Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (1): 76-83.doi: 10.12307/2022.013
Previous Articles Next Articles
Expression and regulation of keratin 6B in CD44+ bladder cancer stem cells
Liu Lu, Yang Yuming, Shang Aichen, Liu Cui, Sun Ce, Wang Jing
- Department of Pathology, Sino-Singapore Eco-City Hospital of Tianjin Medical University, Tianjin 300456, China
-
Received:2020-11-28Revised:2020-12-08Accepted:2021-01-06Online:2022-01-08Published:2021-10-25 -
Contact:Yang Yuming, MD, Associate professor, Department of Pathology, Sino-Singapore Eco-City Hospital of Tianjin Medical University, Tianjin 300456, China -
About author:Liu Lu, Master candidate, Department of Pathology, Sino-Singapore Eco-City Hospital of Tianjin Medical University, Tianjin 300456, China -
Supported by:the Science and Technology Project of Tianjin Binhai New District Health and Family Planning Commission, No. 2017BWKY033 (to YYM)
CLC Number:
Cite this article
Liu Lu, Yang Yuming, Shang Aichen, Liu Cui, Sun Ce, Wang Jing. Expression and regulation of keratin 6B in CD44+ bladder cancer stem cells[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 76-83.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

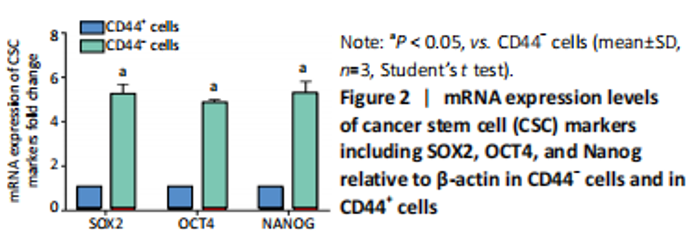
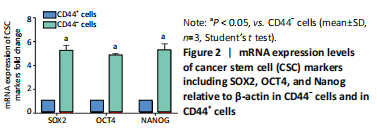
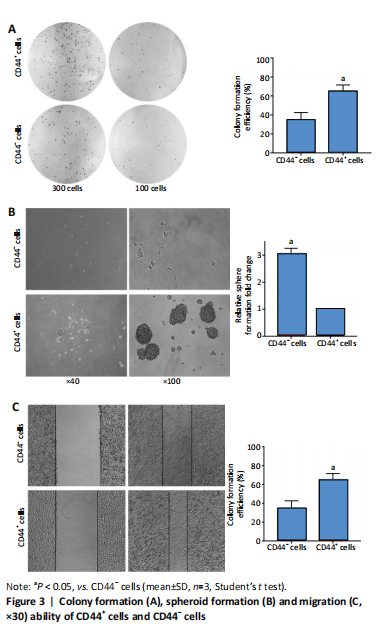
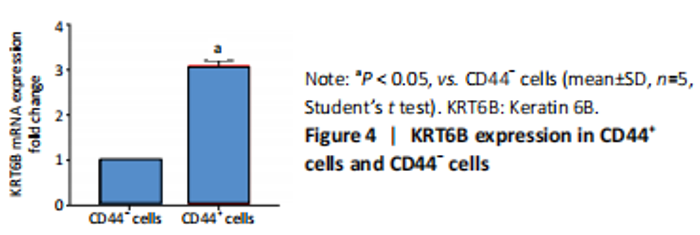
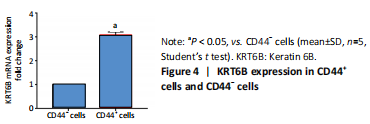
High expression of KRT6B in CD44+ bladder cancer stem cells (in vitro) Initially, the cell suspension was prepared by digesting the 10 bladder cancer samples. The CD44v6+EMA- bladder cancer stem cells were isolated by magnetic bead cell sorting. The results of transcriptome sequencing compared the expression profiles of the bladder cancer genes in the CD44v6+EMA- BCSCs, control subjects, and the KRT6B expression levels in BCSCs were significantly higher. To explore the expression of KRT6B in bladder cancer cell lines, the CD44+ cells were isolated by magnetic bead cell sorting from 5637 cell line. RT-PCR assay was used for detection of CD44 expression after magnetic sorting (P < 0.05; Figure 1). RT-PCR results showed that CD44+ cells expressed higher level of CSCs-related genes (Sox2, Oct4, and Nanog; P < 0.05; Figure 2). In the next step, colony formation assays indicated a higher proliferation and colony formation ability of CD44+ cells (P < 0.05; Figure 3A). Sphere formation