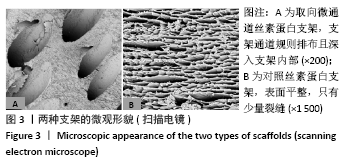
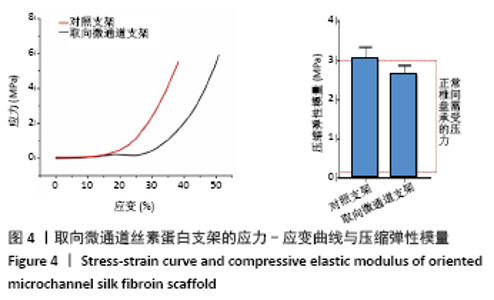
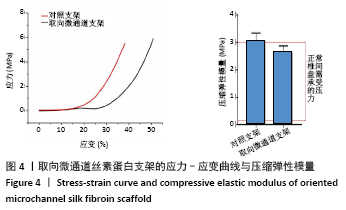
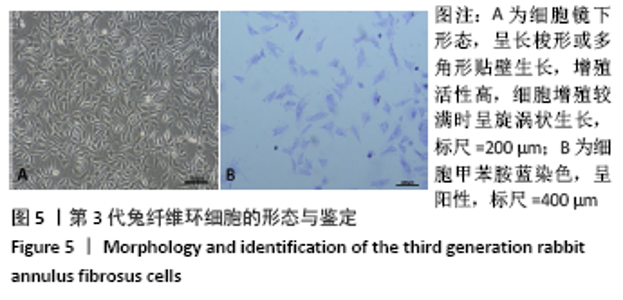
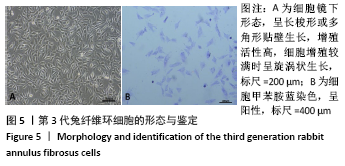
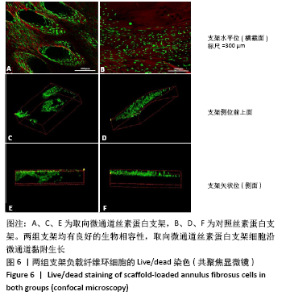
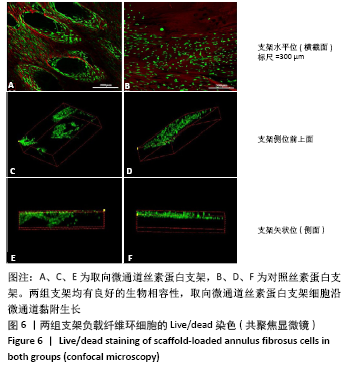
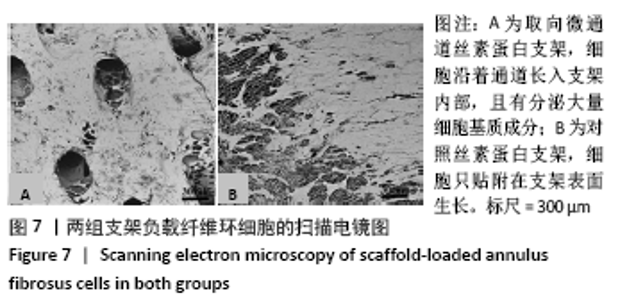
Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (4): 560-566.doi: 10.12307/2022.092
Previous Articles Next Articles
Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin
He Guanyu1, 2, Xu Baoshan2, Du Lilong2, Zhang Tongxing2, Huo Zhenxin1, 2, Shen Li1, 2
- 1Graduate School of Tianjin Medical University, Tianjin 300070, China; 2Department of Minimally Invasive Spine Surgery, Tianjin Hospital, Tianjin 300020, China
-
Received:2021-01-13Revised:2021-01-16Accepted:2021-02-18Online:2022-02-08Published:2021-11-03 -
Contact:He Guanyu, Master candidate, Graduate School of Tianjin Medical University, Tianjin 300070, China; Department of Minimally Invasive Spine Surgery, Tianjin Hospital, Tianjin 300020, China -
About author:He Guanyu, Master candidate, Graduate School of Tianjin Medical University, Tianjin 300070, China; Department of Minimally Invasive Spine Surgery, Tianjin Hospital, Tianjin 300020, China -
Supported by:the National Natural Science Foundation of China, No. 82072491, 31670983 (to XBS), No. 31900967 (to DLL); Natural Science Foundation of Tianjin, No. 19JCQNJC09300 (to DLL)
CLC Number:
Cite this article
He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
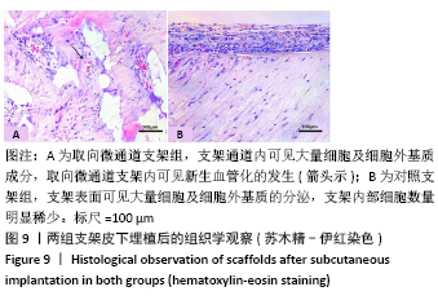
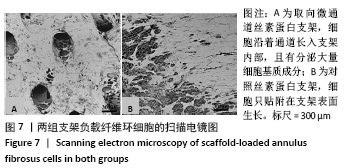
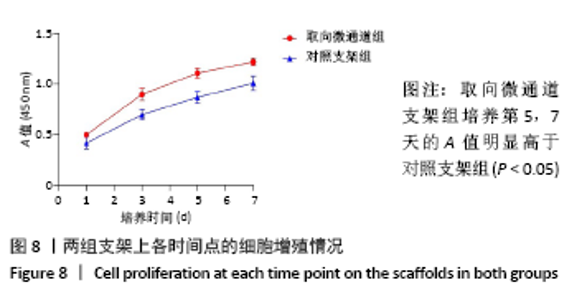
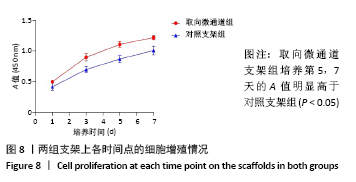
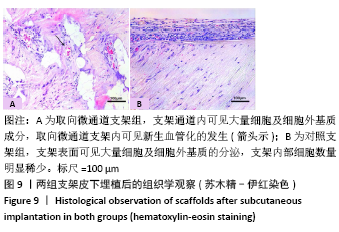
2.5 支架皮下埋植后的体内生物相容性 取材时发现,两组支架与皮下组织长合良好,支架与周围软组织包裹结合紧密不易分开,切口愈合良好没有化脓及感染发生,这初步说明了支架在体内没有发生明显的炎症反应与免疫排斥反应。 两组支架皮下埋植4 周后的苏木精-伊红染色可见,取向微通道支架组支架通道内可见大量细胞及细胞外基质成分,取向微通道支架内可见新生血管化的发生(箭头处可见血管腔内红染的红细胞);对照支架组仅在支架表面可见大量细胞及细胞外基质的分泌,支架内部细胞数量明显稀少,未见新生血管化,见图9。 2.6 支架的生物相容性 由 Live/dead染色与CCK-8实验可知,取向微通道丝素蛋白支架具有良好的细胞相容性;由皮下埋植实验的大体与组织学观察可知,取向微通道丝素蛋白支架具有良好的生物相容性。"
| [1] VOS T, FLAXMAN AD, NAGHAVI M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012; 380(9859):2163-2196. [2] HOY D, MARCH L, BROOKS P, et al. Measuring the global burden of low back pain. Best Pract Res Clin Rheumatol. 2010;24(2):155-165. [3] YANG S, ZHANG F, MA J, et al. Intervertebral disc ageing and degeneration: The antiapoptotic effect of oestrogen. Ageing Res Rev. 2020;57:100978. [4] BAO J, TANG Q, CHEN Y. Individual nursing care for the elderly among China’s aging population. Biosci Trends. 