Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (36): 5884-5890.doi: 10.12307/2023.720
Previous Articles Next Articles
Roles of ferroptosis in intervertebral disc degeneration and osteoarthritis
Xiong Zhilin, Sun Hong, Liu Miao, Zhuang Yong
- Department of Orthopedics, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2022-10-14Accepted:2022-11-17Online:2023-12-28Published:2023-03-27 -
Contact:Zhuang Yong, Master’s supervisor, Associate chief physician, Department of Orthopedics, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Xiong Zhilin, Master candidate, Department of Orthopedics, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:Plan of Guiyang Science and Technology Bureau, No. [2018]1-78 (to ZY); Science and Technology Fund of Guizhou Health Commission, No. gzwjkj2020-1-120 (to SH); Youth Fund Cultivation Program of National Natural Science Foundation of Affiliated Hospital of Guizhou Medical University, No. gyfynsfc-2021-12 (to SH); the Project of Guizhou Postgraduate Scientific Research Fund, No. YJSKYJJ[2021]157 (to SH)
CLC Number:
Cite this article
Xiong Zhilin, Sun Hong, Liu Miao, Zhuang Yong. Roles of ferroptosis in intervertebral disc degeneration and osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5884-5890.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
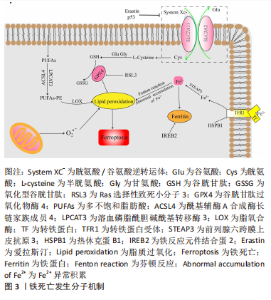
2.1 铁死亡概述及发生机制 细胞死亡在生物体生命活动中扮演至关重要的角色。细胞死亡根据是否受到自身基因调控分为调控性细胞死亡和偶发性细胞死亡[21],其中调控性细胞死亡主要包括凋亡、焦亡和自噬等。随着对细胞死亡机制的深入研究,2012年DIXON等[22]将铁离子依赖性PUFAs过氧化物蓄积引起细胞死亡的方式命名为铁死亡。铁死亡也属于调控性细胞死亡,但在形态学表现和生化特征上区别于其他类型的调控性细胞死亡。铁死亡形态学特征主要表现包括线粒体体积缩小、嵴模糊不清、外膜破裂、膜密度增加,细胞核膜完整[23]。在生化特征上表现为胱氨酸/谷氨酸逆转运体(cystine/glutamate antiporter system,System Xc-)被抑制、谷胱甘肽合成下降、谷胱甘肽过氧化物酶4(glutathione peroxidase 4,GPX4)活性下降、铁离子和PUFAs过氧化物蓄积。随着对铁死亡机制的深入研究,发现其发生与铁离子异常积蓄、PUFAs过氧化和氨基酸异常代谢等因素密切相关,见图3。"

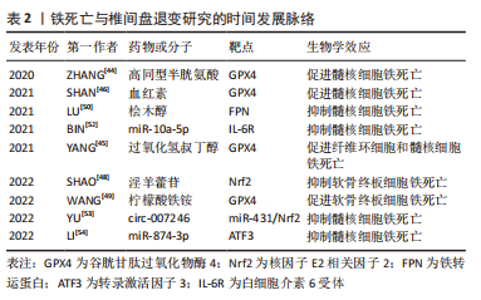
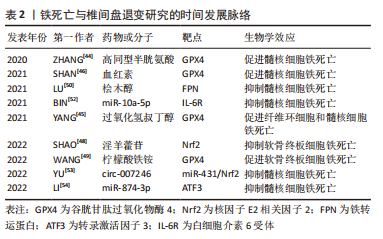
2.1.1 铁代谢与铁死亡 铁是维持哺乳动物正常生长发育的重要微量元素,与多种生命活动密切相关,如通过参与血红蛋白的合成从而参与氧气和二氧化碳的运输[24]。正常生理情况下,机体通过食物摄入铁和机体内铁循环利用从而保持铁的动态平衡。食物中的铁以二价铁离子形式在十二指肠被吸收,吸收进入黏膜细胞的二价铁离子在铁转运蛋白的转运下进入循环,随即被多铜氧化酶氧化为三价铁离子。循环中的三价铁离子与转铁蛋白结合后转运到外周组织[25],在位于细胞膜上的转铁蛋白受体1的介导下进入外周组织细胞后储存于内体。内体中的三价铁被前列腺六跨膜上皮抗原3还原为二价铁离子并在二价金属转运体1(divalent metal transporter1,DMT1)介导下从内体转运到细胞质[26],进入胞质的亚铁离子储存在铁蛋白和不稳定铁池中。铁蛋白由铁蛋白轻链和铁蛋白重链构成,重链中的亚铁氧化酶能将二价铁离子氧化为三价铁离子并储存在铁蛋白中,轻链与铁的矿化和储存相关。铁反应元件结合蛋2基因沉默后能够上调铁蛋白重链和轻链的表达使细胞内铁离子含量下降,从而抑制爱拉斯汀(erastin)、Ras选择性致死小分子3(RAS-selective lethal small molecule 3,RSL3)和 RSL5诱导的铁死亡[21]。此外,热休克蛋白B1可通过抑制转铁蛋白受体1表达来降低细胞内铁离子水平,从而抑制铁死亡[27]。