Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (31): 5058-5063.doi: 10.12307/2023.727
Previous Articles Next Articles
Application of finite element analysis in artificial knee arthroplasty
Li Haoran1, Huang Jian2
- 1Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China; 2Second Affiliated Hospital of Inner Mongolia Medical University (Inner Mongolia Institute of Orthopedics), Hohhot 010000, Inner Mongolia Autonomous Region, China
-
Received:2022-09-17Accepted:2022-11-16Online:2023-11-08Published:2023-01-31 -
Contact:Huang Jian, MD, Professor, Chief physician, Second Affiliated Hospital of Inner Mongolia Medical University (Inner Mongolia Institute of Orthopedics), Hohhot 010000, Inner Mongolia Autonomous Region, China -
About author:Li Haoran, Master candidate, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China -
Supported by:Science and Technology Research Plan Program of Inner Mongolia Autonomous Region, No. 2021GG0194 (to HJ)
CLC Number:
Cite this article
Li Haoran, Huang Jian. Application of finite element analysis in artificial knee arthroplasty[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(31): 5058-5063.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
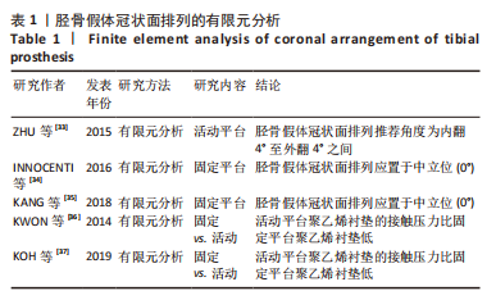
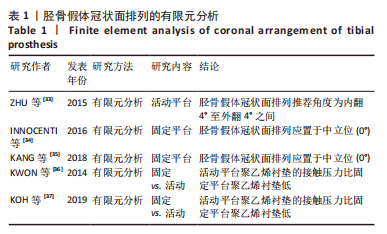
2.1 有限元分析在TKA术中的应用 TKA将人工假体组件置入膝关节来恢复下肢的运动功能,是临床治疗晚期骨性关节炎、关节畸形、关节重大创伤等严重膝关节疾病最有效的方法[7]。尽管TKA的发展日益成熟,但术后假体松动、假体周围骨折、假体磨损等同样困扰着外科医生。如何避免和改善这些情况的发生,国内外学者应用有限元法进行了许多相关研究和测试。目前有限元在TKA术中重点关注假体材质与设计、对位对线等方面的应用[8-9]。 2.1.1 有限元分析在假体材质与设计的应用 全膝关节假体根据聚乙烯垫片与金属胫骨托的连接方式,可分为固定平台和活动平台。固定平台假体是将聚乙烯部件通过锁定机制固定在胫骨平台上;活动平台除了和股骨假体形成活动关节,同时允许聚乙烯垫片与胫骨平台之间有一定程度的活动。TKA术后胫股关节接触压和接触面积的大小与假体设计有关,接触应力与接触面积呈反比[10]。GREENWALD等[11]利用三维有限元步态分析的方法比较了固定平台假体和活动平台假体的胫股接触面积,结果发现活动平台聚乙烯衬垫的接触面积范围为400-800 mm2,而固定平台聚乙烯衬垫的接触面积范围为200-250 mm2,证实活动平台胫股关节的接触面积均高于固定平台,故活动平台的接触应力低于固定平台。STUKENBORG-COLSMAN等[12]在尸体上的有限元分析得到了同样的结论,相较于固定平台,活动平台假体使胫股关节接触面积最大化,减少峰值接触压;胫股关节间的接触应力越大,聚乙烯垫片磨损越严重。林祥波[10]采用三维有限元分析发现旋转平台假体可以显著降低胫股关节接触应力,降低聚乙烯衬垫的磨损的风险。张立超等[13]对比了固定平台和活动平台的应力分布,研究显示旋转平台假体的内外侧接触压低于固定平台假体,旋转平台假体的内外侧接触面积高于固定平台假体。 综上,固定平台的低匹配度假体能够降低膝关节骨和假体界面的应力,降低假体松动,但是膝关节的接触压比较高,增加聚乙烯衬垫的磨损,自由旋转和低关节接触压之间的矛盾是固定平台假体无法解决的问题。而旋转平台假体的高匹配度能够减少膝关节骨和假体界面的应力,减少接触压,从而降低聚乙烯衬垫的磨损,并且旋转平台假体能够自由旋转,可以减少假体松动,弥补固定平台假体的不足。目前大多数有限元研究表明活动平台假体可改善关节匹配度,降低胫股关节接触面应力,从而减少聚乙烯磨损。但临床实际应用中两者的术后疗效和假体生存率孰优孰劣尚未获得一致性认可,因此有待长期随访进一步研究。 不同材质的人工关节假体在人工关节置换术中表现出不同的生物力学特性。