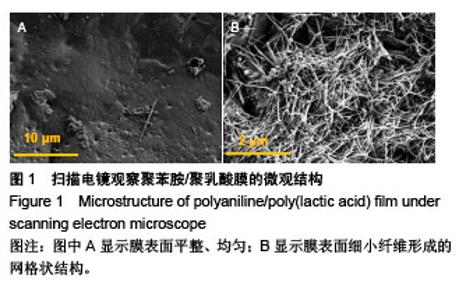
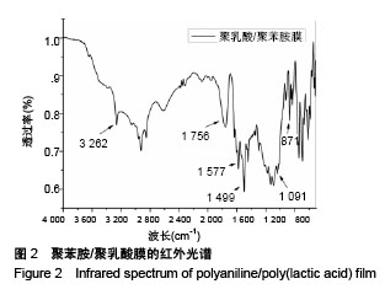
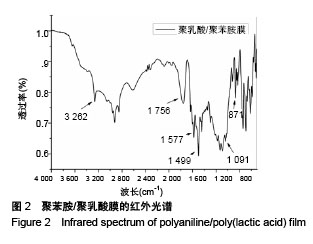
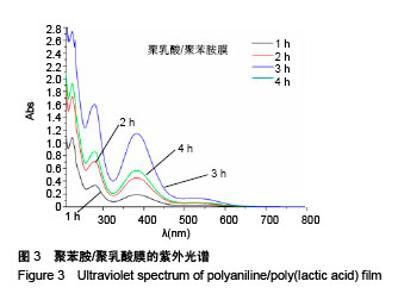
Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (22): 3557-3562.doi: 10.3969/j.issn.2095-4344.0755
Previous Articles Next Articles
Preparation and biocompatibility of an electroactive polyaniline/poly(lactic-acid) scaffold
Wang Ling-ling, Cui Zhi-ming, Xu Guan-hua, Li Wei-dong, Bao Guo-feng, Sun Yu-yu, Chen Jia-jia, Zhang Jin-long
- Department of Spinal Surgery, the Second Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China
-
Received:2017-12-12Online:2018-08-08Published:2018-08-08 -
Contact:Xu Guan-hua, M.D., Associate chief physician, Department of Spinal Surgery, the Second Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China -
About author:Wang Ling-ling, Attending physician, Department of Spinal Surgery, the Second Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China -
Supported by:Health Research Project of Jiangsu Province, No. Z201316; the 10th Batch High-Level Talent Project in Jiangsu Province, No. 2013-WSW-007; the Science and Technology Project of Jiangsu Provincial Traditional Chinese Medicine Administration, No. LB13020; the Major Science and Technology Project of Nantong Municipality, No. HS2014002
CLC Number:
Cite this article
Wang Ling-ling, Cui Zhi-ming, Xu Guan-hua, Li Wei-dong, Bao Guo-feng, Sun Yu-yu, Chen Jia-jia, Zhang Jin-long. Preparation and biocompatibility of an electroactive polyaniline/poly(lactic-acid) scaffold[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(22): 3557-3562.
share this article
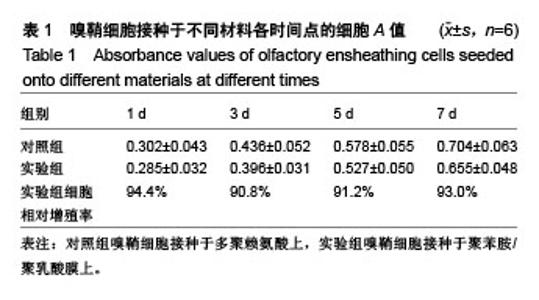
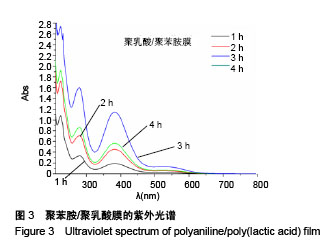
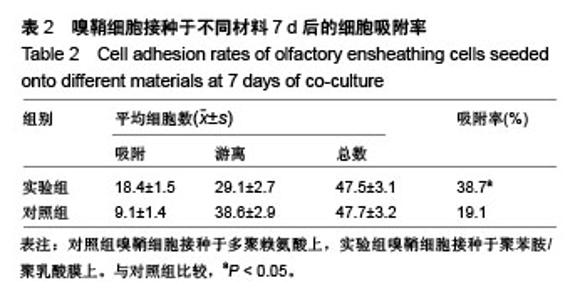
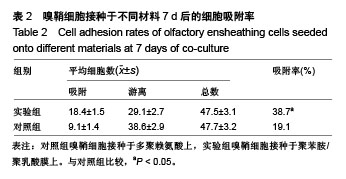
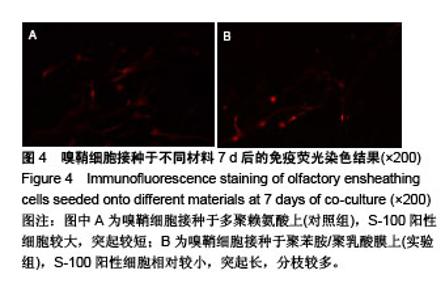
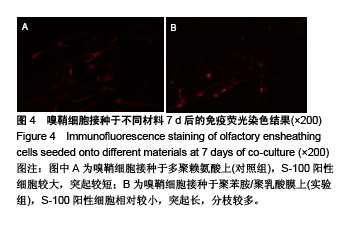
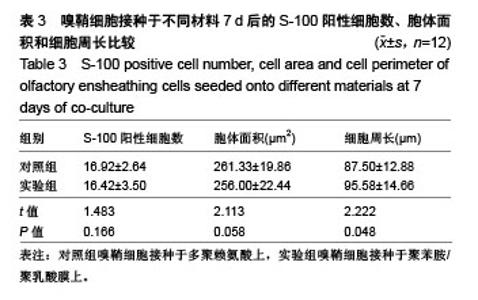
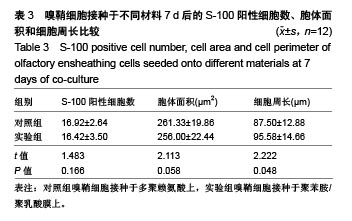
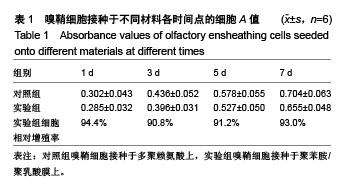
2.4 聚苯胺/聚乳酸膜的电导率测量结果 用循环伏安法测定聚苯胺/聚乳酸膜的电导率为1.5×10-5 S/cm。生物电为微电流,生物组织或器官的电压一般低于150 mV,同时电流的变化电流的变化范围在0.6-400 μA之间。应用于神经修复的平均电压为100 mV,当100 mV电压加到聚苯胺/聚乳酸材料上时,将产生1-100 μA的电流,符合生物电的电流要求。因此,合成的聚苯胺/聚乳酸电导率能满足刺激神经生长的需要。 2.5 嗅鞘细胞增殖检测结果 嗅鞘细胞与聚苯胺/聚乳酸膜共培养不同时间点的吸光度A值见表1,在各时间点实验组的细胞相对增殖率均大于90%,第7天时细胞相对增殖率为93%,显示聚苯胺/聚乳酸膜对嗅鞘细胞的毒性较低,为Ⅰ级。"
| [1] Dong ZY,Pei Z,Li Z,et al. Electric field stimulation induced neuronal differentiation of filum terminale derived neural progenitor cells.Neurosci Lett.2017;651:109-115.[2] Dodel M,Hemmati Nejad N,Bahrami SH,et al.Electrical stimulation of somatic human stem cells mediated by composite containing conductive nanofibers for ligament regeneration.Biologicals.2017;46:99-107.[3] Lee JY,Bashur CA,Goldstein AS,et al.Polypyrrole-Coated Electrospun PLGA Nanofibers for Neural Tissue Applications. Biomaterials.2009;30(26):4325-4335.