assays showed that CD44+ BCSCs could form larger and more spheres than CD44- bladder cancer cells (P < 0.05; Figure 3B). It is demonstrated that CD44+ cells possessed enhanced self-renewal ability. Wound healing assays showed that the migration capacity of CD44+ cells was higher compared with the control group (P < 0.05; Figure 3C). A series of experiments showed that the CD44+ cells possessed the characteristic of BCSC populations, including self-renewal and spheroid formation capacity. Subsequently, the expression of KRT6B in CD44+ BCSCs and CD44- bladder cancer cells was analyzed. RT-PCR indicated that KRT6B was enriched in CD44+ cells of 5637 human bladder cancer cell line (P < 0.05; Figure 4). These results indicated that the KRT6B expression trend remained consistent between RNA sequencing and RT-PCR."


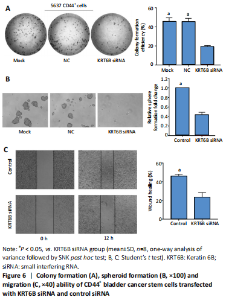
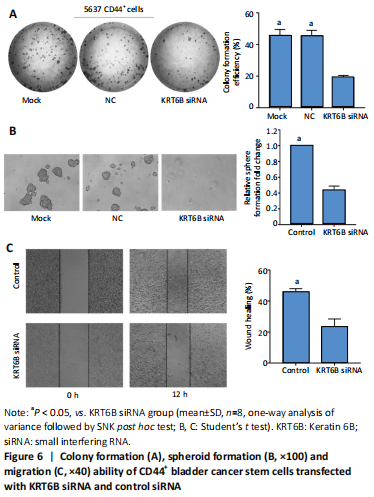
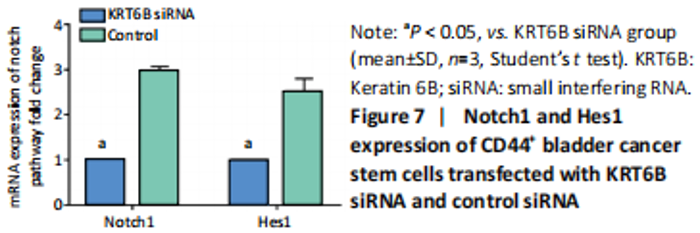
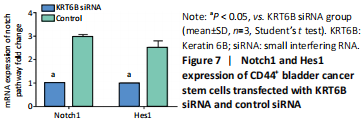
Silencing KRT6B in CD44+ BCSCs inhibited spheroid formation ability (in vitro) We explored the role of KRT6B in the CD44+ bladder cancer stem-like properties by silencing KRT6B in CD44+ BCSCs. KRT6B was knockdown by siRNA in CD44+ BCSCs and the downregulation of KRT6B was confirmed by RT-PCR (P < 0.05; Figure 5). Colony formation assays showed that KRT6B knockdown in CD44+ BCSCs inhibited proliferation and colony formation of CD44+ BCSCs (P < 0.05; Figure 6A). Sphere formation assay indicated that silence of KRT6B expression decreased the spheroid formation and self-renewal ability in CD44+ BCSCs (P < 0.05; Figure 6B). Wound healing assay displayed that ability of migration in KRT6B siRNA cells was decreased compared with the control group (P < 0.05; Figure 6C). This suggested that the downregulated KRT6B inhibited cell self-renewal and spheroid formation ability in CD44+ bladder cancer stem cells. To further explore the effect on a cellular level, we assessed the effect of KRT6B down-regulation in CD44- cells. Next, we aimed to initially explore the potential mechanism by which KRT6B maintains stemness in BCSCs. The RT-PCR analysis demonstrated that the silence of KRT6B in CD44+ cells increased the expression of NOTCH1 and HES1 in mRNA level (P < 0.05; Figure 7). Collectively, we speculated that KRT6B maintained the CD44+ BCSCs stemness which might be related to the Notch signal pathway."

