Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7340-7346.doi: 10.12307/2026.813
Previous Articles Next Articles
Hedgehog signaling pathway and diabetic osteoporosis: a potential target for specific drug therapy
Tian Tan1, Bao Shanjun2
- 1School of Graduate, 2School of Sports Training, Wuhan Sports University, Wuhan 430079, Hubei Province, China
-
Received:2025-08-14Revised:2025-12-05Online:2026-10-08Published:2026-02-12 -
Contact:Bao Shanjun, PhD, Professor, School of Sports Training, Wuhan Sports University, Wuhan 430079, Hubei Province, China -
About author:Tian Tan, MS candidate, School of Graduate, Wuhan Sports University, Wuhan 430079, Hubei Province, China
CLC Number:
Cite this article
Tian Tan, Bao Shanjun. Hedgehog signaling pathway and diabetic osteoporosis: a potential target for specific drug therapy[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7340-7346.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
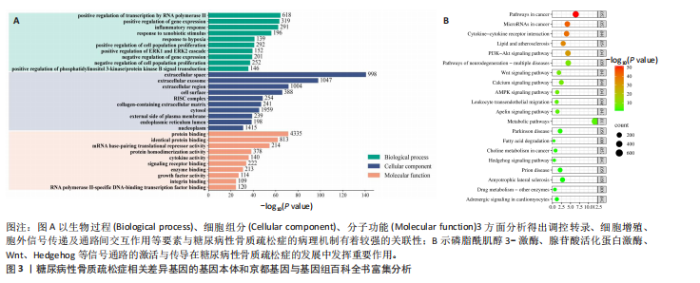
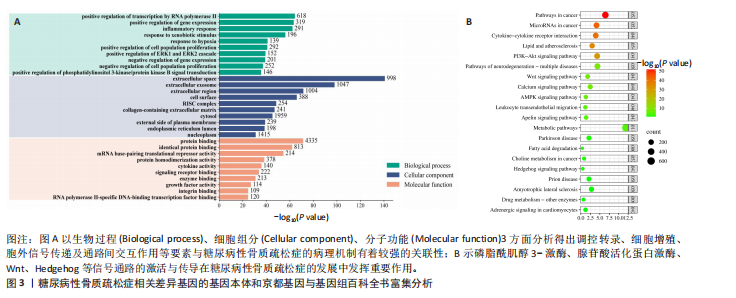
也与该通路的激活程度存在密切联系,在糖尿病性骨质疏松症患者体内因Hedgehog信号通路的核心调控因子表达下调导致通路激活受到抑制,无法发挥其促进成骨形成和分化的作用。当其正常表达时可抑制诱导间充质干细胞向脂肪细胞分化,转而分化为成骨细胞分化升高体内骨含量。因此,阐明该通路的激活机制是防治糖尿病性骨质疏松症重要方向,下面将通过介绍Hedgehog通路的活化机制以及研究进展为其靶向治疗提供理论与参考。 2.1.1 Hedgehog信号通路调节骨细胞增殖分化的作用机制 Hedgehog信号通路的激活是由Hh配体形成开始的,当Hh蛋白进入分泌途径后经过自蛋白水解裂解和脂质修饰使其保留在细胞膜中,在细胞膜中通过与分泌转运蛋白相互作用最终成为成熟的Hh配体[14]。Hh配体在哺乳动物中主要以Shh、Dhh、Ihh三种形式存在,每种配体激活下游靶蛋白所发挥功能各有不同,其中Ihh和Shh配体在骨骼发育、骨形成和骨稳态等过程中起到主要调控作用[16]。经典Hedgehog通路通常分为有Hh配体途径和无Hh配体途径,当没有Hh配体参与通路时,跨膜蛋白受体膜片蛋白对平滑蛋白起到抑制作用,核内转录因子Gli蛋白在全长状态下可促进下游靶基因的转录翻译,当平滑蛋白受到抑制时被蛋白激酶A、糖原合成酶激酶3等蛋白激酶磷酸化和泛素化修饰发生水解失去活性,最终变为转录抑制因子GliR,抑制靶基因表达[17]。当Hh配体存在时,通过Hh与膜片蛋白结合使其失去对受体平滑蛋白的抑制作用,平滑蛋白正常表达时Gli蛋白则变为转录促进因子活化型Gli蛋白激活下游靶基因表达[18]。与人体骨代谢相关的靶基因主要以Runt相关转录因子2、核因子κB受体激活因子配体为主。其中Runt相关转录因子2蛋白在骨代谢中起到促进成骨细胞分化与成熟的作用,而核因子κB受体激活因子配体蛋白则具有调控破骨细胞分化、存活的功能[19],当破骨细胞数量增多打破原平衡时会造成体内骨流失大于骨生成,从而引起骨折、骨质疏松等代谢性骨病[20]。因此,这些与骨骼形成和功能维持密切相关的靶基因表达程度是影响糖尿病性骨质疏松症病理机制的重要因素。 2.1.2 Hedgehog信号通路在骨质疏松症中的研究进展 Hedgehog信号通路作为骨调节及骨生成过程中的重要通路,已在原发性骨质疏松和老年性、绝经后等继发性骨质疏松中获得广泛关注和颇多研究[21],并以此为靶点探究出益为汤、柚皮苷等中药材可有效改善上述类型的骨质疏松症[22-23]。糖尿病性骨质疏松症作为又一类型的继发性骨质疏松,与其他类型相比,患者在骨含量下降的同时还伴有胰岛素抵抗、晚期糖基化终产物积累、氧化和炎症等应激反应造成的微环境损伤[24-25],因此当前针对该疾病病理机制的特异性药物还处于临床前阶段。其他类型骨质疏松症的防治策略主要为补充维生素D及双膦酸盐等抗骨吸收剂维持骨代谢稳态[26],但在糖尿病性骨质疏松症的防治过程中不仅要提高骨形成,还要改善糖尿病所诱发的晚期糖基化终产物积累、胰岛素抵抗以及血糖调节紊乱等问题,因此糖尿病性骨质疏松症的防治策略相较于其他类型骨质疏松症有明显区别。Hedgehog信号通路虽是以调控发育和分化的功能被人们所熟知,但有相关研究称该通路还与糖尿病所诱导的缺氧、炎症应激以及糖代谢紊乱等反应相关[27],在糖尿病患者体内可检测到该通路中核心调控因子的表达受到显著抑制[28],证明Hedgehog通路也是糖尿病病理机制中的重要因素。然而目前对以Hedgehog通路为靶点治疗糖尿病性骨质疏松症的研究还并不十分深入,需进一步加强对该领域的探索研究。 综上所述,Hedgehog通路虽然已被证实是防治骨质疏松症的重要成骨通路,但在糖尿病性骨质疏松症中的研究甚少,比较其在其他类型骨质疏松症防治策略的独特性,该通路在促进骨生成的同时,对于糖尿病进程也有一定的改善作用,可推测其为防治糖尿病性骨质疏松症的重要靶点。因而此文从Hedgehog信号通路角度出发,探讨其转导机制与糖尿病性骨质疏松症发生发展间的潜在联系,填补这一研究领域的空白。 2.2 Hedgehog信号通路与糖尿病性骨质疏松症 首先从Genecards数据库 (https://www. genecards.org/)中检索出与糖尿病性骨质疏松症相关的差异基因,并通过DAVID Bioinformatis (https://david.ncifcrf.gov/)将检索到的差异基因进行基因本体富集分析和京都基因与基因组百科全书富集分析,最终将所得到的基因富集结果在微生信在线平台(https://www. bioinformatics. com.cn/)中绘制成图(图3)。如图所示,基因本体富集分析将基因功能分为生物功能、细胞组分以及分子功能3种类别,可发现细胞增殖调控、缺氧效应以及蛋白结合等因素富集性较为显著,这表明信号通路传导调控的蛋白互作与糖尿病性骨质疏松症的形成发展存在密切联系。