Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7307-7315.doi: 10.12307/2026.773
Previous Articles Next Articles
Xanthohumol combined with swimming ameliorates hepatic injury in rats with metabolic associated fatty liver disease
Wang Zheng1, Wu Weidong1, Zhu Jingsheng2
- 1Henan Sport University, Zhengzhou 450044, Henan Province, China; 2School of Exercise and Health, Shanghai University of Sport, Shanghai 200438, China
-
Received:2025-09-25Revised:2025-11-11Online:2026-10-08Published:2026-02-11 -
Contact:Wu Weidong, PhD, Associate professor, Henan Sport University, Zhengzhou 450044, Henan Province, China -
About author:Wang Zheng, MS, Lecturer, Henan Sport University, Zhengzhou 450044, Henan Province, China -
Supported by:Henan Province Science and Technology Research Project, No. 202102310324 (to WWD)
CLC Number:
Cite this article
Wang Zheng, Wu Weidong, Zhu Jingsheng. Xanthohumol combined with swimming ameliorates hepatic injury in rats with metabolic associated fatty liver disease[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7307-7315.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
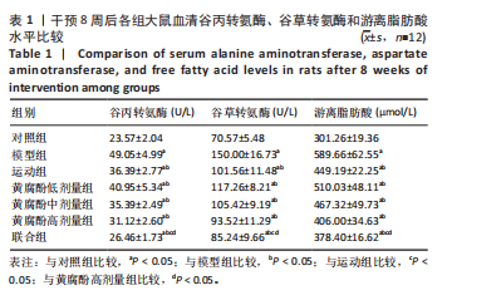
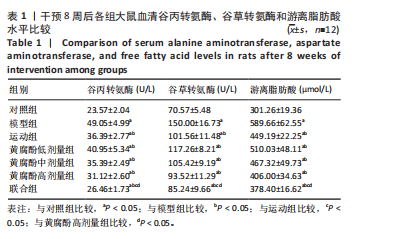
2.1 实验动物数量分析 实验纳入90只大鼠,12只大鼠作为对照,78只用于建模,其中72只建模成功,3只未建模成功,3只处死后观察肝脏组织学改变,因此未纳入分组。 2.2 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平的影响 干预8周后,各组大鼠血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平相比差异有显著性意义(F=78.066,66.667,66.248,P < 0.001)。与对照组比较,模型组大鼠的血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平升高(P < 0.05)。与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠的血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平降低(P < 0.05)。与运动组和黄腐酚高剂量组比较,联合组大鼠的血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平降低(P < 0.05)。两因素方差分析结果显示,黄腐酚(因素一)和游泳(因素二)对大鼠血清谷丙转氨酶、谷草转氨酶和游离脂肪酸水平的影响均存在显著主效应(各指标 F 值分别为 78.066,66.667,66.248,均P < 0.001),且两因素间存在显著交互效应(P < 0.001),即黄腐酚与游泳对上述指标的改善具有协同作用。见表1。"
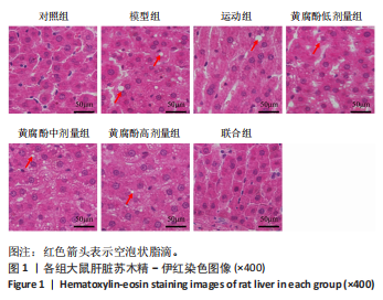
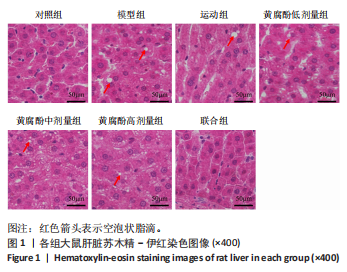
2.3 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠肝脏形态的影响 Olympus BX53生物显微镜下苏木精-伊红染色显示,对照组大鼠肝脏形态和结构正常,无脂肪变性;模型组大鼠肝脏结构紊乱,肝细胞肿胀、变形,存在大量脂肪空泡;与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠肝脏形态明显改善,其中联合组的肝脏形态基本恢复正常。对照组、模型组、运动组、黄腐酚低、中、高剂量组和联合组大鼠的肝脏脂滴面积百分比分别为(5.23±1.15)%,(28.6±3.21)%,(18.76±2.54)%,(15.32±2.10)%,(10.56±1.87)%,(8.23±1.56)%,(6.12±1.23)%,组间差异有显著性意义(F=38.765,P < 0.001)。两因素方差分析显示,黄腐酚和游泳对肝脏脂滴面积百分比的影响均存在显著主效应(F=486.65,P < 0.001),且交互效应显著(P < 0.001),即二者联合具有协同调节作用。见图1。"
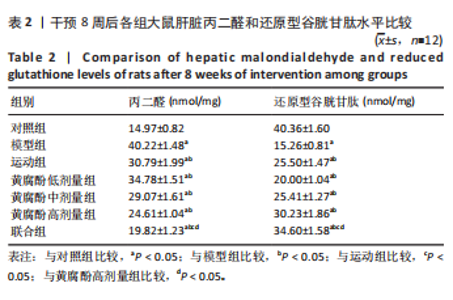
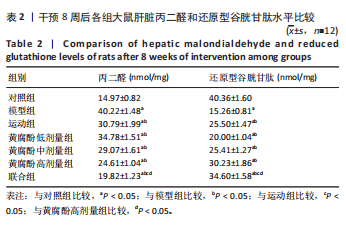