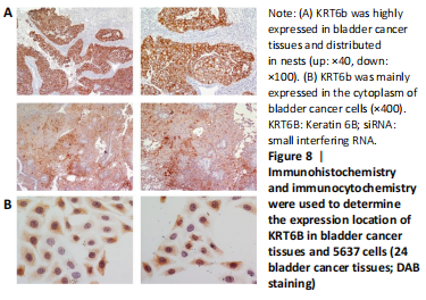
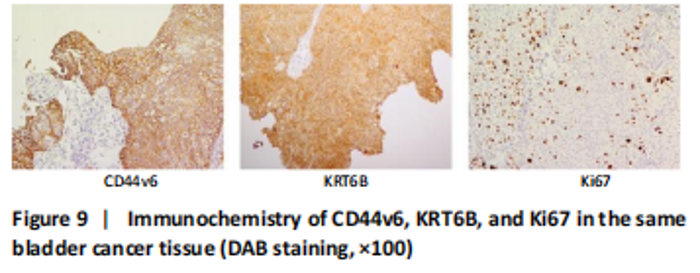
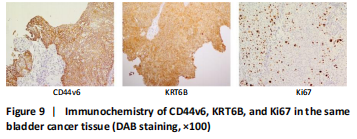
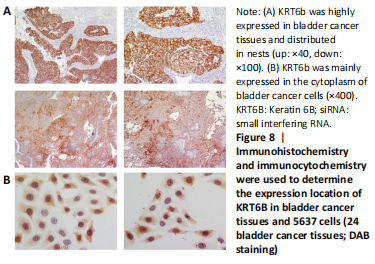
Expression of KRT6B in immunohistochemistry and immunocytochemistry (in vitro) Immunohistochemistry was performed in 24 bladder cancer tissues to analyze the expression of CD44v6 and KRT6B. Immunohistochemical analysis showed that the cells with KRT6B expression located in the basal and intermediate layer of normal bladder uroepithelium. During uroepithelium carcinoma differentiation, immunohistochemical analysis indicated that basal cells expressed KRT6B and KRT6B+ cells distributed in nest-pattern in bladder cancer (Figure 8A). Immunocytochemistry analysis showed that KRT6B was mainly located in the cytoplasm and expressed in different subsets of the bladder cancer cells (Figure 9B). It is proposed that CD44v6+ cells located in the basal layer of bladder cancer. Immunohistochemical analysis confirmed co-expression of CD44v6 and KRT6B defined basal layer cells in bladder cancer and expanded from basal towards differentiated layer. Furthermore, it was observed that KRT6B expression was correlated with CD44v6 expression in bladder cancer specimens (n=24, P=0.006, Bilateral, Fisher’s exact test; Table 3). In summary, these results showed that the expression of KRT6B was correlated with CD44v6 and KRT6B+ cells, which might be related to CD44+ BCSCs."