当体内铁代谢平衡被打破时,累积的亚铁离子通过芬顿(Fenton)或哈伯·韦斯(Haber Weiss)反应产生活性氧,引起PUFAs过氧化物蓄积,从而诱导铁死亡发生[28]。 2.1.2 PUFAs与铁死亡 富含PUFAs尤其是花生四烯酸(Arachidonoyl,AA)和肾上腺酸(Adrenoyl,AdA)的细胞膜、质膜等特别容易发生氧化反应引起PUFAs过氧化导致细胞铁死亡的发生[29]。花生四烯酸和肾上腺酸在长链脂酰辅酶A合成酶4(Acyl-CoA synthetase long-chain family member 4,ACSL4)和溶血磷脂酰胆碱酰基转移酶3(Lysophosphatidylcholine acyltransferase 3,LPCAT3)的催化作下,与磷脂酰乙醇胺反应生成AA/AdA-PE。脂氧合酶是一类含铁蛋白酶,其过表达能够使AA/AdA-PE发生氧化生成PUFAs过氧化物,PUFAs过氧化物在铁死亡中发挥关键作用。外源加入PUFAs可以加速PUFAs过氧化物的蓄积从而增加铁死亡敏感性,此外RSL3可通过直接抑制GPX4活性导致PUFAs过氧化物不能被及时清除而蓄积从而诱发细胞铁死亡发生[30]。相反,通过抑制ACSL4和LPCAT3活性可减少PUFAs过氧化物累积从而抑制铁死亡发生[31],同时也可以通过使用抗氧化剂维生素E来抑制脂氧合酶,从而抑制PUFAs过氧化物生成进而阻止铁死亡进展[32]。 2.1.3 氨基酸与铁死亡 根据是否能在机体内合成,氨基酸被分为必需氨基酸和非必需氨基酸。无论是必需氨基酸还是非必需氨基酸都在细胞增殖、分化中扮演重要的角色。研究发现氨基酸异常代谢与铁死亡相关[33]。细胞内的谷氨酸与细胞外的胱氨酸可通过细胞膜表面的System XC-进行反向1∶1交换[34]。 System XC-由轻链SLC7A11和重链SLC3A2组成,经System XC-介导进入细胞的胱氨酸被还原成半胱氨酸,半胱氨酸在γ-谷氨酰半胱氨酸合成酶、谷胱甘肽合成酶的先后催化下与谷氨酸和甘氨酸合成谷胱甘肽。谷胱甘肽是细胞内重要的还原剂,在保护组织细胞免受氧化发挥重要作用。GPX4以谷胱甘肽为还原剂将PUFAs过氧化物还原为无毒脂质醇。当谷氨酸和胱氨酸在细胞内外异常分布时,谷胱甘肽合成减少,GPX4活性也相应下降。研究表明爱拉斯汀与SLC7A11结合后抑制System XC-活性,导致细胞外的胱氨酸进入细胞内受到抑制,谷胱甘肽合成受阻,从而使GPX4活性下降,导致细胞不能及时清除PUFAs过氧化物,从而引发铁死亡[35]。由此可见氨基酸代谢在铁死亡中尤其重要。 2.1.4 铁死亡其他可能机制 除了铁离子、脂质、氨基酸参与铁死亡的发生机制外,还有其他机制可参与铁死亡发生。线粒体是细胞进行有氧呼吸的主要场所,在有氧时进行氧化磷酸化产生ATP,为细胞生命活动提供能量。当线粒体呼吸链损伤时会产生更多漏电子,使活性氧增加,PUFAs过氧化,从而增加铁死亡敏感性。当抑制线粒体呼吸链时可减轻活性氧水平和PUFAs过氧化,表明线粒体的功能紊乱与铁死亡的发生密切相关[36]。 有研究表明,核因子E2相关因子2(nuclear factor erythroid derived2-like2,Nrf2)和抑癌基因P53、BAP1、BECN1可通过对SLC7A11的调控来参与铁死亡的发生[37-39]。此外辅酶Q10和铁蛋白自噬也在铁死亡发生中发挥重要作用[40-41]。随着对铁死亡研究的深入,铁死亡的机制将在不断补充中得到完善。 2.2 铁死亡与椎间盘退变 椎间盘是体内最大的无血管器官[42],在重力作用下椎间盘细胞处于过度压力、高渗透压、低氧和营养缺乏等恶劣微环境中。因此髓核细胞、纤维环细胞和终板软骨细胞在各种因素影响下极易发生衰老和死亡,其衰老和死亡加速了椎间盘退变进展。 GPX4是铁死亡发生过程中的明星分子,通常被认为是铁死亡的标志分子,其在谷胱甘肽的参与下能将PUFAs过氧化物还原为无毒脂质醇。当各种因素导致GPX4活性下降时会导致PUFAs过氧化物蓄积而增加铁死亡敏感性,反之亦然[43]。同型半胱氨酸通过甲硫氨酸循环在基因甲基化过程中起关键作用,ZHANG等[44]通过高同型半胱氨酸血症模型小鼠,证实了高同型半胱氨酸血症会导致GPX4甲基化,使GPX4活性下降从而增加髓核细胞铁死亡敏感性。使用甲基化酶抑制剂5-杂氮-2-脱氧胞苷和叶酸可抑制GPX4甲基化从而缓解高同型半胱氨酸血症诱导的髓核细胞铁死亡。YANG等[45]将纤维环细胞和髓核细胞暴露于不同浓度的过氧化氢叔丁醇中来模拟椎间盘退变,结果表明随着过氧化氢叔丁醇浓度的增加纤维环细胞和髓核细胞中的GPX4表达下降,纤维环细胞和髓核细胞死亡率增加。外源性加入GPX4抑制剂RSL3会加剧纤维环细胞和髓核细胞死亡,而铁螯合剂去铁胺或铁死亡抑制剂(Ferrostatin-1,Fer-1)则可抑制这个过程。突出髓核中新生血管易发生破裂,导致红细胞外渗使组织暴露在高水平血红素之中,血红素分解代谢增加使活性氧水平上升,GPX4水平下调,从而诱导髓核细胞铁死亡,这种由血红素诱导的细胞死亡可被去铁胺抑制,证明了血红素通过诱导髓核细胞铁死亡参与椎间盘退变发生[46]。 软骨终板位于椎间盘纤维环及髓核的上下两缘,营养物质通过软骨终板的渗透而供给髓核,所以软骨终板中软骨细胞的丢失在椎间盘退变中发挥重要的作用[47]。SHAO等[48]的研究发现,淫羊藿苷处理能够使肿瘤坏死因子α和过氧化氢叔丁醇刺激的终板软骨细胞中GPX4、SLC7A11蛋白水平显著升高,而Nrf2敲除可阻断淫羊藿苷介导的GPX4、SLC7A11上调,表明淫羊藿苷通过激活Nrf2降低终板软骨细胞铁死亡从而缓解椎间盘退变。WANG等[49]用不同浓度柠檬酸铁铵处理终板软骨细胞模拟铁超载,铁死亡标志物GPX4和SLC7A11表达水平与柠檬酸铁铵的浓度呈剂量依赖性下降,而柠檬酸铁铵诱导的GPX4和SLC7A11下调可被Fer-1逆转。