2018;11(6):694-696. [5] FRAPIN L, CLOUET J, DELPLACE V, et al. Lessons learned from intervertebral disc pathophysiology to guide rational design of sequential delivery systems for therapeutic biological factors. Adv Drug Deliv Rev. 2019;149-150:49-71. [6] ATLAS SJ, KELLER RB, WU YA, et al. Long-term outcomes of surgical and nonsurgical management of sciatica secondary to a lumbar disc herniation: 10 year results from the maine lumbar spine study. Spine (Phila Pa 1976). 2005;30(8):927-935. [7] WATTERS WC 3RD, MCGIRT MJ. An evidence-based review of the literature on the consequences of conservative versus aggressive discectomy for the treatment of primary disc herniation with radiculopathy. Spine J. 2009;9(3):240-257. [8] XU J, LIU S, WANG S, et al. Decellularised nucleus pulposus as a potential biologic scaffold for disc tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;99:1213-1225. [9] LEE KH, YUE WM, YEO W, et al. Clinical and radiological outcomes of open versus minimally invasive transforaminal lumbar interbody fusion. Eur Spine J. 2012;21(11):2265-2270. [10] SCHIZAS C, KULIK G, KOSMOPOULOS V. Disc degeneration: current surgical options. Eur Cell Mater. 2010;20:306-315. [11] SAKAI D, ANDERSSON GB. Stem cell therapy for intervertebral disc regeneration: obstacles and solutions. Nat Rev Rheumatol. 2015;11(4): 243-256. [12] DOWDELL J, ERWIN M, CHOMA T, et al. Intervertebral Disk Degeneration and Repair. Neurosurgery. 2017;80(3S):S46-S54. [13] Makhni MC, Caldwell JM, Saifi C, et al. Tissue engineering advances in spine surgery. Regen Med. 2016;11(2):211-222. [14] BOWLES RD, GEBHARD HH, HARTL R, et al. Tissue-engineered intervertebral discs produce new matrix, maintain disc height, and restore biomechanical function to the rodent spine. Proc Natl Acad Sci U S A. 2011;108(32):13106-13111. [15] DU L, YANG Q, ZHANG J, et al. Engineering a biomimetic integrated scaffold for intervertebral disc replacement. Mater Sci Eng C Mater Biol Appl. 2019;96:522-529. [16] ZHOU X, WANG J, HUANG X, et al. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration. Acta Biomater. 2018;81:115-128. [17] ALVIM VC, CESAR CP, FONTANA NN, et al. Design and optimization of biocompatible polycaprolactone/poly (l-lactic-co-glycolic acid) scaffolds with and without microgrooves for tissue engineering applications. J Biomed Mater Res A. 2018;106(6):1522-1534. [18] KELNAR I, ZHIGUNOV A, KAPRALKOVA L, et al. Facile preparation of biocompatible poly (lactic acid)-reinforced poly(epsilon-caprolactone) fibers via graphite nanoplatelets -aided melt spinning. J Mech Behav Biomed Mater. 