[4] Abidian MR,Corey JM,Kipke DR,et al.Conducting-polymer nanotubes improve electrical properties, mechanical adhesion, neural attachment, and neurite outgrowth of neural electrodes[J]. Small.2010;6(3):421-429.[5] 李梦燕,Paul B,Elizabeth GT,等.电活性导电聚合物组织工程支架材料研究进展[J].中国医学科学院学报, 2006,28(6): 845-848.[6] Huang L,Hu J,Lang L,et al.Synthesis and characterization of electroactive and biodegradable ABA block copolymer of polylactide and aniline pentamer.Biomaterials. 2007;28(10): 1741-1751.[7] Sudwilai T,Ng JJ,Boonkrai C,et al.Polypyrrole-coated electrospun poly(lactic acid) fibrous scaffold: effects of coating on electrical conductivity and neural cell growth.J Biomater Sci Polym Ed.2014;25(12):1240-1252.[8] Li L,Adnan H,Xu B,et al.Effects of transplantation of olfactory ensheathing cells in chronic spinal cord injury: a systematic review and meta-analysis.Eur Spine J. 2015; 24(5):919-930.[9] Ekberg JA,St John JA.Crucial roles for olfactory ensheathing cells and olfactory mucosal cells in the repair of damaged neural tracts.Anat Rec(Hoboken). 2014;297(1): 121-128.[10] Veeraraghavan R,Poelzing S,Gourdie RG.Intercellular electrical communication in the heart: a new, active role for the intercalated disk.Cell Commun Adhes. 2014;21(3): 161-167.[11] Spitzer KW,Pollard AE,Yang L,et al.Cell-to-cell electrical interactions during early and late repolarization.J Cardiovasc Electrophysiol.2006;17 Suppl 1:S8-S14.[12] Hamid S,Hayek R.Role of electrical stimulation for rehabilitation and regeneration after spinal cord injury: an overview.Eur Spine J.2008;17(9):1256-1269.[13] Carmel JB,Berrol LJ,Brus-Ramer M,et al.Chronic electrical stimulation of the intact corticospinal system after unilateral injury restores skilled locomotor control and promotes spinal axon outgrowth.J Neurosci.2010;30(32):10918-10926.[14] Ide C, Kanekiyo K.Points regarding cell transplantation for the treatment of spinal cord injury.Neural Regen Res. 2016;11(7): 1046-1049.[15] Straley KS,Foo CW,Heilshorn SC.Biomaterial design strategies for the treatment of spinal cord injuries.J Neurotrauma.2010;27(1):1-19.[16] Salem N, Salem MY, Elmaghrabi MM,et al.Does vitamin C have the ability to augment the therapeutic effect of bone marrow-derived mesenchymal stem cells on spinal cord injury? Neural Regen Res. 2017;12(12):2050-2058.[17] Agbay A,Edgar JM,Robinson M,et al. Biomaterial strategies for delivering stem cells as a treatment for spinal cord injury.Cells Tissues Organs.2016;202(1-2):42-51.[18] Kotwal A,Schmidt CE.Electrical stimulation alters protein adsorption and nerve cell interactions with electrically conducting biomaterials.Biomaterials. 2001;22(10): 1055-1064.[19] Vallejo-Giraldo C,Pugliese E,Larrañaga A,et al. Polyhydroxyalkanoate/carbon nanotube nanocomposites: flexible electrically conducting elastomers for neural applications. Nanomedicine (Lond).2016;11(19):2547-2563.