因此,铁超载下调GPX4和SLC7A11水平促进终板软骨细胞铁死亡加剧椎间盘退变。 铁离子蓄积是铁死亡另外一个重要特征。LU等[50]使用siFPN敲除铁转运蛋白时,过氧化氢叔丁醇诱导的髓核细胞铁水平上升、铁死亡加重。而当在过氧化氢叔丁醇诱导的髓核细胞中转染慢病毒质粒(Lenti-FPN)来过表达铁转运蛋白时,过氧化氢叔丁醇诱导的髓核细胞中细胞铁水平下降。此外,在过氧化氢叔丁醇诱导的髓核细胞中转染慢病毒质粒(Lenti-MTF1)过表达金属调控转录因子1,可上调金属调控转录因子1核易位,从而恢复铁转运蛋白的功能。总之,过氧化氢叔丁醇通过降低金属调控转录因子1蛋白的核易位使铁转运蛋白表达下调,导致髓核细胞内铁蓄积从而诱导铁死亡发生。桧木醇是一种从中国台湾扁柏中提取而得到的芳香族托酚酮,可增加金属调控转录因子1核易位和铁转运蛋白的表达,使细胞内铁水平下降从而抑制铁死亡延缓椎间盘退变。ZHANG等[51]发现椎间盘退变的大鼠髓核组织中铁水平显著增加,为了进一步证明铁死亡在椎间盘退变中的作用,对正常和椎间盘退变的髓核进行单细胞RNA测序,将髓核中的软骨细胞分为纤维软骨细胞祖细胞、软骨祖细胞、稳态软骨细胞、C1-C4 7个不同亚群,与正常髓核相比,椎间盘退变髓核中C2、C4、软骨祖细胞和稳态软骨细胞中的铁死亡相关信号上调。 此外,研究发现微小RNA(micro RNA,miRNA)也可通过调控铁死亡从而参与椎间盘退变病理进展。BIN等[52]研究发现白细胞介素6处理的髓核细胞中丙二醛、活性氧和Fe2+的水平上升,证实了白细胞介素6可诱导软骨细胞氧化应激并中断铁稳态导致铁死亡。在其后续的实验中,证明了白细胞介素6通过下调 miR-10a-5p水平,然后去抑制其受体表达,从而诱导细胞氧化应激和干扰铁稳态引起铁死亡发生。而过表达miR-10a-5p 可下调白细胞介素6受体表达,从而缓解白细胞介素6 诱导的铁死亡。因此,miR-10a-5p/IL-6R 轴在调控髓核细胞铁死亡中发挥重要作用。而去铁胺可通过上调miR-10a-5p水平从而下调白细胞介素6受体表达来削弱铁死亡。来源于骨髓间充质干细胞外囊泡中的circ-007246属于环状RNA,其可作为竞争性内源性RNA与miR-431竞争性结合后下调 miR-431表达。 circ-007246过表达时,髓核细胞中Nrf2和GPX4的表达升高,而miR-431和ACSL4的表达降低;当沉默Nrf2时,circ-007246过表达的髓核细胞中ACSL4表达升高,而GPX4表达降低;所以circ-007246通过下调miR-431和上调 Nrf2的水平来抑制髓核细胞铁死亡,从而促进髓核细胞基质合成和增殖,减缓椎间盘退变[53]。LI等[54]研究表明,miR-874-3p下调可增加转录激活因子3的表达水平。上调转录激活因子3不仅可以通过抑制SLC7A11和增加活性氧产生来增强髓核细胞的铁死亡,而且还可以促进细胞外基质降解和白细胞介素1β分泌,从而加速椎间盘退变进展。因此,miR-874-3p过表达可负向调节转录激活因子3,从而抑制髓核细胞铁死亡,缓解椎间盘退变。 近年来研究发现,铁死亡参与了椎间盘退变的进展,其在椎间盘退变中的研究进展见表2。"
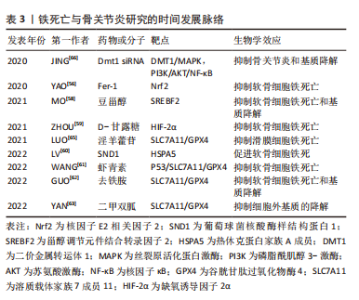
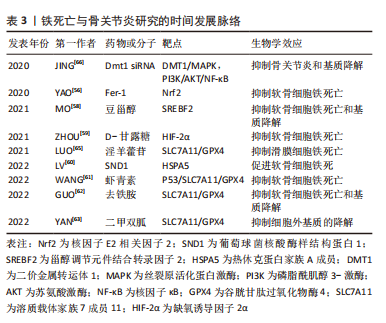
2.3 铁死亡与骨关节炎 骨关节炎是以慢性炎症、关节软骨进行性退变、骨质增生硬化及继发骨赘形成为特征的一种关节疾病[55]。目前,铁死亡与骨关节炎发生的研究较少,现有的研究表明其可通过调控软骨细胞活性、细胞外基质降解和滑膜炎症等病理生理过程,进而参与骨关节炎疾病进程。 软骨细胞是关节软骨中唯一的细胞类型,铁死亡导致软骨细胞丢失在骨关节炎进展中发挥重要的作用。使用白细胞介素1β、柠檬酸铁铵诱导的骨关节炎细胞模型中,软骨细胞中铁死亡标志分子GPX4和SLC7A11表达下降,P53和ACSL4表达上升;下调经白细胞介素1β、柠檬酸铁铵诱导的软骨细胞中Nrf2水平时,GPX4的表达水平下降;而通过关节内注射Fer-1发现其可以逆转软骨细胞中铁死亡相关蛋白表达的变化,并促进Nrf2抗氧化系统激活从而抑制软骨细胞铁死亡延缓骨关节炎,因此Nrf2抗氧化系统和铁死亡是相互调控的,但具体机制尚不清楚[56]。 通过分析骨关节炎和非损伤人软骨组织样本中铁死亡调节因子GPX4、SLC3A2和SLC7A11表达水平,结果显示骨关节炎软骨中GPX4和SLC3A2表达显著降低。进一步用泛半胱天冬酶抑制剂、坏死性凋亡抑制剂、Fer-1和去铁胺分别处理骨关节炎细胞模型时,发现Fer-1和去铁胺可降低细胞内活性氧、PUFAs过氧化物蓄积,恢复GPX4活性,从而抑制软骨细胞铁死亡,而这个过程不能被泛半胱天冬酶抑制剂和坏死性凋亡抑制剂所抑制[57]。 铁死亡对软骨细胞活性的影响受到多种机制调控。MO等[58]研究证实了豆甾醇可通过下调甾醇调节元件结合转录因子2来减轻白细胞介素1β诱导的ATDC5细胞铁死亡发生。ZHOU等[59]学者发现D-甘露糖处理可上调白细胞介素1β诱导的软骨细胞中GPX4和SLC7A11水平,缺氧诱导因子2α过表达则可抑制D-甘露糖的作用,而Fer-1可减弱缺氧诱导因子2α过表达对D-甘露糖的抑制作用,所以D-甘露糖通过抑制缺氧诱导因子2α来降低软骨细胞铁死亡敏感性从而减轻骨关节炎进展。最近的研究表明骨关节炎大鼠软骨组织中GPX4和热休克蛋白家族A成员5表达水平降低,而葡萄球菌核酸酶样结构蛋白1(staphylococcal nuclease domain containing 1,SND1)在骨关节炎中表达升高。