Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 3993-4000.doi: 10.3969/j.issn.2095-4344.0922
Previous Articles Next Articles
Transfusion with human umbilical cord mesenchymal stem cells improves liver function in hepatitis B patients with decompensated liver cirrhosis
Fu Qing-chun1, 2, Jin Yin-peng1, 2, Wang Xiao-jin1, Li Li2, Wang Xi1, Li Hong-chao1, Wang Zhao-jing3, Zhou Feng1, Zang Zu-sheng1, Shi Li-qin1, Li Zhen-yu1, Chen Cheng-wei1
- 1Shanghai Liver Disease Research Center, Nanjing Military Command, Shanghai 200235, China; 2Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China; 3Shenzhen Beike Cell Engineering Research Institute, Shenzhen 518040, Guangdong Province, China
-
Revised:2018-04-05Online:2018-09-08Published:2018-09-08 -
Contact:Chen Cheng-wei, Shanghai Liver Disease Research Center, Nanjing Military Command, Shanghai 200235, China -
About author:Fu Qing-chun, Master, Chief physician, Shanghai Liver Disease Research Center, Nanjing Military Command, Shanghai 200235, China; Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China. Jin Yin-peng, Master, Shanghai Liver Disease Research Center, Nanjing Military Command, Shanghai 200235, China; Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China. Fu Qing-chun and Jin Yin-peng contributed equally to this work. -
Supported by:the Medical Innovation Project of Nanjing Military Command, No. 10MA001
CLC Number:
Cite this article
Fu Qing-chun, Jin Yin-peng, Wang Xiao-jin, Li Li, Wang Xi, Li Hong-chao, Wang Zhao-jing, Zhou Feng, Zang Zu-sheng, Shi Li-qin, Li Zhen-yu, Chen Cheng-wei. Transfusion with human umbilical cord mesenchymal stem cells improves liver function in hepatitis B patients with decompensated liver cirrhosis[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 3993-4000.
share this article

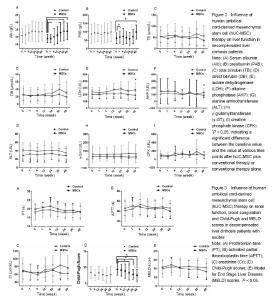
Quantitative analysis of patients The study flow is presented in Figure 1. A total of 138 hepatitis B patients with DLC were recruited. Three patients were excluded because they did not meet the inclusion criteria, and 101 patients were excluded because they did not provide one-on-one correspondence. Seventeen hepatitis B patients were recruited to the human umbilical cord-derived mesenchymal stem cell (hUC-MSC) treatment group and the control group contained 17 clinically matched individuals. Baseline comparisons and clinical symptoms The baseline characteristics are presented in Table 1. The patients in the MSC-treated and control groups were similar regarding sex, age and biochemical data. The baseline Child-Pugh (P=0.149) and MELD (P=0.661) scores were also similar. The patients in the two groups did not differ regarding the course of DLC (P=0.333; Figure 2A). and control groups had undetectable levels of HBV DNA in their serum, with no significant differences detected between the groups (P=0.247). The antiviral length was also comparable between the groups (P=0.704). Ascites gradually decreased in both the MSC-treatment and control groups following the administration of albumin and diuretics and no significant differences were detected between the groups (P=0.203). Liver function tests Forty-eight weeks after treatment, Alb levels increased from (28.9±2.8) g/L to (33.9±5.7) g/L in the MSC-treatment group, which was significantly higher than the baseline (P < 0.05; Figure 2A). PAB increased from (62.1±32.7) g/L to (98.7±41.6) g/L in MSC-treated patients, which was also significantly higher than the baseline value (P < 0.05; Figure 2B). Alb and PAB levels did not differ significantly to the baseline values after 48 weeks of conventional treatment alone (Figure 2A, B). The differences in Alb and PAB levels were not significant between the two groups at different time points (Figure 2A, B). TB levels at 48 weeks after treatment were: MSC-treatment group, 53.9 to 37.0 μmol/L; control group, (54.6±45.3) to (55.7±47.0) μmol/L (Figure 2C, D). The differences between the two groups at each time point were not statistically significant. The levels of LDH and AKP were significantly lower in the MSC-treatment group compared to the control group 48 weeks after treatment (P < 0.05; Figure 2E, F). Forty-eight weeks after treatment, the LDH values were: MSC-treatment group, (191.4±63.7) U/L; control group, (272.2±105.3) U/L. The AKP values 48 weeks after treatment were: MSC-treatment group, (87.2±33.7) U/L; control group, (133.8±58.3) U/L. The levels of ALT, γ-GT and CPK did not differ significantly between