Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (5): 753-759.doi: 10.3969/j.issn.2095-4344.1885
Previous Articles Next Articles
Application of bisphosphonates in avascular necrosis of the femoral head
Ge Juncheng1, Ma Jinhui2, Wang Bailiang1, Yue Debo2, Sun Wei2, Wang Weiguo2, Guo Wanshou2, Li Zirong2
- 1Sino-Japan Friendship Hospital, Peking University, Beijing 100020, China; 2First Department of Orthopedics, Sino-Japan Friendship Hospital, Beijing 100020, China
-
Received:2019-04-27Revised:2019-04-30Accepted:2019-06-27Online:2020-02-18Published:2020-01-10 -
Contact:Wang Bailiang, MD, Associate professor, Master’s supervisor, Sino-Japan Friendship Hospital, Peking University, Beijing 100020, China -
About author:Ge Juncheng, Master candidate, Sino-Japan Friendship Hospital, Peking University, Beijing 100020, China Ma Jinhui, MD, First Department of Orthopedics, Sino-Japan Friendship Hospital, Beijing 100020, China Ge Juncheng and Ma Jinhui contributed equally to this paper. -
Supported by:the National Natural Science Foundation of China, No. 81772350; Project of China-Japan Friendship Hospital, No. 2018-1-QN-9
CLC Number:
Cite this article
Ge Juncheng, Ma Jinhui, Wang Bailiang, Yue Debo, Sun Wei, Wang Weiguo, Guo Wanshou, Li Zirong. Application of bisphosphonates in avascular necrosis of the femoral head[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 753-759.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
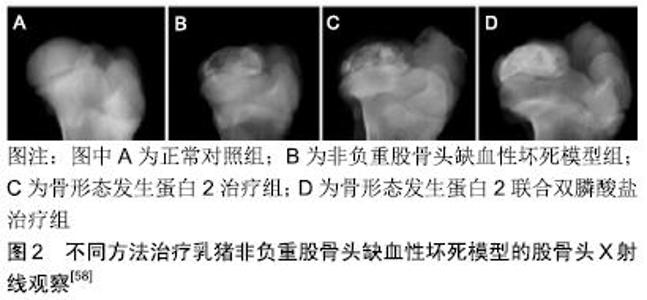
2.1 双膦酸盐的化学结构及发展历史 双膦酸盐是天然焦磷酸盐的一种人工合成类似物,最早于20世纪60年代应用于临床[12]。与焦磷酸盐不同的是,双膦酸盐分子由1个中间碳原子取代氧原子与2个磷酸根相连接,这使其具有抗水解的作用并能在体内保持一定的稳定性[13]。同时,碳原子形成R1和R2两个侧链基团,两个侧链基团的结构影响双膦酸盐对骨基质的亲和力,以及抗骨吸收作用的强度和效力[14]。对碳原子侧链结构的研究推动着双膦酸盐类药物的不断发展。第一代双膦酸盐伊替膦酸钠的侧链为羟基,不含氮原子;第二代药物氯替磷酸盐在侧链基团中加入了氮原子,使其比第一代药物的抗骨吸收作用强度增加了10-100倍[15];第三代药物阿仑磷酸盐和伊班磷酸盐侧链含有脂质及单个氮原子,其抗骨吸收效果进一步增强;目前为止作用最强的第三代双膦酸盐唑来膦酸钠,其侧链结构中含有咪唑环和2个氮原子[5]。相信随着对双膦酸盐药物侧链结构的不断探索,其抗骨吸收作用也将会进一步提高。 2.2 双膦酸盐治疗股骨头缺血性坏死的作用原理及机制 关于股骨头缺血性坏死发生发展的病理生理机制仍不明确,已发现的病理学机制包括股骨头血液供应受损、脂质代谢异常或脂肪栓塞、骨细胞死亡、机械应力负荷增加、骨内压升高及骨重塑失衡,骨重塑失衡在股骨头缺血性坏死的发生发展过程中起到关键的作用。股骨头缺血性坏死发生后可见缓慢的新骨形成、过度的骨吸收以及纤维结缔组织代替新骨等病理性的坏死骨修复过程,这意味着在股骨头缺血性坏死修复中骨重塑出现失衡[2-4]。 双膦酸盐与骨骼羟磷灰石结合力高,能特异性结合到骨转换活跃的骨表面上,抑制破骨细胞活性及其功能,从而抑制骨吸收并提高骨矿物密度[5-6]。在双膦酸盐结合到骨矿盐之后,活化的破骨细胞引起磷酸盐质子化,降低其与骨骼中钙离子的结合力,使双膦酸盐在破骨细胞介导的骨吸收过程中释放[16]。紧接着破骨细胞通过液相胞饮机制使双膦酸盐连同降解的基质蛋白内 化[17]。一旦内化,双膦酸盐便诱导破骨细胞内产生多种形态学改变,包括:皱褶缘的缩小或完全消失、细胞骨架结构的分裂及介导破骨细胞与骨表面接触的F-肌动蛋白环结构消失[18-19]。最终,双膦酸盐导致破骨细胞凋亡,起到抑制骨吸收的作用。此外,含氮双膦酸盐可通过干扰小分子GTPase的功能影响破骨细胞的形态和功能,诱导其凋亡,进而抑制其骨吸收活性[20]。不含氮双膦酸盐可在细胞内被多种胞内酶代谢为非水解性ATP类似物,这些毒性ATP类似物能够通过抑制ATP依赖的胞内酶诱导破骨细胞凋亡,进而抑制骨吸收[21-24]。同时一些动物实验指出,双膦酸盐不仅能够抑制成熟的破骨细胞,还可通过干扰破骨细胞的分化来降低破骨细胞生成,机制可能与双膦酸盐减少破骨细胞前体细胞生成及干扰其功能有关[13,25-27]。 大量体内外研究已证实双膦酸盐能够抑制骨细胞凋亡[26,29],质膜中连接蛋白43半通道的开放,大量钙内流引起的胞外信号调控激酶磷酸化在其中发挥了重要作用[28-29]。此外,双膦酸盐对成骨细胞既有抑制作用也有激活作用,目前存在一定争议。