进一步研究发现,白细胞介素1β刺激的原代软骨细胞中SND1表达增加,增加的SND1抑制了热休克蛋白家族A成员5表达导致GPX4降解增加,而敲除SND1可上调大鼠软骨中热休克蛋白家族A成员5水平和增加GPX4表达。因此,降低SND1的水平可促进热休克蛋白家族A成员5的表达,从而增加GPX4的活性,延缓骨关节炎进展[60]。虾青素是一种具有抗氧化作用的类胡萝卜素,其可以通过下调P53表达来缓解白细胞介素1β诱导的SLC7A11、GPX4、谷胱甘肽和铁蛋白的表达下降,从而抑制软骨细胞铁死亡的发生[61]。 关节软骨的绝大部分成分由细胞外基质组成,抑制铁死亡的发生可减缓细胞外基质降解从而缓解骨关节炎进展。YAO等[56]通过关节内注射Fer-1,发现Fer-1可以缓解Ⅱ型胶原降解从而减轻软骨退化和骨关节炎进展,通过进一步研究表明Fer-1可减轻白细胞介素1β和柠檬酸铁铵对胶原蛋白Ⅱ的抑制,但是Fer-1并不减少白细胞介素1β和柠檬酸铁铵诱导的基质金属蛋白酶13 表达。而又有研究表明Fer-1可以减少白细胞介素1β诱导的基质金属蛋白酶13的表达[60,62]。除了Fer-1可以抑制铁死亡引起的细胞外基质降解外,去铁胺也可逆转爱拉斯汀和白细胞介素1β诱导的基质金属蛋白酶13表达上调、胶原蛋白Ⅱ表达下调[62]。豆甾醇可以通过下调甾醇调节元件结合转录因子2来调节基质金属蛋白酶13、Ⅱ型胶原和蛋白多糖的表达水平,从而减轻白细胞介素1β诱导的铁死亡和细胞外基质降解[58]。二甲双胍治疗也可以部分减轻由关节内注射Erastin而导致的蛋白多糖丢失,从而减轻细胞外基质降解,延缓骨关节炎进展[63]。 骨关节炎另一个重要特征是滑膜炎,滑膜炎会导致软骨退化,还与关节疼痛以及关节功能障碍密切相关[64]。为了探索铁死亡是否与滑膜炎相关,MIAO等[57]检测了骨关节炎患者滑液中铁的水平,结果表明滑液中铁离子水平随着骨关节炎的进展而增加。经过脂多糖诱导的滑膜炎细胞模型中,铁死亡标志分子GPX4、SLC7A11和SLC3A2L的表达明显下降,淫羊藿苷以浓度依赖性方式上调GPX4、SLC7A11和SLC3A2L的表达水平;淫羊藿苷还抵消了RSL3诱导的滑膜细胞中铁死亡相关蛋白表达、PUFAs过氧化物和铁离子的蓄积[65]。JING等[66]的研究发现,骨关节炎中的炎症因子通过上调铁输入蛋白转铁蛋白受体1和DMT1,下调铁流出蛋白铁转运蛋白,干扰铁稳态导致铁超载。铁超载可通过上调基质金属蛋白酶13的表达,加重炎症反应和细胞外基质降解。抑制DMT1可阻断MAPK和PI3K/AKT/NF-κB通路,从而抑制铁超载导致的炎症反应和细胞外基质降解。XIA等[67]通过生物信息学分析,发现7个铁死亡差异表达基因包括ATF3、IL6、CDKN1A、IL1B、EGR1、JUN和CD44具有良好的诊断特性,可作为骨关节炎中滑膜炎的诊断生物标志物和治疗靶点,进一步阐明了骨关节炎的发病机制。滑膜炎中的炎症反应也可直接激活铁死亡,从而参与骨关节炎疾病进展。 铁死亡与骨关节炎的研究机制及时间发展脉络见表3。"
| [1] CHEN HW, ZHOU JW, ZHANG GZ, et al. Emerging role and therapeutic implication of mTOR signalling in intervertebral disc degeneration. Cell Prolif. 2022:e13338. [2] GBD 2017 DISEASE AND INJURY INCIDENCE AND PREVALENCE COLLABORATORS. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159): 1789-1858. [3] XIN J, WANG Y, ZHENG Z, et al. Treatment of Intervertebral Disc Degeneration. Orthop Surg. 2022;14(7):1271-1280. [4] TAKEOKA Y, YURUBE T, NISHIDA K. Gene Therapy Approach for Intervertebral Disc Degeneration: An Update. Neurospine. 2020;17(1): 3-14. [5] LUAN L, LIANG Z. Tanshinone IIA protects murine chondrogenic ATDC5 cells from lipopolysaccharide-induced inflammatory injury by down-regulating microRNA-203a. Biomed Pharmacother. 2018;103:628-636. [6] LANE NE, SHIDARA K, WISE BL. Osteoarthritis year in review 2016: clinical. Osteoarthritis Cartilage. 2017;25(2):209-215. [7] 金涛,刘林,朱晓燕,等.骨关节炎与线粒体异常[J].中国组织工程研究,2022,26(9):1452-1458. [8] LIAO Z, KE W, LIU H, et al. Vasorin-containing small extracellular vesicles retard intervertebral disc degeneration utilizing an injectable thermoresponsive delivery system. J Nanobiotechnology. 2022;20(1):420. [9] 刘岩路,胡炜,艾克拜尔,等.抑制半乳糖凝集素3促进椎间盘软骨终板细胞凋亡诱导椎间盘退变[J].