Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6282-6288.doi: 10.12307/2026.213
Previous Articles Next Articles
Role of chondrocyte ferroptosis in the pathogenesis of osteoarthritis
Su Jiemao1, 2, Qi Yansong2, Kong Keyu3, Zhai Zanjing3, Xu Yongsheng1, 2
- 1Inner Mongolia Clincal Medical College, Inner Mongolia Medical University, Hohhot 010017, Inner Mongolia Autonomous Region, China; 2Orthopedic Center (Sport Medicine Center), Inner Mongolia People’s Hospital, Hohhot 010017, Inner Mongolia Autonomous Region, China; 3Department of Orthopaedic Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China
-
Received:2025-08-04Revised:2025-09-10Online:2026-08-28Published:2026-02-03 -
Contact:Xu Yongsheng, Chief physician, Doctoral supervisor, Inner Mongolia Clincal Medical College, Inner Mongolia Medical University, Hohhot 010017, Inner Mongolia Autonomous Region, China; Orthopedic Center (Sport Medicine Center), Inner Mongolia People’s Hospital, Hohhot 010017, Inner Mongolia Autonomous Region, China Co-corresponding author: Qi Yansong, Associate chief physician, Associate researcher, Orthopedic Center (Sport Medicine Center), Inner Mongolia People's Hospital, Hohhot 010017, Inner Mongolia Autonomous Region, China -
About author:Su Jiemiao, MS candidate, Inner Mongolia Clincal Medical College, Inner Mongolia Medical University, Hohhot 010017, Inner Mongolia Autonomous Region, China; Orthopedic Center (Sport Medicine Center), Inner Mongolia People’s Hospital, Hohhot 010017, Inner Mongolia Autonomous Region, China -
Supported by:Inner Mongolia Autonomous Region Natural Science Foundation, No. 2024ZD32 (to XYS); Inner Mongolia Autonomous Region Natural Science Foundation, No. 2024LHMS08015 (to QYS); Science and Technology Project for the Construction of High-Level Clinical Specialties in Public Hospitals in the Capital Region of Inner Mongolia Autonomous Region, No. 2024SGGZ015 (to XYS)
CLC Number:
Cite this article
Su Jiemao, Qi Yansong, Kong Keyu, Zhai Zanjing, Xu Yongsheng. Role of chondrocyte ferroptosis in the pathogenesis of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6282-6288.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
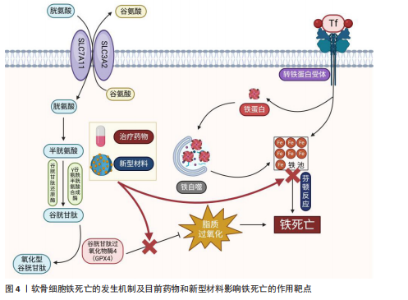
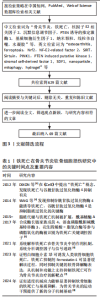
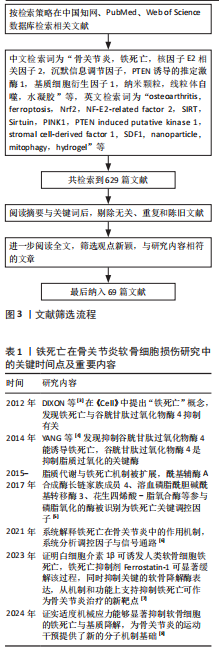
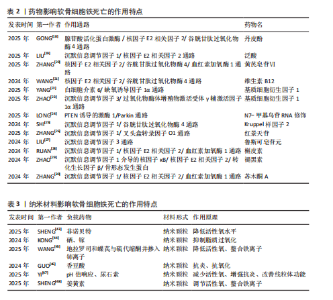
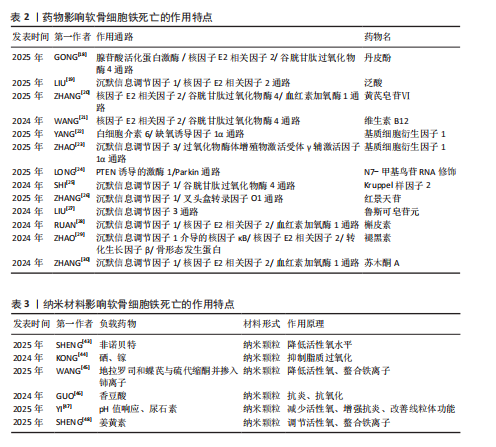
骨细胞迁移能力有限,受损后修复潜力不足,成为软骨再生的一大障碍。软骨细胞的能量主要依赖于糖酵解途径,区别于以有氧氧化为主的多数细胞类型,这种代谢模式虽适应低氧环境,但也使它在面对氧化应激时更为敏感。与此同时,软骨细胞中线粒体数量较少且功能不全,进一步限制了线粒体清除活性氧的能力,增加了细胞损伤风险。 2.2.2 细胞外基质的组成与生物力学作用 在关节软骨中,绝大多数体积被细胞外基质所占据,其中以Ⅱ型胶原蛋白为主,使软骨具备良好的抗拉强度,用来维持软骨良好的机械性能。蛋白多糖通过自身所携带的负电荷与水分子相结合,在基质中形成膨胀压,从而提高软骨组织的抗压能力,并赋予软骨一定的弹性[10]。当关节发生病变时,细胞外基质往往受到破坏,以基质金属蛋白酶13为代表的基质降解酶表达上调,显著加速细胞外基质的分解,这种降解不仅会削弱软骨的结构,还会引发一系列级联反应,如损伤相关分子模式的释放,损伤相关分子模式的积聚可激活局部免疫系统,诱导持续性炎症反应,进而形成炎症放大与基质破坏之间的正反馈机制,加剧软骨损伤的程度[11]。 2.3 铁死亡在软骨损伤中的作用机制 在骨关节炎进程中,软骨细胞功能紊乱与细胞数量减少是主要病理特征之一。在骨关节炎中,软骨细胞铁死亡被认为是导致软骨退变的关键病理机制之一[12]。研究发现,骨关节炎患者软骨组织中铁离子含量显著升高,铁过载可通过芬顿反应生成大量活性氧诱导脂质过氧化反应,从而触发铁死亡[13]。活性氧的过度积累不仅破坏细胞膜结构,还可损伤线粒体及核内DNA,进而削弱细胞抗氧化系统,加剧软骨细胞的功能障碍与死亡[14]。 在软骨细胞铁死亡的分子机制中,谷胱甘肽过氧化物酶4表达下调是关键环节[15]。