大量研究指出,低浓度双膦酸盐能够通过钙离子通道开放及胞外信号调控激酶激活机制促进成骨细胞增殖和形成,还可促进间充质干细胞向成骨细胞分化[7,30-31]。同时体外研究显示,双膦酸盐还能够通过激活肿瘤坏死因子α转换酶来降低人成骨细胞中RANKL表达、增加骨保护素的表达来达到抗骨吸收效应[32-33],RANK主要由骨髓基质细胞和成骨细胞分泌,RANK是RANKL的受体,破骨细胞系所有的细胞均表达RANK。成骨细胞、基质细胞通过分泌RANKL与破骨细胞表面的RANKL结合,激活受体 RANK的效应器肿瘤坏死因子受体相关因子 TARF,TARF激活下游的转录因子如PI3K、AKT/PKB、TAK1和MAPK等,这些转录因子可以诱导NFAT家族成员NFATc1的表达,NFATc1可以调节破骨细胞的特定基因,刺激破骨细胞的分化及增强其骨吸收活性。而骨保护素是RABKL可溶性诱骗受体,也是由成骨细胞、骨基质细胞产生分泌到胞外,骨保护素与 RANKL竞争性结合,从而阻碍RANK与RANKL的结合,进而抑制RANK/RANKL的功能[9]。然而一些体外研究显示,高浓度双膦酸盐具有毒性作用,能够对成骨细胞功能和生存产生抑制作用[34-35]。一般情况下,双膦酸盐浓度在10-9-10-6 mol/L之间对成骨细胞增殖产生促进作用,而浓度高于10-5 mol/L时则产生抑制作用[36]。总而言之,尽管双膦酸盐可能对成骨细胞产生抑制与激活的双重效应,但骨形成与骨吸收之间的关系是决定双膦酸盐对体内骨重塑作用的关键因素。 2.3 双膦酸盐的药代动力学 双膦酸盐的用药方式为口服或者静脉。口服用药后主要通过细胞旁途径以被动扩散方式在小肠进行转运[37],由于较低的脂溶性和具有较高的负电荷,口服给药的生物利用率较低,仅为单次给药剂量的1%-10%。当静脉给药时,最大血浆药物浓度较高,但循环中的半衰期较短[13]。几乎50%循环中的双膦酸盐迅速被骨骼摄取,骨骼摄取双膦酸盐的程度取决于性别、年龄、骨吸收率及成分的不同[13]。骨骼中双膦酸盐的半衰期很长,可长达数十年,没有被骨骼摄取的双膦酸盐则在几小时内以原形从尿液中排出[38]。 2.4 双膦酸盐的药物相互作用 双膦酸盐与氨基糖苷类抗生素、非类固醇类消炎镇痛药、铁剂、抗酸药或含二价金属离子的药物联合应用会降低其生物利用度,应尽量避免与其联合应用。此外,不得同时应用2种及以上的双膦酸盐药物,以减少不良反应的发生[39]。 2.5 双膦酸盐治疗股骨头缺血性坏死的研究进展 非创伤性股骨头缺血性坏死是一种致残率极高的骨科疑难病,多发生在中青年[1]。如不及早诊断及进行有效的治疗,病情将逐渐进展,股骨头最终将变形、塌陷及出现继发髋关节骨关节炎改变。股骨头缺血性坏死发生的病因可能与大剂量使用激素、长期饮酒、感染、减压病、骨髓病变、凝血机制障碍、器官移植、自身免疫性疾病及血红蛋白疾病(如镰状细胞疾病)、Gaucher病、白血病、系统性红斑狼疮、血液透析、妊娠、血液高凝状态等有关[40-41]。目前关于股骨头缺血性坏死发生发展的病理生理机制仍不明确,已发现的病理学机制包括股骨头血液供应受损、脂质代谢异常或脂肪栓塞、骨细胞死亡、机械应力负荷增加、骨内压升高以及骨重塑失衡[2,40]。其中,骨重塑失衡在股骨头缺血性坏死的发生发展过程中起到关键作用。股骨头缺血性坏死发生后可见缓慢的新骨形成、过度的骨吸收及纤维结缔组织代替新骨等病理性的坏死骨修复过程,这意味着在股骨头缺血性坏死修复中骨重塑出现失衡[2-4]。由于病理性骨重塑引起的坏死修复区域软骨下骨机械应力降低,将最终导致股骨头的塌陷。因此,如果在股骨头缺血性坏死修复过程中抑制破骨吸收,同时增加新骨形成,将有利于坏死骨更好地修复,延缓甚至避免股骨头缺血性坏死的进展。 2.5.1 系统应用双膦酸盐治疗股骨头缺血性坏死 双膦酸盐是人工合成的天然焦磷酸盐类似物,与骨骼中羟磷灰石结合力强,不仅能够抑制破骨细胞介导的骨吸收,还能够促进成骨细胞介导的新骨形成,抑制骨转化[8,42-43],从理论上讲能够用于治疗股骨头缺血性坏死。双膦酸盐常用的用药方式为口服或静脉的系统给药途径。系统应用双膦酸盐治疗大鼠、新西兰大白兔、比格犬及乳猪等股骨头坏死动物模型已被证实能够抑制骨吸收,增加骨小梁密度和厚度,改善股骨头外形,延缓股骨头塌陷及影像学进展[5]。同时大量研究显示,临床上系统应用双膦酸盐治疗股骨头缺血性坏死也取得了积极显著的效果[44-48]。LAI等[44]对40例股骨头缺血性坏死患者(SteinbergⅡ期或者Ⅲ期,且坏死灶大于30%)进行了一项随机对照试验,试验组患者每周口服70 mg阿仑膦酸钠,持续25周,对照组口服安慰剂,最短随访24个月,结果指出阿仑膦酸钠能够预防股骨头缺血性坏死早期塌陷,降低全髋关节置换发生率。同样,LUO等[49]的系统综述也支持这一观点。更进一步,AGARWALA等[48]纳入了40例(53髋,FicatⅠ期15髋,Ⅱ期19髋,Ⅲ期19髋)股骨头缺血性坏死患者,每日口服10 mg阿仑膦酸钠,持续3年,进行了为期10年的长期随访,结果发现口服阿仑膦酸钠可延缓股骨头缺血性坏死的自然病程,与其他文献报道的未经治疗的股骨头缺血性坏死患者相比明显降低了临床失败率、影像学进展率及塌陷率。 然而,有一些研究对双膦酸盐治疗股骨头缺血性坏死的效果持有相反意见。CHEN等[50]对52例股骨头缺血性坏死患者进行了一项多中心、前瞻性、随机、双盲临床试验,他们发现阿仑膦酸钠对推迟全髋关节置换时间及延缓股骨头缺血性坏死疾病进展方面无明显效果。此外,LEE等[51]进行了一项前瞻性、随机、对照、多中心试验,他们将110例SteinbergⅠ期或Ⅱ期且坏死灶面积大于30%的患者(110髋)随机分为静脉应用唑来膦酸组和对照组,经过最少2年的随访,发现唑来膦酸组29髋出现塌陷大于2 mm,而对照组22髋出现塌陷大于2 mm(P > 0.05);唑来膦酸组19髋和对照组20髋分别需行全髋关节置换(P > 0.05),他们得出双膦酸盐并不能阻止股骨头塌陷及降低全髋置换发生率。YUAN等[52]纳入5篇随机对照试验的Meta分析指出,双膦酸盐并不能改善术后髋关节评分、推迟股骨头缺血性坏死塌陷的进展及降低全髋置换发生率,并不推荐用来治疗股骨头缺血性坏死。此外,长期系统应用双膦酸盐不但会引起如股骨非典型骨折、下颌骨坏死等不良反应,还会使正在发育中的骨骼暴露于药物,影响长骨生长及未成熟骨的塑形[10-11]。尽管双膦酸盐治疗股骨头缺血性坏死存在一定争议,目前临床上仍把双膦酸盐作为辅助其他保髋方法治疗股骨头缺血性坏死的一项措施。