而京都基因与基因组百科全书富集分析筛选出与糖尿病性骨质疏松症显著相关的信号通路主要集中在磷脂酰肌醇3-激酶/蛋白激酶B信号通路、腺苷酸活化蛋白激酶、Wnt、Hedgehog等,前三者已在糖尿病及其并发症中研究颇为广泛,而Hedgehog信号通路在这一领域研究相对较少,因而此文选择对Hedgehog信号通路在糖尿病性骨质疏松症发生发展中的分子机制总结论述。 2.2.1 Hedgehog信号通路与成骨蛋白Runt相关转录因子2 Runt相关转录因子2蛋白属于Runx家族转录因子,该基因主要在成骨前体细胞、成骨成熟细胞和软骨细胞中表达,当前已被证明在成骨细胞分化、增殖和软骨细胞成熟等过程中发挥重要作用[29]。成骨细胞是骨骼的主要组成部分,是由骨髓间充质干细胞分化而成,具有充当骨形成的细胞调节剂、促进骨细胞外基质矿化和平衡多种骨细胞基因表达的作用[30]。此外,有研究指出成骨细胞还与胰岛素传导过程相关,具有维持体内葡萄糖水平稳态的作用[31]。因此,成骨细胞数量和功能的正常维持与糖尿病性骨质疏松症的形成发展密切联系。 Hedgehog信号通路的正常表达是Runt相关转录因子2蛋白发挥成骨作用的重要因素,且该蛋白的表达与信号通路的激活之间也存在关联[32]。在先前研究中将敲除Runt相关转录因子2基因小鼠和正常小鼠进行对比,发现前"
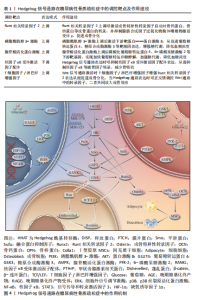
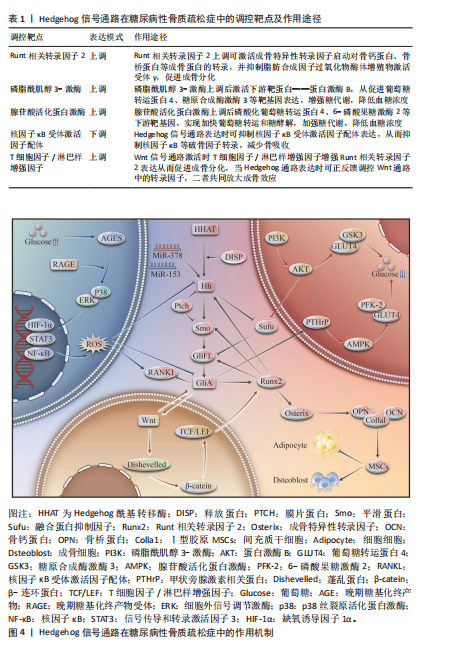
者出现体质量降低、骨骼发育不良的同时伴随体内Ihh、平滑蛋白和GLI家族锌指蛋白1含量下降[33],这表明Runt相关转录因子2除了作为Hedgehog信号通路靶基因促进成骨分化和骨骼生成外,还对该信号通路的表达起到正反馈调控作用,其机制可能为通过抑制刺猬相互作用蛋白、融合蛋白抑制因子等负调控蛋白或在Hh配体加工形成阶段起到促进Hedgehog信号通路激活的作用,但对于这一结论目前并无大量实验结果得以支撑,后续需要检测相关蛋白表达量进行验证。此外,成骨细胞与破骨细胞比例的稳态平衡是维持体内骨骼质量与密度的决定性因素,前面已提及成骨细胞是由骨髓间充质干细胞分化而成,Runt相关转录因子2蛋白作为成骨分化的核心调控因子,在此过程中通过多层级协调相应的成骨基因表达、抑制非成骨谱系分化维持成骨细胞的数量。此前,有报道称骨质疏松症患者骨髓间充质干细胞分化途径与常人相比有明显损伤[34],其原因与患者体内Hedgehog信号通路受到抑制,Runt相关转录因子2蛋白无法正常表达紧密相关。在成骨细胞分化早期阶段,Runt相关转录因子2蛋白可通过自身Runt结构域与骨唾液蛋白、碱性磷酸酶等成骨基因启动子结合从而发挥促进其转录的作用,促进成骨分化过程[35]。当进入分化终期时,Runt相关转录因子2蛋白通过激活成骨特异性转录因子(Osterix)启动Runt相关转录因子2/成骨特异性转录因子信号轴发挥驱动骨基质成熟与矿化的作用,其作用机制为成骨特异性转录因子通过与骨钙蛋白、骨桥蛋白以及Ⅰ型胶原等成骨关键蛋白的启动子特异区域结合促进转录翻译过程以及抑制过氧化物酶体增殖物激活受体γ 和 CCAAT/增强子结合蛋白(C/EBPα/β)等脂肪生成核心调控因子活性介导骨髓间充质干细胞向成骨细胞分化,维持成骨谱系表达[36]。 综上叙述,Runt相关转录因子2蛋白在骨发育、生成及分化过程中起到至关重要的作用,并且其稳定表达依赖于Hedgehog信号通路的磷酸化调控,保障该信号通路的正常激活是防治糖尿病性骨质疏松症的重要靶点。当前,miR-378和miR-155两种基因转录调节因子已被证明可通过调控Hedgehog信号通路表达有效干预股骨头坏死和骨质疏松症等骨类疾病[37-38],并且有研究表明miR-155具有影响胰岛素敏感性和糖脂代谢的功能[39],因此从实验所展现的功能可推测该miRNA具有成为糖尿病性骨质疏松症治疗的潜力靶点,以此设计外泌体联合降糖药物或可成为糖尿病性骨质疏松症的又一有效治疗策略,但其植入后的转化潜力以及表达效果后续仍需大量临床研究加以评估。 2.2.2 Hedgehog信号通路与糖代谢调节 Hedgehog通路对糖尿病性骨质疏松症的防治途径除促进成骨细胞分化、骨基质成熟与矿化外,还有调控糖代谢、维持体内血糖稳态的功能。前文已提到糖尿病性骨质疏松症作为以骨代谢异常为主要特征的糖尿病并发症,在治疗过程中应遵循对糖尿病和骨质疏松症的双重治疗原则,在维持正常骨代谢提高骨含量的同时还要兼顾对糖尿病患者体内糖代谢紊乱的干预治疗。因此下文将从Hedgehog通路调控糖代谢的角度出发,进一步阐述该通路在糖尿病性骨质疏松症中的重要作用。 Hedgehog通路在调节糖代谢的过程中离不开与磷脂酰肌醇3-激酶/蛋白激酶B、腺苷酸活化蛋白激酶两种信号通路的协同调控。磷脂酰肌醇3-激酶/蛋白激酶B信号通路是胰岛素信号传导的重要下游通路,在血糖调节中发挥重要作用[40]。当Hedgehog信号通路正常表达时,Hh配体通过与膜片蛋白结合解除对受体平滑蛋白的抑制作用,从而激活下游因子Gli蛋白,Gli蛋白通过上调磷脂酰肌醇3-激酶蛋白表达并促进蛋白激酶B蛋白分子磷酸化启动磷脂酰肌醇3-激酶/蛋白激酶B通路的信号传导,最终依靠调控葡萄糖转运蛋白4、糖原合成酶激酶3等下游靶基因转录促进葡萄糖的转运、储存并抑制糖异生,起到降低血糖的作用[41]。与此同时,磷脂酰肌醇3-激酶-蛋白激酶B对于Hedgehog通路的表达也具有反馈调控作用,其机制为蛋白激酶B蛋白分子可通过阻止Gli蛋白降解以及抑制融合蛋白抑制因子(Sufu)等Hedgehog信号通路负调控因子表达,增强Hedgehog信号通路靶基因的转录活性,更高效的发挥对糖代谢的正向调节作用。有相关实验结果证明在糖尿病小鼠中检测到Shh、Gli2等蛋白含量显著降低[42],由此可推测其原因为胰岛β细胞损伤、胰岛素抵抗等病理因素通过阻碍胰岛素信号传导抑制了磷脂酰肌醇3-激酶/蛋白激酶B信号通路的激活,最终导致Hedgehog信号通路的表达下调。腺苷酸活化蛋白激酶信号通路作为细胞中的能量感受器,其激活程度随着细胞内能量变化产生改变。当细胞内AMP/ATP升高时,通路中经由腺苷酸活化蛋白激酶激酶激酶至腺苷酸活化蛋白激酶逐级信号传递实现对下游葡萄糖转运蛋白4、6-磷酸果糖激酶2等靶蛋白的磷酸化[43],促进葡萄糖转运的同时增强糖酵解过程,从而调节糖代谢将血糖维持在正常水平,且在糖尿病患者体内可检测到该信号通路的表达显著下调[44],其原因可能与此文所探讨的Hedgehog通路有关。骨骼在人体能量代谢以及葡萄糖代谢中扮演重要角色,在此过程中也离不开Hedgehog与腺苷酸活化蛋白激酶通路间的协同调控效应。当Hedgehog通路上调时可促进骨钙素的分泌,骨钙素已被证明具有增强能量消耗的作用[45],前面已提到腺苷酸活化蛋白激酶通路激活程度是由细胞内ATP浓度所决定,因此当能量消耗增多时,ATP含量下降,腺苷酸活化蛋白激酶通路被激活促进体内糖代谢。此外,Hedgehog通路还可通过产生甲状旁腺激素相关蛋白促进腺苷酸活化蛋白激酶通路表达[46],其作用机制为甲状旁腺激素相关蛋白具有脂联素分泌的功能,而脂联素可激活腺苷酸活化蛋白激酶蛋白活性,从而激活腺苷酸活化蛋白激酶通路表达,改善糖代谢紊乱。