谷胱甘肽过氧化物酶4是调控细胞膜脂质过氧化反应的核心酶,它的活性受损会导致膜磷脂中多不饱和脂肪酸发生氧化,最终破坏细胞膜的稳定性并引发细胞死亡。骨关节炎软骨组织中谷胱甘肽过氧化物酶4表达水平明显下降,与软骨细胞的退变密切相关;此外,谷胱甘肽耗竭也是铁死亡过程中的重要促发因素,系统Xc?(主要由SLC7A11亚基构成)负责转运胞外半胱氨酸进入细胞内参与谷胱甘肽合成[16],在骨关节炎软骨中该系统的活性受到抑制,导致谷胱甘肽合成能力下降、细胞抗氧化水平降低,进一步增强脂质过氧化反应,促进铁死亡的发生(图4)。 2.4 干扰软骨细胞铁死亡的药物研究 铁死亡是一种铁依赖性、由脂质过氧化驱动的程序性细胞死亡形式,核心特征包括线粒体形态异常(如嵴减少、膜密度增加)、抗氧化系统失衡(如谷胱甘肽耗竭)以及铁代谢紊乱。近年来关于铁死亡的研究逐渐增多,谷胱甘肽过氧化物酶4作为铁死亡的标志物成为诸多研究证明和验证铁死亡发生的关键蛋白,例如,牛磺酸能够通过谷胱甘肽过氧化物酶4发挥保护软骨的功能[17]。近年来,通过提取药物中的有效成分或开发新型材料来降低软骨细胞铁死亡程度,已成为大多数研究的主要切入点,并作为治疗软骨损伤的主要突破点,相关研究见表2。 随着对铁死亡机制研究的深入,一些传统药物被证实能够通过铁死亡途径对软骨细胞损伤产生治疗作用。XIE等[31]从骨关节炎患者的手术标本以及内侧半月板动物模型着手,通过免疫组织化学方法观察到在软骨损伤较为严重的患者以及小鼠模型软骨组织中,谷胱甘肽过氧化物酶4和PTEN蛋白激酶1的表达水平有所下降;研究选择JP4-039作为治疗药物,发现该药物能够有效促进PTEN蛋白激酶1的表达,从而促进线粒体自噬作用,进而抑制软骨细胞的铁死亡现象,显示出JP4-039有潜力成为治疗软骨损伤的有效药物之一。JP4-039作为一种线粒体靶向抗氧化剂,在多种疾病的铁死亡相关机制研究中显示出显著的干预效果[32-34]。 在帕金森病研究的过程中,PTEN蛋白激酶1/Parkin通路被首次发现,经过近30年的科学研究,该通路在多种骨科疾病中的重要性得到了广泛认可。PTEN蛋白激酶1与铁死亡均与细胞内的氧化还原反应密切相关,激活PTEN蛋白激酶1通路能够有效抑制铁死亡,减轻软骨细胞损伤,为相关疾病的治疗提供了新的策略。研究表明,提高PTEN蛋白激酶1基因表达可以促进受损线粒体的自噬作用,进而发挥预防骨关节炎[35]、2型糖尿病引起的骨质损伤[36]、糖皮质激素导致的成骨细胞凋亡并促进成骨作用[37-38],缓解椎间盘退变。 铁死亡作为各类疾病研究的热门细胞死亡方式,它在软骨损伤中的分子机制日益受到重视。在相关信号通路中,核因子E2相关因子2在抗氧化防御中发挥了关键作用,研究人员就该通路与铁死亡之间的相关性进行了深入研究。核因子E2相关因子2作为细胞应对氧化应激的核心调控因子,能够调节多种与细胞周期密切相关的下游基因表达减轻由氧化损伤诱导的细胞功能障碍[39]。早期研究主要聚焦于合成药物对核因子E2相关因子2的调控,而近年的研究发现,来自中草药和天然植物来源的活性成分同样具有良好的生物活性,特别是在抑制铁死亡、保护软骨细胞方面表现出积极效果。GONG等[18]报道,紫草中的主要成分丹皮酚可上调核因子E2相关因子2表达水平抑制铁死亡在软骨细胞中的发生。LIU等[19]的研究显示,天然抗氧化剂泛酸通过激活核因子E2相关因子2通路显著减轻骨关节炎所致的软骨细胞损伤程度。ZHANG等[20]发现,黄芪皂苷Ⅵ能够通过调控核因子E2相关因子2信号通路来改善软骨组织的退变。另有研究表明,长春新碱具有调节核因子E2相关因子2活性的作用,能够在体内外模型中起到一定的软骨保护效果[21]。上述研究大多基于细胞实验和动物模型,系统验证了核因子E2相关因子2在软骨细胞铁死亡调控中的核心地位。总体来看,核因子E2相关因子2作为潜在的干预靶点,已显示出在减缓软骨退变、干预骨关节炎进程方面的重要作用,为后续开发以该通路为基础的治疗策略奠定了坚实的理论基础。 基质细胞衍生因子1在骨和软骨代谢中发挥着重要作用,能够指导间充质干细胞的迁移,促进骨和软骨生成,并改变炎性环境;与此相反,基质细胞衍生因子1还能在炎性环境中充当炎性趋化因子,引起骨与软骨变性,导致骨关节炎。由于基质细胞衍生因子1对软骨具有双向影响,不同的研究针对基质细胞衍生因子不同的作用方向进行深入探索,也得出不同的结论。YANG等[22]发现基质细胞衍生因子1能够通过白细胞介素6/缺氧诱导因子1α通路促进软骨细胞铁死亡,引发骨关节炎。ZHAO等[23]发现基质细胞衍生因子1可以通过沉默信息调节因子3/过氧化物酶体增殖物激活受体γ辅激活因子1α通路改善线粒体功能障碍,进一步减轻骨关节炎。在受到异常机械应力时,基质细胞衍生因子1/CXC趋化因子受体4被异常激活,提高细胞表面受体结合效率,导致软骨细胞受到各种炎症因子的损害[40]。也有研究在软骨损伤部位检测到基质细胞衍生因子1表达显著增加,但是在异常应力区域并未发现这些改变[41]。相比于全身用药,关节腔内局部注射基质细胞衍生因子1过表达的软骨源性干细胞能够有效促进兔半月板愈合,预防骨关节炎的发生[42]。与基质细胞衍生因子1类似,对软骨细胞具有双向作用的还有非类固醇抗炎药类药物、机械刺激、血管生成因子、Wnt通路等。 沉默信息调节因子是近期研究软骨细胞铁死亡、铜死亡和焦亡的又一热门靶点,作为一种依赖NAD+的去乙酰化酶蛋白家族,参与氧化应激、调节炎症代谢,骨科研究人员将目光集中于沉默信息调节因子的上下游通路。DENG等[35]通过对线粒体自噬的研究发现,沉默信息调节因子通过使PTEN蛋白激酶1脱乙酰化增强线粒体自噬,从而发挥修复软骨的功能。LONG等[24]通过研究相同的通路和靶点得出类似的结论,N7-甲基鸟苷修饰RNA也是治疗软骨缺损的有效手段之一。Kruppel样因子2通过作用于沉默信息调节因子1来抑制软骨细胞铁死亡的发生,有效改善线粒体功能[25]。随着研究的深入,传统中医药也逐渐成为治疗软骨损伤的重要方式之一。ZHANG等[26]从中药提取红景天苷并对其抗炎能力和改善关节炎症功能进行研究,发现红景天苷能够干预沉默信息调节因子影响软骨细胞铁死亡,改善软骨损伤。鲁斯可皂苷元作为改善血脑屏障功能障碍药物,已被证实能够通过作用于沉默信息调节因子影响软骨细胞铁死亡,进而减轻软骨损伤程度[27]。槲皮素[28]、褪黑素[29]、苏木酮A作为中药中的重要天然化合物[30],已被证实能够通过沉默信息调节因子影响软骨细胞铁死亡。 2.5 干扰软骨细胞铁死亡的材料研究 骨科领域广泛涉及各类材料的临床应用,如骨折内固定装置、人工关节材料以及用于骨缺损修复的生物材料等,随着材料性能的提升,使得治疗的安全性与有效性也显著提升。近年来,随着对软骨损伤铁死亡机制认识的不断加深,材料学在调控软骨细胞铁死亡、减缓软骨退变方面的研究逐渐受到重视,目前相关研究正尝试通过构建具有良好生物相容性、稳定性和靶向释放能力的功能性材料,为软骨损伤的治疗提供新的技术支持。材料的改良不仅有望提高治疗效率,还可以在一定程度上实现缓解患者症状、延缓软骨损伤进展并实现功能恢复的目的,因此,骨科学与材料学之间的密切联系,对推动治疗手段创新、提升临床疗效具有重要意义。纳米材料影响软骨细胞铁死亡的作用特点,见表3。 软骨细胞引发铁死亡的始动因素就是氧化代谢反应失衡,细胞内活性氧累积导致软骨细胞铁死亡的发生。针对活性氧累积,研究人员有针对性地开发抗氧化材料,减少软骨细胞铁死亡的发生。例如,SHENG等[43]合成了一种负载对活性氧敏感的非诺贝特纳米颗粒,该颗粒能够有效降低软骨细胞内活性氧水平,进而发挥保护软骨细胞的作用。KONG团队[44]发现骨关节炎样本中缺失硒元素和镓元素,并以此为基础定制富含硒、镓的抗氧化纳米颗粒,证"