BRIHAULT等[14]应用有限元分析法对比了膝关节在屈曲120°时不同材质胫骨平台假体的应力分布和微动,研究结果显示,全聚乙烯胫骨假体在平台下方的应力分布明显高于金属胫骨假体,另外较金属胫骨假体产生5倍微动。 TOKUNAGA等[15]建立站立位有限元模型,对比金属胫骨假体与全聚乙烯胫骨假体的应力分布,结果显示金属胫骨假体的应力明显低于全聚乙烯假体,应力的分布更加平衡。此外,HYLDAHL等[16-17]利用X射线立体摄影测量分析法对不同材质的假体进行研究,同样证实了上述结果。此外,股骨髁与胫骨平台之间的聚乙烯垫片在膝关节运动起缓冲作用,不同设计类型的聚乙烯垫片具有不同的生物力学影响。CASTELLARIN等[18]应用有限元法回顾性分析303例TKA患者,在使用相同胫骨和股骨假体条件下,其中151例采用对称聚乙烯垫片,另外152例采用非对称假体设计的垫片;通过站立和下蹲,相比于对称垫片,使用非对称垫片组中在胫骨产生更低的接触应力,在后续的临床随访中似乎也证实了上述结论。 除了假体的材质与设计,胫骨假体的型号匹配也影响着TKA术后聚乙烯垫片的使用寿命。COMPLETO等[19]构建有限元模型,将3号股骨髁假体分别与2.5号、3号、4号的胫骨平台假体配对,中间采用对应尺寸10 mm的聚乙烯垫片配对,对各屈曲角度的应力进行研究,结果显示3号股骨髁假体配对2.5号平台假体与3号髁股骨假体配对4号平台假体的接触面应力明显增高。刘志宏等[20]对固定平台的膝关节假体进行有限元分析,假体型号不完全匹配(3/2.5,3/4),假体接触面应力在不同膝关节活动角度和不同运动状态时增高,可能增加聚乙烯垫片的磨损风险。此外,姬林松等[21]对不同型号膝关节假体进行有限元分析时发现了同样的结论,由于应力的增加加速了聚乙烯垫片的磨损,进而影响了假体的使用寿命。 2.1.2 有限元分析在假体对位对线中的应用 TKA的成功与否与下肢力线的恢复密切相关[22]。下肢对线不良和假体位置偏差可导致聚乙烯衬垫异常磨损及假体过早松动,进而影响假体寿命[23-25]。下肢力线是股骨头中心点到踝关节胫骨下端中心点的连线,正常情况下该轴线恰好通过膝关节中心,称为中立位力学轴线[26]。在正常膝关节中,相对于下肢力线胫骨关节面有3°内翻,股骨关节面有9°外翻。在TKA术中目前存在2种对线方式,一种是传统的生物力学对线,另一种是运动学对线,传统的观点认为TKA术后患者下肢力线应重建至与中立位力线偏差小于3°的位置,而运动学对线则认为应以患者膝关节正常状态或病损前状态的运动轴为对线、定位参考进行截骨及假体安置,从而在术后使人工膝关节尽可能的模拟人体膝关节正常的生物力学状态。袁伟[27]运用有限元模型对比发现,相较于机械对线,采用运动对线进行TKA的患者在胫骨关节聚乙烯垫片上的接触面积更大,使人工关节接触面压力分布更均匀,产生的应力更低,从而减少关节接触面的磨损,延长假体寿命。KLASAN等[28]对10例膝骨关节炎患者建立有限元模型,分别模拟机械对线和运动对线TKA,研究结果显示,相较于机械对线,运动对线组在聚乙烯垫片上具有更低的接触压力,而在聚乙烯垫片和胫骨之间的von Mises应力却无明显差异。此外,SONG等[29]研究发现,在膝关节外翻时,运动对线在聚乙烯垫片上产生的接触应力高于机械对线。总的来说,目前TKA最佳的对线方式仍有争议,在运动对线出现之前,机械对线是TKA的“金标准”,而运动力学对线能最大程度恢复患者的膝关节生物力学,从而获得更好的手术效果和功能恢复,但目前的有限元和临床研究依然不能完全符合这一结论,需要进一步实验和研究。 2.2 有限元分析在单髁置换术中的应用 单髁置换相较于TKA是一种新型微创手术。单髁置换只置换病损部分,对膝关节内侧或外侧间室进行表面置换,替代膝关节损坏的软骨表面,该技术不用切除前后交叉韧带,保留了膝关节的本体感觉和关节功能,具有损伤小、恢复快等特点[30-32]。然而,单髁置换是一种技术要求很高的手术方式,必须注意部件的尺寸、截骨和术后对位对线,因为过度矫正或过度松解都可能导致不良的结果。在临床中,许多单髁置换术后翻修的数量增多提示研究者应从单髁假体的对位对线进行研究。 有限元分析在单髁假体对位对线的应用:单髁关节的假体根据运动模式的不同主要分为活动平台假体和固定平台假体,这两种假体在设计原理上有根本区别。活动平台假体是利用球窝状的关节面将股骨髁活动范围限制在关节面之内,利用相同曲率的曲面最大限度地增大接触面,以此来降低接触应力;而固定平台假体的设计理念恰恰相反,固定平台模仿了正常膝关节的运动模式,将活动区域均设定在股骨髁假体与半月板假体之间,更加贴近人体本身的膝关节运动模式。ZHU等[33]对活动平台胫骨假体冠状面排列进行了研究,当胫骨假体外翻> 4°时,内侧皮质骨近端Von Mises应力和压缩应变显著增加;当胫骨假体内翻> 4°时,胫骨假体龙骨切槽处压缩应变高于最高阈值,因此在活动平台胫骨假体冠状面排列推荐角度为内翻 4°至外翻4°之间。INNOCENTI等[34]对固定平台假体冠状面进行了研究,建立了股骨和胫骨假体呈不同内、外翻角度且胫骨假体后倾角均为 6°的模型,研究结论显示在步态周期中胫骨假体内外翻在聚乙烯垫片产生的应力均大于中立位。KANG等[35]的有限元试验同样证实了上述结论,因此在固定平台胫骨假体冠状面排列推荐中立位(0°)。KWON等[36]运用有限元模型模拟完整步态对比了固定平台和活动平台聚乙烯衬垫的接触压力和应力,研究结果表明活动平台聚乙烯衬垫的接触压力比固定平台聚乙烯衬垫低,而固定平台的单髁置换由于对侧室的压力较高,会增加膝关节进行性骨性关节炎的总体风险。KOH等[37]对比了在全步态周期中固定平台和活动平台的接触应力,研究发现活动平台的接触应力更低,聚乙烯假体的磨损量更低。总的来说,有限元分析结果显示活动平台胫骨假体冠状面排列推荐角度为内翻 4°至外翻4°,而固定平台保持中立位(0°)。活动平台股骨和胫骨假体形合性较好,能够减少假体磨损;而固定平台假体单髁置换应避免过度形合引起局部应力集中导致加速假体磨损。见表1。"