GIANAKOS等[53]在2006至2014年期间纳入了49例股骨头缺血性坏死患者,其中29例患者单独应用双膦酸盐,20例患者联合应用髓芯减压干细胞植入和双膦酸盐,平均随访2年得结果显示,联合治疗方案能够延缓早期股骨头缺血性坏死的塌陷进展。KANG等[54]将早期股骨头缺血性坏死患者随机分为单纯髓芯减压组(46例患者,60髋)和髓芯减压联合口服阿仑膦酸钠组(47例患者,67髋),最少随访4年,结论是联合组能够减轻髋关节疼痛,延缓早期股骨头缺血性坏死的进展;即使在FicatⅡA和Ⅲ期病例,联合组也可以推迟全髋关节置换的时间。但另一项研究指出与单纯应用体外冲击波相比,体外冲击波联合阿仑膦酸钠治疗股骨头缺血性坏死在髋关节疼痛、功能改善,全髋关节置换发生率及坏死灶影像学进展率方面无明显优势,联合方案短期疗效并不理想[55]。鉴于双膦酸盐治疗股骨头缺血性坏死目前存在较大争论,未来仍需大样本的随机双盲多中心的临床试验来进一步明确双膦酸盐治疗股骨头缺血性坏死的确切疗效、适应证及最佳的用药方案。 2.5.2 局部应用双膦酸盐治疗股骨头缺血性坏死 股骨头缺血性坏死的治疗对骨科医生来说仍然是一种挑战。股骨头缺血性坏死的治疗目标是预防股骨头塌陷,保存关节功能,延缓全髋关节置换的时间。目前有各种各样的外科手术治疗方法,包括股骨头髓芯减压、病灶刮除植骨手术、截骨手术、钽棒植入术及带或不带血管蒂的腓骨、髂骨移植手术等[56],其中以病灶清除、植骨为主的保髋手术仍是治疗早中期股骨头坏死的主要方法。但研究显示保髋术后股骨头坏死塌陷的进展与局部成骨不足、术后植骨吸收过快均有关[57]。因此抑制局部破骨过快吸收与增加成骨同步进行有利于坏死骨更好地修复,延缓股骨头塌陷,使术后股骨头内的骨重塑平衡。 近期对局部应用双膦酸盐治疗股骨头缺血性坏死的实验研究逐渐增多。大量体外研究已显示双膦酸盐能够促进成骨细胞增殖、分化和形成,起到积极的作用[7,32-33]。同时,体内研究也发现局用应用双膦酸盐不仅能够抑制破骨细胞介导的骨吸收,还可以增加成骨细胞诱导的骨生成,但与浓度相关[8,11,43]。丹麦奥胡斯骨科研究中心的系列研究发现,局部应用唑来膦酸浸泡处理后的同种异体骨可有效降低移植骨的吸收,且不影响成骨水平[8];同时他们还发现低浓度的唑来膦酸(0.005 g/L)不仅可有效抑制破骨,而且还促进成骨。VANDERMEER等[11]在小猪Legg-Perthes病模型中发现,局部同时植入双膦酸盐和骨形态发生蛋白2(560 μg伊班膦酸盐和1 mg重组人骨形态发生蛋白2溶于1 mL生理盐水,经皮注射到股骨头内)的股骨头坏死修复效果最佳,联合用药组能够明显减轻股骨头变形,同时刺激股骨头内新骨形成。另外,KIM 等[58]在乳猪非负重股骨头缺血性坏死模型中发现,股骨头局部联合应用双膦酸盐(0.6 mg伊班膦酸盐溶于0.6 mL生理盐水中)与骨形态发生蛋白2(0.6 mg溶于 0.6 mL生理盐水)能够明显减少骨吸收及增加新骨形成,见图2。同时,CHENG等[59]在股骨头局部联合应用唑来膦酸和骨形态发生蛋白2治疗猪Legg-Perthes病的模型中发现,联合应用能够阻止股骨头塌陷,并且指出单纯骨形态发生蛋白2无法防止股骨头塌陷。MA等[57]对兔股骨头缺血性坏死模型行经头颈开窗打压植骨手术,对股骨头局用应用负载低浓度(0.005 g/L)唑来膦酸的羟基磷灰石,发现该方法不仅能够抑制股骨头局部破骨吸收,还可以促进新骨形成。AYAAY等[60]在猪股骨头缺血性坏死模型的实验中发现,骨内局部应用双膦酸盐能够有效维持股骨头结构、改善股骨头外形,并指出局部应用双膦酸盐可能是修复股骨头结构的最有效给药途径,因为其能在骨坏死修复启动前使双膦酸盐结合到骨表面发挥作用。总体来说,与静脉或者口服的系统用药方式相比,局部用药所需治疗剂量较小,局部可达到较高有效药物浓度,能够避免重复给药。同时,局部用药可使药物直接作用于靶器官,减少全身的药物吸收,避免了系统用药导致的全身不良反应[10-11]。此外有研究指出,系统用药的生物利用度和药物分配程度取决于股骨头的血运状态,而大部分股骨头坏死患者的股骨头局部血运受损,但局部用药不依赖于股骨头血运状态[61]。 "

但是,目前对局部应用双膦酸盐治疗股骨头缺血性坏死的最佳浓度仍存在争议。体外研究已显示低浓度双膦酸盐对成骨细胞产生激活作用,而高浓度则导致抑制作用[32-35]。但体内实验结果并不一致。JAKOBSEN等[8]关于双膦酸盐的剂量-反应实验研究发现,低浓度唑来膦酸(0.005 g/L)能够促进新骨形成,但抑制骨吸收作用较弱,而高浓度(0.005 g/L)能够明显地抑制骨吸收,但同时也抑制新骨形成。MA等[57]的研究指出,对兔缺血性坏死股骨头局部应用低浓度(0.005 g/L)唑来膦酸不仅能够抑制破骨吸收,还可以促进新骨形成。S?RENSEN等[62]的类似实验研究支持这一观点。该研究指出,低浓度(0.005 g/L)唑来膦酸能够抑制关节翻修术中移植骨的吸收,并且提高假体的稳定性。但也有研究提出不同的意见[43,60]。GOU等[43]利用聚乳酸羟基乙酸共聚物作为载体对鼠股骨远端骨缺损处释放不同剂量的双膦酸盐,发现中、高剂量组(30,300 μg)的双膦酸盐能够提高骨矿盐密度和矿化骨体积分数,还可减少破骨细胞数量、增加成骨细胞数量,同时可以加速骨形成标记物的表达,促进局部新骨形成。另一研究同样指出,对猪缺血性坏死股骨头内局部应用高剂量(560 μg)双膦酸盐能够增加骨小梁体积、骨小梁数量及结构,维持股骨头外形[60]。局部应用双膦酸盐治疗股骨头缺血性坏死的最佳浓度还需要高质量的实验及大样本的临床研究来明确。 "

| [1] KOROMPILIAS AV, BERIS AE, LYKISSAS MG, et al.Femoral head osteonecrosis: why choose free vascularized fibula grafting. Microsurgery.2011;31:223-228. [2] OHZONO K, TAKAOKA K, SAITO S, et al.Intraosseous arterial architecture in nontraumatic avascular necrosis of the femoral head: microangiographic and histologic study.Clin Orthop Relat Res. 1992; 277:79-88. [3] KIM HK.Pathophysiology and new strategies for the treatment of Legg-Calvé-Perthes disease. J Bone