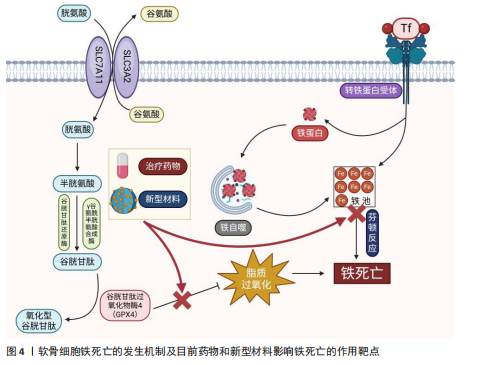
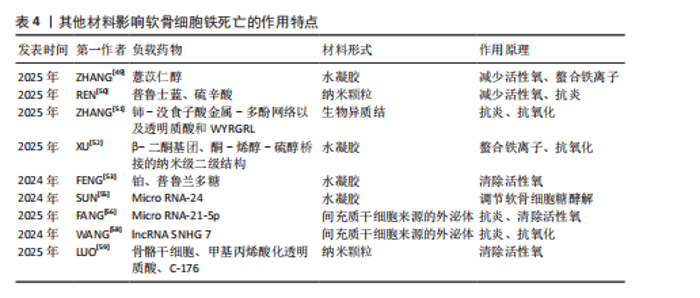
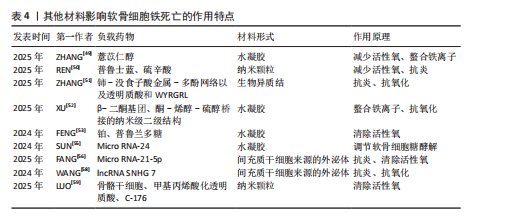
明该纳米颗粒可通过抑制铁死亡来改善软骨细胞状态,减少软骨损伤。WANG等[45]开发了一种活性氧响应性纳米颗粒对铁死亡进行多靶点治疗。GUO等[46]研发了聚香豆酸纳米颗粒,利用它的抗氧化特性通过抑制软骨细胞铁死亡治疗软骨损伤。YI等[47]开发了一种pH值响应脂质纳米粒并负载尿素石,可以有效减少软骨细胞内的活性氧聚集、促进巨噬细胞向M2极化、有效改善线粒体结构、抑制软骨细胞铁死亡,为治疗软骨损伤提供更有效的方案。SHENG等[48]研发一种负载姜黄素的纳米颗粒,通过清除软骨细胞内活性氧和避免脂质过氧化来发挥保护软骨细胞的功能。 近年来,多项研究正在尝试通过构建具有抗氧化性能或调控金属离子分布的材料减缓骨关节炎所致的软骨损伤并改善疾病进程。例如,ZHANG等[49]构建了一种载有薏苡醇的明胶水凝胶,该材料在体内能够持续释放药物,通过降低软骨细胞内氧化应激水平有效抑制铁死亡的发生,起到保护软骨的作用。REN等[50] 联合应用中空普鲁士蓝和硫辛酸合成了一种具备抗氧化能力的新型复合物,发现该水凝胶可显著干预软骨细胞铁死亡过程、改善膝关节局部微环境,减轻软骨损伤。此外,ZHANG等[51]利用自组装技术合成了一种具有生物异质结构的材料,该材料具备多种抗氧化酶样活性,包括类超氧化物歧化酶、类过氧化氢酶和谷胱甘肽过氧化物酶,可有效降低细胞内活性氧水平,实现阻断铁死亡诱导路径的目的。XU等[52]研发的铁离子螯合型水凝胶,通过螯合铁离子来限制铁离子积聚以抑制相关氧化反应,从而延缓骨关节炎所致的软骨细胞损伤。FENG等[53]设计的含铂材料能够模拟抗氧化酶的性能,在消除活性氧的同时对软骨细胞形成一定程度的保护。总体而言,当前相关研究多聚焦于研发具备抗氧化或调控铁稳态功能的材料,尝试通过改善软骨细胞的胞内环境来抑制铁死亡的发生,为骨关节炎治疗提供了新的研究方向(图4)。 除合成抗氧化材料外,研制通过其他途径干扰铁死亡的材料也是目前研究骨关节炎治疗的重点。干细胞是再生疗法最理想的选择之一,备受临床医师和科研工作者的青睐。相比于干细胞治疗,干细胞来源外泌体从伦理审查和储存条件来说具有更显著的优势,自2003年获得诺贝尔奖后逐渐成为科研工作者的研究重点[54],2024年再一次获得诺贝尔奖,充分证明干细胞外泌体的发展前景广阔。SUN等[55]合成一种负载miR-24的水凝胶,通过影响软骨细胞铁死亡修复受损软骨。FANG等[56]通过提取间充质干细胞来源的外泌体miR-21-5p来干扰铁死亡。MA等[57]发现,miR-140-3p能够通过软骨中间层蛋白调节软骨细胞铁死亡,减轻炎症水平和发挥抗氧化功能。miR-485-5p能够抑制白细胞介素1β诱导的软骨细胞铁死亡[58]。除外泌体外,通过信号通路进行免疫治疗也是目前研究的热点之一。干扰素基因刺激因子通路作为影响细胞周期的关键蛋白之一,在多种疾病都进行过深入研究。LUO等[59]研发涂有C-176的聚多巴胺纳米颗粒,该颗粒通过抑制环磷酸鸟苷-腺苷合成酶-干扰素基因刺激因子通路来避免细胞内活性氧积累,从而减少铁死亡的发生(图4)。 在现有研究中,多种抗氧化纳米材料、多功能水凝胶及干细胞来源外泌体在缓解软骨细胞铁死亡方面均表现出积极的干预效果,显现出良好的临床应用前景,然而,各种材料的生物安全性仍有待进一步评估,例如,部分材料如明胶基水凝胶、中空普鲁士蓝等具有较好的天然来源和生物相容性,为后续转化研究提供了可靠基础;但是,某些纳米颗粒在体内可能存在诱发免疫反应或因无法有效代谢而发生长期滞留的问题,而且纳米材料相关代谢途径及毒性分布尚不明确。与纳米材料相比,干细胞外泌体因低免疫原性及较强的生物适应性在安全性方面更具优势。总体来看,相关材料仍处于实验研究阶段,未来应注重它们的长期毒理效应及组织相容性,以推动它们在骨关节炎治疗中的安全高效应用。其他材料影响软骨细胞铁死亡的作用特点,见表4。 2.6 铁死亡与其他细胞死亡方式的交叉机制 铁死亡作为一种热门研究的细胞死亡方式,在多种病理状态中均扮演着重要角色,并且与传统的细胞死亡形式如凋亡、坏死以及自噬等存在广泛的交叉和联系。铁死亡与凋亡和坏死之间均涉及活性氧的异常积累[60],细胞内活性氧水平的升高可引发一系列损伤反应,如线粒体功能紊乱[61]、膜脂质的过氧化、DNA链断裂及蛋白质结构改变[62],从而激活多个细胞死亡相关的信号通路,最终导致软骨细胞死亡和软骨组织损伤;与此同时,铁蛋白自噬会促进铁离子释放,造成细胞内铁负荷升高,进一步加剧软骨细胞内部的脂质过氧化反应,是诱导铁死亡的重要机制之一[63]。此外,谷胱甘肽和谷胱甘肽过氧化物酶 4作为细胞内抗氧化系统的关键组成,参与清除过氧化脂质,它们的表达或活性下降不仅可以直接触发铁死亡,也会增强其他类型细胞死亡形式的敏感性[64]。这些机制之间的协同与交错,为深入理解细胞命运调控提供了新的视角。"
| [1] GLYN-JONES S, PALMER AJ, AGRICOLA R, et al. Osteoarthritis. Lancet. 2015;386(9991):376-387. [2] LI D, LI S, CHEN Q, et al. The Prevalence of Symptomatic Knee Osteoarthritis in Relation to Age, Sex, Area, Region, and Body Mass Index in China: A Systematic Review and Meta-Analysis. Front Med (Lausanne). 2020;7:304. [3] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060-1072. [4] YANG WS, SRIRAMARATNAM R, WELSCH ME, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156(1-2):317-331. [5] XIE Y, HOU W, SONG X, et al. Ferroptosis: process and function. Cell Death Differ. 2016; 23(3):369-379. [6] LU S, LIU Z, QI M, et al. Ferroptosis and its role in osteoarthritis: mechanisms, biomarkers, and therapeutic perspectives. Front Cell Dev Biol. 2024;12:1510390. [7] XU W, ZHANG B, XI C, et al. Ferroptosis Plays a Role in Human Chondrocyte of Osteoarthritis Induced by IL-1β In Vitro. Cartilage. 