2.4 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠肝脏丙二醛和还原型谷胱甘肽的影响 干预8周后,各组大鼠的肝脏丙二醛和还原型谷胱甘肽水平相比差异有显著性意义(F=441.738,436.872,P < 0.001)。与对照组比较,模型组大鼠的肝脏丙二醛水平升高,还原型谷胱甘肽水平降低(P < 0.05);与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠的肝脏丙二醛水平降低,还原型谷胱甘肽水平升高(P < 0.05);与运动组和黄腐酚高剂量组比较,联合组大鼠的肝脏丙二醛水平降低,还原型谷胱甘肽水平升高(P < 0.05)。两因素方差分析显示,黄腐酚和游泳对肝脏丙二醛(F=441.738,P < 0.001)和还原型谷胱甘肽(F=436.872,P < 0.001)水平的影响均存在显著主效应,且交互效应显著(P < 0.001),即二者联合具有协同调节作用。见表2。"
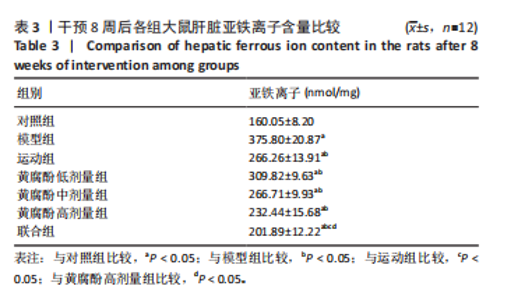
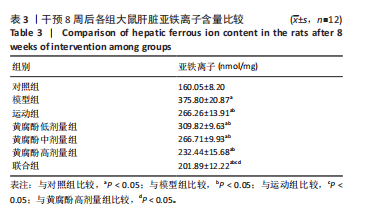
2.5 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠肝脏亚铁离子含量的影响 干预8周后,各组大鼠的肝脏亚铁离子含量相比,差异有显著性意义(F=328.427,P < 0.001)。与对照组比较,模型组大鼠的肝脏亚铁离子含量升高(P < 0.05);与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠的肝脏亚铁离子含量降低(P < 0.05);与运动组和黄腐酚高剂量组比较,联合组大鼠的肝脏亚铁离子含量降低(P < 0.05)。两因素方差分析结果显示,黄腐酚与游泳对肝脏亚铁离子含量的影响存在显著主效应(F=328.427,P < 0.001)及交互效应(P < 0.001),即二者联合具有协同降铁作用。见表3。"
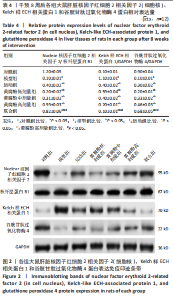
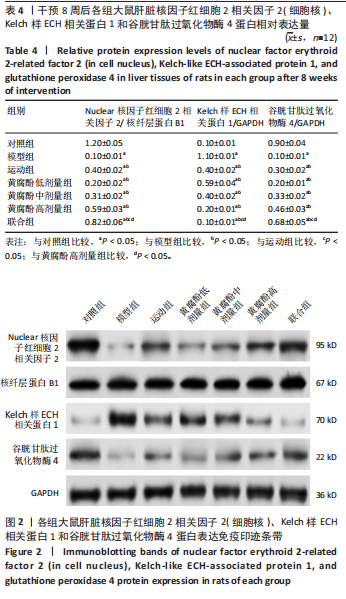
2.6 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠肝脏核因子红细胞2相关因子2(细胞核)、Kelch样ECH相关蛋白1和谷胱甘肽过氧化物酶4的影响 干预8周后,各组大鼠肝脏核因子红细胞2相关因子2(细胞核)、Kelch样ECH相关蛋白1和谷胱甘肽过氧化物酶4蛋白表达水平相比差异有显著性意义(F=1 465.512,3 382.528,1 181.989,P < 0.001)。与对照组比较,模型组大鼠的肝脏核因子红细胞2相关因子2(细胞核)和谷胱甘肽过氧化物酶4蛋白表达水平降低,Kelch样ECH相关蛋白1蛋白表达水平升高(P < 0.05);与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠的肝脏核因子红细胞2相关因子2(细胞核)和谷胱甘肽过氧化物酶4蛋白表达水平升高,Kelch样ECH相关蛋白1蛋白表达水平降低(P < 0.05);与运动组和黄腐酚高剂量组比较,联合组大鼠的肝脏核因子红细胞2相关因子2(细胞核)和谷胱甘肽过氧化物酶4蛋白表达水平升高,Kelch样ECH相关蛋白1蛋白表达水平降低(P < 0.05)。两因素方差分析显示,黄腐酚与游泳对肝脏核因子红细胞2相关因子2(细胞核)、Kelch样ECH相关蛋白1和谷胱甘肽过氧化物酶4蛋白表达的影响均存在显著主效应(F值分别为1 465.512,3 382.528,1 181.989,均P < 0.001),且交互效应显著(P < 0.001),即二者联合具有协同调控作用。见表4及图2。 2.7 黄腐酚联合游泳对代谢相关脂肪性肝病大鼠肝脏铁死亡相关基因的影响 干预8周后,与对照组比较,模型组大鼠的肝脏谷胱甘肽过氧化物酶4、溶质载体家族7成员11、铁蛋白重链 1和膜铁转运蛋白1 mRNA水平降低,阳离子转运调控样蛋白1 mRNA水平升高(P < 0.05);与模型组比较,运动组、黄腐酚低、中、高剂量组和联合组大鼠的肝脏谷胱甘肽过氧化物酶4、溶质载体家族7成员11、铁蛋白重链 1和膜铁转运蛋白1 mRNA"

水平升高,阳离子转运调控样蛋白1 mRNA水平降低(P < 0.05);与运动组和黄腐酚高剂量组比较,联合组大鼠的肝脏谷胱甘肽过氧化物酶4、溶质载体家族7成员11、铁蛋白重链 1和膜铁转运蛋白1 mRNA水平升高,阳离子转运调控样蛋白1 mRNA水平降低(P < 0.05)。两因素方差分析结果显示,黄腐酚与游泳对肝脏铁死亡相关基因mRNA水平的影响存在显著主效应(谷胱甘肽过氧化物酶4:F=1 286.354,溶质载体家族7成员11:F=1 152.783,阳离子转运调控样蛋白1:F=1 890.426,铁蛋白重链 1:F=986.215,膜铁转运蛋白1:F=876.532,均P < 0.001),且存在显著交互效应(均P < 0.001),即二者联合具有协同调控作用。见表5。"

| [1] ENGIN A. Nonalcoholic fatty liver disease and staging of hepatic fibrosis. Adv Exp Med Biol. 2024;1460:539-574. [2] NIE K, GAO Y, CHEN S, et al. Diosgenin attenuates non-alcoholic fatty liver disease in type 2 diabetes through regulating SIRT6-related fatty acid uptake. Phytomedicine. 2023;111:154661. [3] RONG L, ZOU J, RAN W, et al. Advancements in the treatment of non-alcoholic fatty liver disease (NAFLD). Front Endocrinol (Lausanne). 2022;13:1087260. [4] TONG J, LAN XT, ZHANG Z, et al. Ferroptosis inhibitor liproxstatin-1 alleviates metabolic dysfunction-associated fatty liver disease in mice: potential involvement of PANoptosis. Acta Pharmacol Sin. 2023;44(5):1014-1028. [5] WANG S, LIU Z, GENG J, et al. An overview of ferroptosis in non-alcoholic fatty liver disease. Biomed PHARMACOTHER. 