Joint Surg Am. 2012; 94:659-669. [4] KIM HK, SU PH. Development of flattening and apparent fragmentation following ischemic necrosis of the capital femoral epiphysis in a piglet model.J Bone Joint Surg Am. 2002;84:1329-1334. [5] RODAN GA, RESZKA AA.Bisphosphonate mechanism of action. Curr Mol Med.2002;2:571-577. [6] RESZKA AA, RODAN GA.Bisphosphonate mechanism of action. Curr Rheumatol Rep.2003;5:65-74. [7] MATHOV I, PLOTKIN LI, SGARLATA CL, et al.Extracellular signal-regulated kinases and calcium channels are involved in the proliferative effect of bisphosphonates on osteoblastic cells in vitro.J Bone Miner Res.2001;16:2050-2056. [8] JAKOBSEN T, BAAS J, BECHTOLD JE, et al.The effect of soaking allograft in bisphosphonate: A pilot dose response study.Clin Orthop Relat Res.2010;468:867874. [9] 谢小伟,谭振,裴福兴,等.二磷酸盐在防治股骨头坏死中的研究现状[J].中国矫形外科杂志,2013,21(9):893-896. [10] SHARMA D, IVANOVSKI S, SLEVIN M, et al. Bisphosphonate-related osteonecrosis of jaw (BRONJ): diagnostic criteria and possible pathogenic mechanisms of an unexpected anti-angiogenic side effect.Vasc Cell.2013;5(1):1. [11] VANDERMEER JS, KAMIYA N, AYA AY J, et al.Local administration of ibandronate and bone morphogenetic protein 2 after ischemic osteonecrosis of the immature femoral head: A combined therapy that stimulates bone formation and decreases femoral head deformity.J Bone Joint Surg Am. 2011;93:905-913. [12] FLEISCH H, BISAZ S.Isolation from urine of pyrophosphate, a calcification inhibitor. Am J Physiol.1962;203:671-675. [13] ROELOFS AJ, THOMPSON K, EBETINO FH, et al. Bisphosphonates: molecular mechanisms of action and effects on bone cells, monocytes andmacrophages. Curr Pharm Des. 2010;16(27):2950-2960. [14] RESZKA AA, RODAN GA.Mechanism of action of bisphosphonates. Curr Osteoporos Rep. 2003;1(2):45-52. [15] SHINODA H, ADAMEK G, FELIX R, et al.Structure-activity relationships of various bisphosphonates.Calcif Tissue Int. 1983;35(1):87-99. [16] EBETINO FH, FRANCIS MD, ROGERS MJ, et al. Mechanisms of action of etidronate and other bisphosphonates. Rev Contemp Pharmacother.1998;9:233-243. [17] THOMPSON K, ROGERS MJ, COXON FP, et al.Cytosolic entry of bisphosphonate drugs requires acidification of vesicles after fluid-phase endocytosis.Mol Pharmacol. 2006;69:1624-1632. [18] MURAKAMI H, TAKAHASHI N, SASAKI T, et al.A possible mechanism of the specific action of bisphosphonates on osteoclasts: Tiludronate preferentially affects polarized osteoclasts having ruffled borders.Bone.1995;17:137-144. [19] HIROI-FURUYA E, KAMEDA T, HIURA K, et al.Etidronate (EHDP) inhibits osteoclastic-bone resorption, promotes apoptosis and disrupts actin rings in isolate-mature osteoclasts.Calcif Tissue Int.1999;64:219-223. [20] COXON FP, ROGERS MJ.The role of prenylated small GTP-binding proteins in the regulation