2023;14(4):455-466. [8] HAN J, ZHAN LN, HUANG Y, et al. Moderate mechanical stress suppresses chondrocyte ferroptosis in osteoarthritis by regulating NF-κB p65/GPX4 signaling pathway. Sci Rep. 2024; 14(1):5078. [9] DÖNGES L, DAMLE A, MAINARDI A, et al. Engineered human osteoarthritic cartilage organoids. Biomaterials. 2024;308:122549. [10] CORREIA CR, REIS RL, MANO JF. Multiphasic, Multistructured and Hierarchical Strategies for Cartilage Regeneration. Adv Exp Med Biol. 2015;881:143-160. [11] MEI J, XIAO N, XI Y, et al. Regulation of apoptosis and interaction with cartilage degeneration in osteoarthritis. Front Cell Dev Biol. 2025;13: 1571448. [12] SUN K, GUO Z, HOU L, et al. Iron homeostasis in arthropathies: From pathogenesis to therapeutic potential. Ageing Res Rev. 2021;72:101481. [13] WANG X, PAN L, NIU D, et al. Jingfang Granules alleviates the lipid peroxidation induced ferroptosis in rheumatoid arthritis rats by regulating gut microbiota and metabolism of short chain fatty acids. J Ethnopharmacol. 2025;339:119160. [14] YAO X, SUN K, YU S, et al. Chondrocyte ferroptosis contribute to the progression of osteoarthritis. J Orthop Translat. 2021;27: 33-43. [15] MIAO Y, CHEN Y, XUE F, et al. Contribution of ferroptosis and GPX4’s dual functions to osteoarthritis progression. EBioMedicine. 2022; 76:103847. [16] RUAN Q, WANG C, ZHANG Y, et al. Brevilin A attenuates cartilage destruction in osteoarthritis mouse model by inhibiting inflammation and ferroptosis via SIRT1/Nrf2/GPX4 signaling pathway. Int Immunopharmacol. 2023;124(Pt B): 110924. [17] ZHOU X, YANG Y, QIU X, et al. Antioxidant taurine inhibits chondrocyte ferroptosis through upregulation of OGT/Gpx4 signaling in osteoarthritis induced by anterior cruciate ligament transection. J Adv Res. 2025:S2090-1232(25)00029-3. doi: 10.1016/j.jare.2025.01.010. [18] GONG S, LANG S, JIANG X, et al. Paeonol ameliorates ferroptosis and inflammation in chondrocytes through AMPK/Nrf2/GPX4 pathway. Front Pharmacol. 2025;16:1526623. [19] LIU Y, WANG Y, CHENG S, et al. Pantothenic acid alleviates osteoarthritis progression by inhibiting inflammatory response and ferroptosis through the SIRT1/Nrf2 signaling pathway. Chem Biol Interact. 2025;413:111494. [20] ZHANG Z, YUAN D, JIN X, et al. Asperosaponin VI suppresses ferroptosis in chondrocytes and ameliorates osteoarthritis by modulating the Nrf2/GPX4/HO-1 signaling pathway. Front Pharmacol. 2025;16:1539092. [21] WANG J, YANG J, FANG Y, et al. Vinpocetine protects against osteoarthritis by inhibiting ferroptosis and extracellular matrix degradation via activation of the Nrf2/GPX4 pathway. Phytomedicine. 2024;135:156115. [22] YANG T, YANG X, WANG G, et al. Unraveling the crucial role of SDF-1 in osteoarthritis progression: IL6/HIF-1α positive feedback and chondrocyte ferroptosis. Int Immunopharmacol. 