[20] Scholkmann F.Two emerging topics regarding long-range physical signaling in neurosystems: Membrane nanotubes and electromagnetic fields.J Integr Neurosci. 2015;14(2): 135-153.[21] Wu Y,Wang L,Guo B,et al.Electroactive biodegradable polyurethane significantly enhanced Schwann cells myelin gene expression and neurotrophin secretion for peripheral nerve tissue engineering. Biomaterials.2016;87:18-31.[22] Qazi TH,Rai R,Dippold D,et al.Development and characterization of novel electrically conductive PANI-PGS composites for cardiac tissue engineering applications.Acta Biomater.2014; 10(6):2434-2445.[23] Vallejo-Giraldo C,Kelly A,Biggs MJ.Biofunctionalisation of electrically conducting polymers. Drug Discov Today. 2014; 19(1):88-94.[24] Bidez PR 3rd,Li S,Macdiarmid AG,et al.Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts.J Biomater Sci Polym Ed. 2006;17(1-2): 199-212.[25] Wang L,Huang Q,Wang JY.Nanostructured polyaniline coating on ITO glass promotes the neurite outgrowth of PC12 cells by electrical stimulation.Langmuir. 2015;31(44): 12315-12322.[26] Muller D,Silva JP,Rambo CR,et al.Neuronal cells' behavior on polypyrrole coated bacterial nanocellulose three-dimensional (3D) scaffolds.J Biomater Sci Polym Ed. 2013;24(11): 1368-1377.[27] Pedrotty DM,Koh J,Davis BH,et al.Engineering skeletal myoblasts: roles of three dimensional culture and electrical stimulation.Am J Physiol Heart Circ Physiol. 2005;288(4): H1620-1626.[28] Qazi TH,Rai R,Boccaccini AR.Tissue engineering of electrically responsive tissues using polyaniline based polymers: a review.Biomaterials.2014;35(33):9068-9086.[29] You C,Wu H,Wang M,et al.A strategy for photothermal conversion of polymeric nanoparticles by polyaniline for smart control of targeted drug delivery.Nanotechnology. 2017;28(16): 165102.[30] Attia NF, Geckeler KE.Polyaniline as a material for hydrogen storage applications. Macromol Rapid Commun. 2013;34(13): 1043-1055.[31] Li M,Guo Y,Wei Y,et al.Electrospinning polyaniline-contained gelatin nanofibers for tissue engineering applications. Biomaterials.2006;27(13):2705-2715.[32] Kamalesh S,Tan P,Wang J,et al. Biocompatibility of electroactive polymers in tissues.J Biomed Mater Res. 2000; 52(3):467-478.[33] Bergström JS,Hayman D.An overview of mechanical properties and material modeling of polylactide (PLA) for medical applications.Ann Biomed Eng.2016;44(2):330-340.[34] Prabhakaran MP,Ghasemi-Mobarakeh L,Jin G,et al. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells.J Biosci Bioeng. 2011;112(5): 501-507.[35] Liu Y,Cui H,Zhuang X,et al.Electrospinning of aniline pentamer-graft-gelatin/PLLA nanofibers for bone tissue engineering.Acta Biomater.2014;10(12):5074-5080.[36] 李卫东,崔志明,徐冠华,等.壳聚糖增强型左旋聚乳酸与嗅鞘细胞的生物相容性[J].中国组织工程研究,2013,17(29):5316-5322. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||