the MSC-treatment and control groups at all time points (Figure 2G, H, I). Blood coagulation tests Cirrhosis patients have an increased risk for bleeding. We assessed the coagulation function in HBV patients with DLC by measuring the PT and APTT. The PT did not differ significantly between the MSC-treated and control patients at various time points after treatment (Figure 3A). We observed a faster APTT in the MSC-treatment group 4 weeks after treatment and the APTT continued to decrease in the MSC-treated patients until 48 weeks after treatment (Figure 3B). The APPT values at 48 weeks after treatment were: MSC-treatment group, 39.9 seconds (7.5-second decrease); control group, 43.1 seconds (7.5-second increase). The differences between the two groups at each time point were not statistically significant. Renal function Cr levels in the blood were measured to assess renal function[35]. We detected a decline in Cr levels in the blood of MSC-treated patients compared to control patients (Figure 3C), which indicated improved renal function. However these differences were not statistically significant, although this may reflect the low number of participants, rather than a genuine lack of significance. Child-Pugh and MELD scores The Child-Pugh and MELD scores were calculated to evaluate the severity of cirrhosis in the patients[30]. The Child-Pugh score declined significantly from 9.4 points at the baseline to 7.5 points measured 48 weeks after treatment in the MSC-treatment group, (P < 0.05; Figure 3D), indicating a better prognosis. The MELD score also declined from 15.8 to 13.3 points in MSC-treated patients, while in the control group, the MELD score rose slightly from 15.1 points at the baseline to 15.5 points 48 weeks after treatment (Figure 3E). This indicates a better prognosis in patients treated with hUC-MSCs compared to patients who received conventional treatment alone, although the differences between the two groups in each time point were not statistically significant. Evaluation of potential complications following hUC-MSC transfusion No abnormal changes were detected in the vital signs, electrocardiogram results and renal function of patients following transfusion with hUC-MSCs which were observed at the time of transfusion. Two patients developed a fever and a reduced platelet count was detected in two further patients. In one patient we observed an increase in the INR to 8.21, which recovered baseline after 72 hours without any treatment, although this decreased within 4 days and no hemorrhage-related complications were observed. No tumor development was observed during follow-up examinations in MSC-treated patients. Other potential serious adverse reactions associated with end-stage liver diseases, such as hepatorenal syndrome, hepatic encephalopathy and pleural effusion, were not related to MSC-treatment. There was no statistically significant difference in the adversity of the reaction between the two groups."

| [1] Mast EE, Alter MJ, Margolis HS. Strategies to prevent and control hepatitis B and C virus infections: a global perspective. Vaccine. 1999;17(13-14):1730-1733.[2] Park TW, Park YM, Bae SH, et al. Efficacy and safety of long-term lamivudine therapy in the patients with decompensated liver cirrhosis secondary to hepatitis B. Taehan Kan Hakhoe Chi. 2002;8(4):428-435.[3] Krautbauer S, Rein-Fischboeck L, Haberl EM, et al. Circulating fibroblast growth factor 21 in patients with liver cirrhosis. Clin Exp Med. 2018;18(1):63-69. [4] Fraser AR, Pass C, Burgoyne P, et al. Development, functional characterization and validation of methodology for GMP-compliant manufacture of phagocytic macrophages: A novel cellular therapeutic for liver cirrhosis. Cytotherapy. 2017;19(9):1113-1124..[5] Alexopoulou A, Agiasotelli D, Vasilieva LE, et al. Bacterial translocation markers in liver cirrhosis. Ann Gastroenterol. 2017;30(5):486-497.[6] de Jongh FE, Janssen HL, de Man RA, et al. Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology. 1992;103(5):1630-1635.[7] Mehta N, Dodge JL, Roberts JP, et al. Outcomes after liver transplantation for patients with hepatocellular carcinoma and a low risk of dropout from the transplant waiting list. Liver Transpl. 