中国组织工程研究,2021, 25(35):5599-5603. [10] HAO X, ZHAO J, JIA L, et al. XMU-MP-1 attenuates osteoarthritis via inhibiting cartilage degradation and chondrocyte apoptosis. Front Bioeng Biotechnol. 2022;10:998077. [11] CHAO-YANG G, PENG C, HAI-HONG Z. Roles of NLRP3 inflammasome in intervertebral disc degeneration. Osteoarthritis Cartilage. 2021;29(6): 793-801. [12] 张皓博,赵宇楠,杨学军.细胞焦亡在椎间盘退变中的作用及治疗意义[J].中国组织工程研究,2022,26(9):1445-1451. [13] KRITSCHIL R, SCOTT M, SOWA G, et al. Role of autophagy in intervertebral disc degeneration. J Cell Physiol. 2022;237(2):1266-1284. [14] DUAN R, XIE H, LIU ZZ. The Role of Autophagy in Osteoarthritis. Front Cell Dev Biol. 2020;8:608388. [15] MOU Y, WANG J, WU J, et al. Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J Hematol Oncol. 2019;12(1):34. [16] JIANG X, STOCKWELL BR, CONRAD M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22(4):266-282. [17] YAN HF, ZOU T, TUO QZ, et al. Ferroptosis: mechanisms and links with diseases. Signal Transduct Target Ther. 2021;6(1):49. [18] 朱蕊,曾庆,黄国志.铁死亡与脑卒中[J].中国组织工程研究,2021, 25(23):3734-3739. [19] YANG J, HU S, BIAN Y, et al. Targeting Cell Death: Pyroptosis, Ferroptosis, Apoptosis and Necroptosis in Osteoarthritis. Front Cell Dev Biol. 2022; 9:789948. [20] OHNISHI T, IWASAKI N, SUDO H. Causes of and Molecular Targets for the Treatment of Intervertebral Disc Degeneration: A Review. Cells. 2022;11(3):394. [21] TANG D, KANG R, BERGHE TV, et al. The molecular machinery of regulated cell death. Cell Res. 2019;29(5):347-364. [22] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060-1072. [23] GAN B. Mitochondrial regulation of ferroptosis. J Cell Biol. 2021;220(9): e202105043. [24] BOGDAN AR, MIYAZAWA M, HASHIMOTO K, et al. Regulators of Iron Homeostasis: New Players in Metabolism, Cell Death, and Disease. Trends Biochem Sci. 2016;41(3):274-286. [25] GAO M, MONIAN P, QUADRI N, et al. Glutaminolysis and Transferrin Regulate Ferroptosis. Mol Cell. 2015;59(2):298-308. [26] TANG D, CHEN X, KANG R, et al. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31(2):107-125. [27] GAO M, MONIAN P, JIANG X. Metabolism and iron signaling in ferroptotic cell death. Oncotarget. 2015;6(34):35145-35146. [28] FISCHBACHER A, VON SONNTAG C, SCHMIDT TC. Hydroxyl radical yields in the Fenton process under various pH, ligand concentrations and hydrogen peroxide/Fe(II) ratios. Chemosphere. 2017;182:738-744. [29] YANG WS, KIM KJ, GASCHLER MM, et al. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci U S A. 2016;113(34):E4966-E4975. [30] YANG WS, SRIRAMARATNAM R, WELSCH ME, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156(1-2):317-331. [31] FENG H, STOCKWELL BR. Unsolved mysteries: How does lipid peroxidation cause ferroptosis? PLoS Biol. 2018;16(5):e2006203. [32] KAGAN VE, MAO G, QU F, et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol. 2017;13(1):81-90. [33] ANGELI JPF, SHAH R, PRATT DA, et al. Ferroptosis Inhibition: Mechanisms and Opportunities. Trends Pharmacol Sci. 2017;38(5):489-498. [34] PITMAN KE, ALLURI SR, KRISTIAN A, et al. Influx rate of 18F-fluoroaminosuberic acid reflects cystine/glutamate antiporter expression in tumour xenografts. Eur J Nucl Med Mol Imaging. 2019; 46(10):2190-2198. [35] CHEN L, LI X, LIU L, et al. Erastin sensitizes glioblastoma cells to temozolomide by restraining xCT and cystathionine-γ-lyase function. Oncol Rep. 2015;33(3):1465-1474. [36] GAO M, YI J, ZHU J, et al. Role of Mitochondria in Ferroptosis. Mol Cell. 2019;73(2):354-363.e3. [37] XIE Y, ZHU S, SONG X, et al. The Tumor Suppressor p53 Limits Ferroptosis by Blocking DPP4 Activity. Cell Rep. 2017;20(7):1692-1704. [38] ZHANG Y, KOPPULA P, GAN B. Regulation of H2A ubiquitination and SLC7A11 expression by BAP1 and PRC1. Cell Cycle. 2019;18(8):773-783. [39] FAN Z, WIRTH AK, CHEN D, et al. Nrf2-Keap1 pathway promotes cell proliferation and diminishes ferroptosis. Oncogenesis. 2017;6(8):e371. [40] BERSUKER K, HENDRICKS JM, LI Z, et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature. 2019;575(7784): 688-692. [41] MANCIAS JD, WANG X, GYGI SP, et al. Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy. Nature. 2014; 509(7498):105-109. [42] BOUBRIAK OA, WATSON N, SIVAN SS, et al. Factors regulating viable cell density in the intervertebral disc: blood supply in relation to disc height. J Anat. 2013;222(3):341-348. [43] WEI Y, LV H, SHAIKH AB, et al. Directly targeting glutathione peroxidase 4 may be more effective than disrupting glutathione on ferroptosis-based cancer therapy. Biochim Biophys Acta Gen Subj. 2020;1864(4):129539. [44] ZHANG X, HUANG Z, XIE Z, et al. Homocysteine induces oxidative stress and ferroptosis of nucleus pulposus via enhancing methylation of GPX4. Free Radic Biol Med. 2020;160:552-565. [45] YANG RZ, XU WN, ZHENG HL, et al. Involvement of oxidative stress-induced annulus fibrosus cell and nucleus pulposus cell ferroptosis in intervertebral disc degeneration pathogenesis. J Cell Physiol. 