2018;84:108-115. [19] BHUNIA BK, KAPLAN DL, MANDAL BB. Silk-based multilayered angle-ply annulus fibrosus construct to recapitulate form and function of the intervertebral disc. Proc Natl Acad Sci U S A. 2018;115(3):477-482. [20] ANITUA E, PADILLA S. Biologic therapies to enhance intervertebral disc repair. Regen Med. 2018;13(1):55-72. [21] ALINEJAD Y, ADOUNGOTCHODO A, GRANT MP, et al. Injectable Chitosan Hydrogels with Enhanced Mechanical Properties for Nucleus Pulposus Regeneration. Tissue Eng Part A. 2019;25(5-6):303-313. [22] PEREIRA CL, GONCALVES RM, PEROGLIO M, et al. The effect of hyaluronan-based delivery of stromal cell-derived factor-1 on the recruitment of MSCs in degenerating intervertebral discs. Biomaterials. 2014;35(28):8144-8153. [23] BHATTACHARJEE M, MIOT S, GORECKA A, et al. Oriented lamellar silk fibrous scaffolds to drive cartilage matrix orientation: towards annulus fibrosus tissue engineering. Acta Biomater. 2012;8(9):3313-3325. [24] NEO PY, SHI P, GOH JC, et al. Characterization and mechanical performance study of silk/PVA cryogels: towards nucleus pulposus tissue engineering. Biomed Mater. 2014;9(6): 065002. [25] 佘荣峰,张一,陈龙,等.丝素蛋白-壳聚糖支架复合骨髓间充质干细胞体内构建组织工程化软骨的生物相容性[J].中国组织工程研究,2020,24(1):27-32. [26] SHANG L, MA B, WANG F, et al. Nanotextured silk fibroin/hydroxyapatite biomimetic bilayer tough structure regulated osteogenic/chondrogenic differentiation of mesenchymal stem cells for osteochondral repair. Cell Prolif. 2020;53(11):e12917. [27] SARAVANAN S, LEENA RS, SELVAMURUGAN N. Chitosan based biocomposite scaffolds for bone tissue engineering. Int J Biol Macromol. 2016;93(Pt B):1354-1365. [28] O’BRIEN FJ, HARLEY BA, YANNAS IV, et al. Influence of freezing rate on pore structure in freeze-dried collagen-GAG scaffolds. Biomaterials. 2004;25(6):1077-1086. [29] AGRAWAL CM, RAY RB. Biodegradable polymeric scaffolds for musculoskeletal tissue engineering. J Biomed Mater Res. 2001;55(2): 141-150. [30] HOOGENDOORN RJ, LU ZF, KROEZE RJ, et al. Adipose stem cells for intervertebral disc regeneration: current status and concepts for the future. Cell Mol Med. 2008;12(6a):2205-2216. [31] CHU G, YUAN Z, ZHU C, et al. Substrate stiffness- and topography-dependent differentiation of annulus fibrosus-derived stem cells is regulated by Yes-associated protein. Acta Biomater. 