of osteoclast function. Calcif Tissue Int.2003;72:80-84. [21] FRITH JC, MÖNKKÖNEN J, AURIOLA S,et al.The molecular mechanism of action of the antiresorptive and antiinflammatory drug clodronate: Evidence for the formation in vivo of a metabolite that inhibits bone resorption and causes osteoclast and macrophage apoptosis.Arthritis Rheum.2001;44:2201-2210. [22] LEHENKARI PP, KELLINSALMI M, NÄPÄNKANGAS JP, et al. Further insight into the mechanism of action of clodronate: Inhibition of mitochondrial ADP/ATP translocase by a nonhydrolyzable, adeninecontaining metabolite.Mol Pharmacol. 2002;62:1255-1262. [23] VAN BEEK ER, LÖWIK CWGM, PAPAPOULOS SE. Bisphosphonates suppress bone resorption by a direct effect on early osteoclast precursors without affecting the osteoclastogenic capacity of osteogenic cells: The role of protein geranylgeranylation in the action of nitrogen-containing bisphosphonates on osteoclast precursors.Bone. 2002;30: 64-70. [24] D'AMELIO P, GRIMALDI A, DI BELLA S, et al.Risedronate reduces osteoclast precursors and cytokine production in postmenopausal osteoporotic women.J Bone Miner Res. 2008;23:373-379. [25] D'AMELIO P, GRIMALDI A, CRISTOFARO MA, et al. Alendronate reduces osteoclast precursors in osteoporosis. Osteoporos Int. 2010;21:1741-1750. [26] PLOTKIN LI, WEINSTEIN RS, PARFITT AM, et al.Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin.J Clin Invest.1999;104:1363-1374. [27] FOLLET H, LI J, PHIPPS RJ, et al.Risedronate and alendronate suppress osteocyte apoptosis following cyclic fatigue loading.Bone.2007;40:1172-1177. [28] PLOTKIN LI, MANOLAGAS SC, BELLIDO T.Transduction of cell survival signals by connexin-43 hemichannels.J Biol Chem. 2002;277:8648-8657. [29] PLOTKIN LI, LEZCANO V, THOSTENSON J, et al.Connexin 43 is required for the anti-apoptotic effect of bisphosphonates on osteocytes and osteoblasts in vivo.J Bone Miner Res.2008; 23:1712-1721. [30] REINHOLZ GG, GETZ B, PEDERSON L, et al. Bisphosphonates directly regulate cell proliferation, differentiation, and gene expression in human osteoblasts. Cancer Res. 2000;60:6001-6007. [31] DUQUE G, RIVAS D.Alendronate has an anabolic effect on bone through the differentiation of mesenchymal stem cells.J Bone Miner Res.2007;10:1603-1611. [32] VIERECK V, EMONS G, LAUCK V, et al.Bisphosphonates pamidronate and zoledronic acid stimulate osteoprotegerin production by primary human osteoblasts.Biochem Biophys Res Commun.2002; 291: 680-686. [33] PAN B, FARRUGIA AN, TO LB, et al.The nitrogen-containing bisphosphonate, zoledronic acid, influences RANKL expression in human osteoblast-like cells by activating TNF-alpha converting enzyme (TACE) J.Bone Miner Res. 2004;19:147-154. [34] IDRIS AI, ROJAS J, GREIG IR, et al.Aminobisphosphonates cause osteoblast apoptosis and inhibit bone nodule formation in vitro.Calcif Tissue Int.2008;82:191-201. [35] ORRISS IR, KEY ML, COLSTON KW, et al.Inhibition of osteoblast function in vitro by aminobisphosphonates.J Cell Biochem. 