2014;20(5):627-628. [8] Takaki A, Yasunaka T, Yagi T. Molecular mechanisms to control post-transplantation hepatitis B recurrence. Int J Mol Sci. 2015;16(8):17494-17513. [9] Navarro Burgos JB, Lee KW, Shin YC, et al. Inexplicable outcome of early appearance of hepatocellular carcinoma in the allograft after deceased donor liver transplantation: a case report. Transplant Proc. 2015;47(10):3012-3015. [10] Lorenzini S, Gitto S, Grandini E, et al. Stem cells for end stage liver disease: how far have we got? World J Gastroenterol. 2008;14(29): 4593-4599.[11] Tatsumi K, Okano T. Hepatocyte transplantation: cell sheet technology for liver cell transplantation. Curr Transplant Rep. 2017;4(3):184-192. [12] Belaschk E, Rohn S, Mukiibi R, et al. Isolation, characterization and cold storage of cells isolated from diseased explanted livers. Int J Artif Organs. 2017;40(6):294-306.[13] Tacke F, Zimmermann HW. Macrophage heterogeneity in liver injury and fibrosis. J Hepatol. 2014;60(5):1090-1096. [14] Melief SM, Schrama E, Brugman MH, et al. Multipotent stromal cells induce human regulatory T cells through a novel pathway involving skewing of monocytes toward anti-inflammatory macrophages. Stem Cells. 2013;31(9):1980-1991. [15] Eggenhofer E, Hoogduijn MJ. Mesenchymal stem cell-educated macrophages. Transplant Res. 2012;1(1):12. [16] Jones S, Horwood N, Cope A, et al. The antiproliferative effect of mesenchymal stem cells is a fundamental property shared by all stromal cells. J Immunol. 2007;179(5):2824-2831.[17] Choi H, Lee RH, Bazhanov N, et al. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-κB signaling in resident macrophages. Blood. 2011;118(2):330-338. [18] Ortiz LA, Dutreil M, Fattman C, et al. Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc Natl Acad Sci U S A. 2007;104(26):11002-11007. [19] Bianco P. "Mesenchymal" stem cells. Annu Rev Cell Dev Biol. 2014;30:677-704.[20] Shi R, Jin Y, Cao C, et al. Localization of human adipose-derived stem cells and their effect in repair of diabetic foot ulcers in rats. Stem Cell Res Ther. 2016;7(1):155.[21] Fan CG, Zhang QJ, Zhou JR. Therapeutic potentials of mesenchymal stem cells derived from human umbilical cord. Stem Cell Rev. 2011;7(1):195-207. [22] Zhang X, Li J, Ye P, et al. Coculture of mesenchymal stem cells and endothelial cells enhances host tissue integration and epidermis maturation through AKT activation in gelatin methacryloyl hydrogel-based skin model. Acta Biomater. 2017;59:317-326.[23] Wang HX, Gao XW, Ren B, et al. Comparative analysis of different feeder layers with 3T3 fibroblasts for culturing rabbits limbal stem cells. Int J Ophthalmol. 2017;10(7):1021-1027. [24] Li T, Liu Y, Yu L, et al. Human umbilical cord mesenchymal stem cells protect against SCA3 by modulating the level of 70 kD heat shock protein. Cell Mol Neurobiol. 2017. doi: 10.1007/s10571-017-0513-1.[25] Campard D, Lysy PA, Najimi M, et al. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology. 2008;134(3):833-848.[26] Li Y, Wu Q, Wang Y, et al. Construction of bioengineered hepatic tissue derived from human umbilical cord mesenchymal stem cells via aggregation culture in porcine decellularized liver scaffolds. Xenotransplantation. 2017;24(1). [27] Chen Z, Kuang Q, Lao XJ, et al. Differentiation of UC-MSCs into hepatocyte-like cells in partially hepatectomized model rats. Exp Ther Med. 2016;12(3):1775-1779. [28] Chitrangi S, Nair P, Khanna A. 3D engineered in vitro hepatospheroids for studying drug toxicity and metabolism. Toxicol In Vitro. 2017;38:8-18. [29] Gai XD, Wu WF. Effect of entecavir in the treatment of patients with hepatitis B virus-related compensated and decompensated cirrhosis. Exp Ther Med. 2017;14(4):3908-3914.[30] Peng Y, Qi X, Guo X. Child-Pugh versus MELD score for the assessment of prognosis in liver cirrhosis: a systematic review and meta-analysis of observational studies. Medicine (Baltimore). 2016;95(8):e2877. [31] Zhang C, Yin X, Zhang J, et al. Clinical observation of umbilical cord mesenchymal stem cell treatment of severe idiopathic pulmonary fibrosis: A case report. Exp Ther Med. 2017;13(5):1922-1926.[32] Chao K, Zhang S, Qiu Y, et al. Human umbilical cord-derived mesenchymal stem cells protect against experimental colitis via CD5(+) B regulatory cells. Stem Cell Res Ther. 2016;7(1):109. [33] Xu Y, Wu XN, Shi YW, et al. Baseline hepatitis B virus DNA level is a promising factor for predicting the 3 (rd) month virological response to entecavir therapy: a study of strict defined Hepatitis B virus induced cirrhosis. Chin Med J (Engl). 2015;128(14):1867-1872. [34] Guan R, Lui HF. Treatment of hepatitis B in decompensated liver cirrhosis. Int J Hepatol. 2011;2011:918017.