2.3 有限元分析在RTKA术中的应用 RTKA是人工膝关节初次置换因各种原因失败后的一种补救性手术,其具有手术难度高、患者创伤大、恢复时间长、围术期临床管理复杂等特点。因初次膝关节置换的患者人数逐年快速增长,RTKA逐渐成为关节外科领域未来的研究重点。运用有限元分析研究手术失败的原因,从生物力学角度更好地优化RTKA手术方案及假体的设计[38-40]。有限元分析在人工膝关节翻修中的研究主要集中在假体设计和材质选择。 2.3.1 有限元分析在延长杆设计的应用 对于全膝关节翻修手术,按固定方式可以分为骨水泥假体和非骨水泥假体(压配型假体),绝大多数假体髁面均使用骨水泥固定,延长杆可以用骨水泥固定或压配型固定。压配型有便于翻修的特点,但对于明显骨质疏松、干骺端畸形的患者应使用骨水泥假体。 CONLISK等[41]运用有限元分析了骨水泥型假体,他们构建了4种不同长度股骨假体,分别为无延长杆股骨假体、短柄延长杆假体(50 mm)、中柄延长杆假体(75 mm)和长柄延长杆假体(100 mm),将延长杆假体分别植入股骨,在步态循环中分析延长杆股骨周围的应力和应变。研究表明,配有延长杆的假体都能减少假体周围的应力,短柄、中柄和长柄延长杆对应股骨的应力分别减少11%,26%和29%,中柄延长杆(75 mm)被认为在手术时保留骨质和植入后减少假体周围应力之间可以保持最佳平衡。EL-ZAYAT等[42]运用有限元分析对比了骨水泥型和压配型延长杆在股骨上的应力分布,在步态循环和下蹲时与骨水泥假体相比,压配型延长杆的应力分布较大,同时容易产生微动,这可能解释了RTKA术后髓内延长杆尖端痛的原因。INNOCENTI等[43]在EI-ZAYAT等[42]研究的基础上,进一步分析在不同固定方式下延长杆的形状对应力分布的影响,研究发现由于股骨前弓的存在,弓形延长杆相较于传统延长杆整体应力分布更低,此外在髓内延长杆尖端开槽比实心延长杆降低了在髓腔内的应力,这与HIRSCHMANN等[44]和BRARACK等[45]的研究一致。在RTKA术中,骨水泥的存在减少了沿假体每个区域的应力。综上,延长杆的设计长度应在保留骨质和植入后减少假体周围应力之间保持最佳平衡;与骨水泥柄相比,压配式延长杆显示出更高的微动。此外接近于解剖结构的弓形延长杆和尖端开槽的髓腔杆有利于降低前端应力分布,减少术后尖端痛的出现,这些发现将有助于骨科医生选择最合适的翻修假体。 2.3.2 有限元分析在金属加强块材质的应用 在RTKA过程中,经常需要重建不完整的骨缺损,这是由于TKA的失败往往与胫骨近端或股骨远端的骨缺损有关[46-48]。金属加强块是重建非封闭性骨缺损的主要选择之一,因为它具有广泛的模块化,可以快速和简单的使用[49-51]。 INNOCENTI等[52]应用有限元模型对不同材质的加强块(骨水泥、多孔金属和实心金属)进行了生物力学分析,结果表明任何材质的加强块都会引起应力的变化,特别是在靠近骨缺损的区域,其中多孔金属和骨水泥产生的应力明显低于实心金属(Ti 和CoCr),此外多孔金属允许骨长入的特性进一步减少了松动的发生。KANG等[53]进一步研究了在大面积(10-20 mm)骨缺损中,相较于单个金属加强块,使用双金属加强块可以明显降低应力分布。此外,定制金属加强块可以实现与皮质骨的完全接触,实现更好的应力传递,从而减少应力屏蔽和骨水泥失效带来的骨吸收风险。目前,RTKA术中治疗骨缺损的主要方法包括骨水泥、带螺钉加固的骨水泥、金属加强块、压配型骨移植、结构性异体移植,具体取决于缺损的位置和大小。由于金属加强块简单易用、具有良好的应力传递等特性,其越来越多的应用于骨缺损的治疗中,此外定制个性化的金属加强块将优化骨缺损的治疗。"

| [1] POPESCU R, HARITINIAN EG, CRISTEA S. Relevance of Finite Element in Total Knee Arthroplasty - Literature Review. Chirurgia (Bucur). 2019; 114(4):437-442. [2] RYBICKI EF, SIMONEN FA, WEIS EB JR. On the mathematical analysis of stress in the human femur. J Biomech. 1972;5(2): 203-215. [3] BREKELMANS WA, POORT HW, SLOOFF TJ. A new method to analyse the mechanical behaviour of skeletal parts. Acta Orthop Scand. 1972;43(5): 301-317. [4] SAKSENA J, PLATTS AD, DOWD GS. Recurrent haemarthrosis following total knee replacement. Knee. 2010;17(1):7-14. [5] PRICE AJ, ALVAND A, TROELSEN A, et al. Knee replacement. Lancet. 