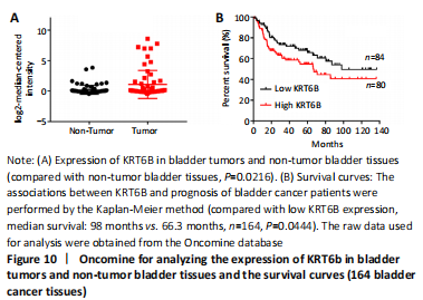
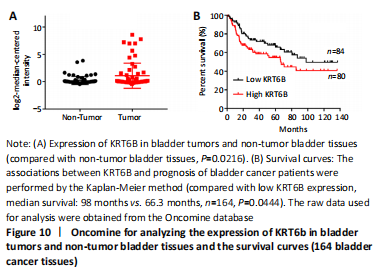
Expression and prognosis of KRT6B in normal bladder tissue and bladder cancer (in vitro) To further explore the effect of KRT6B expression on the clinical outcome of bladder cancer patients, we used the Lee database from ONCOMINE aimed at mining bladder gene information. We analyzed the expression of KRT6B at the protein level by GraphPad Prism 7.0 statistical software. The result displayed that the expression of KRT6B in bladder cancer tissues was significantly increased compared with normal bladder tissues (P=0.022; Figure 10A). Subsequently, we evaluated the possible association between KRT6B expression and the prognostic role of KRT6B in human bladder cancer tissues. The result showed that bladder cancer patients with high KRT6B expression had a shorter overall survival rate than that of patients with low KRT6B expression (median survival=66.3 months vs. 98 months, n=164, P=0.044; Figure 10B). Therefore, these results indicate that the high expression of KRT6B is related to the poor prognosis of bladder cancer patients. "
| [1] BAJAJ J, DIAZ E, REYA T. Stem cells in cancer initiation and progression. J Cell Biol. 2020;219(1):e201911053. [2] AZZARELLI R. Organoid models of glioblastoma to study brain tumor stem cells. Front Cell Dev Biol. 2020;8:220. [3] JAHANAFROOZ Z, MOSAFER J, AKBARI M, et al. Colon cancer therapy by focusing on colon cancer stem cells and their tumor microenvironment. J Cell Physiol. 2020;235(5):4153-4166. [4] XIAOBIN Z, CHENGXIAO L, JIE Y, et al. Breast cancer stem cells, heterogeneity, targeting therapies and therapeutic implications. Pharmacol Res. 2020;163: 105320. [5] COLES GL, CRISTEA S, WEBBER JT, et al. Unbiased proteomic profiling uncovers a targetable GNAS/PKA/PP2A axis in small cell lung cancer stem cells. Cancer Cell. 2020;38(1):129-143. [6] BARBATO L, BOCCHETTI M, DI BIASE A, et al. Cancer stem cells and targeting strategies. Cells. 2019;8(8):926. [7] AFIFY SM, CALLE AS, HASSAN G, et al. A novel model of liver cancer stem cells developed from induced pluripotent stem cells. Br J Cancer. 2020; 122(9):1378-1390. [8] O’CONOR CJ, CHEN T, GONZÁLEZ I, et al. Cancer stem cells in triple-negative breast cancer: a potential target and prognostic marker. Biomark Med. 2018;12(7):813-820. [9] GZIL A, ZARĘBSKA I, BURSIEWICZ W, et al. Markers of pancreatic cancer stem cells and their clinical and therapeutic implications. Mol Biol Rep. 2019;46(6):6629-6645. [10] NAGARE RP, SNEHA S, KRISHNAPRIYA S, et al. ALDH1A1+ ovarian cancer stem cells co-expressing surface markers CD24, EPHA1 and CD9 form tumours in vivo. Exp Cell Res. 2020;392(1):112009. [11] ALMANAA TN, GEUSZ ME, JAMASBI RJ. A new method for identifying stem-like cells in esophageal cancer cell lines. J Cancer. 2013;4(7):536. [12] HUNTLY BJP, GILLILAND DG. Leukaemia stem cells and the evolution of cancer-stem-cell research. Nat Rev Cancer. 2005;5(4):311-321. [13] ZHU J, HUANG G, HUA X, et al. CD44s is a crucial ATG7 downstream regulator for stem-like property, invasion, and lung metastasis of human bladder cancer (BC) cells. Oncogene. 