2009;106:109-118. [36] BELLIDO T, PLOTKIN LI.Novel actions of bisphosphonates in bone: Preservation of osteoblast and osteocyte viability. Bone. 2011;49:50-55. [37] BOULENC X, MARTI E, JOYEUX H, et al.Importance of the paracellular pathway for the transport of new bisphosphonate using the human CACO-2 monolayers model.Biochem Pharmacol. 1993;46:1591-1600. [38] KASTING GB, FRANCIS MD.Retention of etidronate in human, dog, and rat.J Bone Miner Res. 1992;7:513-522. [39] 商红,张石革,孙定人.双膦酸盐类药物的研究进展与临床合理应用[J].中国药房,2003,14(4):245-247. [40] ASSOULINE-DAYAN Y, CHANG C, GREENSPAN A, et al. Pathogenesis and natural history of osteonecrosis.Semin Arthritis Rheum. 2002;32:94-124. [41] BOSS JH, MISSELEVICH I.Osteonecrosis of the femoral head of laboratory animals: the lessons learned from a comparative study of osteonecrosis in man and experimental animals.Vet Pathol.2003;40:345-354. [42] KIM HK, KIM JH, ABBAS AA, et al.Alendronate enhances osteogenic differentiation of bone marrow stromal cells: A preliminary study.Clin Orthop Relat Res.2009;467:3121-3128. [43] GOU W, WANG X, PENG J, et al.Controlled delivery of zoledronate improved bone formation locally in vivo.PLoS One.2014;9:e91317. [44] LAI KA, SHEN WJ, YANG CY, et al.The use of alendronate to prevent early collapse of the femoral head in patients with nontraumatic osteonecrosis. A randomized clinical study.J Bone Joint Surg Am.2005;87(10):2155-2159. [45] NISHII T, SUGANO N, MIKI H, et al.Does alendronate prevent collapse in osteonecrosis of the femoral head? Clin Orthop Relat Res.2006;443:273-279. [46] AGARWALA S, JAIN D ,JOSHI VR, et al.Efficacy of alendronate, a bisphosphonate,in the treatment of AVN of the hip. A prospective open-label study. Rheumatology (Oxford). 2005;44(3):352-359. [47] AGARWALA S, SULE A, PAI BU, et al.Alendronate in the treatment of avascular necrosis of the hip.Rheumatology (Oxford). 2002;41(3):346-347. [48] AGARWALA S, SHAH SB.Ten-year follow-up of avascular necrosis of femoral head treated with alendronate for 3 years.J Arthroplasty.2011;26(7):1128-1134. [49] LUO RB, LIN T, ZHONG HM, et al.Evidence for using alendronate to treat adult avascular necrosis of the femoral head: a systematic review.Med Sci Monit.2014;20:2439-2447. [50] CHEN CH, CHANG JK, LAI KA, et al.Alendronate in the prevention of collapse of the femoral head in nontraumatic osteonecrosis: a two-year multicenter, prospective, randomized, double-blind, placebo-controlled study.Arthritis Rheum. 