2025;152:114400. [23] ZHAO Y, LIN D, ZHU X, et al. SDF-1 alleviates osteoarthritis by resolving mitochondrial dysfunction through the activation of the Sirt3/PGC-1α signalling pathway. Arthritis Res Ther. 2025;27(1):51. [24] LONG D, DENG Z, ZHAO X, et al. m(7)G-modified mt-tRF3b-LeuTAA regulates mitophagy and metabolic reprogramming via SUMOylation of SIRT3 in chondrocytes. Biomaterials. 2025;314: 122903. [25] SHI J, CHEN L, WANG X, et al. KLF2 Inhibits Ferroptosis and Improves Mitochondrial Dysfunction in Chondrocyte Through SIRT1/GPX4 Signaling to Improve Osteoarthritis. Drug Dev Res. 2024;85(7):e70015. [26] ZHANG X, HUANG L, FENG W, et al. Salidroside ameliorates lipopolysaccharide‑induced ferroptosis in chondrocytes via regulation of the sirt1/foxo1 axis. Mol Med Rep. 2025;31(5):138. [27] LIU Y, LI W, TANG H, et al. Ruscogenin attenuates osteoarthritis by modulating oxidative stress-mediated macrophage reprogramming via directly targeting Sirt3. Int Immunopharmacol. 2024;143(Pt 1): 113336. [28] RUAN H, ZHU T, WANG T, et al. Quercetin Modulates Ferroptosis via the SIRT1/Nrf-2/HO-1 Pathway and Attenuates Cartilage Destruction in an Osteoarthritis Rat Model. Int J Mol Sci. 2024;25(13):7461. [29] ZHAO M, QIU D, MIAO X, et al. Melatonin Delays Arthritis Inflammation and Reduces Cartilage Matrix Degradation through the SIRT1-Mediated NF-κB/Nrf2/TGF-β/BMPs Pathway. Int J Mol Sci. 2024;25(11):6202. [30] ZHANG Z, ZHANG N, LI M, et al. Sappanone a alleviates osteoarthritis progression by inhibiting chondrocyte ferroptosis via activating the SIRT1/Nrf2 signaling pathway. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(11):8759-8770. [31] XIE Y, LV Z, LI W, et al. JP4-039 protects chondrocytes from ferroptosis to attenuate osteoarthritis progression by promoting Pink1/Parkin-dependent mitophagy. J Orthop Translat. 2025;51:132-144. [32] AIRIK M, CLAYTON K, WIPF P, et al. JP4-039 Mitigates Cisplatin-Induced Acute Kidney Injury by Inhibiting Oxidative Stress and Blocking Apoptosis and Ferroptosis in Mice. Antioxidants (Basel). 2024;13(12);13(12):1534. [33] WU YL, CHRISTODOULOU AG, BEUMER JH, et al. Mitigation of Fetal Radiation Injury from Mid-Gestation Total-body Irradiation by Maternal Administration of Mitochondrial-Targeted GS-Nitroxide JP4-039. Radiat Res. 2024;202(3):565-579. [34] ADEGHATE JO, EPPERLY MW, DAVOLI KA, et al. JP4-039, a Mitochondria-Targeted Nitroxide, Mitigates the Effect of Apoptosis and Inflammatory Cell Migration in the Irradiated Mouse Retina. Int J Mol Sci. 2024;25(12);25(12):6515. [35] DENG Y, HOU M, WU Y, et al. SIRT3-PINK1-PKM2 axis prevents osteoarthritis via mitochondrial renewal and metabolic switch. Bone Res. 2025; 13(1):36. [36] XIAN Y, LIU B, SHEN T, et al. Enhanced SIRT3 expression restores mitochondrial quality control mechanism to reverse osteogenic impairment in type 2 diabetes mellitus. Bone Res. 2025;13(1):30. [37] KHANKA S, RASTOGI SK, SINGH KB, et al. Pym-18a, a novel pyrimidine derivative ameliorates glucocorticoid induced osteoblast apoptosis and promotes osteogenesis via autophagy and PINK 1/Parkin mediated mitophagy induction. Biochem Pharmacol. 2025;233:116751. [38] ZHANG Y, XING D, LIU Y, et al. CREG1 attenuates intervertebral disc degeneration by alleviating nucleus pulposus cell pyroptosis via the PINK1/Parkin-related mitophagy pathway. Int Immunopharmacol. 