恩格列宁作为钠-葡萄糖共转运蛋白2抑制剂类新型降糖药物,在癌症治疗中发现可通过激活腺苷酸活化蛋白激酶抑制Hedgehog通路[47],达到抑癌的作用,这与前面所讲述的Hedgehog正向调控腺苷酸活化蛋白激酶通路相违背,但由于Hedgehog通路的过表达已被证实是促进癌细胞生长的重要因素,并且两种疾病的病理机制存在诸多差异,因此该实验不能否认Hedgehog与腺苷酸活化蛋白激酶在糖代谢中存在正反馈调控机制,但这一结论亟待大量研究得以验证。 综上所述,Hedgehog信号通路除调控成骨分化外,还可以促进能量消耗,增强糖代谢,将血糖维持在正常水平。因此可说明以该通路为靶点防治糖尿病性骨质疏松症可同时改善糖尿病和骨质疏松症的双重病理机制,尽管目前以Hedgehog通路靶向治疗糖尿病的相关研究较为稀少,但考虑其可与磷脂酰肌醇3-激酶-蛋白激酶B、腺苷酸活化蛋白激酶等已在糖尿病临床治疗中被广泛应用的信号通路间存在协同调控作用,因此在后续研究中可以Hedgehog与上述经典糖代谢通路间的反馈调控角度出发,为糖尿病性骨质疏松症的临床治疗提供多机制、多角度、多途径的防治策略。 2.2.3 Hedgehog信号通路与晚期糖基化终末产物积累 晚期糖基化终产物是蛋白质、氨基酸和脂类等大分子物质在非酶促条件下与还原糖一系列反应后生成的稳定产物[48]。高糖环境可诱导晚期糖基化终产物在体内的不断积累,当晚期糖基化终产物水平升高时,其可与晚期糖基化终产物特异性受体结合启动晚期糖基化终产物/晚期糖基化终产物特异性受体信号轴诱发氧化、炎症等应激反应,进一步破坏血糖稳态并加重糖尿病并发症的进程[49],因此控制晚期糖基化终产物在患者体内的浓度是改善糖尿病性骨质疏松症的重要策略。 尽管当前并无直接研究可证明当糖尿病患者体内晚期糖基化终产物堆积时,Hedgehog信号通路表达受到抑制,从而诱导糖尿病性骨质疏松症的发生这一致病机制。但晚期糖基化终产物与其特异性受体反应是产生氧化、炎症应激反应的重要原因,并且在糖尿病神经病变、内皮损伤等其他糖尿病并发症研究中发现氧化、炎症应激反应可抑制Shh、GLI家族锌指蛋白1等Hedgehog通路相关基因表达造成内皮血管生成障碍、神经受损等影响[50-51],因此可推测晚期糖基化终产物与Hedgehog通路下调、糖尿病性骨质疏松症的形成存在密切关联。其作用机制可能为当晚期糖基化终产物含量升高时,晚期糖基化终产物受体与晚期糖基化终产物结合激活后自身受体表达也得到促进,形成正反馈回路。此后,二者相互作用激活细胞外信号调节激酶、p38丝裂原活化蛋白激酶(p38)等与氧化应激密切联系的下游信号通路,导致核因子κB、信号传导和转录激活因子3、缺氧诱导因子1α等转录因子持续表达[52],当这些转录因子水平持续升高时通过激活烟酰胺腺嘌呤二核苷酸氧化酶介导活性氧的产生[53],活性氧具有超负荷过氧化物歧化酶、过氧化氢酶活性以及阻碍谷胱甘肽储存的作用,随着活性氧的逐步积累导致细胞内的氧化还原稳态受到破坏,最终造成氧化应激以及炎症反应,推动糖尿病性骨质疏松症的形成发展过程。氧化应激干预糖尿病性骨质疏松症的发病进程可分为破坏血糖稳态和抑制成骨细胞两个角度,其中破坏血糖稳态这一角度包括降低胰岛素敏感性、诱导胰岛β细胞损伤凋亡以及产生胰岛素抵抗等途径[54-55],通过进一步加重患者体内糖代谢紊乱从而降低骨细胞质量;而氧化应激损伤成骨过程主要依靠抑制成骨分化过程以及破坏成骨与破骨细胞平衡所实现,有研究表明晚期糖基化终产物和高血糖对于成骨细胞分化和矿化具有抑制作用[56],前面已提到Runt相关转录因子2是成骨分化过程重要的调控基因,且该基因的表达对于Hedgehog通路存在正反馈作用;而晚期糖基化终产物破坏骨生成、增强骨吸收则是依靠核因子κB受体激活因子配体基因表达所实现的,核因子κB受体激活因子配体作为Hedgehog通路的靶基因在正常表达时被抑制,当通路表达减弱时核因子κB受体激活因子配体被激活从而促进破骨细胞的分化与表达,有报道称当晚期糖基化终产物与晚期糖基化终产物受体结合时可通过激活核因子κB受体激活因子配体促进破骨前体细胞向成熟破骨细胞的转化[57],加剧糖尿病性骨质疏松症的发展进程。由此可推断出晚期糖基化终产物对于Hedgehog通路存在抑制作用。 综上所述,可以推断出糖尿病患者体内堆积的晚期糖基化终产物对于Hedgehog通路的正常表达有着直接抑制作用,最终可诱导糖尿病性骨质疏松症的发生。并且其作用机制可分为抑制信号通路中Ihh、GLI家族锌指蛋白等核心调控因子和利用Runt相关转录因子2和核因子κB受体激活因子配体等靶基因表达对信号通路的反馈作用两种途径,但其实际作用机制需结合相关实验进一步验证和阐明。与其他种类骨质疏松症相比,糖尿病性骨质疏松症作用机制相对复杂,而Hedgehog作为重要成骨信号通路,与糖尿病相关致病因素的关联性研究是防治该疾病的重要策略,因此亟待相关研究填补该领域的空白。 2.2.4 Hedgehog与Wnt信号通路交叉协同效应 Wnt信号通路已被证实与多种疾病的发生发展相关,且有研究表明在糖尿病和骨质疏松症患者体内伴随该信号通路的异常表达[58-59],因此可推断其与糖尿病性骨质疏松症的发生发展过程相关。考虑到Wnt通路在发挥促进间充质干细胞向成骨细胞分化、矿化以及促进骨细胞成熟等作用时,高度依赖于其他成骨信号通路的协同作用,而此文着重探讨的Hedgehog通路也在此过程中与Wnt通路间存在密切联系,因此下面将通过阐述二者间的协同效应为后续糖尿病性骨质疏松症的临床研究提供理论基础。 前面已叙述到Runt相关转录因子2是成骨分化的重要调控因子,在骨代谢多个环节中发挥了至关重要的作用。除Hedgehog通路外,经典Wnt通路也可通过激活β-连环蛋白(β-catenin),使其与T细胞因子/淋巴样增强因子转录因子结合促进Runt相关转录因子2蛋白表达调控骨代谢,Runt相关转录因子2的表达对Hedgehog通路具有正反馈作用[60],因此当Wnt通路表达时也可促进Hedgehog通路的激活,二者相互激活彼此的转录活性,形成正反馈环路,最终放大Runt相关转录因子2的表达效应调节骨生成[30]。由此看出,这两种通路调控骨代谢过程中存在着共同的靶基因,通过增强Wnt通路表达促进Runt相关转录因子2的同时也对Hedgehog通路激活有一定的促进作用。此外,当Wnt通路激活时可促进Hedgehog通路中GLI家族锌指蛋白1蛋白、平滑蛋白等核心调控因子的活性;反之亦然,Hedgehog通路中的GLI家族锌指蛋白也可促进Wnt配体的分泌[61],两者形成协同激活效应。骨形态发生蛋白通路也是成骨细胞形成与分化过程中的重要通路,有着骨修复与再生以及促进骨基质矿化等作用,当该通路激活时可通过抑制硬化蛋白(sclerostin)等Wnt抑制剂和增强GLI家族锌指蛋白1促进Wnt和Hedgehog通路的激活;而Hedgehog和Wnt通路也可通过激活胰岛素样生长因子-哺乳动物雷帕霉素靶蛋白复合体2通路和稳定Smad1/5蛋白等骨形态发生蛋白下游信号分子等方式促进骨形态发生蛋白通路表达[62],三者间形成多通路协同网络共同调控体内的骨代谢以及骨组织平衡。 综上所述,此文从4个角度概括Hedgehog信号通路传导在糖尿病性骨质疏松症病理机制中的作用(表1,图4)。这表明骨代谢过程除受到单一通路靶基因的调控作用外,还依赖于多条与成骨相关的信号通路协同调控。因此,当以Hedgehog通路为靶点治疗糖尿病性骨质疏松症时,可参考其与其他通路间的交互作用进行联合靶向治疗,例如可通过联合Wnt激活剂在成骨分化过程中激活Hedgehog通路或设计同时调节多通路的小分子激活剂或抑制剂,同时避免因通路的过度激活诱导新的骨损伤,起到事半功倍的治疗效果。"