2012;64(5):1572-1578. [51] LEE YK, HA YC, CHO YJ, et al.Does Zoledronate Prevent Femoral Head Collapse from Osteonecrosis? A Prospective, Randomized, Open-Label, Multicenter Study.J Bone Joint Surg Am.2015;97(14):1142-1148. [52] YUAN HF, GUO CA, YAN ZQ.The use of bisphosphonate in the treatment of osteonecrosis of the femoral head: a meta-analysis of randomized control trials.Osteoporos Int. 2016;27(1):295-299. [53] GIANAKOS AL, MOYA-ANGELER J, DUGGAL S, et al.The Efficacy of Bisphosphonates with Core Decompression and Mesenchymal Stem Cells Compared with Bisphosphonates Alone in the Treatment of Osteonecrosis of the Hip: a Retrospective Study.HSS J.2016;12:137-144. [54] KANG P, PEI F, SHEN B, et al.Are the results of multiple drilling and alendronate for osteonecrosis of the femoral head better than those of multiple drilling? A pilot study. Joint Bone Spine. 2012;79:67-72. [55] WANG CJ, WANG FS, YANG KD, et al.Treatment of osteonecrosis of the hip: comparison of extracorporeal shockwave with shockwave and alendronate. Arch Orthop Trauma Surg. 2008;128(9):901-908. [56] MA J,SUN W,GAO F,et al.Porous Tantalum Implant in Treating Osteonecrosis of the Femoral Head: Still a Viable Option? Sci Rep.2016;6:28227. [57] MA JH, GUO WS, LI ZR, et al.Local Administration of Bisphosphonate-soaked Hydroxyapatite for the Treatment of Osteonecrosis of the Femoral Head in Rabbit. Chin Med J (Engl).2016;129(21): 2559-2566. [58] KIM HK, ARUWAJOYE O, DU J, et al.Local administration of bone morphogenetic protein-2 and bisphosphonate during non-weight-bearing treatment of ischemic osteonecrosis of the femoral head: an experimental investigation in immature pigs.J Bone Joint Surg Am.2014; 96(18):1515-1524. [59] CHENG TL, MURPHY CM, CANTRILL LC, et al.Local delivery of recombinant human bone morphogenetic proteins and bisphosphonate via sucrose acetate isobutyrate can prevent femoral head collapse in Legg-Calve-Perthes disease: a pilot study in pigs.Int Orthop.2014;38(7):1527-1533. [60] AYAAY J, ATHAVALE S, MORGAN BAGLEY S, et al. Retention, distribution, and effects of intraosseously administered ibandronate in the infarcted femoral head.J Bone Miner Res. 2007;22:93-100. [61] KIM HK, SANDERS M, ATHAVALE S, et al.Local bioavailability and distribution of systemically (parenterally) administered ibandronate in the infarcted femoral head.Bone. 2006;39:205-212. [62] SØRENSEN M, BARCKMAN J, BECHTOLD JE, et al. Preclinical evaluation of zoledronate to maintain bone allograft and improve implant fixation in revision joint replacement. J Bone Joint Surg Am.2013;95:1862-1868. |
| [1] | Wang Qiufei, Gu Ye, Peng Yuqin, Xue Feng, Ju Rong, Zhu Feng, Wang Yijun, Geng Dechun, Xu Yaozeng. Effect of Wnt/beta-catenin signaling pathway on osteoblasts under the action of wear particles [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3894-3901. |