2018;392(10158):1672-1682. [6] LI X, WANG C, GUO Y, et al. An Approach to Developing Customized Total Knee Replacement Implants. J Healthc Eng. 2017;2017:9298061. [7] MARTIN JR, BEAHRS TR, STUHLMAN CR, et al. Complex Primary Total Knee Arthroplasty: Long-Term Outcomes. J Bone Joint Surg Am. 2016; 98(17):1459-1470. [8] PARK HJ, BAE TS, KANG SB, et al. A three-dimensional finite element analysis on the effects of implant materials and designs on periprosthetic tibial bone resorption. PLoS One. 2021;16(2):e0246866. [9] INNOCENTI B, BELLEMANS J, CATANI F. Deviations From Optimal Alignment in TKA: Is There a Biomechanical Difference Between Femoral or Tibial Component Alignment? J Arthroplasty. 2016;31(1): 295-301. [10] 林祥波. 固定与旋转平台膝关节假体有限元、体外生物力学分析及临床应用研究[D]. 上海:第二军医大学,2009. [11] GREENWALD AS, HEIM CS. Mobile-bearing knee systems: ultra-high molecular weight polyethylene wear and design issues. Instr Course Lect. 2005;54:195-205. [12] STUKENBORG-COLSMAN C, OSTERMEIER S, HURSCHLER C, et al. Tibiofemoral contact stress after total knee arthroplasty: comparison of fixed and mobile-bearing inlay designs. Acta Orthop Scand. 2002;73(6): 638-646. [13] 张立超, 张立敏, 武丽珠. 全膝关节置换固定和旋转平台假体的有限元分析[J]. 中国组织工程研究,2016,20(39):5801-5806. [14] BRIHAULT J, NAVACCHIA A, PIANIGIANI S, et al. All-polyethylene tibial components generate higher stress and micromotions than metal-backed tibial components in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2016;24(8):2550-2559. [15] TOKUNAGA S, ROGGE RD, SMALL SR, et al. A Finite-Element Study of Metal Backing and Tibial Resection Depth in a Composite Tibia Following Total Knee Arthroplasty. J Biomech Eng. 2016;138(4):041001. [16] HYLDAHL H, REGNéR L, CARLSSON L, et al. All-polyethylene vs. metal-backed tibial component in total knee arthroplasty-a randomized RSA study comparing early fixation of horizontally and completely cemented tibial components: part 1. Horizontally cemented components: AP better fixated than MB. Acta Orthop. 2005;76(6):769-777. [17] HYLDAHL H, REGNéR L, CARLSSON L, et al. All-polyethylene vs. metal-backed tibial component in total knee arthroplasty-a randomized RSA study comparing early fixation of horizontally and completely cemented tibial components: part 2. Completely cemented components: MB not superior to AP components. Acta Orthop. 