[35] Zeng SY, Xing LY, Hou HB, et al. The clinical diagnostic value of cystatin C, creatinine, urea and uric acid levels in renal function injury was investigated. Zhongguo Yixue Chuangxin. 2011;8(10):26-27.[36] Forraz N, McGuckin CP. The umbilical cord: a rich and ethical stem cell source to advance regenerative medicine. Cell Prolif. 2011;44 Suppl 1:60-69. [37] Kim YJ, Yoo SM, Park HH, et al. Exosomes derived from human umbilical cord blood mesenchymal stem cells stimulates rejuvenation of human skin. Biochem Biophys Res Commun. 2017;493(2):1102-1108.[38] Dehghani-Soltani S, Shojaee M, Jalalkamali M, et al. Effects of light emitting diode irradiation on neural differentiation of human umbilical cord-derived mesenchymal cells. Sci Rep. 2017;7(1):9976. [39] Dong HJ, Shang CZ, Li G, et al. The distribution of transplanted umbilical cord mesenchymal stem cells in large blood vessel of experimental design with traumatic brain injury. J Craniofac Surg. 2017;28(6):1615-1619.[40] Hassan G, Kasem I, Soukkarieh C, et al. A simple method to isolate and expand human umbilical cord derived mesenchymal stem cells: using explant method and umbilical cord blood serum. Int J Stem Cells. 2017 ;10(2):184-192. [41] Hodgkinson CP, Naidoo V, Patti KG, et al. Abi3bp is a multifunctional autocrine/paracrine factor that regulates mesenchymal stem cell biology. Stem Cells. 2013;31(8):1669-1682.[42] Alvarez-Dolado M, Martínez-Losa M. Cell fusion and tissue regeneration. Adv Exp Med Biol. 2011;713:161-175. [43] Chen W, Liu X, Chen Q, et al. Angiogenic and osteogenic regeneration in rats via calcium phosphate scaffold and endothelial cell co-culture with human bone marrow mesenchymal stem cells (MSCs), human umbilical cord MSCs, human induced pluripotent stem cell-derived MSCs and human embryonic stem cell-derived MSCs. J Tissue Eng Regen Med. 2017. doi: 10.1002/term.2395.[44] Higashiyama R, Inagaki Y, Hong YY, et al. Bone marrow-derived cells express matrix metalloproteinases and contribute to regression of liver fibrosis in mice. Hepatology. 2007 ;45(1):213-222.[45] Wang J, Bian C, Liao L, et al. Inhibition of hepatic stellate cells proliferation by mesenchymal stem cells and the possible mechanisms. Hepatol Res. 2009;39(12):1219-1228. [46] Shen Q, Chen B, Xiao Z, et al. Paracrine factors from mesenchymal stem cells attenuate epithelial injury and lung fibrosis. Mol Med Rep. 2015;11(4):2831-2837.[47] Houtgraaf JH, de Jong R, Kazemi K, et al. Intracoronary infusion of allogeneic mesenchymal precursor cells directly after experimental acute myocardial infarction reduces infarct size, abrogates adverse remodeling, and improves cardiac function. Circ Res. 2013;113(2):153-166.[48] Parekkadan B, van Poll D, Megeed Z, et al. Immunomodulation of activated hepatic stellate cells by mesenchymal stem cells. Biochem Biophys Res Commun. 2007;363(2):247-252.[49] Zhang Z, Lin H, Shi M, et al. Human umbilical cord mesenchymal stem cells improve liver function and ascites in decompensated liver cirrhosis patients. J Gastroenterol Hepatol. 2012;27 Suppl 2:112-120. [50] Parekkadan B, van Poll D, Suganuma K, et al. Mesenchymal stem cell-derived molecules reverse fulminant hepatic failure. PLoS One. 2007;2(9):e941.[51] Brückner S, Tautenhahn HM, Winkler S, et al. A fat option for the pig: hepatocytic differentiated mesenchymal stem cells for translational research. Exp Cell Res. 2014;321(2):267-275. [52] Chen G, Jin Y, Shi X, et al. Adipose-derived stem cell-based treatment for acute liver failure. Stem Cell Res Ther. 2015;6:40.[53] Spaeth E, Klopp A, Dembinski J, et al. Inflammation and tumor microenvironments: defining the migratory itinerary of mesenchymal stem cells. Gene Ther. 2008;15(10):730-738. [54] Jung JW, Kwon M, Choi JC, et al. Familial occurrence of pulmonary embolism after intravenous, adipose tissue-derived stem cell therapy. Yonsei Med J. 2013;54(5):1293-1296.[55] Glassberg MK, Minkiewicz J, Toonkel RL, et al. Allogeneic human mesenchymal stem cells in patients with idiopathic pulmonary fibrosis via intravenous delivery (AETHER): a phase I safety clinical trial. Chest. 2017;151(5):971-981. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Ruan Guangping, Yao Xiang, Liu-Gao Miyang, Cai Xuemin, Li Zian, Pang Rongqing, Wang Jinxiang, Pan Xinghua. Umbilical cord mesenchymal stem cell transplantation for traumatic systemic inflammatory response syndrome in tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3994-4000. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||