| [2] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [3] | Huo Hua, Cheng Yuting, Zhou Qian, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Jian, Hong Wei. Effects of drug coating on implant surface on the osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3558-3564. |
| [4] | Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299. |
| [5] | Li Ping, Lin Yu, Chen Xiang, Liu Zhentao, Xiao Lili, Lin Xueyi, Hua Peng . Characteristics of bone remodeling in female ovariectomized rat models of osteoporosis undergoing Erzhi Pill extract intervention [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 191-195. |
| [6] | Wei Qin, Zhang Xue, Ma Lei, Li Zhiqiang, Shou Xi, Duan Mingjun, Wu Shuo, Jia Qiyu, Ma Chuang. Platelet-derived growth factor-BB induces the differentiation of rat bone marrow mesenchymal stem cells into osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2953-2957. |
| [7] | Guo Zhibin, Wu Chunfang, Liu Zihong, Zhang Yuying, Chi Bojing, Wang Bao, Ma Chao, Zhang Guobin, Tian Faming. Simvastatin stimulates osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2963-2968. |
| [8] | Zhang Shengmin, Cao Changhong, Liu Chao. Adipose-derived stem cells integrated with concentrated growth factors prevent bisphosphonate-related osteonecrosis of the jaws in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2982-2987. |
| [9] | Zhu Shiqiang, Xu Jianfeng, Hei Xiaoyan, Chen Yundong, Tian Xinbao, Zhang Jinchen, Lin Ruizhu. Effect of internal heat-type acupuncture needle therapy on the expression of type I collagen, matrix metalloproteinase-3 and osteopontin in the subchondral bone of rabbit knee osteoarthritis model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2636-2642. |
| [10] | Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279. |
| [11] | Li Dongdong, Liao Hongbing . MicroRNA-214 is involved in the regulation of bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1779-1784. |
| [12] | Chen Qiang, Zhuo Hongwu, Xia Tian, Ye Zhewei . Toxic effects of different-concentration isoniazid on newborn rat osteoblasts in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1162-1167. |
| [13] | Xu Nuo, Cao Zhen, Li Xiaojie, Shi Chun. MicroRNA-21 regulates proliferation and differentiation of osteoclasts in periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1225-1230. |
| [14] | Li Jinyu, Yu Xing, Jiang Junjie, Xu Lin, Zhao Xueqian, Sun Qi, Zheng Chenying, Bai Chunxiao, Liu Chuyin, Jia Yusong. Promoting effect of osteopractic total flavone combined with nano-bone materials on proliferation and differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1030-1036. |
| [15] | Qiao Jiutao, Guan Dehong, Wang Dongyan, Liu Aiyun. Zuogui Pill has protective effect against oxidative stress injury in osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1052-1056. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||