):1589-1598. [74] WANG H, ZHENG Y, SUN Q, et al. Ginsenosides emerging as both bifunctional drugs and nanocarriers for enhanced antitumor therapies. J Nanobiotechnology. 2021;19:322. [75] KIM H, LEE JH, KIM JE, et al. Micro-/nano-sized delivery systems of ginsenosides for improved systemic bioavailability. J Ginseng Res. 2018;42(3):361-369. |
| [1] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [2] | Cao Wenqi, Feng Xiuzhi, Zhao Yi, Wang Zhimin, Chen Yiran, Yang Xiao, Ren Yanling. Effect of macrophage polarization on osteogenesis-angiogenesis coupling in type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 917-925. |
| [3] | Wang Sifan, He Huiyu, Yang Quan, Han Xiangzhen. miRNA-378a overexpression of macrophage cell line composite collagen sponge: anti-inflammation and tissue repair promotion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 789-799. |
| [4] | Dai Yueyou, Guo Dandan, Wang Qianqian, Wang Baiyan, Feng Shuying. Anti-tumor effects of engineered exosomes for targeted drug delivery [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(31): 6753-6764. |
| [5] | Zhang Zihan¹, Wang Jiaxin¹, Yang Wenyi², Zhu Lei¹. Regulatory mechanism of exercise promoting mitochondrial biogenesis in skeletal muscle [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(30): 6499-6508. |
| [6] | Wang Shuangmin, Wang Xianyao, He Zhixu. Application and problems in targeted delivery of antitumor drugs by exosomes derived from engineered mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(23): 4975-4983. |
| [7] | Li Guangzhao, Pei Xibo, Wang Jian. Application of nanoparticles in gene modification therapy for bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(22): 4771-4783. |
| [8] | Zhang Wensheng, Guo Haiwei, Weng Rui, Mo Ling, Song Zhenjie, Tian Han, Zhong Yelin, Wang Yuancheng, Tang Hanwu, Liu Caijun, Yuan Chao, Li Ying. Liquiritin inhibits osteoclast differentiation and alleviates bone loss [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(12): 2429-2437. |
| [9] | Xue Chunyang, Wang Xiuhui. Icariin regulates acidic microenvironment to alleviate pain caused by postmenopausal osteoporosis in the elderly [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4461-4468. |
| [10] | Kong Jianda, Xie Yingao, Ma Wen, Liu Youhan, Wang Qinglu. Mitochondrial dysfunction in Parkinson’s disease and the potential ameliorative effects of exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(27): 4413-4420. |
| [11] | Wei Yuxing, Dong Hao, Wei Huiping, Xie Cheng, Tuo Yangjuan, Qin Hao, Cao Yong. Screening and verification of differentially expressed genes in inflammatory tissues with peri-implantitis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4844-4849. |
| [12] | Ma Yujiu, Zhang Xudong, Tan Jichun. Application status and prospect of menstrual blood-derived stem cells and their exosomes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2427-2434. |
| [13] | Liu Zhuoran, Jiang Ming, Li Yourui. Extracellular vesicles in chronic periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 99-104. |
| [14] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [15] | Zou Wanghui, Qian Nannan, Zhang Meng, Pang Qiming, Yang Yichun, Zhang Tao. Implications of preclinical research on mesenchymal stem cells: relationship between cell function of mesenchymal stem cells and the JAK/STAT signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3048-3055. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||