| [1] 郭曼, 黄炜, 伍棋, 等. 铁死亡在2型糖尿病性骨质疏松症中的作用机制和研究进展[J].中华骨质疏松和骨矿盐疾病杂志,2024,17(5):498-505. [2] 阮泽松, 陈云丰. 间充质干细胞源性外泌体在糖尿病性骨质疏松中的研究进展[J]. 中国骨质疏松杂志,2022,28(1):104-108. [3] 文晓晨, 修静, 刘子英, 等. 糖尿病性骨质疏松症与糖尿病肾病机制及异病同治探析[J]. 辽宁中医药大学学报,2025,27(7):162-170. [4] 柴家超, 杨卫芳, 扈艳雯, 等. SGLT2i对2型糖尿病患者骨代谢影响的研究进展[J]. 中国骨质疏松杂志,2023,29(12):1866-1872. [5] 高浩. 降糖药对糖尿病患者骨代谢的影响[J]. 黑龙江医学,2023,47(22):2815-2817. [6] 马玉萍. 二甲双胍对1型糖尿病患者骨代谢标志物及骨密度影响的研究[D]. 兰州:兰州大学,2021. [7] SUCHORS C, KIM J. Canonical Hedgehog Pathway and Noncanonical GLI Transcription Factor Activation in Cancer. Cells. 2022;11(16):2523. [8] JING J, WU Z, WANG J, et al. Hedgehog signaling in tissue homeostasis, cancers, and targeted therapies. Signal Transduct Target Ther. 2023;8(1):315. [9] ZHOU H, ZHANG L, CHEN Y, et al. Research progress on the hedgehog signalling pathway in regulating bone formation and homeostasis. Cell Prolif. 2022;55(1):e13162. [10] 王雨荷, 刘红, 史婧儒, 等. 由Hedgehog信号通路探讨左归丸对卵巢切除大鼠骨质疏松症的影响[J]. 中国骨质疏松杂志,2023,29(6):781-785+791. [11] 阿布都克热木·达吾提, 柯义兵, 金裕林, 等. 骨质疏松症相关信号通路研究进展[J]. 国际骨科学杂志,2025,46(3):178-182. [12] NGUYEN NM, CHO J. Hedgehog Pathway Inhibitors as Targeted Cancer Therapy and Strategies to Overcome Drug Resistance. Int J Mol Sci. 2022;23(3):1733. [13] JIANG J. Hedgehog signaling mechanism and role in cancer. Semin Cancer Biol. 2022;85:107-122. [14] ZARZOSA P, GARCIA-GILABERT L, HLADUN R, et al. Targeting the Hedgehog Pathway in Rhabdomyosarcoma. Cancers (Basel). 2023;15(3):727. [15] FENG Z, ZHU S, LI W, et al. Current approaches and strategies to identify Hedgehog signaling pathway inhibitors for cancer therapy. Eur J Med Chem. 2022;244:114867. [16] MORSCZECK C, RECK A, BECK HC. The hedgehog-signaling pathway is repressed during the osteogenic differentiation of dental follicle cells. Mol Cell Biochem. 2017;428(1-2):79-86. [17] ZHANG L, FU X, NI L, et al. Hedgehog Signaling Controls Bone Homeostasis by Regulating Osteogenic/Adipogenic Fate of Skeletal Stem/Progenitor Cells in Mice. J Bone Miner Res. 2022; 37(3):559-576. [18] HU Z, CHEN B, ZHAO Q. Hedgehog signaling regulates osteoblast differentiation in zebrafish larvae through modulation of autophagy. Biol Open. 2019;8(5):bio040840. [19] NIRALA BK, YAMAMICHI T, YUSTEIN JT. Deciphering the Signaling Mechanisms of Osteosarcoma Tumorigenesis. Int J Mol Sci. 2023;24(14):11367. [20] LU W, ZHENG C, ZHANG H, et al. Hedgehog signaling regulates bone homeostasis through orchestrating osteoclast differentiation and osteoclast-osteoblast coupling. Cell Mol Life Sci. 2023;80(6):171. [21] 杨仁慧, 王怡璇, 甘胜露, 等. 基于Hedgehog信号通路探讨Shh和Ihh蛋白在原发性骨质疏松症患者血清中的表达[J]. 贵州中医药大学学报,2024,46(1):82-86. [22] FAN W, MENG Y, ZHANG J, et al. To investigate the mechanism of Yiwei Decoction in the treatment of premature ovarian insufficiency-related osteoporosis using transcriptomics, network pharmacology and molecular docking techniques. Sci Rep. 2023;13(1):19016. [23] LIN FX, DU SX, LIU DZ, et al. Naringin promotes osteogenic differentiation of bone marrow stromal cells by up-regulating Foxc2 expression via the IHH signaling pathway. Am J Transl Res. 2016;8(11): 5098-5107. [24] ZHANG C, LI H, LI J, et al. Oxidative stress: A common pathological state in a high-risk population for osteoporosis. Biomed Pharmacother. 