2021; 236(4):2725-2739. [46] SHAN L, XU X, ZHANG J, et al. Increased hemoglobin and heme in MALDI-TOF MS analysis induce ferroptosis and promote degeneration of herniated human nucleus pulposus. Mol Med. 2021;27(1):103. [47] 张艳琳,黄国付,邹璟,等.终板软骨细胞衰老在腰椎间盘退变中的研究进展[J].医学研究杂志,2022,51(7):173-176. [48] SHAO Y, SUN L, YANG G, et al. Icariin protects vertebral endplate chondrocytes against apoptosis and degeneration via activating Nrf-2/HO-1 pathway. Front Pharmacol. 2022;13:937502. [49] WANG W, JING X, DU T, et al. Iron overload promotes intervertebral disc degeneration via inducing oxidative stress and ferroptosis in endplate chondrocytes. Free Radic Biol Med. 2022;190:234-246. [50] LU S, SONG Y, LUO R, et al. Ferroportin-Dependent Iron Homeostasis Protects against Oxidative Stress-Induced Nucleus Pulposus Cell Ferroptosis and Ameliorates Intervertebral Disc Degeneration In Vivo. Oxid Med Cell Longev. 2021;2021:6670497. [51] ZHANG Y, HAN S, KONG M, et al. Single-cell RNA-seq analysis identifies unique chondrocyte subsets and reveals involvement of ferroptosis in human intervertebral disc degeneration. Osteoarthritis Cartilage. 2021;29(9):1324-1334. [52] BIN S, XIN L, LIN Z, et al. Targeting miR-10a-5p/IL-6R axis for reducing IL-6-induced cartilage cell ferroptosis. Exp Mol Pathol. 2021;118:104570. [53] YU X, XU H, LIU Q, et al. circ_0072464 Shuttled by Bone Mesenchymal Stem Cell-Secreted Extracellular Vesicles Inhibits Nucleus Pulposus Cell Ferroptosis to Relieve Intervertebral Disc Degeneration. Oxid Med Cell Longev. 2022;2022:2948090. [54] LI Y, PAN D, WANG X, et al. Silencing ATF3 Might Delay TBHP-Induced Intervertebral Disc Degeneration by Repressing NPC Ferroptosis, Apoptosis, and ECM Degradation. Oxid Med Cell Longev. 2022;2022: 4235126. [55] FINDLAY DM, ATKINS GJ. Osteoblast-chondrocyte interactions in osteoarthritis. Curr Osteoporos Rep. 2014;12(1):127-134. [56] YAO X, SUN K, YU S, et al. Chondrocyte ferroptosis contribute to the progression of osteoarthritis. J Orthop Translat. 2020;27:33-43. [57] MIAO Y, CHEN Y, XUE F, et al. Contribution of ferroptosis and GPX4’s dual functions to osteoarthritis progression. EBioMedicine. 2022;76: 103847. [58] MO Z, XU P, LI H. Stigmasterol alleviates interleukin-1beta-induced chondrocyte injury by down-regulatingsterol regulatory element binding transcription factor 2 to regulateferroptosis. Bioengineered. 2021;12(2):9332-9340. [59] ZHOU X, ZHENG Y, SUN W, et al. D-mannose alleviates osteoarthritis progression by inhibiting chondrocyte ferroptosis in a HIF-2alpha-dependent manner. Cell Prolif. 