2019;38(17):3301-3315. [14] CHAN KS, ESPINOSA I, CHAO M, et al. Identification, molecular characterization, clinical prognosis, and therapeutic targeting of human bladder tumor-initiating cells. Proc Natl Acad Sci U S A. 2009;106(33): 14016-14021. [15] TYNER AL, EICHMAN MJ, FUCHS E. The sequence of a type II keratin gene expressed in human skin: conservation of structure among all intermediate filament genes. Proc Natl Acad Sci U S A. 1985;82(14):4683-4687. [16] FUCHS EV, COPPOCK SM, GREEN H, et al. Two distinct classes of keratin genes and their evolutionary significance. Cell. 1981;27(1):75-84. [17] LIU YR, YIN PN, SILVERS CR, et al. Enhanced metastatic potential in the MB49 urothelial carcinoma model. Sci Rep. 2019;9(1):1-9. [18] DYRSKJØT L, KRUHØFFER M, THYKJAER T, et al. Gene expression in the urinary bladder: a common carcinoma in situ gene expression signature exists disregarding histopathological classification. Cancer Res. 2004;64(11):4040-4048. [19] HOGGARTH ZE, OSOWSKI DB, SLUSSER-NORE A, et al. Enrichment of genes associated with squamous differentiation in cancer initiating cells isolated from urothelial cells transformed by the environmental toxicant arsenite. Toxicol Appl Pharmacol. 2019;374:41-52. [20] HOGGARTH ZE, OSOWSKI DB, FREEBERG BA, et al. The urothelial cell line UROtsa transformed by arsenite and cadmium display basal characteristics associated with muscle invasive urothelial cancers. PLoS One. 2018;13(12):e0207877. [21] SJÖDAHL G, LAUSS M, LÖVGREN K, et al. A molecular taxonomy for urothelial carcinoma. Clin Cancer Res. 2012;18(12):3377-3386. [22] GU C, WANG Z, ZHOU N, et al. Mettl14 inhibits bladder TIC self-renewal and bladder tumorigenesis through N 6-methyladenosine of Notch1. Mol Cancer. 2019;18(1):1-12. [23] XU Z, HUANG B, ZHANG Q, et al. NOTCH1 regulates the proliferation and migration of bladder cancer cells by cooperating with long non‐coding RNA HCG18 and microRNA‐34c‐5p. J Cellular Biochem. 2019;120(4):6596-6604. [24] ARSHI A, RAEISI F, MAHMOUDI E, et al. A comparative study of HOTAIR expression in breast cancer patient tissues and cell lines. Cell J. 2020;22(2): 31721532. [25] LEE JS, LEEM SH, LEE SY, et al. Expression signature of E2F1 and its associated genes predict superficial to invasive progression of bladder tumors. J Clin Oncol. 2010;28(16):2660-2667. [26] SIEGEL RL, MILLER KD, JEMAL A. Cancer statistics, 2020. CA: a cancer journal for clinicians. 2020;70(1):7-30. [27] OHISHI T, KOGA F, MIGITA T. Bladder cancer stem-like cells: their origin and therapeutic perspectives. Int J Mol Sci. 2016;17(1):43. [28] BRANDT WD, MATSUI W, ROSENBERG JE, et al. Urothelial carcinoma: stem cells on the edge. Cancer Metastasis Rev. 2009;28(3):291-304. [29] JUNG M, KIM B, MOON KC. Immunohistochemistry of cytokeratin (CK) 5/6, CD44 and CK20 as prognostic biomarkers of non-muscle-invasive papillary upper tract urothelial carcinoma. Histopathology. 2019;74(3):483-493. [30] WU CT, LIN WY, CHEN WC, et al. Predictive Value of CD44 in Muscle-Invasive Bladder Cancer and Its Relationship with IL-6 Signaling. Ann Surg Oncol. 2018;25(12):3518-3526. [31] YANG YM, CHANG JW. Bladder cancer initiating cells (BCICs) are among EMA-CD44v6+ subset: novel methods for isolating undetermined cancer stem (initiating) cells . Cancer Invest. 2008;26(7):725-733. [32] WANG F, CHEN S, LIU HB, et al. Keratin 6 regulates collective keratinocyte migration by altering cell–cell and cell–matrix adhesion. J Cell Biol. 2018; 217(12):4314-4330. [33] WONG P, COULOMBE PA. Loss of keratin 6 (K6) proteins reveals a function for intermediate filaments during wound repair. J Cell Biol. 2003;163(2): 327-337. [34] VOLKMER JP, SAHOO D, CHIN RK, et al. Three differentiation states risk-stratify bladder cancer into distinct subtypes. Proc Natl Acad Sci U S A. 2012;109(6):2078-2083. [35] MAZZUCCHELLI R, SCARPELLI M, GALOSI AB, et al. Pathology of upper tract urothelial carcinoma with emphasis on staging. Int J Immunopathol Pharmacol. 