2022;153:113374. [6] GUO T, YAN W, CUI X, et al. Liraglutide attenuates type 2 diabetes mellitus-associated non-alcoholic fatty liver disease by activating AMPK/ACC signaling and inhibiting ferroptosis. Mol Med. 2023;29(1):132. [7] SHEN X, YU Z, WEI C, et al. Iron metabolism and ferroptosis in nonalcoholic fatty liver disease: what is our next step?. Am J Physiol Endocrinol Metab. 2024;326(6):E767-E775. [8] WU J, WANG Y, JIANG R, et al. Ferroptosis in liver disease: new insights into disease mechanisms. Cell Death Discov. 2021;7(1):276. [9] SHEN K, WANG X, WANG Y, et al. miR-125b-5p in adipose derived stem cells exosome alleviates pulmonary microvascular endothelial cells ferroptosis via Keap1/Nrf2/GPX4 in sepsis lung injury. Redox Biol. 2023;62:102655. [10] GAO G, XIE Z, LI EW, et al. Dehydroabietic acid improves nonalcoholic fatty liver disease through activating the Keap1/Nrf2-ARE signaling pathway to reduce ferroptosis. J Nat Med. 2021;75(3):540-552. [11] JIANG Y, JIANG K, SUN P, et al. Oroxylin A ameliorates non-alcoholic fatty liver disease by modulating oxidative stress and ferroptosis through the Nrf2 pathway. Biochim Biophys Acta Mol Cell Biol Lipids. 2025;1870(5):159628. [12] COSTA R, RODRIGUES I, GUARDÃO L, et al. Xanthohumol and 8-prenylnaringenin ameliorate diabetic-related metabolic dysfunctions in mice. J Nutr Biochem. 2017;45:39-47. [13] LEGETTE LL, LUNA AY, REED RL, et al. Xanthohumol lowers body weight and fasting plasma glucose in obese male Zucker fa/fa rats. Phytochemistry. 2013; 91:236-241. [14] TAKAHASHI K, OSADA K. Effect of dietary purified xanthohumol from hop (humulus lupulus l.) pomace on adipose tissue mass, fasting blood glucose level, and lipid metabolism in kk-ay mice. J Oleo Sci. 2017;66(5):531-541. [15] MAHLI A, SEITZ T, FREESE K, et al. Therapeutic application of micellar solubilized xanthohumol in a western-type diet-induced mouse model of obesity, diabetes and non-alcoholic fatty liver disease. Cells. 2019;8(4):359. [16] DENG Y, CHU X, LI Q, et al. Xanthohumol ameliorates drug-induced hepatic ferroptosis via activating Nrf2/xCT/GPX4 signaling pathway. Phytomedicine. 2024;126:155458. [17] LIN JH, YANG KT, LEE WS, et al. Xanthohumol protects the rat myocardium against ischemia/reperfusion injury-induced ferroptosis. Oxid Med Cell Longev. 2022;2022:9523491. [18] WANG S, ZHOU Y, WU Y, et al. Long-term aerobic exercise improves learning memory capacity and effects on oxidative stress levels and Keap1/Nrf2/GPX4 pathway in the hippocampus of APP/PS1 mice. Front Neurosci. 2024;18:1505650. [19] GAO Y, LING Y, WU H, et al. Swimming training attenuates doxorubicin induced cardiomyopathy by targeting the mir-17-3p/KEAP1/NRF2 axis. Biochem Biophys Res Commun. 