2005; 76(6):778-784. [18] CASTELLARIN G, PIANIGIANI S, INNOCENTI B. Asymmetric polyethylene inserts promote favorable kinematics and better clinical outcome compared to symmetric inserts in a mobile bearing total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2019;27(4):1096-1105. [19] COMPLETO A, REGO A, FONSECA F, et al. Biomechanical evaluation of proximal tibia behaviour with the use of femoral stems in revision TKA: an in vitro and finite element analysis. Clin Biomech (Bristol, Avon). 2010;25(2):159-165. [20] 刘志宏, 刘利, 韦林, 等. 不同型号配对固定平台型膝关节假体生物力学有限元分析研究[J]. 国际骨科学杂志,2012,33(5):322-325. [21] 姬林松, 李彦林, 黄赞, 等. 不同型号膝关节固定平台假体匹配的三维有限元生物力学分析[J]. 实用医学杂志,2016,32(2):222-225. [22] DE STEIGER RN, LIU YL, GRAVES SE. Computer navigation for total knee arthroplasty reduces revision rate for patients less than sixty-five years of age. J Bone Joint Surg Am. 2015;97(8):635-642. [23] LONGSTAFF LM, SLOAN K, STAMP N, et al. Good alignment after total knee arthroplasty leads to faster rehabilitation and better function. J Arthroplasty. 2009;24(4):570-578. [24] BARGREN JH, BLAHA JD, FREEMAN MA. Alignment in total knee arthroplasty. Correlated biomechanical and clinical observations. Clin Orthop Relat Res. 1983;(173):178-183. [25] HOOD RW, VANNI M, INSALL JN. The correction of knee alignment in 225 consecutive total condylar knee replacements. Clin Orthop Relat Res. 1981;(160):94-105. [26] DONALDSON J, JOYNER J, TUDOR F. Current Controversies of Alignment in Total Knee Replacements. Open Orthop J. 2015;9:489-494. [27] 袁伟. 运动力学对线的人工膝关节有限元建模和生物力学研究[D]. 上海:第二军医大学,2017. [28] KLASAN A, KAPSHAMMER A, MIRON V, et al. Kinematic Alignment in Total Knee Arthroplasty Reduces Polyethylene Contact Pressure by Increasing the Contact Area, When Compared to Mechanical Alignment-A Finite Element Analysis. J Pers Med. 2022;12(8):1285. [29] SONG YD, NAKAMURA S, KURIYAMA S, et al. Biomechanical Comparison of Kinematic and Mechanical Knee Alignment Techniques in a Computer Simulation Medial Pivot Total Knee Arthroplasty Model. J Knee Surg. 2021. doi: 10.1055/s-0041-1740392. [30] JENNINGS JM, KLEEMAN-FORSTHUBER LT, BOLOGNESI MP. Medial Unicompartmental Arthroplasty of the Knee. J Am Acad Orthop Surg. 2019;27(5):166-176. [31] MURRAY DW, PARKINSON RW. Usage of unicompartmental knee arthroplasty. Bone Joint J. 2018;100-b(4):432-435. [32] MITTAL A, MESHRAM P, KIM WH, et al. Unicompartmental knee arthroplasty, an enigma, and the ten enigmas of medial UKA. J Orthop Traumatol. 2020;21(1):15. [33] ZHU GD, GUO WS, ZHANG QD, et al. Finite Element Analysis of Mobile-bearing Unicompartmental Knee Arthroplasty: The Influence of Tibial Component Coronal Alignment. Chin Med J (Engl). 