2014;27(4):509-516. [36] SESTERHENN AM, MANDIC R, DüNNE AA, et al. Cytokeratins 6 and 16 are frequently expressed in head and neck squamous cell carcinoma cell lines and fresh biopsies . Anticancer Res. 2005;25(4):2675-2680. [37] LIM SC, PARAJULI KR, HAN SI. Keratin 6, induced by chronic cisplatin exposure, confers chemoresistance in human gastric carcinoma cells. Oncol Rep. 2019;42(2):797-804. [38] KUDO I, ESUMI M, KUSUMI Y, et al. Particular gene upregulation and p53 heterogeneous expression in TP53-mutated maxillary carcinoma. Oncol Lett. 2017;14(4):4633-4640. [39] TSAI FJ, LAI MT, CHENG J, et al. Novel K6-K14 keratin fusion enhances cancer stemness and aggressiveness in oral squamous cell carcinoma. Oncogene. 2019;38(26):5113-5126. [40] FLESKEN-NIKITIN A, HWANG CI, CHENG CY, et al. Ovarian surface epithelium at the junction area contains a cancer-prone stem cell niche. Nature. 2013; 495(7440):241-245. [41] HU J, ZHANG LC, SONG X, et al. KRT6 interacting with notch1 contributes to progression of renal cell carcinoma, and aliskiren inhibits renal carcinoma cell lines proliferation in vitro. Int J Clin Exp Pathol. 2015;8(8):9182-9188. [42] ARUMUGAM A, WENG Z, CHAUDHARY SC, et al. Keratin-6 driven ODC expression to hair follicle keratinocytes enhances stemness and tumorigenesis by negatively regulating Notch. Biochem Biophys Research Commun. 2014;451(3):394-401. [43] Abdou AG, El-Wahed MMA, Kandil MAE, et al. Immunohistochemical analysis of the role and relationship between Notch‐1 and Oct‐4 expression in urinary bladder carcinoma. Apmis. 2013;121(10):982-996. [44] LU N, PIAO MH, FENG CS, et al. Isoflurane promotes epithelial-to-mesenchymal transition and metastasis of bladder cancer cells through HIF-1α-β-catenin/Notch1 pathways. Life Sci. 2020;258:118154. [45] MARAVER A, FERNANDEZ-MARCOS PJ, CASH TP, et al. NOTCH pathway inactivation promotes bladder cancer progression. J Clin Invest. 2015;125(2): 824-830. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [3] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1033-1039. |
| [4] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1040-1046. |
| [5] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1053-1059. |
| [6] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1060-1067. |
| [7] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1183-1190. |
| [8] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1191-1197. |
| [9] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1074. |
| [10] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1075-1079. |
| [11] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1085. |
| [12] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1086-1092. |
| [13] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1098. |
| [14] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1099-1104. |
| [15] | Xiong Tinglin, Ying Menghui, Zhang Lisha, Zhang Xiaogang, Yang Yan. Electrophysiological characteristics of cardiomyocytes differentiated from induced pluripotent stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1111-1115. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||