2024;739: 50568. [20] ZHENG F, CAI Y. Concurrent exercise improves insulin resistance and nonalcoholic fatty liver disease by upregulating PPAR-γ and genes involved in the beta-oxidation of fatty acids in ApoE-KO mice fed a high-fat diet. Lipids Health Dis. 2019;18(1):6. [21] ROMERO-GÓMEZ M, ZELBER-SAGI S, TRENELL M. Treatment of NAFLD with diet, physical activity and exercise. J Hepatol. 2017;67(4):829-846. [22] 冷亮, 蒋卓勤, 纪桂元. 大豆异黄酮对非酒精性脂肪肝大鼠肝脏脂代谢的影响[J]. 中华预防医学杂志,2011,45(4):335-339. [23] WANG CC, HO YH, HUNG CF, et al. Xanthohumol, an active constituent from hope, affords protection against kainic acid-induced excitotoxicity in rats. Neurochem Int. 2020;133:104629. [24] 申甜, 徐碧林, 盛霞, 等. 非酒精性脂肪肝大鼠肝组织FGF21mRNA表达与脂质沉积、胰岛素抵抗的关系[J]. 山东医药,2016,56(8):10-13. [25] 徐立, 胡瑞斌, 李兆波, 等. STZ诱导糖尿病大鼠胰腺及肝脏石蜡切片HE染色改进方法[J]. 临床与实验病理学杂志,2019,35(9):1122-1124. [26] 周迪, 陈章健, 胡贵平, 等. 纳米二氧化钛亚急性经口暴露对大鼠氧化/抗氧化生物标志和炎性因子的影响[J]. 北京大学学报(医学版),2020,52(5):821-827. [27] 王玉鑫, 袁梦淑, 田小荣, 等. 铁死亡在大鼠骨髓间充质干细胞减轻肝硬化中的作用[J]. 中华实验外科杂志,2022,39(10):1879-1883. [28] 李璐, 李玉萍, 陆娜, 等. 激动线粒体ALDH2抑制脓毒症小鼠肝损伤铁死亡机制的研究[J]. 中华危重病急救医学,2023,35(7):684-689. [29] WANG W, CHEN Z, ZHENG T, et al. Xanthohumol alleviates T2DM-induced liver steatosis and fibrosis by mediating the NRF2/RAGE/NF-κB signaling pathway. Future Med Chem. 2021;13(23):2069-2081. [30] GAO WY, CHEN PY, HSU HJ, et al. Xanthohumol, a prenylated chalcone, regulates lipid metabolism by modulating the LXRα/RXR-ANGPTL3-LPL axis in hepatic cell lines and high-fat diet-fed zebrafish models. Biomed Pharmacother. 2024;174:116598. [31] 张振刚, 王峰. 有氧运动联合补充D-核糖对小鼠抗疲劳和抗氧化的作用[J]. 食品工业科技,2022,43(2):368-375. [32] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060-1072. [33] FRIEDMANN ANGELI JP, SCHNEIDER M, PRONETH B, et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol. 2014;16(12):1180-1191. [34] TSURUSAKI S, TSUCHIYA Y, KOUMURA T, et al. Hepatic ferroptosis plays an important role as the trigger for initiating inflammation in nonalcoholic steatohepatitis. Cell Death Dis. 2019;10(6):449. [35] CARLSON BA, TOBE R, YEFREMOVA E, et al. Glutathione peroxidase 4 and vitamin E cooperatively prevent hepatocellular degeneration. Redox Biol. 2016;9:22-31. [36] QI J, KIM JW, ZHOU Z, et al. Ferroptosis affects the progression of nonalcoholic steatohepatitis via the modulation of lipid peroxidation-mediated cell death in mice. Am J Pathol. 2020;190(1):68-81. [37] GAO M, MONIAN P, QUADRI N, et al. Glutaminolysis and transferrin regulate ferroptosis. Mol Cell. 2015;59(2):298-308. [38] BOGDAN AR, MIYAZAWA M, HASHIMOTO K, et al. Regulators of iron homeostasis: new players in metabolism, cell death, and disease. Trends Biochem Sci. 2016; 41(3):274-286. [39] WANG YQ, CHANG SY, WU Q, et al. The Protective role of mitochondrial ferritin on erastin-induced ferroptosis. Front Aging Neurosci. 