2019;92:254-264 [32] ZHU M, WANG Z, ZHANG J, et al. Circumferentially aligned fibers guided functional neoartery regeneration in vivo. Biomaterials. 2015;61:85-94. [33] 夏金健,徐宝山,马信龙,等.仿生可降解PCL-PLGA纤维支架负载人脐带间充质干细胞构建组织工程纤维环[J].天津医药,2019, 47(6):594-599. [34] 张维昊,徐宝山,马信龙,等.仿生可降解组织工程纤维环支架的制备与评估[J].中国组织工程研究,2020,24(4):524-531. [35] ZHANG XY, CHEN YP, HAN J, et al. Biocompatiable silk fibroin/carboxymethyl chitosan/strontium substituted hydroxyapatite/cellulose nanocrystal composite scaffolds for bone tissue engineering. Int J Biol Macromol. 2019;136:1247-1257. [36] SARAVANAN S, LEENA RS, SELVAMURUGAN N. Chitosan based biocomposite scaffolds for bone tissue engineering. Int J Biol Macromol. 2016;93(Pt B):1354-1365. [37] JULIANO RL. Signal transduction by cell adhesion receptors and the cytoskeleton: functions of integrins, cadherins, selectins, and immunoglobulin-superfamily members. Annu Rev Pharmacol Toxicol. 2002;42:283-323. [38] CHANG G, KIM HJ, VUNJAK-NOVAKOVIC G, et al. Enhancing annulus fibrosus tissue formation in porous silk scaffolds. J Biomed Mater Res A. 2010;92(1):43-51. [39] BOWLES RD, SETTON LA. Biomaterials for intervertebral disc regeneration and repair. Biomaterials. 2017;129:54-67. |
| [1] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [2] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Hou Wanxing, Li Hongwei, Zheng Xin, Zhu Xianren. Correlation between preoperative magnetic resonance imaging findings and bone cement leakage after percutaneous vertebral augmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 504-509. |
| [6] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [7] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [8] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [9] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [10] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [11] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [12] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [13] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [14] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [15] | Li Yanting, Chen Jian, Liu Menglan, Ren Manman, Zhong Weihong, Chen Changxing. Three-dimensional finite element analysis of Daogaijinbei manipulation on lumbar intervertebral disc biomechanics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 340-343. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||