2015;128(21): 2873-2878. [34] INNOCENTI B, PIANIGIANI S, RAMUNDO G, et al. Biomechanical Effects of Different Varus and Valgus Alignments in Medial Unicompartmental Knee Arthroplasty. J Arthroplasty. 2016;31(12): 2685-2691. [35] KANG KT, SON J, KWON SK, et al. Preservation of femoral and tibial coronal alignment to improve biomechanical effects of medial unicompartment knee arthroplasty: Computational study. Biomed Mater Eng. 2018;29(5):651-664. [36] KWON OR, KANG KT, SON J, et al. Biomechanical comparison of fixed- and mobile-bearing for unicomparmental knee arthroplasty using finite element analysis. J Orthop Res. 2014;32(2):338-345. [37] KOH YG, PARK KM, LEE HY, et al. Influence of tibiofemoral congruency design on the wear of patient-specific unicompartmental knee arthroplasty using finite element analysis. Bone Joint Res. 2019;8(3): 156-164. [38] HAMILTON DF, HOWIE CR, BURNETT R, et al. Dealing with the predicted increase in demand for revision total knee arthroplasty: challenges, risks and opportunities. Bone Joint J. 2015;97-b(6):723-728. [39] THIENPONT E. Revision knee surgery techniques. EFORT Open Rev. 2016;1(5):233-238. [40] SCULCO PK, ABDEL MP, HANSSEN AD, et al. The management of bone loss in revision total knee arthroplasty: rebuild, reinforce, and augment. Bone Joint J. 2016;98-b(1 Suppl A):120-124. [41] CONLISK N, HOWIE CR, PANKAJ P. Optimum stem length for mitigation of periprosthetic fracture risk following primary total knee arthroplasty: a finite element study. Knee Surg Sports Traumatol Arthrosc. 2018; 26(5):1420-1428. [42] EL-ZAYAT BF, HEYSE TJ, FANCIULLACCI N, et al. Fixation techniques and stem dimensions in hinged total knee arthroplasty: a finite element study. Arch Orthop Trauma Surg. 2016;136(12):1741-1752. [43] INNOCENTI B, BORI E, PIANIGIANI S. Biomechanical Analysis of the Use of Stems in Revision Total Knee Arthroplasty. Bioengineering (Basel). 2022;9(6):259. [44] HIRSCHMANN MT, BECKER R. The unhappy total knee replacement: a comprehensive review and management guide. Springer. 2015. [45] BARRACK RL, STANLEY T, BURT M, et al. The effect of stem design on end-of-stem pain in revision total knee arthroplasty. J Arthroplasty. 2004;19(7 Suppl 2):119-124. [46] MALHOTRA R, GARG B, KUMAR V. Dual massive skeletal allograft in revision total knee arthroplasty. Indian J Orthop. 