2016;8:308. [40] SALOMAO MA. Pathology of hepatic iron overload. Clin Liver Dis (Hoboken). 2021;17(4):232-237. [41] NELSON JE, WILSON L, BRUNT EM, et al. Relationship between the pattern of hepatic iron deposition and histological severity in nonalcoholic fatty liver disease. Hepatology. 2011;53(2):448-457. [42] VALENTI L, MOSCATIELLO S, VANNI E, et al. Venesection for non-alcoholic fatty liver disease unresponsive to lifestyle counselling-a propensity score-adjusted observational study. Qjm. 2011;104(2):141-149. [43] REISMAN SA, YEAGER RL, YAMAMOTO M, et al. Increased Nrf2 activation in livers from Keap1-knockdown mice increases expression of cytoprotective genes that detoxify electrophiles more than those that detoxify reactive oxygen species. Toxicol Sci. 2009;108(1):35-47. [44] OSBURN WO, WAKABAYASHI N, MISRA V, et al. Nrf2 regulates an adaptive response protecting against oxidative damage following diquat-mediated formation of superoxide anion. Arch Biochem Biophys. 2006;454(1):7-15. [45] SALAZAR M, ROJO AI, VELASCO D, et al. Glycogen synthase kinase-3beta inhibits the xenobiotic and antioxidant cell response by direct phosphorylation and nuclear exclusion of the transcription factor Nrf2. J Biol Chem. 2006; 281(21):14841-14851. [46] 房国梁, 赵杰修, 张漓, 等. 有氧运动对APP/PS1小鼠大脑皮质和海马组织Keap1/Nrf2信号通路的影响[J]. 中国运动医学杂志,2018,37(10):839-846. [47] HAN YM, YUN SM, LEE DY, et al. Targeting KEAP1-mediated IKKβ degradation strategy for colitis-associated colorectal carcinogenesis: The potential of xanthohumol. Biomed Pharmacother. 2025;184:117879. [48] LIU Y, WAN Y, JIANG Y, et al. GPX4: The hub of lipid oxidation, ferroptosis, disease and treatment. Biochim Biophys Acta Rev Cancer. 2023;1878(3):188890. [49] WANG L, LIU Y, DU T, et al. ATF3 promotes erastin-induced ferroptosis by suppressing system Xc. Cell Death Differ. 2020;27(2):662-675. [50] LONG Q, LI T, ZHU Q, et al. SuanZaoRen decoction alleviates neuronal loss, synaptic damage and ferroptosis of AD via activating DJ-1/Nrf2 signaling pathway. J Ethnopharmacol. 2024;323:117679. [51] SUN J, REN H, WANG J, et al. CHAC1: a master regulator of oxidative stress and ferroptosis in human diseases and cancers. Front Cell Dev Biol. 2024;12:1458716 |
| [1] | Lyu Guoqing, Aizimaitijiang·Rouzi, Xiong Daohai. Irisin inhibits ferroptosis in human articular chondrocytes: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1359-1367. |
| [2] | Zou Rongji, Yu Fangfang, Wang Maolin, Jia Zhuopeng. Triptolide inhibits ferroptosis and improves cerebral ischemia-reperfusion injury in a rat model of cerebral artery occlusion/reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 873-881. |