2023;163:114834. [25] WANG B, VASHISHTH D. Advanced glycation and glycoxidation end products in bone. Bone. 2023; 176:116880. [26] 文晓晨, 刘若实, 朱克, 等. 补肾健脾法调控自噬相关PINK1/Parkin信号通路治疗糖尿病性骨质疏松症[J]. 中国中医基础医学杂志,2025, 31(4):649-654. [27] GARG C, KHAN H, KAUR A, et al. Therapeutic implications of sonic hedgehog pathway in metabolic disorders: Novel target for effective treatment. Pharmacol Res. 2022;179:106194. [28] BRAUNE J, WEYER U, MATZ-SOJA M, et al. Hedgehog signalling in myeloid cells impacts on body weight, adipose tissue inflammation and glucose metabolism. Diabetologia. 2017;60(5):889-899. [29] KOMORI T. Regulation of Skeletal Development and Maintenance by Runx2 and Sp7. Int J Mol Sci. 2024;25(18):10102. [30] ZHU S, CHEN W, MASSON A, et al. Cell signaling and transcriptional regulation of osteoblast lineage commitment, differentiation, bone formation, and homeostasis. Cell Discov. 2024;10(1):71. [31] SUN H, WANG S, YANG Z, et al. Chordin Like-1 Regulates Osteoblast and Adipocyte Differentiation Through Stabilizing Insulin-Like Growth Factor Binding Protein 3. Stem Cells. 2023;41(4):400-414. [32] PEI Y, LIU F, ZHAO Y, et al. Role of hedgehog signaling in the pathogenesis and therapy of heterotopic ossification. Front Cell Dev Biol. 2024; 12:1454058. [33] WANG Y, DONG Z, YANG R, et al. Inactivation of Ihh in Sp7-Expressing Cells Inhibits Osteoblast Proliferation, Differentiation, and Bone Formation, Resulting in a Dwarfism Phenotype with Severe Skeletal Dysplasia in Mice. Calcif Tissue Int. 2022; 111(5):519-534. [34] HUO S, TANG X, CHEN W, et al. Epigenetic regulations of cellular senescence in osteoporosis. Ageing Res Rev. 2024;99:102235. [35] ZHANG YX, SUN HL, LIANG H, et al. Dynamic and distinct histone modifications of osteogenic genes during osteogenic differentiation. J Biochem. 2015;158(6):445-457. [36] CHI B, LIU G, XING L, et al. [Research progress of hedgehog signaling pathway in regulating bone formation and osteogenic differentiation of bone mesenchymal stem cells]. Chin J Repar Reconstr Surg. 2016;30(12):1545-1550. [37] NAN K, ZHANG Y, ZHANG X, et al. Exosomes from miRNA-378-modified adipose-derived stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head by enhancing angiogenesis and osteogenesis via targeting miR-378 negatively regulated suppressor of fused (Sufu). Stem Cell Res Ther. 2021;12(1):331. [38] GAO X, ZHOU J, WANG J, et al. Mechanism of exosomal miR-155 derived from bone marrow mesenchymal stem cells on stemness maintenance and drug resistance in myeloma cells. J Orthop Surg Res. 2021;16(1):637. [39] JANKAUSKAS SS, GAMBARDELLA J, SARDU C, et al. Functional Role of miR-155 in the Pathogenesis of Diabetes Mellitus and Its Complications. Noncoding RNA. 2021;7(3):39. [40] FONTANA F, GIANNITTI G, MARCHESI S, et al. The PI3K/Akt Pathway and Glucose Metabolism: A Dangerous Liaison in Cancer. Int J Biol Sci. 2024; 20(8):3113-3125. [41] BENCHOULA K, PARHAR IS, WONG EH. The crosstalk of hedgehog, PI3K and Wnt pathways in diabetes. Arch Biochem Biophys. 