2025;147:113974. [39] HE F, RU X, WEN T. NRF2, a Transcription Factor for Stress Response and Beyond. Int J Mol Sci. 2020;21(13): 4777. [40] 匡斌,王庆昱,宋容,等. 基质细胞衍生因子-1刺激对周期性牵张力作用下ATDC5细胞的趋化因子受体4、白介素-6、胶原X表达的影响[J].华西口腔医学杂志,2014,32(6):592-595. [41] RAPP AE, BINDL R, HEILMANN A, et al. Systemic mesenchymal stem cell administration enhances bone formation in fracture repair but not load-induced bone formation. Eur Cell Mater. 2015;29:22-34. [42] TRIVEDI J, DESAI S, MOLINO J, et al. Intra-articular Injections of CXCR4-Overexpressing Human Cartilage-Derived Progenitor Cells Improve Meniscus Healing and Protect Against Posttraumatic Osteoarthritis in Immunocompetent Rabbits. Am J Sports Med. 2025;53(2):396-405. [43] SHENG WB, LIAO S, CAO SY, et al. Reactive oxygen species-sensitive fenofibrate-loaded dextran nanoparticles in alleviation of osteoarthritis. Carbohydrate Polymers. 2025;347:122768. [44] KONG KY, YANG YQ, CHANG YY, et al. A Ubiquitin-Competitive Strategy Based on the Element Microenvironment to Treat Osteoarthritis. Advanced Functional Materials. 2024;34(51):2409707. [45] WANG D, PAN YL, CHEN WZ, et al. Nanodrugs Targeting Key Factors of Ferroptosis Regulation for Enhanced Treatment of Osteoarthritis. Adv Sci (Weinh). 2025;12(11):e2412817. [46] GUO JX, SU K, WANG LY, et al. Poly(p-coumaric acid) nanoparticles alleviate temporomandibular joint osteoarthritis by inhibiting chondrocyte ferroptosis. Bioact Mater. 2024;40:212-226. [47] YI GL, LI M, ZHOU JY, et al. Novel pH-responsive lipid nanoparticles deliver UA-mediated mitophagy and ferroptosis for osteoarthritis treatment. Materials Today Bio. 2025;32:101697 [48] SHENG W, LI A, YUE Y, et al. A Novel Curcumin-Loaded Nanoplatform Alleviates Osteoarthritis by Inhibiting Chondrocyte Ferroptosis. Macromol Rapid Commun. 2025;46(7):e2400495. [49] ZHANG L, ZHENG G, ZHAO W, et al. Coixol-Loaded Hydrogels Promote Osteochondral Defect Repair via Modulation of Ferroptosis and Autophagy in Chondrocytes. ACS Biomater Sci Eng. 2025; 11(2):1096-1105. [50] REN DZ, LIU MJ, CAO M, et al. HPB@LA@PDA nanoplatform ameliorates osteoarthritis by scavenging reactive oxygen species and remodelling the inflammatory microenvironment: An in vitro and in vivo study. Chem Eng J. 2025; 507:160592. [51] ZHANG Z, CHENG DB, LIU D, et al. Bioengineered Versatile Heterojunctions as Stress Busters Targeting Matrix Degradation and Ferroptosis for Osteoarthritis Therapy. Adv Funct Mater. 2025;35(16):2419400. [52] XU Y, GU X, LI X, et al. β-Diketone Functionalized Microspheres Chelate Reactive Iron via Metal Coordination for Cartilage Repair. Adv Healthc Mater. 2025;14(10):e2403933. [53] FENG JW, DENG X, HAO P, et al. Intra-articular injection of platinum nanozyme-loaded silk fibroin/pullulan hydrogels relieves osteoarthritis through ROS scavenging and ferroptosis suppression.Int J Biol Macromol. 2024;280(Pt 2): 135863. [54] SU J, QI Y, NIU L, et al. The role of synovial mesenchymal stem cell-derived exosomes in cartilage repair: a systematic review. Frontiers in Pharmacology. 