| [3] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [4] | Su Jiemao, Qi Yansong, Kong Keyu, Zhai Zanjing, Xu Yongsheng. Role of chondrocyte ferroptosis in the pathogenesis of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6282-6288. |
| [5] | Xu Dongfang, Zhao Kun, Lu Changzhu, Wang Yuge, Bai Lianjie, Meng Fanmou, Wang Yang, , Yao Hongbo. m6A-related ferroptosis gene expression and its association with immune infiltration in Alzheimer’s disease: machine learning and molecular biology validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6421-6432. |
| [6] | Zhou Wen, Yang Hongwei. Molecular mechanism and natural drug screening for ferroptosis-targeted therapy in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6051-6061. |
| [7] | Zheng Peng, Jia Xiaoning, Tao Jingwei, Fan Xiao. Tetramethylpyrazine improves iron metabolism disorders in a rat model of spinal cord injury via the Keap-1/Nrf2 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6134-6141. |
| [8] | Huang Yushan, Wang Rongrong, Li Xiangmiao, Bai Jinzhu. Prostaglandin E1 pretreatment inhibits ferroptosis in endothelial cells in a rat model of spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5716-5727. |
| [9] | Tao Xiangyu, Wang Shuang, Li Yuhan, Cao Jimin, Sun Teng. Effects of piRNA CFAPIR in doxorubicin-induced ferroptosis models of rat and human cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5756-5769. |
| [10] | Fan Longyu, Yuan Xiao, Xie Yanan, Yin Xiaoxuan . Action mechanism of mesenchymal stem cells and their derivatives in the treatment of liver fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4990-4999. |
| [11] | Liao Xingzhuan, Li Guangdi, Wu Yabin, Liu Xingyu, Wan Jiajia. Molecular mechanisms underlying non-coding RNA regulation of ferroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4713-4725. |
| [12] | Yu Le, Nan Songhua, Shi Zijian, He Qiqi, Li Zhenjia, Cui Yinglin. Mechanisms underlying mitophagy, ferroptosis, cuproptosis, and disulfidptosis in Parkinson’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4446-4456. |
| [13] | Niu Qi, Chen Junji, Tu Haining, Mo Weibin, Zhong Yujin, Li Mingliang. Effect of swimming exercise combined with probiotic intervention on anti-inflammatory and apoptotic gene expression in renal tissue of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4105-4114. |
| [14] | Chen Xinlong, Meng Tao, Wang Yaomin, Zhang Kefan, Li Jian, Shi Hui, Zhang Chenchen. Ferroptosis inhibitors in the treatment of osteoarthritis: diversity and multitarget characteristics [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4166-4179. |
| [15] | Liu Annan, Li Jianhui, Gao Wei, Li Xue, Song Jing, Xing Liping, Li Honglin. Bibliometric analysis of ferroptosis and Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4278-4288. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||