2011;45(4):368-371. [47] STOCKLEY I, MCAULEY JP, GROSS AE. Allograft reconstruction in total knee arthroplasty. J Bone Joint Surg Br. 1992;74(3):393-397. [48] ANDERSON JA, SCULCO TP. Implant Selection in Revision Total Knee Replacement. Tech Orthop. 2011;26(2):94-98. [49] CUCKLER JM. Bone loss in total knee arthroplasty: graft augment and options. J Arthroplasty. 2004;19(4 Suppl 1):56-58. [50] VASSO M, BEAUFILS P, CERCIELLO S, et al. Bone loss following knee arthroplasty: potential treatment options. Arch Orthop Trauma Surg. 2014;134(4):543-553. [51] TSUKADA S, WAKUI M, MATSUEDA M. Metal block augmentation for bone defects of the medial tibia during primary total knee arthroplasty. J Orthop Surg Res. 2013;8:36. [52] INNOCENTI B, FEKETE G, PIANIGIANI S. Biomechanical Analysis of Augments in Revision Total Knee Arthroplasty. J Biomech Eng. 2018; 140(11):111006. [53] KANG KS, TIEN TN, LEE MC, et al. Suitability of Metal Block Augmentation for Large Uncontained Bone Defect in Revision Total Knee Arthroplasty (TKA). J Clin Med. 2019;8(3):384. |
| [1] | Peng Zhixin, Yan Wengang, Wang Kun, Zhang Zhenjiang. Finite element analysis and structural optimization design of 3D printed forearm braces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1340-1345. |
| [2] | Wu Tianliang, Tao Xiuxia, Xu Hongguang. Influence of different bone mineral densities on cage subsidence after stand-alone oblique lateral interbody fusion: three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1352-1358. |
| [3] | Liu Jinyu, Zhang Hanshuo, Cui Hongpeng, Pan Lingzhi, Zhao Boran, Li Fei, Ding Yu. Finite element biomechanical analysis of minimally invasive treatment of cervical spondylotic myelopathy and accurate exercise rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1359-1364. |
| [4] | He Yujie, Kang Zhijie, Xue Mingming, Jin Feng, Li Zhijun, Wang Xing, Xu Yangyang, Gao Mingjie, Li Jiawei, Li Xiaohe, Wang Haiyan. Finite element analysis of transarticular screw fixation of adolescent thoracic vertebra [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1365-1370. |
| [5] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [6] | Zhang Lichuang, Gao Huali, Wang Jingchao, Lin Huijun, Wu Chonggui, Ma Yinghui, Huang Yunfei, Fang Xue, Zhai Weitao. Effect of tendon manipulation with equal emphasis on muscles and bones on accelerating the functional rehabilitation of quadriceps femoris after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1383-1389. |