2025;16:1617874.. [55] SUN Y, YOU YQ, WU Q, et al. Senescence-targeted MicroRNA/Organoid composite hydrogel repair cartilage defect and prevention joint degeneration via improved chondrocyte homeostasis. Bioact Mater. 2024;39:427-442. [56] FANG MJ, WANG YX, LI ZY, et al. Decidual mesenchymal stem cells derived extracellular vesicles regulate ferroptosis to alleviate intervertebral disc degeneration through the HIF-1α signaling pathway. Chemical Engineering Journal. 2025;509:161357. [57] MA F, WANG L, CHI H, et al. Exploring the Therapeutic Potential of MIR-140-3p in Osteoarthritis: Targeting CILP and Ferroptosis for Novel Treatment Strategies. Cell Prolif. 2025; 58(11):e70018. [58] WANG Y, HU K, LIAO C, et al. Exosomes-Shuttled lncRNA SNHG7 by Bone Marrow Mesenchymal Stem Cells Alleviates Osteoarthritis Through Targeting miR-485-5p/FSP1 Axis-Mediated Chondrocytes Ferroptosis and Inflammation. Tissue Eng Regen Med. 2024;21(8):1203-1216. [59] LUO LW, ZHANG SY, GONG JF, et al. 3-D Sustained-Release Culture Carrier Alleviates Rat Intervertebral Disc Degeneration by Targeting STING in Transplanted Skeletal Stem Cells. Adv Sci (Weinh). 2025;12:2410151. [60] WAWRZEŃCZYK A, NAPIÓRKOWSKA-BARAN K, ALSKA E, et al. Ferroptosis in Gastrointestinal Diseases: A New Frontier in Pathogenesis and Therapy. J Clin Med. 2025;14(12):4035. [61] CHU T, HUANG Z, MA W. Mitophagy: A double-edged sword in tumor cell death regulation and therapeutic response. Biochem Biophys Res Commun. 2025;777:152254. [62] MA C, WANG J, CUI S, et al. Crotonoside induces ferroptosis and mitochondrial dysfunction in AML. Eur J Pharmacol. 2025:1002:177859. [63] ASCHNER M, SKALNY AV, RONGZHU L, et al. The Role of Epigenetics in Manganese Neurotoxicity: An Update with a Focus on Non-Coding RNAs and Histone Modifications. Neurochem Res. 2025;50(3):195. [64] XU J, LI Y, PAN F, et al. BAOXIN Granules Reverse Ferroptosis Through the Solute Carrier Family 7 Member -Glutathione-Glutathione Peroxidase 4 Axis in Ischemia-Induced Heart Failure in Rats. J Ethnopharmacol. 2025;352:120119. [65] ZHOU X, PAN Y, LI J, et al. Notopterol mitigates osteoarthritis progression and relieves pain in mice by inhibiting PI3K/Akt/GPX4-mediated ferroptosis. Int Immunopharmacol. 2025;151:114323. [66] CAO S, WEI Y, XIONG A, et al. Paeonol inhibits ACSL4 to protect chondrocytes from ferroptosis and ameliorates osteoarthritis progression. J Orthop Translat. 2025;50:1-13. [67] GONG Z, WANG Y, LI L, et al. Cardamonin alleviates chondrocytes inflammation and cartilage degradation of osteoarthritis by inhibiting ferroptosis via p53 pathway. Food Chem Toxicol. 2023;174:113644. [68] YANG J, CHEN D, HE Q, et al. Arctiin alleviates knee osteoarthritis by suppressing chondrocyte oxidative stress induced by accumulated iron via AKT/NRF2/HO-1 signaling pathway. Sci Rep. 2024;14(1):31935. [69] XU J, ZHI X, ZHANG Y, et al. Tanshinone IIA alleviates chondrocyte apoptosis and extracellular matrix degeneration by inhibiting ferroptosis. Open Life Sci. 2023;18(1):20220666. |