2021;698:108743. [42] LI M, WANG Y, WU X, et al. KIAA0753 enhances osteoblast differentiation suppressed by diabetes. J Cell Mol Med. 2024;28(17):e70035. [43] HERMAN R, KRAVOS NA, JENSTERLE M, et al. Metformin and Insulin Resistance: A Review of the Underlying Mechanisms behind Changes in GLUT4-Mediated Glucose Transport. Int J Mol Sci. 2022;23(3):1264. [44] ZHANG L, ZHOU X, CHEN H, et al. Mulberry extract ameliorates T2DM-related symptoms via AMPK pathway in STZ-HFD-induced C57BL/6J mice. J Ethnopharmacol. 2023;313:116475. [45] MERA P, FERRON M, MOSIALOU I. Regulation of Energy Metabolism by Bone-Derived Hormones. Cold Spring Harb Perspect Med. 2018; 8(6):a031666. [46] ZHANG X, CHENG Q, WANG Y, et al. Hedgehog signaling in bone regulates whole-body energy metabolism through a bone-adipose endocrine relay mediated by PTHrP and adiponectin. Cell Death Differ. 2017;24(2):225-237. [47] XIE Z, WANG F, LIN L, et al. An SGLT2 inhibitor modulates SHH expression by activating AMPK to inhibit the migration and induce the apoptosis of cervical carcinoma cells. Cancer Lett. 2020;495: 200-210. [48] SHEN CY, LU CH, WU CH, et al. The Development of Maillard Reaction, and Advanced Glycation End Product (AGE)-Receptor for AGE (RAGE) Signaling Inhibitors as Novel Therapeutic Strategies for Patients with AGE-Related Diseases. Molecules. 2020;25(23):5591. [49] PASSARELLI M, MACHADO UFF. AGEs-Induced and Endoplasmic Reticulum Stress/Inflammation-Mediated Regulation of GLUT4 Expression and Atherogenesis in Diabetes Mellitus. Cells. 2021; 11(1):104. [50] DEWANJEE S, DAS S, DAS AK, et al. Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets. Eur J Pharmacol. 2018;833:472-523. [51] NIU C, CHEN Z, KIM KT, et al. Metformin alleviates hyperglycemia-induced endothelial impairment by downregulating autophagy via the Hedgehog pathway. Autophagy. 2019;15(5):843-870. [52] KHALID M, PETROIANU G, ADEM A. Advanced Glycation End Products and Diabetes Mellitus: Mechanisms and Perspectives. Biomolecules. 2022;12(4):542. [53] GASIOROWSKI K, BROKOS B, ECHEVERRIA V, et al. RAGE-TLR Crosstalk Sustains Chronic Inflammation in Neurodegeneration. Mol Neurobiol. 2018;55(2): 1463-1476. [54] DU H, MA Y, WANG X, et al. Advanced glycation end products induce skeletal muscle atrophy and insulin resistance via activating ROS-mediated ER stress PERK/FOXO1 signaling. Am J Physiol Endocrinol Metab. 2023;324(3):E279-E287. [55] WYSHAM C, SHUBROOK J. Beta-cell failure in type 2 diabetes: mechanisms, markers, and clinical implications. Postgrad Med. 2020;132(8):676-686. [56] GE W, JIE J, YAO J, et al. Advanced glycation end products promote osteoporosis by inducing ferroptosis in osteoblasts. Mol Med Rep. 2022; 25(4):140. [57] DING X, HU Y, LUO D, et al. [Effects of advanced glycation end products on osteoclasts at different stages of differentiation]. J South Med Univ. 2020; 40(4):573-579. [58] MAIESE K. Cornerstone Cellular Pathways for Metabolic Disorders and Diabetes Mellitus: Non-Coding RNAs, Wnt Signaling, and AMPK. Cells. 