2021;54(11):e13134. [60] LV M, CAI Y, HOU W, et al. The RNA-binding protein SND1 promotes the degradation of GPX4 by destabilizing the HSPA5 mRNA and suppressing HSPA5 expression, promoting ferroptosis in osteoarthritis chondrocytes. Inflamm Res. 2022;71(4):461-472. [61] WANG X, LIU Z, PENG P, et al. Astaxanthin attenuates osteoarthritis progression via inhibiting ferroptosis and regulating mitochondrial function in chondrocytes. Chem Biol Interact. 2022;366:110148. [62] GUO Z, LIN J, SUN K, et al. Deferoxamine Alleviates Osteoarthritis by Inhibiting Chondrocyte Ferroptosis and Activating the Nrf2 Pathway. Front Pharmacol. 2022;13:791376. [63] YAN J, FENG G, MA L, et al. Metformin alleviates osteoarthritis in mice by inhibiting chondrocyte ferroptosis and improving subchondral osteosclerosis and angiogenesis. J Orthop Surg Res. 2022;17(1):333. [64] SCANZELLO CR, GOLDRING SR. The role of synovitis in osteoarthritis pathogenesis. Bone. 2012;51(2):249-257. [65] LUO H, ZHANG R. Icariin enhances cell survival in lipopolysaccharide-induced synoviocytes by suppressing ferroptosis via the Xc-/GPX4 axis. Exp Ther Med. 2021;21(1):72. [66] JING X, LIN J, DU T, et al. Iron Overload Is Associated With Accelerated Progression of Osteoarthritis: The Role of DMT1 Mediated Iron Homeostasis. Front Cell Dev Biol. 2020;8:594509. [67] XIA L, GONG N. Identification and verification of ferroptosis-related genes in the synovial tissue of osteoarthritis using bioinformatics analysis. Front Mol Biosci. 2022;9:992044. |
| [1] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [2] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [4] | He Xi, Wan Yu, Tang Yuting, Yang Anning, Wu Kai, Jiao Yun, Bai Zhigang, Jiang Yideng, Shen Jiangyong. Erastin inhibits proliferation of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-. |
| [5] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [6] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [7] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [8] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [9] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [10] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [11] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [12] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [13] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [14] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [15] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||