| [1] | Liu Dawei, Cui Yingying, Wang Fanghui, Wang Zixuan, Chen Yuhan, Li Yourui, Zhang Ronghe. Epigallocatechin gallate-mediated bidirectional regulation of reactive oxygen species and its application in nanomaterials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2101-2112. |
| [2] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [3] | Wang Songpeng, Liu Yusan, Yu Huanying, Gao Xiaoli, Xu Yingjiang, Zhang Xiaoming, Liu Min. Bidirectional regulation of reactive oxygen species based on zeolitic imidazolate framework-8 nanomaterials: from tumor therapy and antibacterial activity to cytoprotection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2033-2013. |
| [4] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [5] | Wen Guangwei, Zhen Yinghao, Zheng Taikeng, Zhou Shuyi, Mo Guoye, Zhou Tengpeng, Li Haishan, Lai Yiyi. Effects and mechanisms of isoginkgetin on osteoclastogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1348-1358. |
| [6] | Lyu Guoqing, Aizimaitijiang·Rouzi, Xiong Daohai. Irisin inhibits ferroptosis in human articular chondrocytes: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1359-1367. |
| [7] | Li Hao, Tao Hongcheng, Zeng Ping, Liu Jinfu, Ding Qiang, Niu Chicheng, Huang Kai, Kang Hongyu. Mitogen-activated protein kinase signaling pathway regulates the development of osteoarthritis: guiding targeted therapy with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1476-1485. |
| [8] | Zhang Qian, Huang Dongfeng. Weighted gene co-expression network analysis combined with machine learning to screen and validate biomarkers for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1096-1105. |
| [9] | Yin Yongcheng, Zhao Xiangrui, Yang Zhijie, Li Zheng, Li Fang, Ning Bin. Effect and mechanism of peroxiredoxin 1 in microglial inflammation after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1106-1113. |
| [10] | Hu Jing, Zhu Ling, Xie Juan, Kong Deying, Liu Doudou. Autophagy regulates early embryonic development in mice via affecting H3K4me3 modification [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1147-1155. |
| [11] | Bu Yangyang, Ning Xinli, Zhao Chen. Intra-articular injections for the treatment of osteoarthritis of the temporomandibular joint: different drugs with multiple combined treatment options [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1215-1224. |
| [12] | Zou Rongji, Yu Fangfang, Wang Maolin, Jia Zhuopeng. Triptolide inhibits ferroptosis and improves cerebral ischemia-reperfusion injury in a rat model of cerebral artery occlusion/reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 873-881. |
| [13] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [14] | Xu Dongfang, Zhao Kun, Lu Changzhu, Wang Yuge, Bai Lianjie, Meng Fanmou, Wang Yang, , Yao Hongbo. m6A-related ferroptosis gene expression and its association with immune infiltration in Alzheimer’s disease: machine learning and molecular biology validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6421-6432. |
| [15] | Zhou Wen, Yang Hongwei. Molecular mechanism and natural drug screening for ferroptosis-targeted therapy in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6051-6061. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||