| [7] | Bi Gengchao, Zhang Yanlong, Li Qiuyue, Hu Longwei, Zhang Yu. Knee joint mechanics and activation characteristics of surrounding muscles during deep jumps at different heights and distances [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1211-1218. |
| [8] | Zhu Lin, Gu Weiping, Wang Can, Chen Gang. Biomechanical analysis of All-on-Four and pterygomaxillary implants under different maxillary bone conditions [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 985-991. |
| [9] | Sun Jiangwei, Wang Junxiang, Baibujiafu·Yellisi, Dai Huijuan, Nijati·Turson. Three-dimensional finite element analysis of stress distribution in different smooth collar implants [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1004-1011. |
| [10] | Jiang Yifang, Cai Qimin, Chu Zhengyi, Qin Min, Shen Yurong, Gu Yuanping. Simulation analysis of stress distribution of NRT FILES in curved root canals [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1038-1042. |
| [11] | Li Huiqin, Wang Chunjuan, Wang Yu, Wang Weifeng, Chen Dinggen, Li Na. Clear aligner orthodontic therapy of rotated mandibular teeth with different shapes: a three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1050-1054. |
| [12] | Xiong Bohan, Yu Yang, Lu Xiaojun, Wang Xu, Yang Tengyun, Zhang Yaozhang, Liao Xinyu, Zhou Xiaoxiang, He Lu, Li Yanlin. Research progress in promoting tendon to bone healing during anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 779-786. |
| [13] | Liu Qinghua, Cai Yongqiang, Jin Feng, Yu Jinghong, Wang Haiyan, Zhang Yunfeng, Wang Lidong, Li Jiawei, Wang Xing, He Yujie, Dai Lina, Wang Jianzhong, Wu Chao, Tong Ling, Kang Zhijie, Li Zhijun, Li Xiaohe. Finite element model of the 12-year-old child whole cervical spine: establishment and validity verification based on CT data [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 500-504. |
| [14] | Wei Bo, Yao Qingqiang, Tang Cheng, Li Xuxiang, Xu Yan, Wang Liming. Advantage of medial pivot prosthesis in total knee arthroplasty via medial subvastus approach [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 552-557. |
| [15] | Wan Guoli, Shi Chenhui, Wang Weishan, Li Ang, Shi Xunda, Cai Yi. Retrospective analysis of the influencing factors of chronic pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 558-564. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||