2023;12(22):2595. [59] RONG X, KOU Y, ZHANG Y, et al. ED-71 Prevents Glucocorticoid-Induced Osteoporosis by Regulating Osteoblast Differentiation via Notch and Wnt/β-Catenin Pathways. Drug Des Devel Ther. 2022;16:3929-3946. [60] KOMORI T. Bone development by Hedgehog and Wnt signaling, Runx2, and Sp7. J Bone Miner Metab. 2025;43(1):33-38. [61] XUE C, CHU Q, SHI Q, et al. Wnt signaling pathways in biology and disease: mechanisms and therapeutic advances. Signal Transduct Target Ther. 2025;10(1)106. [62] LI SY, XUE ST, LI ZR. Osteoporosis: Emerging targets on the classical signaling pathways of bone formation. Eur J Pharmacol. 2024;973:176574. [63] LI J, YAN R, SHI S, et al. Recent progress and application of the tetrahedral framework nucleic acid materials on drug delivery. Expert Opin Drug Deliv. 2023;20(11):1511-1530. [64] ZHAO Y, LI S, FENG M, et al. Effects of Puerarin-Loaded Tetrahedral Framework Nucleic Acids on Osteonecrosis of the Femoral Head. Small. 2023; 19(41):e2302326. [65] LI Y, TANG Y, SHI S, et al. Tetrahedral Framework Nucleic Acids Ameliorate Insulin Resistance in Type 2 Diabetes Mellitus via the PI3K/Akt Pathway. ACS Appl Mater Interfaces. 2021;13(34):40354-40364. [66] DENG TT, DING WY, LU XX, et al. Pharmacological and mechanistic aspects of quercetin in osteoporosis. Front Pharmacol. 2024;15:1338951. [67] DHANYA R. Quercetin for managing type 2 diabetes and its complications, an insight into multitarget therapy. Biomed Pharmacother. 2022; 146:112560. [68] CHEN Y, WEI Z, SHI H, et al. BushenHuoxue formula promotes osteogenic differentiation via affecting Hedgehog signaling pathway in bone marrow stem cells to improve osteoporosis symptoms. PLoS One. 2023;18(11):e0289912. [69] CUI Z, ZHAO X, AMEVOR FK, et al. Therapeutic application of quercetin in aging-related diseases: SIRT1 as a potential mechanism. Front Immunol. 2022;13:943321. [70] WANG N, WANG L, YANG J, et al. Quercetin promotes osteogenic differentiation and antioxidant responses of mouse bone mesenchymal stem cells through activation of the AMPK/SIRT1 signaling pathway. Phytother Res. 2021;35(5):2639-2650. [71] 徐嵩昊, 董德龙. 有氧与抗阻运动对老年人慢性病的影响[J]. 中国老年学杂志,2025,45(11): 2798-2802. [72] 代新宇, 李斌, 靳丹, 等. 运动通过调节骨自噬途径防治骨质疏松症的作用及可能机制 [J]. 生物化学与生物物理进展,2024,51(7):1589-1603. [73] 张帅军, 张锦, 杜旭辉, 等. 跑台运动对切除卵巢大鼠骨质疏松症的保护机制[J]. 中国骨质疏松杂志,2024,30(6):824-828. [74] ZHANG M, LI Y, LIU L, et al. The effects on type 2 diabetes mellitus mouse femoral bone achieved by anti-osteoporosis exercise interventions. Front Endocrinol (Lausanne). 2022;13:914872. [75] 文武, 侯铁奇. 不同运动强度对去势大鼠骨质疏松软骨形态的影响[J]. 中国骨质疏松杂志, 2020,26(6):876-880. [76] 侯慧卿, 王晓曦, 叶天鸣, 等. 八段锦对中年人群血脂、血糖及血尿酸水平的影响[J]. 现代中西医结合杂志,2025,34(5):685-689. [77] 邵霞, 王宁, 王花欣, 等. 中医导引术在骨质疏松症中的临床应用概述[J]. 山东中医药大学学报,2025,49(2):249-255. [78] 董子萱, 马占科. Piezo1信号通路在运动干预改善2型糖尿病中的作用及其机制[J]. 生物化学与生物物理进展,2025,52(2):290-298. [79] ZHANG J, TAM WWS, HOUNSRI K, et al. Effectiveness of Combined Aerobic and Resistance Exercise on Cognition, Metabolic Health, Physical Function, and Health-related Quality of Life in Middle-aged and Older Adults With Type 2 Diabetes Mellitus: A Systematic Review and Meta-analysis. Arch Phys Med Rehabil. 2024;105(8):1585-1599. [80] 王惠国, 杨高源, 谢仙燕, 等. 运动诱导的代谢重塑对免疫功能的调控作用[J]. 生物化学与生物物理进展,2025,52(6):1574-1586. [81] STOCKS B, ZIERATH JR. Post-translational Modifications: The Signals at the Intersection of Exercise, Glucose Uptake, and Insulin Sensitivity. Endocr Rev. 2022;43(4):654-677. |
| [1] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [2] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [3] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [4] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [5] | Liu Huan, Zeng Shaopeng, Chen Jun, He Linqian, Yang Ying, Zhang Jing. Aging-related dysregulation of glucose metabolism: crossroads of cancer and neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1527-1538. |
| [6] | Zhou Zixiang, Zhao Baoxiang. Research progress in the relationship between nontraumatic necrosis of the femoral head and lipid metabolism and its treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 680-690. |
| [7] | Sun Danhe, Guo Xiaoling, Zhao Lingzhou. Construction and osteogenic activity of titanium dioxide nanotube and polydopamine composite coating on titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5167-5177. |
| [8] | Zhong Zhuolan, Peng Zhina, Tian Xiaohong, Han Cuifei, Zhang Zihan, Chu Jiaqi. Acanthopanax exosome-like nanovesicles promote osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4825-4835. |
| [9] | Wang Hengxin, Li Hongkun, Xu Nuo, Li Anping, Wang Xinjing, Zhang Tong. Quercetin promotes osteogenic differentiation of senescent jaw bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4843-4852. |
| [10] | Zhou Rui, Zhang Xuesong, Yang Donghong, Jin Yinan, Yang Yuqi, Ye Zhihui. Overexpression of collagen triple helix repeat-containing protein 1 promotes proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4918-4925. |
| [11] | Wang Siwei, Yao Xiaosheng, Qi Xiaonan, Wang Yu, Cui Haijian, Zhao Jiaxuan. Matrix metalloproteinase 9 mediates mitophagy to regulate osteogenesis and myogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4557-4567. |
| [12] | Li Pengli, Yang Yanmei, Hu Yawen, Liu Hongqi, Wang Manyi, Yan Jianfei, Gu Bin. Effect of zoledronic acid on jaw bone marrow mesenchymal stem cells in mice with bisphosphonate-related osteonecrosis of the jaw [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3217-3225. |
| [13] | Wu Jiazhou, Qian Tao, Liu Zexian, Wu Yanbin, He Ying, Li Yazhou, Peng Jiang. Three-dimensional culture of stromal vascular fraction self-assembles into complex vascularized osteogenic organoids [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2681-2690. |
| [14] | Zou Yuxi, Chen Yanyan, Jiang Peng, Chen Ting, Ding Lingling. Critical role of lysosomal enzymes in metabolic diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2823-2833. |
| [15] | Chen Qiheng, Weng Tujun, Peng Jiang. Effect of dimethylglyoxal glycine on osteogenic, adipogenesis differentiation, and mitophagy of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 50-57. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||